Abstract
Background:
Neuromyelitis optica spectrum disorder (NMOSD) is a group of inflammatory diseases affecting the central nervous system, characterized by optic neuritis and myelitis. The complex nature of NMOSD and varied patient response necessitates personalized treatment and efficient patient stratification strategies.
Objective:
To provide a comprehensive review of recent advances in clinical and biomarker research related to aquaporin-4 (AQP4)-immunoglobulin G (IgG)-seropositive NMOSD prognosis and identify key areas for future research.
Methods:
A comprehensive review and synthesis of recent literature were conducted, focusing on demographic factors and laboratory investigations.
Results:
Demographic factors, such as age, ethnicity, and sex, influence NMOSD prognosis. Key biomarkers for NMOSD prognosis include homocysteine, antinuclear antibodies, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, thyroid hormone levels, neurofilament light chain levels, and serum glial fibrillary acidic protein might also predict NMOSD attack prognosis.
Conclusion:
Further investigation is required to understand sex-related disparities and biomarker inconsistencies. Identification and understanding of these factors can aid in the development of personalized therapeutic strategies, thereby improving outcomes for NMOSD patients. Future studies should focus on unifying research design for consistent results.
Keywords
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is a collective of inflammatory demyelinating diseases of the central nervous system. These diseases are hallmarked by optic neuritis (ON) and myelitis, with their etiology rooted in the presence of aquaporin-4 (AQP4) antibodies. Patients affected with NMOSD experience high rates of disease recurrence and disability, constituting a major challenge to health systems worldwide. 1
Management of acute NMOSD primarily employs intravenous methylprednisolone pulse therapy (IVMP), which has demonstrated considerable success. However, not all patients respond favorably to IVMP, necessitating alternative therapeutic approaches. In such instances, plasma exchange or immune adsorption (PE/IA) has proven to effectively alleviate symptoms, underlining the diversity in patient response and treatment outcomes. 2
Given this variability, the importance of promptly identifying high-risk and highly disabled patients cannot be overstated. Timely identification and intervention could improve the prognosis and quality of life for these patients. 3 Unfortunately, the heterogeneity of NMOSD manifestations and responses to treatment complicate these efforts, making it a prominent area requiring extensive research.
In recent years, significant advancements have been made in our understanding of NMOSD, particularly in the field of clinical and biomarker research. These efforts are driving more personalized and effective treatment approaches. Despite these advancements, gaps remain in our knowledge, necessitating a comprehensive review of the current literature.
This article aims to review and synthesize recent advances in research pertaining to clinical and biomarkers related to AQP4-immunoglobulin G (IgG) seropositive NMOSD prognosis. Through a comprehensive analysis of current findings, we seek to highlight key areas for future research, provide guidance for clinical practice, and ultimately contribute to improving outcomes for NMOSD patients. The importance of this review is emphasized by the complex nature of NMOSD, the variability in treatment response, and the critical need for more efficient patient stratification strategies.
Methods
For this narrative review, we searched English-language publications in PubMed published mostly in the last decade. Our main search terms were “neuromyelitis optica,” “prognostic factor,” “biomarker,” and “outcome prediction.” While our focus was on AQP4-IgG+ NMOSD, we did not exclusively limit our search to studies addressing only AQP4-IgG+ patients. Reference lists of the identified publications were also examined. Biomarker candidates that have been investigated in studies considered meaningful by the authors were selected for inclusion in our review. And the “NMOSD” we discussed in the present review refers to “AQP4-IgG+ NMOSD.”
Demographic factors
Age
The influence of age on NMOSD is increasingly recognized. The 30s and 40s are high-incidence age ranges for females with NMOSD,4,5 although cases in individuals above 70 have been reported. 6 Patients with an onset before 50 are categorized as early-onset neuromyelitis optica spectrum disorder (EO-NMOSD), while those 50 and above are classified as late-onset neuromyelitis optica spectrum disorder (LO-NMOSD).6,7 Patients aged 70 or above are defined as very late-onset neuromyelitis optica spectrum disorder (VLO-NMOSD). 8 Recent research suggests LO-NMOSD patients experience more frequent initial transverse myelitis attacks and fewer ON and brain/brainstem attacks compared to EO-NMOSD patients. 9 A multicenter study also reveals that patients with younger age of onset are more likely to have ON relapses, while those with older age of onset are more prone to developing ambulatory disability. 5 Similar findings have been reported in previous studies.8–13 Age-dependent anatomical susceptibility may be associated with differing fragility of the blood–brain barrier and blood–spinal cord barrier during aging. 9
Notably, age is also a significant factor in the broader context of disability in the general population. Age-related physiological changes, combined with a possible increase in comorbidities and the natural process of aging, often lead to increased disability. 14 In the context of NMOSD, a British study reveals that onset age is an independent predictor of disability. Every decade increase in age escalates the risk of an Expanded Disability Status Scale (EDSS) ⩾ 4 by 34%. 15 Numerous studies have also indicated a correlation between NMOSD onset age and prognosis, with older patients having poorer outcomes and higher disability scores after follow-up, particularly in AQP4-IgG+ patients.6,7,13,16
Ethnicity
Ethnic differences in NMOSD have started to gain research traction in recent years. The disease manifestation, frequency, and severity can vary among different ethnic groups, potentially influencing the prognosis. Overall, NMOSD appears to be more common in non-White populations and tends to have a more severe disease course among non-White patients compared to Caucasians. 17
Afro-descendant patients with NMOSD tend to have more severe disease activity than Caucasians, experiencing more frequent relapses and greater disability. This observation is supported by Flanagan et al., 18 who found a higher rate of recurrent myelitis and diminished visual outcomes after ON episodes. Asian populations, particularly from East Asia, often display NMOSD in association with AQP4-IgG antibodies and exhibit a heightened incidence of ON throughout the disease course, possibly linked to region-specific genetic and environmental factors.19,20 Meanwhile, Latin American (LATAM) NMOSD presentations seem to bridge the characteristics of the Asian and Afro-descendant groups. Studies from Brazil and Argentina indicate symptom overlap between these two ethnicities, likely due to the diverse genetics of the LATAM region, with environmental aspects also deserving further investigation. 21
Sex
It is well known that women constitute a significant portion of NMOSD patients, leading to differences in incidence rates by sex. However, the impact of sex on clinical prognosis remains controversial. This may be due to both the lack of studies addressing sex’s impact on NMOSD prognosis and differences in research design. In a recent retrospective study, Sun et al. 22 found no significant differences in the annual relapse rate between male and female patients. These rates were similar to those reported in other studies conducted in China.23,24 Kim et al. 19 initially found that sex can independently affect the site of onset in AQP4-IgG+ NMOSD patients, regardless of onset age. Male patients had a lower rate of ON at onset, fewer ON recurrences during follow-up, and better visual evoked potential (VEP) outcomes. These findings contradict previous research in diverse populations (including British and Japanese patients), which suggested worse visual outcomes in men. 20 The reason for these disparities in study findings remains enigmatic, and the exact mechanisms through which sex influences the site of onset in AQP4-IgG+ NMOSD patients continue to evade understanding.
The female predominance in NMOSD may be attributed to a combination of hormonal, genetic, and environmental factors. Research suggests that sex hormones, particularly estrogen and progesterone, can modulate immune responses, 25 potentially explaining sex-related differences in autoimmune diseases, including NMOSD. 26 In addition, specific genetic factors such as X-chromosome inactivation and the phenomenon of microchimerism might also contribute to the female preponderance in NMOSD.27,28 Environmental factors and lifestyle differences between genders may also be potential contributing factors. However, these remain speculative until further research is conducted.
The inconsistent and often contradictory findings in sex-based differences in NMOSD underscore the need for larger, more diverse, and methodologically uniform studies. Thorough examination and understanding of the sex-related factors could potentially aid in the development of personalized, sex-specific therapeutic strategies, thereby improving outcomes for all NMOSD patients.
Laboratory investigations
Serum AQP4 titer level
The relationship between serum AQP4 titer levels and the prognosis of seropositive NMOSD patients remains a subject of considerable debate. A diverse range of findings has emerged from various studies examining the implications of serum titer levels in seropositive patients. For instance, a study by Takahashi et al. identified a positive correlation between AQP4 titer levels and the degree of disability in NMOSD patients. 29 In contrast, much other research reported no definitive correlation between these two variables.30–33 Given the heterogeneous nature of these findings, it underscores the complexity of NMOSD and suggests that factors beyond AQP4 titer levels may influence disease prognosis.
Recent research has explored the seroreversion of AQP4-IgG over time. One study from the Mayo Clinic found that most patients maintain consistent AQP4-IgG levels. However, 11% showed a decrease to negative levels, especially those with lower initial levels and younger age. Interestingly, some of these patients still experienced disease attacks even after their levels decreased. 34 Another study found that patients who became negative for AQP4-IgG had fewer relapses, but certain factors like younger age at disease onset and specific initial symptoms made relapses more likely. 35
Given the current evidence, it is clear that AQP4-IgG titer levels cannot be recommended as a disease activity biomarker to guide treatment in individual patients. Clinicians and researchers must therefore approach the potential prognostic role of AQP4-IgG titer levels with a degree of circumspection.
Homocysteine
Homocysteine (Hcy) is a crucial intermediate product of folic acid, vitamin B12, and one-carbon metabolism.
36
While Hcy’s role in cellular physiology and pathophysiology is complex, elevated serum levels are known to induce oxidative stress and mitochondrial dysfunction by increasing reactive oxygen species (ROS) production37,38 and stimulate N-methyl-
However, it is essential to consider that Hcy serum levels are influenced by various external factors, some of which might also be associated with NMOSD prognosis, such as smoking and diabetes.48,49 Moreover, there is a potential association between Hcy and cerebrovascular disease, which could confound disability prognostication in NMOSD patients. 50 In light of these findings and considerations, a comprehensive understanding of Hcy’s multifaceted role is imperative for precise diagnosis, prognosis, and therapeutic strategies in NMOSD patients.
Antinuclear antibodies
As the understanding of NMOSD expands, it is evident that the disease often coexists with other autoimmune diseases, and relevant antibodies, such as antinuclear antibodies (ANAs), anti-Sjögren syndrome A (SSA) antibodies, and anti-Sjögren syndrome B (SSB) antibodies, can frequently be detected in the blood. The rationale behind measuring ANA and other antibodies in NMOSD is partly rooted in the observation of significant comorbidity between AQP4-IgG+ NMOSD and certain rheumatoid disorders. Here, we mainly review the influence of ANA on the prognosis of NMOSD. Recent studies on the relationship between ANA and NMOSD have yielded different results. A recent study found that patients with ANA (+) reached an EDSS score of 4.0, indicative of moderate disability, faster than ANA (−) patients, indicating that ANA (+) patients may reach moderate disability faster after onset, implying a correlation between ANA and poor NMOSD prognosis. 51 However, another study found that ANA (+) NMOSD patients had a lower annual relapse rate than the negative group, and the median time from onset to an EDSS score of 6.0 was significantly longer than in ANA (−) NMOSD patients, suggesting a protective role of ANA in NMOSD. 52 Discrepancies in study results may be due to differences in the number of patients included and the EDSS threshold used. Another study showed that ANAs are not related to benign or malignant NMOSD prognosis and might be non-specific antibodies. 53
Neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio
While neutrophils are believed to have a role in NMOSD pathogenesis, 54 there are limited studies exploring the potential of the neutrophil-to-lymphocyte ratio (NLR) in peripheral blood as an inflammatory marker in this context. A study suggests that the NLR might be a more reliable indicator of inflammation than individual white blood cell counts or their subtypes. 55 One study on NMOSD patients suggested that a higher NLR could be associated with a greater recurrence rate and poorer recovery. 56 And another investigation found a correlation between NLR and the severity of initial neurological damage. 57 Yet, a multinational study from Latin America found that, while AQP4-IgG+ NMOSD patients did exhibit elevated NLRs, these high ratios were not necessarily predictive of adverse clinical outcomes within the first 1 or 2 years. 58 Given these varying results and the scarcity of data, conclusions drawn on the significance of NLR in NMOSD prognosis should be approached with caution.
Moreover, the platelet-to-lymphocyte ratio (PLR) has recently emerged as a noteworthy potential diagnostic and prognostic marker for NMOSD. A research endeavor conducted in Argentina delineated that PLR levels in NMOSD patients were notably higher when compared to those diagnosed with multiple sclerosis and highlighted PLR’s role as an independent predictor of severe neurological disability (EDSS ⩾ 4) in NMOSD patients at the 2-year mark. 59
Thyroid hormone levels
In previous reports, we found a higher incidence of coexisting thyroid disease in NMOSD patients.11,60,61 Low triiodothyronine (T3) syndrome is quite common among these patients. Initial studies have discovered a negative correlation between T3 levels in NMOSD patients and their EDSS scores, suggesting that serum T3 levels may be a useful indicator of disease activity and disability in these patients, but a cause–effect relationship has yet to be elucidated.
62
The related mechanism might be associated with interleukin (IL)-6, which acts to promote NMOSD onset, reduces thyroid hormone levels, and whose elevated levels can be observed in the cerebrospinal fluid (CSF) during disease recurrence.
63
He et al.
64
’s research indicated that high free tetraiodothyronine (FT4) levels might be linked to an increased risk of relapse and poor EDSS outcomes in NMOSD patients, with a notable inflection point at 12.01 pmol/L where the risk peaks before losing correlation. On one hand,
Neurofilament light chain levels
Neurofilament light chain (NfL), a major structural protein of neurons, is released into the CSF and blood in response to axonal damage. 67 Wang et al. 68 found that serum NfL levels were significantly elevated in AQP4-IgG+ NMOSD patients, especially during relapses, suggesting its potential as a biomarker for disease activity. Another study indicated that higher NfL levels were associated with more severe disability in NMOSD patients. 69 Watanabe et al. 70 emphasized the potential of serum neurofilament light chain (sNfL) as a biomarker of disease activity and disability in NMOSD, suggesting that the serum glial fibrillary acidic protein (sGFAP)/sNfL quotient at relapse could serve as a potential diagnostic marker for NMOSD. Liu et al. 71 investigated the levels of sNfL in Chinese patients with AQP4-IgG+ NMOSD and found it could be crucial for diagnosing and monitoring NMOSD.
Serum glial fibrillary acidic protein
Glial fibrillary acidic protein (GFAP), a kind of intermediate filament in astrocytes, is significantly elevated in the CSF during NMOSD attacks.71–75 Studies have consistently shown a correlation between GFAP levels in both serum and CSF with EDSS scores, and these levels could decrease following treatment.70,72,73,76 In studies focusing on patients with AQP4-IgG+ NMOSD in clinical remission, higher sGFAP levels were associated with poorer clinical disability scores and a shorter time to future attacks.68,77 Watanabe et al. 70 found a strong association between sGFAP levels, EDSS scores, and recent relapse occurrence, suggesting that elevated sGFAP levels during some NMOSD patient remission periods might indicate subclinical disease activity.
The evidence from various studies suggests that sGFAP is a more robust biomarker for prognosis in AQP4-IgG+ NMOSD than NfL or the GFAP/NfL ratio. This is supported by findings that show a more pronounced correlation between blood GFAP and EDSS in NMOSD than that between NfL and EDSS.70,77–79 Furthermore, blood GFAP has demonstrated better performance compared to the GFAP-to-NfL ratio in distinguishing NMOSD patients from healthy controls and in differentiating between attack and remission phases in NMOSD.79,80
All the prognostic markers included in this review and their effects were shown in Table 1.
Effects of prognostic factors of NMOSD.
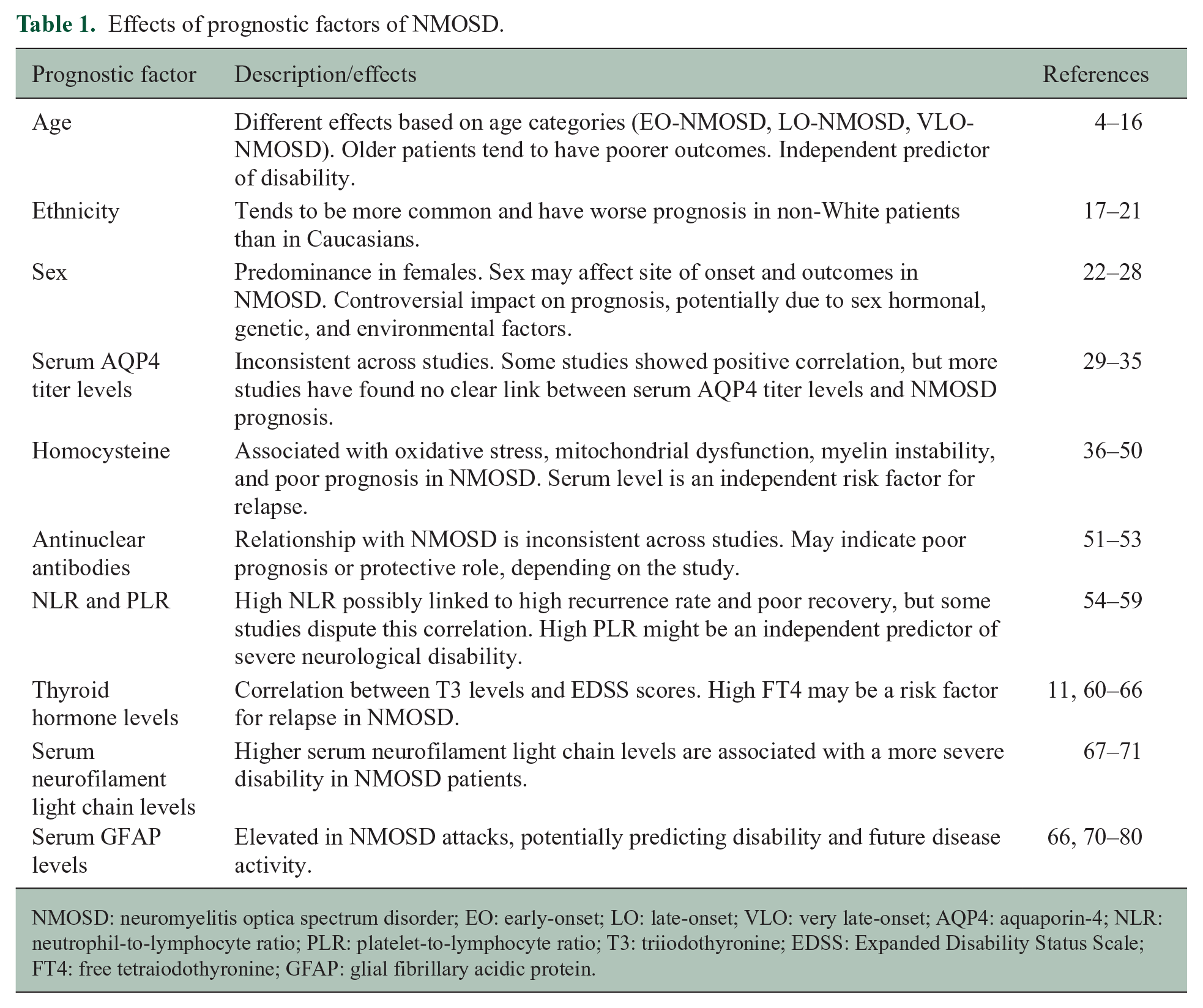
NMOSD: neuromyelitis optica spectrum disorder; EO: early-onset; LO: late-onset; VLO: very late-onset; AQP4: aquaporin-4; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; T3: triiodothyronine; EDSS: Expanded Disability Status Scale; FT4: free tetraiodothyronine; GFAP: glial fibrillary acidic protein.
Conclusion
NMOSD is a disease prone to relapse and often carries a poor prognosis without appropriate management; hence, finding tools that can predict prognosis is of high clinical importance. Early identification of NMOSD patients with poor prognosis will help clinicians monitor changes in indicators closely, adjust treatment strategies in a timely manner, reduce relapse rate, and improve patients’ quality of life. This review examines various factors influencing the prognosis of NMOSD. Variables identified include age, ethnicity, sex, serum AQP4 titer levels, Hcy levels, ANA, NLR, PLR, thyroid hormone levels, GFAP levels, and NfL levels. Older onset age correlates with poorer outcomes and higher disability. Ethnic differences play a crucial role, with varying disease manifestations and severities observed across Afro-descendant, Asian, and LATAM populations. The impact of sex is inconsistent, with varying results reported. Elevated Hcy, GFAP, and NfL levels suggest a higher risk of relapse and disability, while the implications of AQP4 titer levels, ANA, and NLR remain uncertain due to conflicting studies. The relationship between NMOSD prognosis and thyroid hormones is emerging but requires further research. There is no single biomarker that has been repeatedly verified to be an independent predictor of prognosis, and several markers are in the process of clinical research confirmation. However, the complexity of NMOSD and the variability in treatment responses necessitate additional research to solidify these findings and to uncover other potential prognostic markers.
Outlook
Looking forward, the future of NMOSD prognosis research should prioritize large-scale, diverse, and methodologically consistent studies. Such investigations could help resolve current contradictions, consolidate our understanding of established prognostic factors, and reveal novel ones. In addition, research into the impact of demographic factors like sex and age on NMOSD prognosis may enable the development of more personalized, effective therapeutic approaches. A deeper understanding of the role of hormonal, genetic, and environmental factors in NMOSD could also facilitate this objective. The increasing awareness of NMOSD comorbidity with other diseases necessitates a more holistic approach to patient care, integrating multiple disease management strategies. Finally, advancements in imaging techniques and biomarker identification could improve our ability to predict disease progression and evaluate treatment effectiveness, aiding in the evolution of NMOSD patient care strategies. This concerted and multidisciplinary approach to NMOSD research holds great promise for enhancing the quality of life for affected patients and their families.
Footnotes
Author Contributions
R.-Y.G. and W.-Y.W. contributed to conceptualization, investigation, and writing—original draft. J.-Y.H., Z.J., Y.-F.S. contributed to investigation and writing—review and editing. B.L. contributed to conceptualization, supervision, writing—review and editing, and funding acquisition.
Data Availability Statement
Data sharing not applicable to this article as no data sets were generated or analyzed during this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by S&T Program of Hebei (no. 22377711D) and Hebei Natural Science Foundation (no. H2023206107).
