Abstract
Background:
Current guidance on the selection of appropriate contraception for people with multiple sclerosis (PwMS) is lacking.
Objective:
To address this gap, an expert-led consensus program developed recommendations to support clinicians in discussing family planning and contraception with women and men with multiple sclerosis (MS).
Methods:
A multidisciplinary steering committee (SC) of 13 international clinical experts led the program, supported by an extended faculty of 32 experts representing 18 countries. A modified Delphi methodology was used for decision-making and consensus-building. The SC drafted 15 clinical questions focused on patient-centered care, selection of contraception, and timing of stopping/starting contraception and disease-modifying therapies (DMTs). Statements addressing each question were drafted based on evaluation of published evidence and the experts’ clinical experience. Consensus was reached if ⩾75% of respondents agreed (scoring 7–9 on a 9-point scale) with each recommendation.
Results:
Consensus was reached on 24 of 25 proposed recommendations, including how and when to discuss contraception, types and safety of contraceptives, and how to evaluate the most appropriate contraceptive options for specific patient groups, including those with significant disability or being treated with DMTs.
Conclusion:
These expert recommendations provide the first practical, relevant, and comprehensive guidance for clinicians on the selection of contraception in PwMS.
Keywords
Introduction
Multiple sclerosis (MS) typically occurs during childbearing years.1,2 Women and men with MS have a unique set of factors to consider during family planning and when choosing contraception.1–3
People with multiple sclerosis (PwMS) may want to delay or prevent pregnancy for several reasons.3,4 Conception is not advised during treatment with potentially gonadotoxic or teratogenic disease-modifying therapies (DMTs) because of potential risks to the embryo/fetus.5–7 However, stopping certain DMTs (e.g. natalizumab, fingolimod) without providing an alternative effective treatment can increase the risk of rebound relapse. 8 Furthermore, pregnancy can affect the course of MS, lowering the risk of relapse between the first and third trimester but increasing the risk of relapse postpartum in some patients.3,4 Counseling with a multidisciplinary team (MDT) can help PwMS evaluate their options for safe and effective contraception and other family planning decisions, including optimal timing of discontinuation/resumption of MS therapies.2,5,9,10
Current contraceptive guidelines do not include specific recommendations for PwMS and provide limited guidance on the options and practicalities that are most appropriate for this patient group.11–15 Consensus-based recommendations can guide clinicians where there are gaps in published guidelines. Here, we describe a consensus-based program, led by a group of international experts, designed to provide practical recommendations relating to contraception for women and men with MS. The recommendations focus on three topics: patient-centered care, selection of contraception for PwMS, and timing of stopping/starting contraception and DMTs.
A plain language summary video of the program and results can be found in the supplementary materials.
Materials and methods
The consensus program was based on a modified Delphi methodology as described by Sørensen et al. 16 (Figure 1). A multidisciplinary steering committee (SC) of 13 international clinical experts led the program, co-chaired by Jan Hillert and Manuela Simoni. Three meetings took place between November 2021 and April 2022.
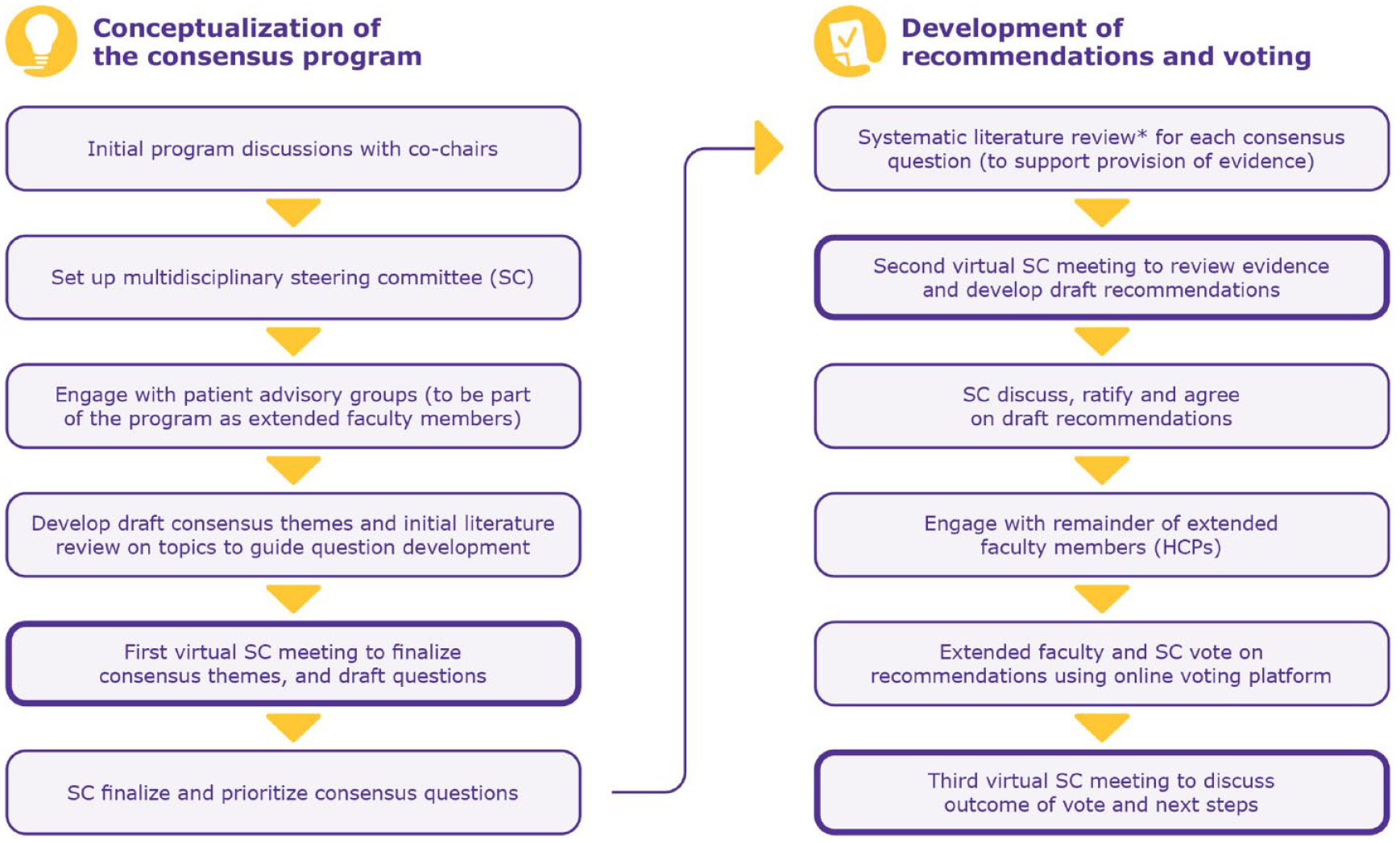
Overview of the modified Delphi process for achieving consensus (modified from Sørensen et al. 16 ).
After an initial review of published literature on contraception and MS, the SC convened to discuss and agree on the consensus themes, and developed 15 clinical questions. An in-depth systematic literature review (SLR; Supplementary methodology) was performed to address the 15 questions, using the PICOS (population, interventions and comparisons, outcomes of interest, and study design) framework (Supplementary Table S1). Evidence levels were assessed using the GRADE (Grading of Recommendations Assessment, Development, and Evaluation) rating scale. 17 The level of evidence for each question was dependent on the number of references identified in the SLR: low (0–9), medium (10–19), and high (⩾20).
After reviewing the evidence, the SC drafted clinical recommendations for each question. An extended faculty (EF) of 91 clinical experts were invited to vote on the clinical recommendations, with the aim of an EF participation rate of ~25%. EF members were selected based on their publications and research interests, and recommendations from the SC. Of the EF, 32 experts from 18 countries participated, and included nurses, patient advisory groups, neurologists, and gynecologists (participation rate, 35.2%).
SC (n = 12) and EF (n = 32) members voted on clinical recommendations (N = 44) using an online platform. Consensus was achieved when ⩾75% of respondents agreed in the range of 7–9 (9-point scale). Each statement/recommendation received a strength score (median) and consensus level (% of votes with a score of 7–9). 18 Responders were asked to provide their rationale for scores of ⩽6; those who felt unqualified to vote could select “not applicable.”
Results
The process of identifying and selecting literature is summarized in Supplementary methodology and Supplementary Figure S1. First-pass screening (title and/or abstract based) of 142 citations identified 75 relevant articles. Most articles (84%) were published between 2011 and 2022. Of the 72 full-text publications and 3 congress abstracts, there were 33 original articles (Supplementary results), 34 reviews, 3 SLRs, 4 recommendations, and 1 commentary.
The SC drafted 25 recommendations for voting on three themes: patient-centered care, selection of contraception for PwMS, and timing of stopping/starting contraception and DMTs. After one round of voting, consensus was reached on 24/25 recommendations, with 11 reaching 90%–100%, 12 achieving 80%–89%, and 1 in the range of 75%–79%. All but one SC member voted on all questions. Twenty-three (52.3%) respondents deemed themselves “not qualified” to vote on ⩾1 recommendation and selected “not applicable.” Therefore, the number of experts voting on each recommendation ranged from 27 to 44 participants.
Key evidence for each question is summarized below, and the clinical recommendations are provided in Tables 1–3. Supplementary Tables S2–S4 list respondent reasons for scores ⩽6. Supplementary Table S5 summarizes SLR outputs for each question.
Clinical recommendations to address clinical questions on patient-centered care.

PwMS: people with multiple sclerosis; CR: clinical recommendation; DMT: disease-modifying therapy; MS: multiple sclerosis; SLR: systematic literature review.
Median score on a 1–9 scale.
Percentage of votes with 7–9 on a 9-point scale.
“Level of evidence” defined based on the following grading of references identified by SLR review: high when supported by ⩾ 20 references; medium when supported by 10–19 references; and low when supported by 0–9 references.
Clinical recommendations to address clinical questions on selection on contraception for PwMS.

CR: clinical recommendation; DMT: disease-modifying therapy; WOCBP: women of childbearing potential; MS: multiple sclerosis; LARC: long-acting reversible contraception; IUD: intrauterine device; CHC: combined hormonal contraception; VTE: venous thromboembolism; DMPA: depot-medroxyprogesterone acetate; SLR: systematic literature review; PwMS: people with multiple sclerosis.
Median score on a 1–9 scale.
Percentage of votes with 7–9 on a 9-point scale.
“Level of evidence” defined based on the following grading of references identified by SLR review: high when supported by ⩾ 20 references; medium when supported by 10–19 references; and low when supported by 0–9 references.
Clinical recommendations to address clinical questions on the timing of stopping/starting contraception and disease-modifying therapies.
DMT: disease-modifying therapy; PwMS: people with multiple sclerosis; CR: clinical recommendation; MS: multiple sclerosis; SLR: systematic literature review.
Median score on a 1–9 scale.
Percentage of votes with 7–9 on a 9-point scale.
“Level of evidence” defined based on the following grading of references identified by SLR review: high when supported by ⩾ 20 references; medium when supported by 10–19 references; and low when supported by 0–9 references.
Patient-centered care
Question 1: what is the population of PwMS with whom contraception issues should be discussed, and when should discussion take place?
The effects of MS on fertility have not been fully determined. The general consensus is that fertility is not significantly affected in PwMS.6,7,19 For this reason, contraception in PwMS is important for those who want to avoid pregnancy and/or to avoid any DMT risk on a potential pregnancy.5,19 Family planning is an important step for women and men with MS and counseling on contraception should be offered at diagnosis and regularly throughout a patient’s follow-up.1,5,9,19
Question 2: who should be included in the discussion and decision-making process with PwMS about contraception?
Discussing contraception for PwMS may involve an MDT of neurologists, obstetricians/gynecologists, and nurses. However, in some practices, certain MDT members may be more relevant and necessary for the conversation than others.1,2,4,9,19 Although neurologists are not expected to prescribe contraceptives, they may help patients make reasonable contraceptive choices while considering MS-specific factors, such as level of disability and co-administered medications. 1
Question 3: what topics should be included in the contraception discussion for PwMS?
A wide range of topics should be discussed, including the benefit–risk profile of DMTs before, during, and after pregnancy, the patient’s desired timing of pregnancy in relation to disease course and activity, and the full range and effectiveness of contraceptive methods suitable for their individual profile.1,19
Some important topics listed within the recommendations for Question 3 are not specific to PwMS. Not all PwMS will be affected by issues, including teratogenic DMTs and sexually transmitted infections (STIs), but it is important to provide the opportunity to discuss if needed.
Selection of contraception for PwMS
Question 4: what factors should be taken into account when deciding the choice of contraception in PwMS?
It is important to consider safety, availability, acceptability, and effectiveness, as well as relevant disabilities, such as dysphagia, when choosing contraception.1,15,19 The role of male and female condoms in preventing STIs should also be considered. 15
Question 5: what is the key consideration for prescribing a DMT that will be used in women of childbearing potential (WOCBP) with MS?
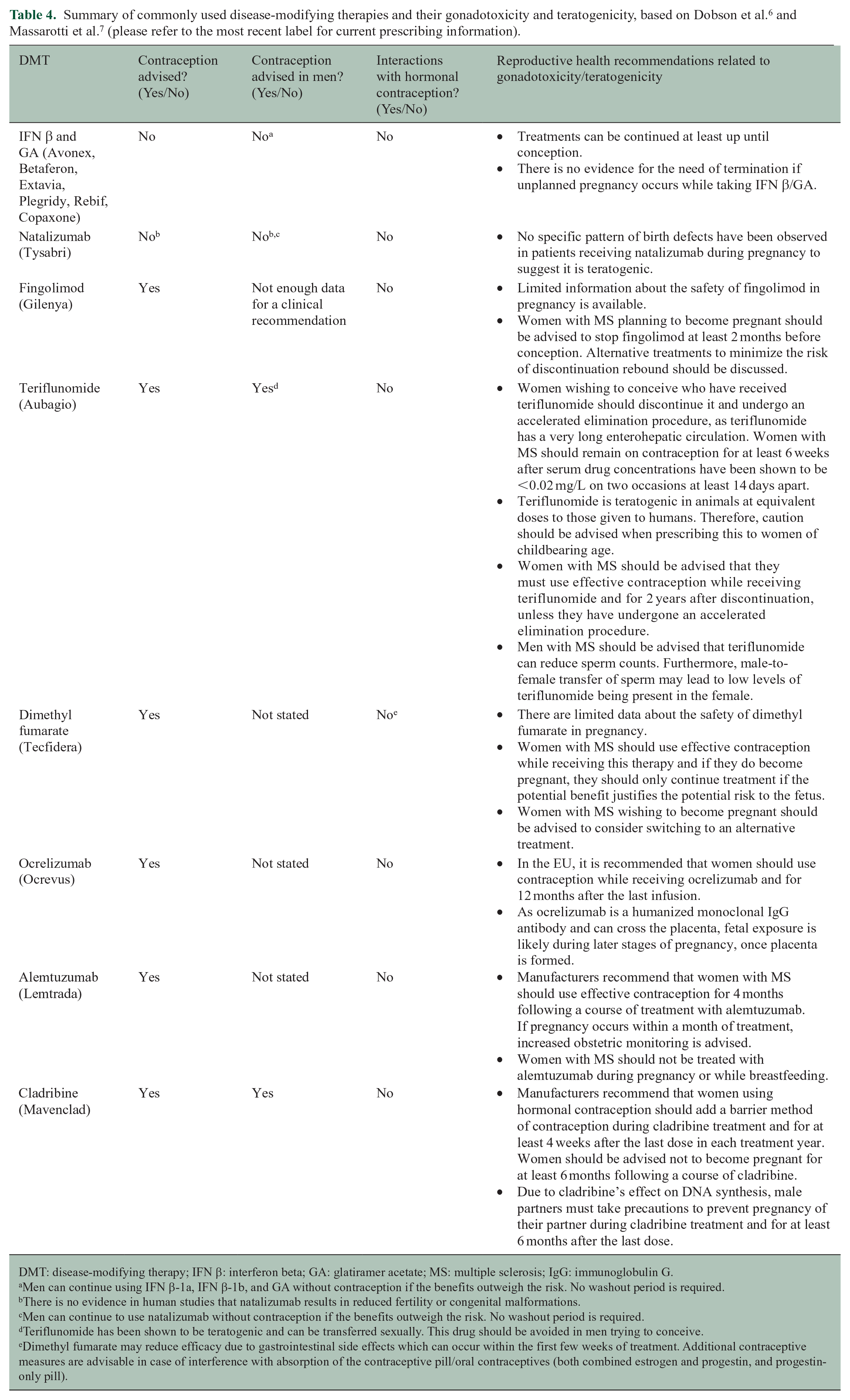
Most DMTs are not known to interact with or decrease the effectiveness of hormonal contraception.1,6,20 Some DMTs, for example, cladribine tablets, teriflunomide, and sphingosine-1-phosphate receptor modulators (such as fingolimod) have known risks in pregnancy, while for others (natalizumab, ocrelizumab, dimethyl fumarate, alemtuzumab) there is not enough evidence about their safety in pregnancy and contraception should still be advised.5,6,21,22 If interferon β and/or glatiramer acetate are prescribed to WOCBP, contraception is not necessary as these drugs can be used until conception. 6 Table 4 summarizes recommendations for reproductive health for PwMS based on known gonadotoxicity/teratogenicity of DMTs detailed in Massarotti et al. 7 and Dobson et al. 6
DMT: disease-modifying therapy; IFN β: interferon beta; GA: glatiramer acetate; MS: multiple sclerosis; IgG: immunoglobulin G.
Men can continue using IFN β-1a, IFN β-1b, and GA without contraception if the benefits outweigh the risk. No washout period is required.
There is no evidence in human studies that natalizumab results in reduced fertility or congenital malformations.
Men can continue to use natalizumab without contraception if the benefits outweigh the risk. No washout period is required.
Teriflunomide has been shown to be teratogenic and can be transferred sexually. This drug should be avoided in men trying to conceive.
Dimethyl fumarate may reduce efficacy due to gastrointestinal side effects which can occur within the first few weeks of treatment. Additional contraceptive measures are advisable in case of interference with absorption of the contraceptive pill/oral contraceptives (both combined estrogen and progestin, and progestin-only pill).
Question 6: what considerations should be given when discussing long-acting reversible contraception (LARC) as an option for WOCBP with MS?
In women receiving potentially teratogenic DMTs, LARC may be recommended to prevent drug-exposed pregnancies due to its long-lasting high efficacy, reliability, and convenience.1,3,9,23 LARC may be particularly suitable for PwMS who cannot be prescribed combined hormonal contraception (CHC) (see Question 7).
Question 7: what are the considerations when discussing CHC as an option for WOCBP with MS?
The use of CHC has not been shown to worsen MS disease course. 1 However, users of CHC may have increased risk of venous thromboembolism (VTE)—these risks are enhanced with immobility, and therefore, CHC should be avoided in MS patients with reduced mobility and/or those at risk of VTE. 9 Furthermore, if PwMS are immobile but do not have a history of blood clots, CHC is still not advisable.
Question 8: can CHC or progestin-only contraception be prescribed to women with MS to help regulate their MS symptoms?
There are limited data in the literature surrounding this topic. Some studies suggest an improvement in MS symptoms with CHC or progestin-only contraception, although these studies were observational, so bias may have influenced the results.24–26 Others have proposed that premenstrual hormonal changes may worsen symptoms in people with relapsing-remitting MS. 27
Question 9: what are the considerations when discussing progestin-only contraception as an option for WOCBP with MS?
Breakthrough bleeding, or unscheduled bleeding, is an important factor to consider with progestin-only contraceptives and PwMS should be made aware of this. 28 As depot-medroxyprogesterone acetate (DMPA) may compromise bone health,1,19 its use should be carefully considered in patients who are at risk of osteopenia (patients with a family history of, or who are older in age), despite reports that the effects of DMPA on bone mineral density are reversible. 29 Women with MS and a history of repeated and excessive use of corticosteroids, and immobility may also be at risk of osteopenia and osteoporosis; bone mineral density in these individuals should be assessed before initiation of DMPA.
Question 10: what considerations should be given when discussing contraception options for male PwMS?
The effects of MS and DMTs on fertility are not fully understood and are particularly under-investigated in men. 7 There is, however, clear contraceptive guidance for men using cladribine tablets and/or teriflunomide. Men taking cladribine tablets must use effective contraception with female partners to avoid pregnancy during treatment and up to 6 months after the last dose. 9 Men who wish to conceive with their female partners should discontinue teriflunomide. 9
Question 11: what methods, if any, of contraception should be recommended against in PwMS?
In PwMS receiving potentially teratogenic or gonadotoxic DMTs, an effective method of contraception is recommended.6,9,10 Oral contraceptives do not increase the risk of MS or increase the risk of relapse. 3 CHC is not recommended for women with MS with prolonged immobility, due to an increased risk of VTE, or who have difficulty swallowing. 10 DMPA is associated with decreased bone mineral density and not recommended in women with osteopenia or osteoporosis. 1 Female patients with impaired fine motor function may experience difficulty with vaginal rings and diaphragms, so another method of contraception may be preferred. 1
Question 12: what are the key overall considerations for methods of contraception in WOCBP with MS who have high levels of disability?
For women with MS with prolonged immobility, CHC is not usually recommended due to VTE risk, and DMPA may further compromise bone mineral density. 1 Tolerability and bleeding patterns are also important to consider with all methods of contraception.
Question 13: what factors should be considered before prescribing a method of emergency contraception to WOCBP with MS?
There is a lack of evidence on this topic for PwMS. In general, emergency oral contraception options are typically levonorgestrel or ulipristal acetate. The copper intrauterine device (IUD) appears to be more effective than oral methods as an emergency contraceptive. 30 A study has shown that the levonorgestrel IUD was noninferior to the copper IUD for emergency contraception. 31
Timing of stopping/starting contraception and DMTs
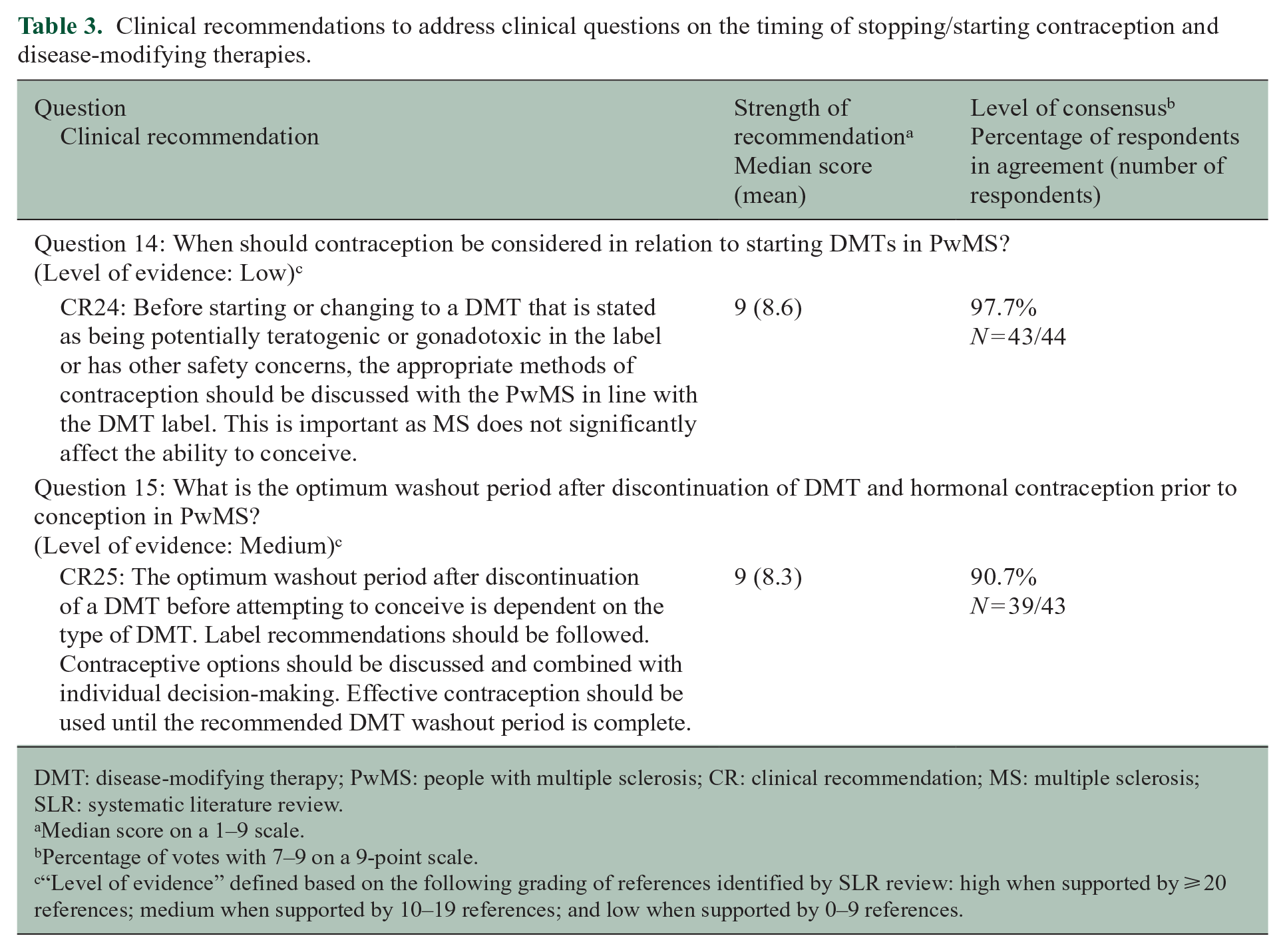
Question 14: when should contraception be considered in relation to starting DMTs in PwMS?
Each DMT label outlines the precautions to be taken when starting therapy. 6 In most cases, contraception is recommended when starting a DMT due to limited safety information.6,20,32 The efficacy of hormonal contraceptives is not affected by the use of DMTs.2,3,6
Question 15: what is the optimum washout period after discontinuation of DMT and hormonal contraception prior to conception in PwMS?
Each DMT label has recommendations on washout before conception.1,2,5,6 DMTs with potential teratogenicity or those contraindicated in pregnancy should be discontinued and replaced with acceptable alternative treatment before conception.2,19,20 If the decision is made to stop DMT or to switch to an alternative treatment before conception, PwMS will usually be required to complete a washout period; contraception should be continued until the washout is complete. 33 Patients may be advised for their DMT washout periods to be as short as possible to prevent relapses.10,23
However, healthcare professionals (HCPs) should be cautious and not underestimate the time required for washout. Once the DMT washout is completed, contraception can be stopped to minimize the period without treatment before conception. 33 Consequently, preconception care, such as folic acid supplementation, may be needed before stopping contraception.34,35
Discussion
The clinical recommendations described by this consensus provide practical advice on contraception to support HCPs involved in the care of PwMS. The current data and therefore recommendations made apply to cisgender women and men with MS. Additional data are needed on gender-diverse patient populations, including transgender and non-binary populations. In this consensus, the faculty of 44 clinical experts from 19 countries voted on the 25 recommendations, giving quality and strength to their relevance. All but one of the recommendations achieved consensus (⩾75%).
The one recommendation that did not achieve consensus (74.1%) was around the considerations when discussing progestin-only contraception as an option for WOCBP with MS (clinical recommendation [CR] 13, Table 2). The faculty commented that the specific type of progestin is rarely considered by HCPs, the relevance of different types of progestins is unknown, and effectiveness and cost are usually of greatest importance (Supplementary Table S3). It is important to note that consensus was missed by a small margin (0.9%).
The strongest clinical recommendations supported by the highest levels of evidence (and 83%–95% expert consensus) were recommendations on which factors to consider when making contraception decisions, including contraceptive efficacy, impact on mood, libido and physical health, potential risks, and patient clinical characteristics and preferences (CR6, 7, and 8), and on the key considerations for DMT prescription for WOCBP (CR9). There is also strong evidence to support the recommendation for contraceptive conversations with PwMS, which emphasize the need for discussion on effective contraception during DMT use and address safety concerns, including consequences of unplanned pregnancy and protection against STIs, while evaluating effectiveness and risk–benefit of each contraceptive method (CR3, 4, and 5). However, a number of recommendations are based predominantly on expert opinion and clinical practice. Therefore, further clinical research is advocated to investigate:
The benefits of LARC for patients with mobility issues and WOCBP on potentially teratogenic DMTs
The risks of VTE with CHCs and reduced bone mineral density with DMPA in PwMS
Emergency contraception in this patient population
Further research is also needed on the teratogenic risks associated with the use of certain DMTs in men, particularly as advice on the use of contraceptive methods differs in the product labeling across countries.
The faculty commented that CHC or progestin-only contraception use for the regulation of MS symptoms reflects inter-patient variation, so should be individualized and may not be evidence-based. HCPs should refer to the Faculty of Sexual and Reproductive Healthcare guidelines and approved contraceptive labels,12,14 and contraceptive devices, such as the IUD or patch, may be considered.
The SC agreed that family planning should be discussed with PwMS. Conversations regarding contraception must be handled sensitively, be driven by the patient, and include partners, depending on patient preference. A balanced discussion should be undertaken covering the risk–benefit of all contraceptive methods, and patients should not feel restricted in their choice. Contraceptive coercion should be avoided and the patient’s preferred method of contraception always considered. As most female PwMS are diagnosed during their childbearing years, these conversations are particularly relevant and disease stabilization before a planned pregnancy is recommended. All PwMS should be made aware of the risks with DMT use and the effect of pregnancy on MS, particularly postpartum.3,6 Experts agreed that patients should make the final decision and they need to feel comfortable with their chosen method of contraception, proceeding with caution in some instances.
The SC highlighted that certain DMT labels may be overly conservative and inconsistent, and noted that European Medicines Agency (EMA) and Food and Drug Administration guidelines may differ for the same product, for example, ocrelizumab.36,37 The SC also noted that not all labels reflect real-world practice; therefore, expert opinion and additional data for DMT use in pregnancy should be considered. For example, the EMA suggests waiting 12 months from last ocrelizumab infusion before pregnancy, but data and expert opinion support a much shorter time. 36 Off-label advice may also be considered at the judgment of the clinical team, but HCPs must be aware of the legal landscape in their jurisdiction regarding off-label use. This should be openly discussed with the patient and their family, if desired by the patient, and all discussions documented. Key factors to consider when prescribing DMTs to WOCBP are disease control, the potential to cause fetal harm, and if the DMT will be used long term, irrespective of the fact that it may be teratogenic or gonadotoxic. PwMS should remain on their current non-teratogenic DMT until conception and the risk of an MS relapse should be balanced against the potential risk to the embryo/fetus. When prescribing a DMT that may be present in semen to male PwMS, it is important to provide guidance on effective contraception to prevent transference of the active substance to a female partner. Barrier contraceptives, such as condoms, are recommended, although erectile dysfunction should be considered when providing this advice. It is important to emphasize that fertility awareness-based contraceptive methods are generally not recommended for PwMS receiving teratogenic/gonadotoxic medications as the risk of failure/pregnancy is high.
It was reflected in the voting and comments that some neurologists feel they do not have the necessary knowledge of contraception to support patients’ decisions; therefore, education is recommended if required. Most contraceptive advice does not require advanced specialist knowledge. It may be beneficial to refer to the medical eligibility criteria when discussing contraception to highlight any contraindications, for example, migraines with aura symptoms. Another key consideration is time constraints; in cases where specialist advice is required, it may not be possible to have comprehensive MDT discussions with patients.
Conclusion
These consensus-based clinical recommendations represent the opinions and perspectives of 44 international clinical experts and are based on current evidence. Joining other consensus recommendations around reproductive health and MS,6,9,15,19,32 namely evolving guidelines for MS treatment during pregnancy and for family planning and pregnancy in PwMS,6,9,15 the current recommendations provide the first specific guidance around the most important aspects of contraception for PwMS.
Supplemental Material
sj-doc-1-msj-10.1177_13524585241228103 – Supplemental material for Expert opinion on the use of contraception in people with multiple sclerosis
Supplemental material, sj-doc-1-msj-10.1177_13524585241228103 for Expert opinion on the use of contraception in people with multiple sclerosis by Jan Hillert, Riley Bove, Lisa B Haddad, Kerstin Hellwig, Maria Houtchens, Melinda Magyari, Gabriele S Merki-Feld, Scott Montgomery, Rossella E Nappi, Egon Stenager, Heidi Thompson, Zeliha Tulek, Elisabetta Verdun Di Cantogno and Manuela Simoni in Multiple Sclerosis Journal
Footnotes
Acknowledgements
Caroline Herbert of Bedrock Healthcare Communications provided medical writing support, funded by Merck (CrossRef Funder ID: 10.13039/100009945). Assistance with the systematic literature review was provided by AccuScript and supported by Merck.
The Steering Committee would like to thank all the experts who contributed their knowledge to this program by voting on the draft recommendations. Listed below are 28 extended faculty members and patient advisory groups who are happy to be acknowledged in this document:
Pamela McCombe1, Therese Burke2, Vilija G. Jokubaitis3, Thomas Berger4, Bart Van Wijmeersch5, Kristen M. Krysko6, Sarah A. Morrow7, Warren Berger8, Dana Horakova9, Juha-Pekka Erälinna10, Thomas Müller11, Nikolaos Grigoriadis12, Marcello Moccia13, Elisabeth G. Celius14, Kjell-Morten Myhr15, Mohammed Aljumah16, Tatjana Pekmezovic17, Ester Moral18, Silvia Sánchez-Ramón19, Virginia Meca-Lallana20, Kristina Gemzell Danielsson21, Jürg Kesselring22, Aksel Siva23, Ide Smets24, Wallace J. Brownlee25, Annette F. Okai26, Bianca Weinstock-Guttman27, Jacqueline A. Nicholas28
1The University of Queensland, Brisbane, QLD, Australia; 2MS Australia (patient advocacy and research organization), Sydney, NSW, Australia; 3Department of Neuroscience, Monash University, Melbourne, VIC, Australia; 4Department of Neurology, Medical University of Vienna, Austria; 5Universitair MS Centrum, Hasselt-Pelt, Hasselt University, Pelt, Belgium; 6Division of Neurology, Department of Medicine, St. Michael’s Hospital, University of Toronto, Li Ka Shing Knowledge Institute, Toronto, Ontario, Canada; 7Western University, London, Ontario, Canada; 8Department of Clinical Neurological Sciences, London Health Sciences Center, London, Ontario, Canada; 9Department of Neurology and Center of Clinical Neuroscience, First Faculty of Medicine Charles University and General University Hospital, Prague, Czech Republic; 10Mehiläinen NEO, Turku, Finland; 11Department of Neurology, St. Joseph Hospital Berlin-Weissensee, Berlin, Germany; 12B' Department of Neurology, Aristotle University of Thessaloniki, Thessaloniki, Greece; 13Department of Neuroscience, Federico II University of Naples, Naples, Italy; 14Department of Neurology, Oslo University Hospital, Oslo, Norway; 15Department of Clinical Medicine, University of Bergen, Bergen, Norway, Department of Neurology, Haukeland University Hospital, Bergen, Norway; 16King Fahad Medical City, MOH, Riyadh, Saudi Arabia; 17University of Belgrade, Faculty of Medicine, Belgrade, Serbia; 18Moises Broggi Hospital, Barcelona, Spain; 19Departamento de Inmunología Clínica Instituto de Medicina de Laboratorio Hospital Clínico San Carlos, Universidad Complutense of Madrid, Madrid, Spain; 20Hospital Universitario de la Princesa, Madrid, Spain; 21Department of Women’s and Children’s Health, Karolinska Institutet and Karolinska University Hospital, Stockholm, Sweden; 22Kliniken Valens, CH 7317 Valens, Switzerland; 23Istanbul University-Cerrahpasa, School of Medicine, Department of Neurology, Clinical Neuroimmunology Unit and MS Clinic, Istanbul, Turkey; 24MS Centre ErasMS, Department of Neurology, Erasmus MC Rotterdam, Rotterdam, Netherlands; 25Queen Square MS Centre, UCL Institute of Neurology, London, UK; 26North Texas Institute of Neurology and Headache, Plano, TX, USA; 27Jacobs School of Medicine and Biomedical Sciences University of Buffalo, Buffalo, NY, USA; 28Ohio Health Multiple Sclerosis Center, Columbus, OH, USA
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article and/or its supplementary materials.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.H. has received honoraria for serving on advisory boards for Biogen, Bristol Myers Squibb/Celgene, Janssen, Merck, Sandoz, and Sanofi-Genzyme and speaker’s fees from Biogen, Janssen, Novartis, Merck, Teva, Sandoz, and Sanofi-Genzyme. He has served as principal investigator for projects sponsored by, or received unrestricted research support from, Biogen, Bristol Myers Squibb/Celgene, Janssen, Merck, Novartis, Roche, and Sanofi-Genzyme. His MS research is funded by the Swedish Brain Foundation. R.B. has received research support from Department of Defense, National Institutes of Health, National MS Society, as well as Biogen, Novartis, and Roche Genentech; consulting and advisory board fees for Alexion, Biogen, EMD Serono Research & Development Institute, Inc., Billerica, MA, USA, an affiliate of Merck KGaA, Janssen, Genzyme Sanofi, Novartis, Roche Genentech, and TG Therapeutics. L.B.H. is employed by Population Council, a global non-profit with several contraceptive products in development or currently marketed globally. No other disclosures related to this work presented. K.H. has received personal compensation as a speaker/consultant from Bayer, Bristol Myers Squibb, Biogen, INC Research, Merck, Novartis, Roche, Teva, and Sanofi-Genzyme, and research funding from Biogen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. M.H. has received research support from Genentech and Biogen. M.M. has served on scientific advisory boards for Sanofi, Novartis, and Merck, and has received honoraria for lecturing from Biogen, Merck, Novartis, Roche, Genzyme, and Bristol Myers Squibb. G.S.M.-F. has received personal compensation as a speaker/consultant from Novartis, Teva, Gedeon Richter, HRA Pharma, Lundbeck, and Merck. S.M. has received research support from Roche, Novartis, and AstraZeneca; received speaker’s honorarium from Teva; and served on a study advisory board for IQVIA. R.E.N. has past financial relationships (lecturer, member of advisory boards, and/or consultant) with Boehringer Ingelheim, Eli Lilly, Endoceutics, Gedeon Richter, Merck Sharpe & Dohme, Palatin Technologies, Procter & Gamble Co., TEVA Women’s Health Inc., and Zambon SpA. At present, she has ongoing relationships with Abbott, Astellas, Bayer HealthCare AG, Exceltis, Fidia, HRA Pharma, Merck, Novo Nordisk, Organon & Co., Pfizer Inc., Shionogi Limited, Theramex, and Viatris. E.S. declares that there is no conflict of interest apart from the funding of this project. H.T. has received honoraria and travel grants from Merck, Biogen Idec, and Novartis. Z.T. declares that there is no conflict of interest apart from the funding of this project. E.V.D.C. was an employee of EMD Serono Research & Development Institute, Inc., Billerica, MA, USA, an affiliate of Merck KGaA, at the time of study. M.S. has received honoraria for lectures, advisory boards, and research grants from Merck and Ferring.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Merck (CrossRef Funder ID: 10.13039/100009945), who provided funding for the project, but had no input into the development of clinical questions nor recommendations. No company representative voted on the recommendations. The authors received no financial support for the authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.