Abstract
Background:
Multiple sclerosis (MS) negatively affects health-related quality of life (HRQoL).
Objective:
To evaluate HRQoL in people with highly active relapsing MS treated with cladribine tablets (CladT; 3.5 mg/kg cumulative dose over 2 years) in CLARIFY-MS.
Methods:
Changes in the MS quality of life (MSQoL)-54 scores were analysed using a repeated mixed-effects linear model. Subgroup analyses were performed for participants who were pretreatment-naïve and those pretreated with disease-modifying therapies (DMTs) before initiating CladT. Safety and tolerability of CladT were also assessed.
Results:
MSQoL-54 physical (mean change = 4.86; 95% confidence interval (CI) = 3.18, 6.53) and mental health (4.80; 95% CI = 3.13, 6.46) composite scores (primary endpoints) showed significant improvement at Month 24 versus Baseline (
Conclusions:
CladT treatment significantly improved the mean MSQoL-54 physical and mental health composite scores over 2 years. CladT efficacy in HRQoL, relapse rates and Expanded Disability Status Scale scores demonstrates its multidimensional effects in MS treatment.
Introduction
Many people with multiple sclerosis (PwMS) exhibit a highly active disease course characterised by frequent relapses, magnetic resonance imaging activity and disability progression. 1 The symptoms, disability burden and comorbidities experienced by PwMS have a detrimental effect on their health-related quality of life (HRQoL) 2 and can disrupt their family, social and work lives. 3
Early initiation of effective treatment may delay disease progression and disability accumulation in people with highly active relapsing MS (RMS).1,4–6 Cladribine tablets (CladT) at a cumulative dose of 3.5 mg/kg over 2 years is approved for highly active RMS, as defined by clinical and imaging features. 7
While clinical development programmes for CladT provide valuable information regarding its efficacy and safety,4,8 its effects on HRQoL and treatment satisfaction were not evaluated as primary or secondary outcomes and have not been explored fully. Analysis of HRQoL data collected during the phase III CLARITY study revealed potential improvements in European Quality of Life-Five Dimensions (EQ-5D) scores; however, data were insufficient to draw clear inferences about outcomes using the disease-specific Multiple Sclerosis Quality of Life (MSQoL)-54 instrument. 9
The CLARIFY-MS study evaluated changes in the HRQoL of people with highly active RMS treated with CladT over 24 months. The 6-month interim analysis from this study demonstrated that CladT was well tolerated and exhibited high treatment satisfaction levels according to the Treatment Satisfaction Questionnaire for Medication (TSQM; version 1.4) scores in both treatment-naïve and disease-modifying therapy (DMT) experienced participants during the first treatment course. 10 Here we report changes in MSQoL-54 physical and mental health composite scores (PCS and MCS) at 24 months from baseline as the primary outcome measure, along with other efficacy and safety findings from CLARIFY-MS.
Participants and methods
Study design
CLARIFY-MS was a 2-year, prospective, open-label, exploratory, single-arm, multicentre, phase IV study (Supplementary Figure S1) conducted at 85 centres across 18 countries between June 2018 and August 2021.
Eligible participants were aged ⩾ 18 years with an Expanded Disability Status Scale (EDSS) score of ⩽ 5.0 and highly active RMS (defined as one relapse in the previous year and ⩾ 1 T1 gadolinium-enhanced lesions or ⩾ 9 T2 lesions while receiving treatment with other DMTs or ⩾ 2 relapses in the previous year regardless of receiving any DMT).7,10 Detailed inclusion and exclusion criteria have been reported previously. 10 Eligible participants received CladT at a cumulative dose of 3.5 mg/kg over 2 years, with 2 weeks of active treatment per course (Weeks 1 and 5 of each year). Each treatment week comprised 4 or 5 days on which a participant received 10 or 20 mg (one or two tablets) of cladribine as a single daily dose, depending on body weight. The study was completed at the 24-month visit.
The MSQoL-54 instrument is a combination of the 36-Item Short Form Health Survey (SF-36) and 18 additional MS-specific items. 11 Based on expert recommendations, a prespecified improvement of ⩾ 5 points was considered as minimal clinically important difference (MCID) for the SF-36 standardised physical component scale and MSQoL-54 composite scores.
All screening, Baseline, Month 2 and 6 visits were done before the coronavirus disease 2019 (COVID-19) pandemic. For most participants, Month 12 and 12b visits were completed pre-COVID-19, while Month 14 and 18 visits were completed post-COVID-19. All Month 24 visits were completed post-COVID-19 (Supplementary Figure S1). The HRQoL outcomes reported by the participants were collected consistently throughout the study without using any bimodal data collection methods. Patient-reported outcomes (PROs) were completed on hand-held tablet devices, onsite at the clinics.
Endpoints
The primary objective of this study was to assess the HRQoL using the MSQoL-54 instrument in people with highly active RMS treated with CladT over 24 months. The primary endpoints were changes in MSQoL-54 PCS and MCS at 24 months compared with baseline. The secondary objective was to evaluate treatment satisfaction with CladT using the TSQM (version 1.4) at 6 months. The full list of tertiary endpoints has been reported previously. 10 Tertiary endpoints discussed here are changes from baseline in MSQoL-54 PCS and MCS at Month 12 and overall quality of life (QoL) scores at Months 12 and 24; TSQM (version 1.4) global satisfaction scores at Months 12 and 24; TSQM (version 1.4) treatment effectiveness, side effects and convenience scores at Months 6, 12 and 24; annualised relapse rates (ARRs) over 24 months; EDSS scores at Months 6, 12, 18 and 24; 6-month confirmed disability progression (6mCDP); and treatment-emergent adverse events (TEAEs), serious TEAEs and lymphocyte counts gradings (based on National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0) 12 up to Month 24 visit.
Herein, qualifying relapses were required to meet several standard criteria described previously. 10 6mCDP was defined as an increase in ⩾ 1.5, ⩾ 1 or ⩾ 0.5 points if baseline EDSS scores were 0, 0.5–4.5 (inclusive) or ⩾ 5 points, respectively, confirmed after 6 months.
Statistical analysis
Participants who received ⩾1 dose of CladT were included in the treated set (TS) and were further grouped into two subgroups: those who did not receive any DMTs before CladT treatment (pretreatment-naïve subgroup) and those who received DMTs at any time before CladT treatment (DMT-pretreated subgroup).
Changes in MSQoL-54 composite scores were analysed using a repeated-measures mixed-effects linear model that was adjusted for baseline MSQoL-54 composite scores, baseline EDSS scores, age and within-country correlations (pre-planned analyses), along with gender and time since diagnosis (post hoc analyses). In addition, separate post hoc analyses were performed to compare baseline demographics and disease characteristics of participants with and without data available for MSQoL-54 assessments and to evaluate MSQoL-54 subscale data.
The secondary and tertiary endpoints were assessed using methods similar to those used for the primary endpoints, wherever applicable. ARRs were estimated using Poisson regression. Data from the second year of treatment were excluded from the efficacy analyses if a participant delayed their second course of treatment by > 3 months. For the analysis of all the secondary and tertiary efficacy endpoints, differences with a
Results
Participants
Overall, 554 people with highly active RMS were enrolled, 485 of whom met the eligibility criteria. Among them, 482 received ⩾ 1 dose of CladT and were included in the TS. In total, 461 participants started the second annual treatment course. Twenty participants had a delay of > 3 months in starting the second treatment course owing to lymphopenia (

Participant disposition.
Baseline participant demographic and disease characteristics are summarised in Table 1. Participants in the TS had a mean (± SD) age of 37.4 (± 10.39) years and were predominantly female (70.1%). Overall, the mean (± SD) time since the onset of MS was 99.1 (± 89.8) months. Most participants (94.1%) experienced one-to-two relapses within the previous 12 months before starting CladT. The mean (± SD) MSQoL-54 PCS and MCS for the TS at baseline were 60.2 (± 19.7) and 61.2 (± 21.7), respectively.
Baseline participant demographic and disease characteristics for the TS.
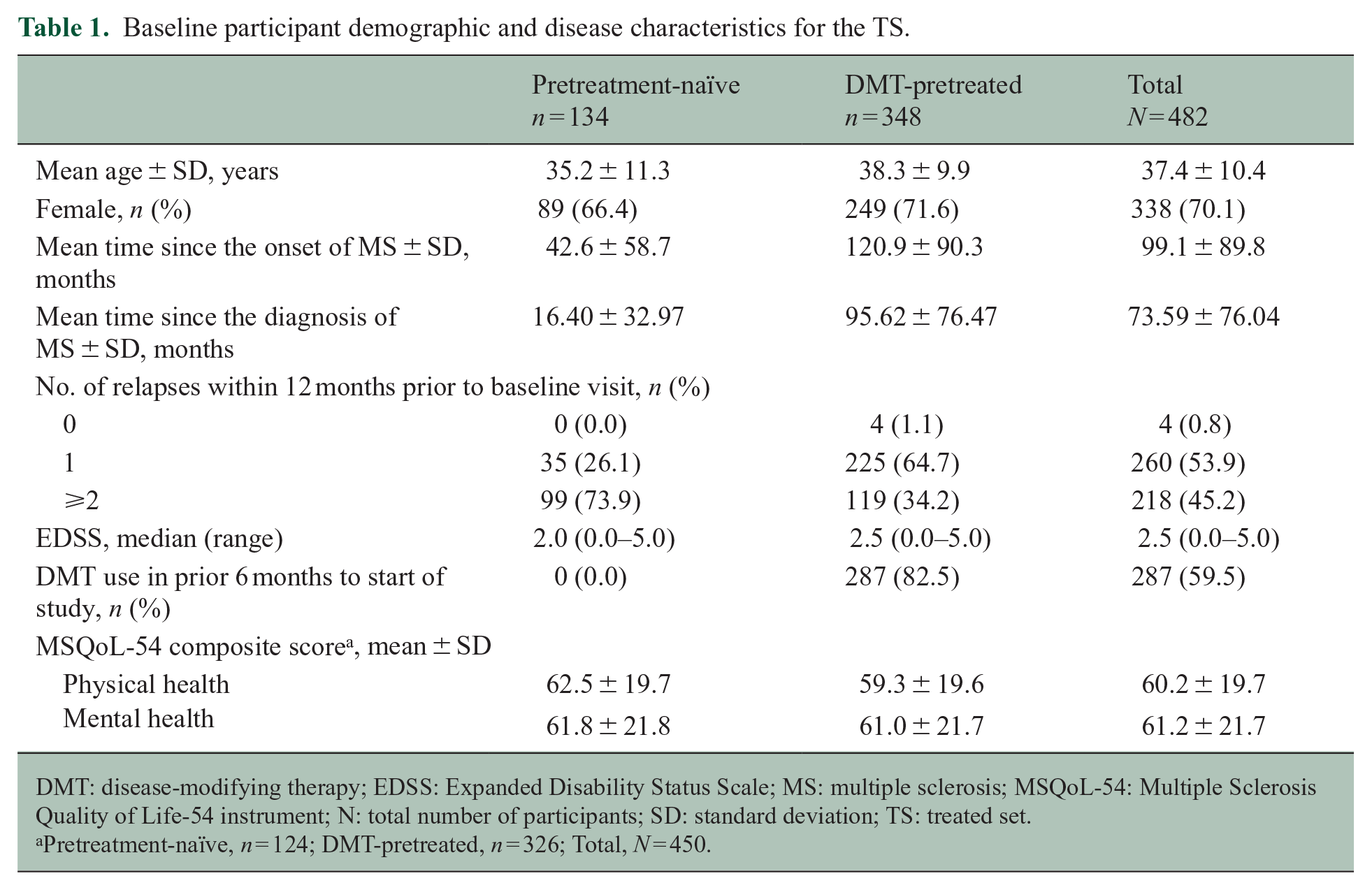
DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; MS: multiple sclerosis; MSQoL-54: Multiple Sclerosis Quality of Life-54 instrument; N: total number of participants; SD: standard deviation; TS: treated set.
Pretreatment-naïve,
Overall, 348 participants had previous DMT exposure matching the criteria for the DMT-pretreated subgroup, whereas the remaining 134 were included in the pretreatment-naïve subgroup during the analyses. The mean time (± SD) since the onset of MS was 42.6 (± 58.7) and 120.9 (± 90.3) months in the pretreatment-naïve and DMT-pretreated subgroups, respectively. The mean MSQoL-54 PCS and MCS were similar in both subgroups at baseline (Table 1). Among the 348 participants who previously received DMTs, 287 (82.5%) received DMTs within 6 months before starting CladT. Most participants switched from interferons (IFNs;
MSQoL-54 composite scores
Of the 482 participants treated with CladT, 433 were included in the mixed-model analysis of the MSQoL-54 data; as those without MSQoL-54 baseline data or any follow-up assessments (
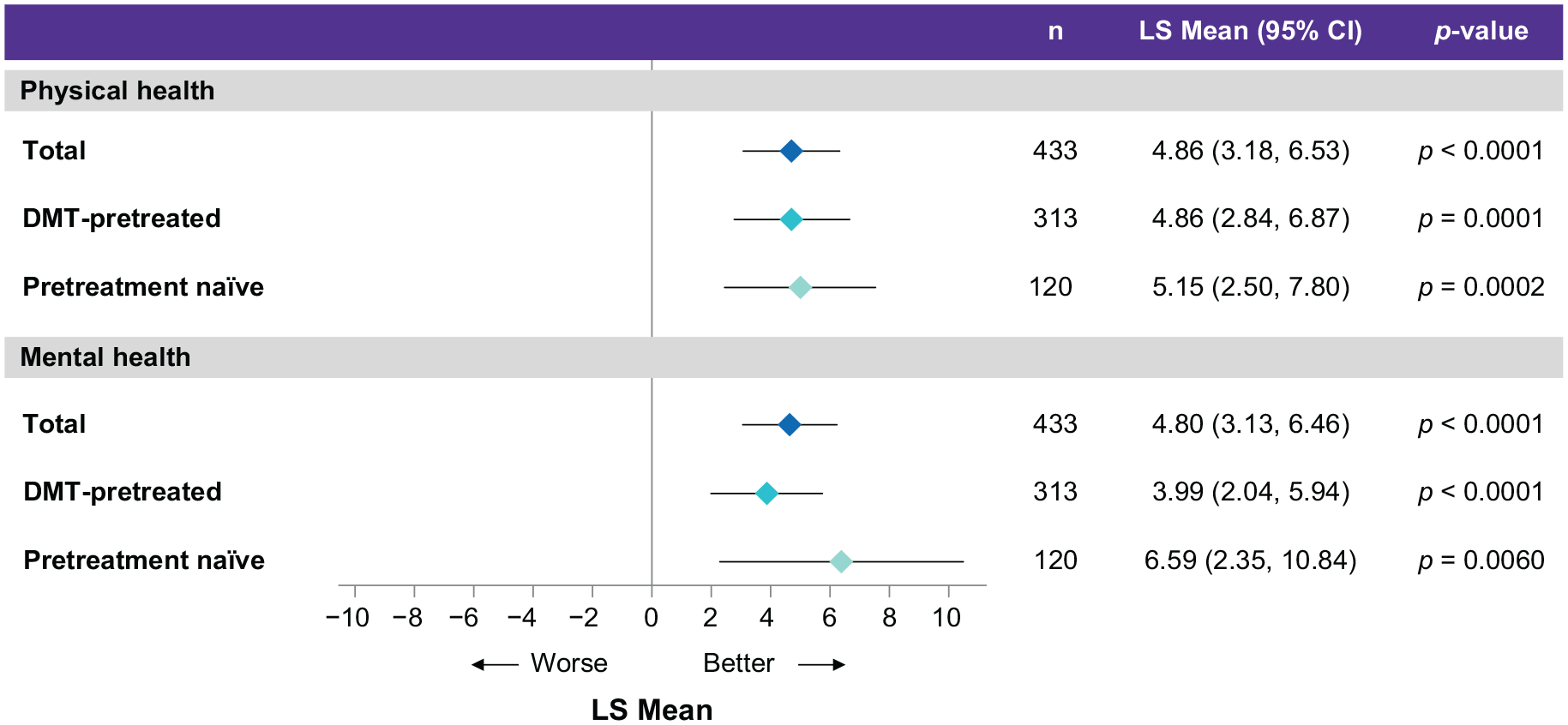
MSQoL-54 PCS and MCS: change from baseline to the Month 24 according to the subgroups in the TS.
MSQoL-54 overall QoL and individual subscale scores
The LS mean MSQoL-54 overall QoL scores improved significantly by 2.71 (95% CI = 1.32, 4.10;
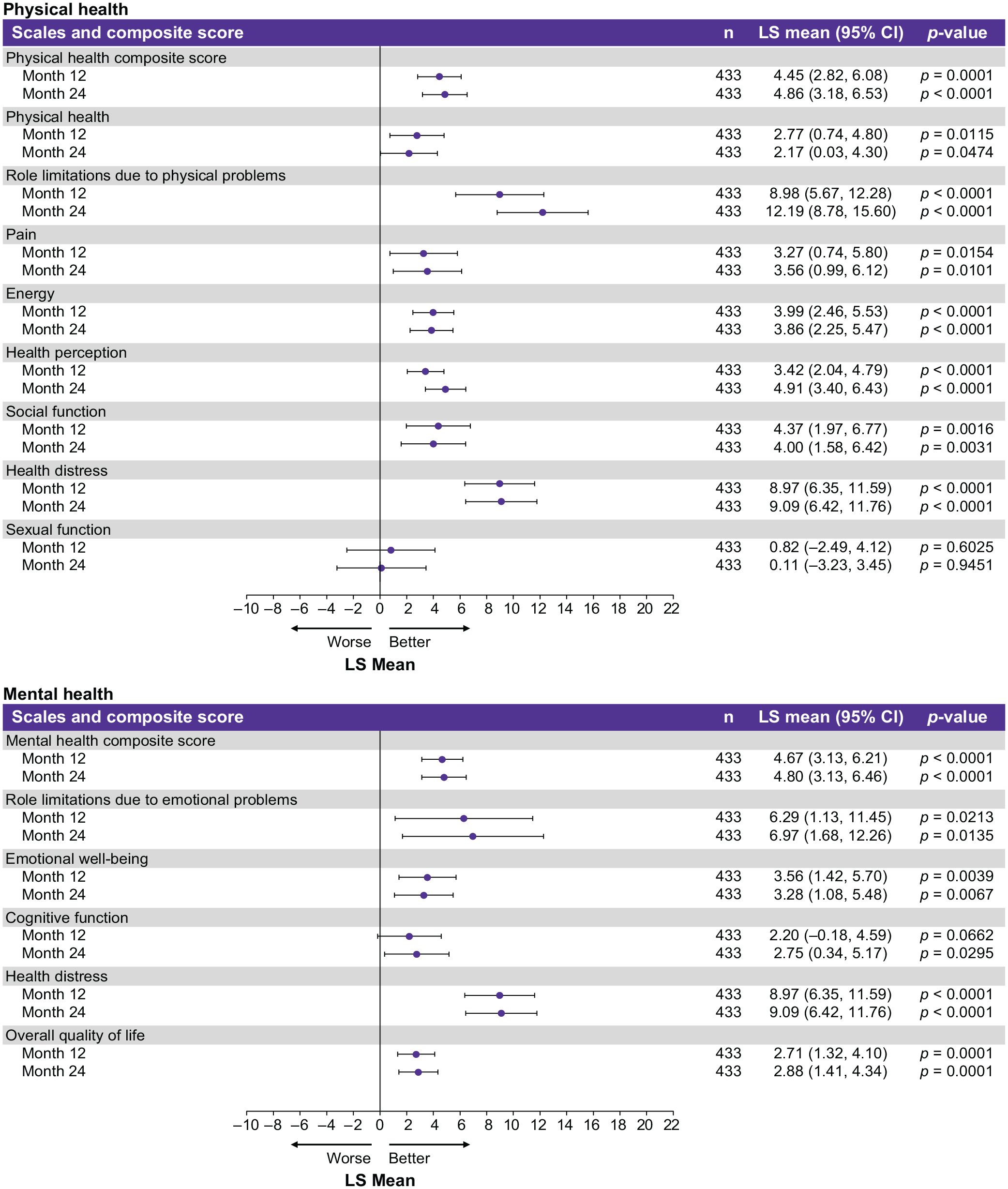
Changes from baseline for each MSQoL-54 scale and composite score for the TS.
TSQM
The 6-month LS mean TSQM (version 1.4) global satisfaction score (secondary endpoint) was 72.02 (95% CI = 68.76, 75.28). The LS mean global satisfaction, effectiveness, side effects and convenience scores remained similar across Months 6, 12 and 24 (Supplementary Figure S2).
Qualifying relapse/ARR
The estimated ARRs of the qualifying relapses were 0.13 (95% CI = 0.11, 0.16), 0.08 (95% CI = 0.05, 0.13) and 0.15 (95% CI = 0.12, 0.18) in the TS, pretreatment-naïve and DMT-pretreated subgroups, respectively. In the TS, 91/482 participants (18.9%; pretreatment-naïve: 21/134, 15.7%; DMT-pretreated: 70/348, 20.1%) experienced a qualifying relapse during the study. Overall, 83 participants (17.2%; pretreatment-naïve:
EDSS and 6mCDP
The median EDSS score at all the timepoints was 2.5 for the TS and DMT-pretreated subgroup, and 2.0 for the pretreatment-naïve subgroup (Supplementary Figure S3). During the treatment period, most participants in the TS (88.0%; 424/482), pretreatment-naïve (88.1%; 118/134) and DMT-pretreated (87.9%; 306/348) subgroups were free from 6mCDP.
Safety
Of the 482 participants in the TS, 376 (78.0%) experienced ⩾ 1 TEAE after starting CladT (Table 2). The most common TEAEs were headache (21.8%; 105/482 participants), lymphopenia (15.1%; 73/482 participants) and nasopharyngitis (13.5%; 65/482 participants). Overall, 3.1% (15/482) participants experienced COVID-19-associated TEAEs. Most participants experienced mild (39.6%; 191/482) or moderate (35.1%; 169/482) TEAEs. Severe TEAEs were observed in 3.3% (16/482) participants.
Summary of TEAEs in the TS.
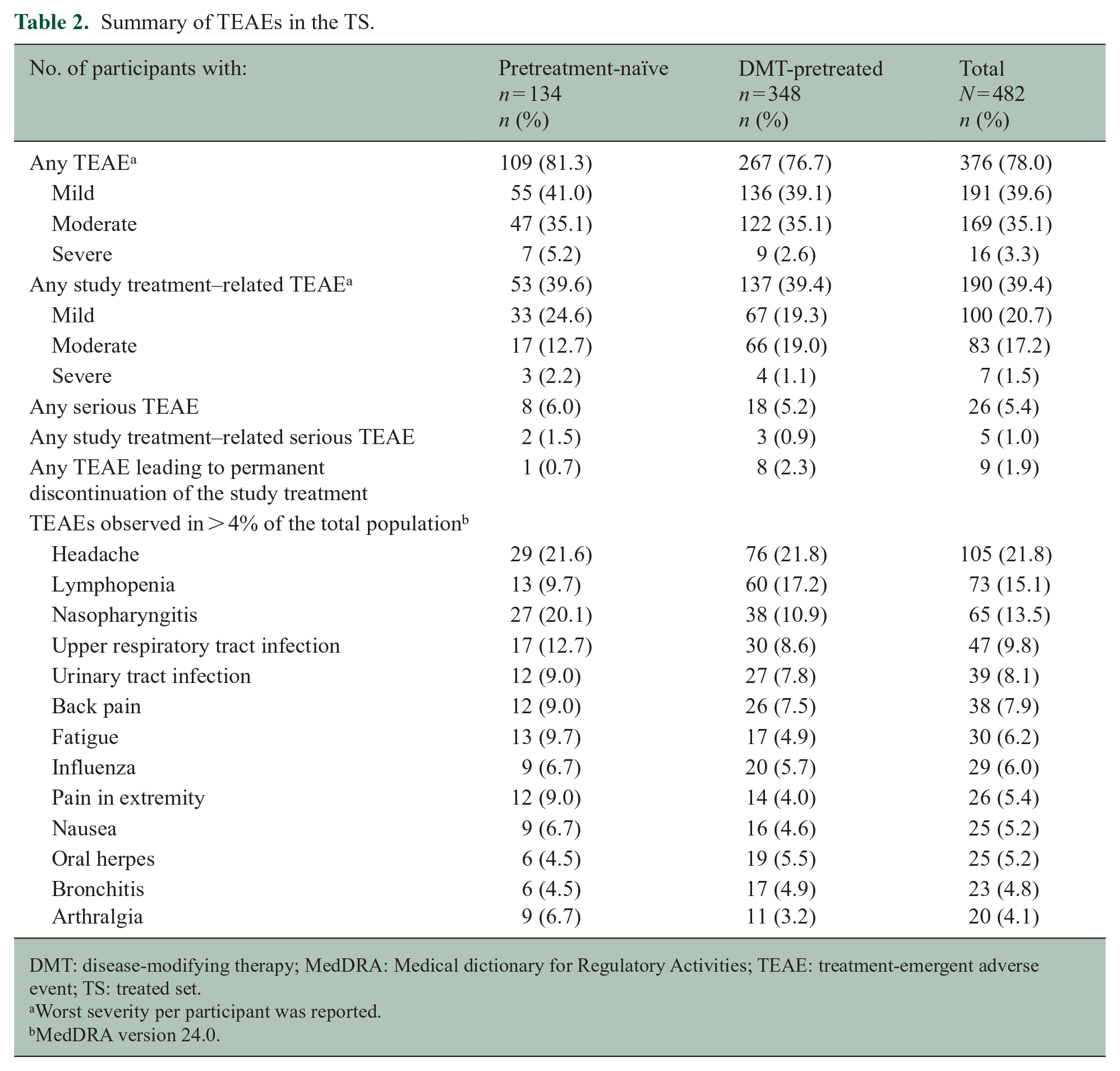
DMT: disease-modifying therapy; MedDRA: Medical dictionary for Regulatory Activities; TEAE: treatment-emergent adverse event; TS: treated set.
Worst severity per participant was reported.
MedDRA version 24.0.
Serious TEAEs (Supplementary Table S4) were observed in 26/482 (5.4%) participants, of which 5 (1.0%) were reported to be potentially related to the study treatment (overdose,
Nine out of 482 (1.9%) participants permanently discontinued study treatment due to TEAEs. No deaths were reported during this study.
As per the laboratory assessments, most post-baseline lymphopenia events were Grade 1–2 in severity. Transient Grade-3 lymphopenia was observed in 19.7% (95/482) of participants. No cases of Grade-4 lymphopenia were observed. Frequency of Grade-3 lymphopenia was higher in the DMT-pretreated subgroup (23.0%; 80/348) than the pretreatment-naïve subgroup (11.2%; 15/134) (Figure 4).
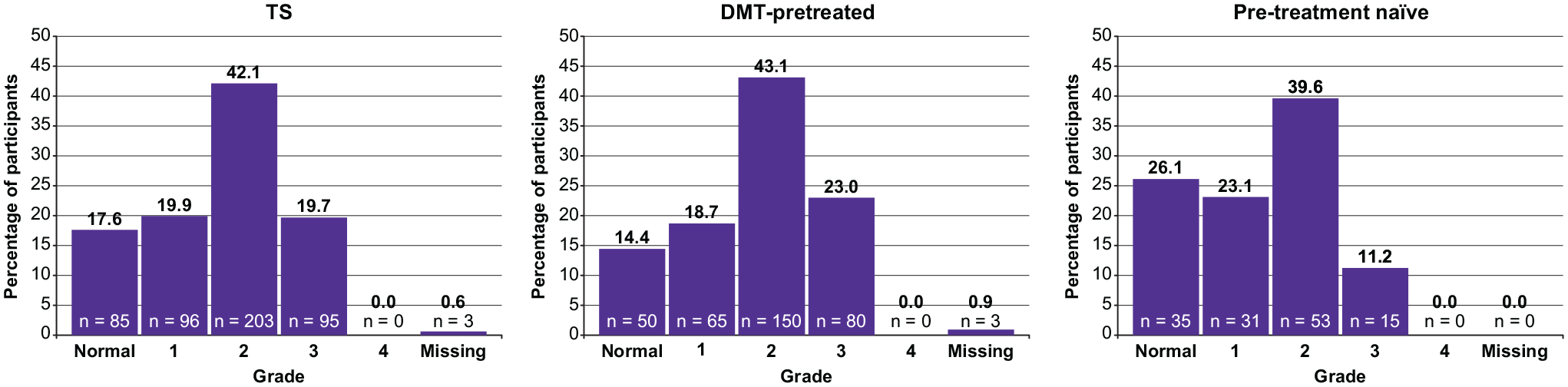
Highest post-baseline grade of lymphopenia.
Discussion
The CLARIFY-MS study assessed changes in HRQoL of participants treated with CladT 3.5 mg/kg over 24 months. The mean MSQoL-54 PCS and MCS scores at Month 24 improved significantly from baseline. ARRs of the qualifying relapses in CLARIFY-MS were comparable with those reported by the phase III CLARITY study; 4 median EDSS scores remained stable throughout this study. No new safety signals were revealed; in particular, there were no new severe or opportunistic infections. These data from CLARIFY-MS supplement the extensively published clinical trial findings regarding the efficacy and safety of CladT,4,8 while substantially enhancing our understanding of the effect of CladT on the participants’ HRQoL.
HRQoL assessment provides essential information regarding the possible benefits and risks of DMTs beyond the clinician-reported outcomes.11,13 Measuring HRQoL and other PROs can increase awareness of clinicians regarding the concerns and priorities of patients, ultimately enhancing shared decision-making and patient-centred care.11,14 HRQoL improvement is a key component of drug efficacy in MS. 15 Findings from small randomised controlled trials and observational studies involving PwMS indicate that treatment with DMTs is linked to either prevention of worsening, stabilisation or improvement of HRQoL. 11 However, limited prospective data are available regarding HRQoL in large trials involving PwMS, particularly with regard to disease-specific measures. In the phase IV EPOC study, PwMS who switched from injectable platform therapies (glatiramer acetate and IFNs) to fingolimod exhibited higher SF-36 PCS and MCS compared with the PCS of those continuing therapy with IFN-β1b and IFN-β1a.16,17 The HRQoL data from the phase III CARE-MS I and II studies showed that more PwMS who received alemtuzumab recorded improvements in the SF-36 PCS than those who received IFN-β1a. 18 In the phase III OPERA-II study, the PwMS who received ocrelizumab demonstrated a greater adjusted mean change in the SF-36 PCS scores from baseline than those who received IFN-β1a. 19 These findings indicate that high-efficacy therapies are linked to greater improvements in HRQoL than platform therapies in PwMS. In the CLARIFY-MS study, CladT significantly improved the MSQoL-54 PCS and MCS over 2 years, but in a dedicated HRQoL study in a cohort with highly active RMS, using a disease-specific scale.
Despite the growing interest in exploring HRQoL-related endpoints in clinical studies, their subjective nature and potential for large inter-individual variation renders it difficult to interpret the clinical relevance of such changes. MCID sets a threshold value for changes in a scale or instrument considered clinically relevant.11,20–22 Several studies have selected changes of 0.5 times the SD for HRQoL scores as clinically relevant.11,21,23 Based on these findings and expert recommendations, the MCID was defined (from baseline to Week 24) as ⩾ 5 for the MSQoL-54 MCS and PCS in our study. Notably, > 40% participants in CLARIFY-MS achieved an MCID of ⩾ 5 points for the MSQoL-54 PCS and MCS. The HRQoL benefits of CladT observed in the CLARIFY-MS study confirm and expand the initial HRQoL EQ-5D and MSQoL-54 instrument findings of the CLARITY study. 9
In PwMS, higher treatment satisfaction is typically linked to fewer relapses, lower disability scores and increases in HRQoL.24,25 Data from the cross-sectional, observational, multicentre THEPAMS study in PwMS treated with DMTs showed that the mean SF-36 PCS and MCS were significantly higher in those with high treatment satisfaction scores (TSQM global score ⩾ 75) versus those being less satisfied (TSQM global score < 75; all
The MSQoL-54 subscale data from CLARIFY-MS show that CladT acts on multiple individual domains of HRQoL to bring about holistic improvements in overall QoL of people with highly active RMS. Hence, the participants experienced improvements across multiple physical health domains, including physical function, limitations due to physical problems, pain, energy levels, social function and health perception. Similarly, improvements in role limitations due to emotional problems, emotional well-being, cognitive function and health distress domains contributed to the overall improvement in mental health.
Study limitations
As CLARIFY-MS was a single-arm study, there was no control group to compare with the treated group. Results for each timepoint were compared to baseline values, wherever applicable. As this was an open-label study, the participants and investigators possessed prior knowledge regarding the study treatment, which may have influenced certain responses of participants to questions in the MSQoL-54 instrument and TSQM (version 1.4), and observations made by the investigators.
Conclusions
CladT treatment significantly improved the MSQoL-54 PCS and MCS with a substantial proportion of participants achieving clinically relevant improvements in HRQoL over 2 years. The ARRs and EDSS data in this study were in accordance with the findings of randomised controlled trials in the CladT clinical development programme. No new safety signals impacting the established benefit-to-risk profile of CladT in people with highly active RMS were observed. CladT efficacy in QoL, relapse rates and EDSS scores demonstrates its multidimensional effects in MS treatment.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231205962 – Supplemental material for Improvements in quality of life over 2 years with cladribine tablets in people with relapsing multiple sclerosis: The CLARIFY-MS study
Supplemental material, sj-docx-1-msj-10.1177_13524585231205962 for Improvements in quality of life over 2 years with cladribine tablets in people with relapsing multiple sclerosis: The CLARIFY-MS study by Bruno Brochet, Alessandra Solari, Jeannette Lechner-Scott, Fredrik Piehl, Dawn Langdon, Raymond Hupperts, Krzysztof Selmaj, Francesco Patti, Luis Brieva, Eva Maria Maida, Nektaria Alexandri, Andrzej Smyk, Axel Nolting, Birgit Keller, Xavier Montalban and Eva Kubala Havrdova in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors thank the participants and their families, investigators, co-investigators and the study teams at each of the participating centres and at Merck KGaA, Darmstadt, Germany. Medical writing assistance was provided by Pritorthi Bhattacharjee of Merck Specialties Pvt. Ltd., Bengaluru, India, an affiliate of Merck KGaA. Trial Registry Details: URL: ![]() ; ClinicalTrials.gov Identifier: NCT03369665.
; ClinicalTrials.gov Identifier: NCT03369665.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: B.B. has received consultancy fees, speaker fees, research grants (non-personal) or honoraria from Biogen, Celgene (Bristol Myers Squibb (BMS)), Merck, Novartis, Roche and Sanofi. A.S. has served on advisory boards for Merck, Novartis and Sanofi, and has been invited to speak on behalf of Almirall, Biogen, EXCEMED, Merck and Teva. J.L.-S. has accepted travel compensation from Biogen, Merck and Novartis; her institution receives the honoraria for talks and advisory board commitment, and research grants from Biogen, Celgene (BMS), Merck, Novartis, Roche, Sanofi and Teva. F.P. has received research grants from Janssen, Merck and Sanofi, and fees for serving as a member of the data monitoring committee (DMC) in clinical trials with Lundbeck, Chugai and Roche, and preparation of witness statement for Novartis. D.L. has participated in speaker bureau for Almirall, Bayer, Biogen, BMS, Merck, Novartis, Roche, Sanofi and Teva; has received consultancy fees from Bayer, Biogen, BMS, Merck, Novartis and Teva; and has received research grants from Bayer, Biogen, Merck and Novartis. R.H. has received institutional research grants and fees for lectures and advisory boards from Biogen, Merck and Sanofi. K.S. has received honoraria for speaking, consulting and serving for advisory boards for Biogen, Celgene (BMS), Merck, Novartis, Roche and TG Therapeutics. F.Pa. has served on scientific Advisory Boards for Almirall, Bayer, Biogen, Celgene (BMS), Merck, Novartis, Roche, Sanofi and Teva; he also received speaker honoraria from the same companies and non-personal research grants for his department from Biogen, Merck, Novartis and Sanofi. L.B. has received honoraria, travel expenses, speaker fees and advisory fees from Almirall, Bayer, Celgene (BMS), Biogen, Merck, Novartis, Roche, Sanofi and Teva. E.M.M. has received honoraria for participating as primary investigator in clinical trials from Actelion (Janssen/J&J), Merck, Novartis and Teva. N.A., A.Sm., A.N. and B.K. are employees of Merck Healthcare KGaA, Darmstadt, Germany. X.M. has received speaking honoraria and travel expenses for participation in scientific meetings, has been a steering committee member of clinical trials or participated in advisory boards of clinical trials in the past years with AbbVie, Actelion, Alexion, Biogen, BMS/Celgene, EMD Serono Research & Development Institute, Inc., Billerica, MA, USA, an affiliate of Merck KGaA, Genzyme, Hoffmann-La Roche, Immunic, Janssen Pharmaceuticals, MedDay, Merck, Mylan, NervGen, Novartis, Sandoz, Sanofi-Genzyme, Teva Pharmaceutical, TG Therapeutics, EXCEMED, MSIF and NMSS. E.K.H. has received honoraria/research support from Actelion (Janssen/J&J), Biogen, Celgene (BMS), Merck, Novartis, Roche, Sanofi and Teva; has served on advisory boards for Actelion (Janssen/J&J), Biogen, Celgene (BMS), Merck, Novartis, Roche and Sanofi; has been supported by the Czech Ministry of Education – project Cooperatio LF1, research area Neuroscience and the project National Institute for Neurological Research (Programme EXCELES, ID project No LX22NPO5107) – funded by the European Union-Next Generation EU.
Funding
This work was supported by Merck (CrossRef Funder ID: 10.13039/100009945).
Ethical Approval
The study was conducted in accordance with the International Conference on Harmonisation Guidelines for Good Clinical Practice and the principles of the Declaration of Helsinki. The Institutional Review Board or ethics committee for each trial site approved the protocol.
Consent to Participate
Written informed consent was obtained from all participants before initiating any study-related procedures.
Supplemental Material
Supplemental material for this article is available online.
Data Availability Statement
Any requests for data by qualified scientific and medical researchers for legitimate research purposes will be subject to Merck’s Data Sharing Policy. All requests should be submitted in writing to Merck’s data sharing portal ![]() . When Merck has a co-research, co-development, or co-marketing or co-promotion agreement, or when the product has been out-licensed, the responsibility for disclosure might be dependent on the agreement between parties. Under these circumstances, Merck will endeavour to gain agreement to share data in response to requests.
. When Merck has a co-research, co-development, or co-marketing or co-promotion agreement, or when the product has been out-licensed, the responsibility for disclosure might be dependent on the agreement between parties. Under these circumstances, Merck will endeavour to gain agreement to share data in response to requests.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
