Abstract
Background:
Late-onset neutropenia (LON), defined as an absolute neutrophil count (ANC) < 1500/mm3 that develops between 4 weeks and 6 months after the last drug administration, is a rare side effect of anti-CD20 drugs including ocrelizumab. Although continuation of ocrelizumab after LON is not contraindicated, the risk of LON recurrence is not well known.
Cases:
We report three cases of recurrent symptomatic agranulocytosis (ANC < 500/mm3) occurring under ocrelizumab.
Conclusion:
Given the risk of recurrence of symptomatic agranulocytosis and the availability of other treatments, a therapeutic switch may be discussed after the first episode of LON.
Introduction
Ocrelizumab is an anti-CD20 antibody that is widely used as treatment for relapsing-remitting multiple sclerosis (RRMS). A relatively rare but expected complication of anti-CD20 drugs is the occurrence of late-onset neutropenia (LON) in autoimmune diseases, including RRMS. LON is defined as an absolute neutrophil count (ANC) < 1500/mm3 that develops between 4 weeks and 6 months after last drug administration, preceded by a normal neutrophil count, without other identifiable causes. 1
Recurrence of LON is well known in systemic autoimmune diseases, 2 but not in RRMS. We report three symptomatic recurrent agranulocytosis (ANC < 500/mm3) with ocrelizumab.
Cases
A 28-year-old male patient was diagnosed with RRMS in 2019 and treated with dimethylfumarate. The persistence of clinical and radiological activity over the next 6 months led to change in treatment, switching to ocrelizumab in February 2020. In November 2021, 8 weeks after the fourth ocrelizumab infusion, he developed severe neutropenia (neutrophils = 322/mm3) along with a febrile stomatitis. The evolution was rapidly favourable with antibiotic prophylactic coverage. Neutropenia resolved spontaneously within 1 week. He received a new infusion in March 2022 that resulted in the recurrence of febrile agranulocytosis (neutrophils = 41/mm3) spontaneously favourable in September 2022. This second episode of neutropenia led to a switch to natalizumab.
The second case concerns a 38-year-old woman affected by RRMS since 2010 initially treated with natalizumab and then fingolimod due to JC virus seroconversion. Because of the disease activity on fingolimod, treatment with ocrelizumab was introduced in March 2020. Infusions were carried out without any adverse effects until the fifth infusion received on 10 March 2022. On 22 April 2022, stomatitis and increased fatigue revealed a febrile agranulocytosis (neutrophils = 30/mm3). Treatment with granulocyte colony-stimulating factor (G-CSF) led to restoration of the neutrophil count within 3 days. On 23 June 2022, a new episode of febrile agranulocytosis occurred (neutrophils = 41/mm3): ocrelizumab was stopped and ofatumumab was introduced in October 2022.
A 35-year-old female patient diagnosed with RRMS since 2007 started ocrelizumab in June 2021 following a severe medullary and cerebellar relapse. The second infusion was administered on 15 December 2021. Asymptomatic agranulocytosis (neutrophils = 20/mm3) was discovered on 15 January 2022 with spontaneous normalization in March 2022 without any therapeutic intervention. The third infusion was performed on 23 June 2022. While she was diagnosed with COVID-19 on 19 September 2022, a febrile agranulocytosis (neutrophils = 20/mm3) was detected, complicated by a pulmonary infection. The clinical course was positive after antibiotic treatment and G-CSF. Natalizumab was introduced in January 2023.
The characteristics of the three cases of LON recurrence are summarized in Figure 1.
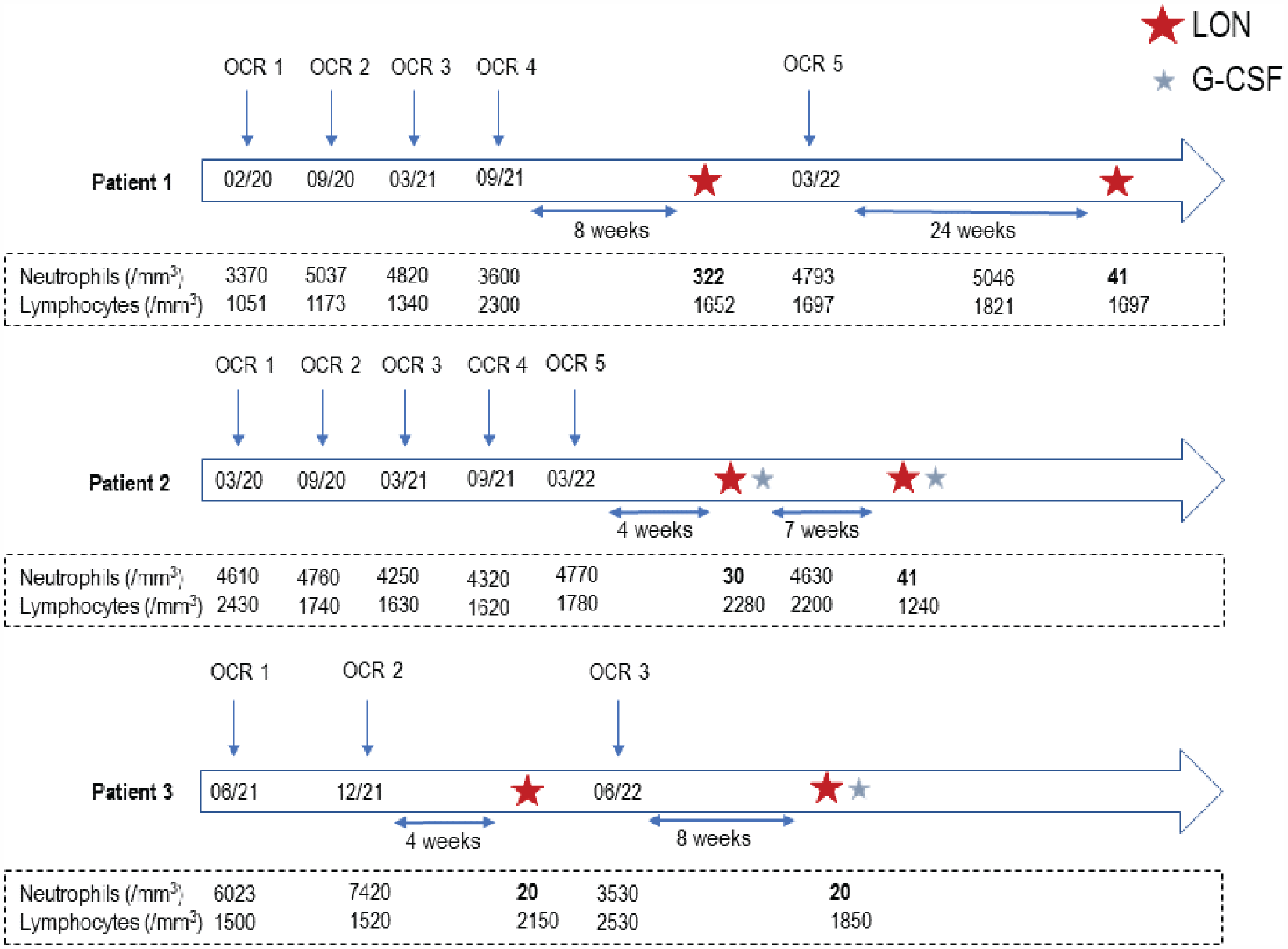
Summary of the characteristics of the three recurrent LON cases (nadir in bold).
Discussion
LON is a rare and known adverse event of anti-CD20 drugs. In a large retrospective cohort of 738 patients treated with rituximab for nephrological and systemic autoimmune diseases, 2 the cumulative incidence of LON at 5 years was 13.5% (95% confidence interval (CI) = 10.4–17.4) with a higher risk in the first year of treatment. Forty percent of cases were associated with infection. The cumulative incidence of recurrent LON at 1, 2 and 5 years after rechallenge was 11.5% (95% CI = 5.6–22.6), 23.4% (95% CI = 13.8–38.2) and 30.4% (95% CI = 16.9–50.9), respectively. 2 Incomplete understanding of the LON mechanism contributes to the lack of a clear recommendation for further treatment. Although suggested, the role of anti-CD20 antibodies is unlikely as the CD20 receptor is neither expressed on granulocyte nor on progenitor cells. Moreover, increased B-cell activating factor (BAFF) blood levels have been detected at the onset of LON, but whether these are causal remains uncertain. 3 Genetic factors may also be involved. The 158V allele of high-affinity FcgammaRIIIa has been associated with rituximab-induced neutropenia. 4
Concerning multiple sclerosis patients, several reports have also described the occurrence of LON on ocrelizumab.5–8 In a recent prospective study including 35 patients on ocrelizumab, followed for 6 months, only one asymptomatic neutropenia (670/mm3) was identified, occurring 119 days after the first infusion. 1 In the pivotal clinical trials of ocrelizumab (OPERA and ORATORIA), neutropenia (defined as ANC < 1500/mm3) was reported in about 4% of patients on ocrelizumab. In the all-exposure population (n = 5680 patients, 18,218 patient-years), 9 serious neutropenia cases (exact neutrophil count not specified) were encountered of which 7 were symptomatic. All continued on ocrelizumab without recurrence. 9
To our knowledge, there is no study evaluating the risk of recurrence of LON with ocrelizumab, and only one single case has been reported. 10 The first episode occurred after the fifth infusion and the recurrence after the sixth infusion, without agranulocytosis (nadir of neutrophils 700 and 900/mm3). In this article, the three recurrences also occurred rapidly after the first episode of LON (between 7 and 40 weeks). All three recurrences were symptomatic agranulocytosis with a very low neutrophil count (nadir < 50/mm3). The clinical course was rapidly favourable.
We performed a retrospective chart of all patients who received ocrelizumab between January 2019 and December 2022 in our centre. Ocrelizumab was introduced to 651 patients, representing a total of approximately 3200 infusions and 1600 years of cumulative exposure. Except these three cases described (i.e. six cases of LON), we did not find any other episode of symptomatic LON in patients treated with ocrelizumab.
Given the recurrence risk of symptomatic LON and the availability of other treatments, therapeutic switch is an open question. A prospective study will be necessary to precisely assess the prevalence of LON and the risk of recurrence. To our knowledge, there is no literature on the risk of neutropenia with other anti-CD20 therapy (ofatumumab, ublituximab) in patients who developed LON with ocrelizumab.
Conclusion
Recurrences of LON previously described with rituximab occur also on ocrelizumab with possible symptomatic agranulocytosis. Due to the risk of infection, close monitoring of complete blood counts and education seems appropriate. After a first episode of LON, a therapeutic alternative may be discussed given the risk of recurrence.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
