Abstract
Background:
A review of the safety profile of exercise training in multiple sclerosis (MS) has not been conducted since 2013.
Objective:
We undertook a systematic review and meta-analysis of randomised controlled trials (RCTs) of exercise training published since 2013 and quantified estimated population risks of clinical relapse, adverse events (AE) and serious adverse event (SAE).
Methods:
Articles reporting safety outcomes from comparisons of exercise training with non-exercise among persons with MS were identified. The risk of bias was established from study’s internal validity assessed using Physiotherapy Evidence Database (PEDro). Rates and estimated mean population relative risks (RRs; 95% confidence interval (CI)) of safety outcomes were calculated, and random-effects meta-analysis estimated the mean RR.
Results:
Forty-six interventions from 40 RCTs (N = 1780) yielded 46, 40 and 39 effects for relapse, AE, adverse effects and SAE, respectively. The mean population RRs ((95% CI), p-value) for relapse, AE and SAE were 0.95 ((0.61, 1.48), p = 0.82), 1.40 ((0.90, 2.19), p = 0.14) and 1.05 ((0.62, 1.80), p = 0.85), respectively. No significant heterogeneity is observed for any outcome.
Conclusion:
In studies that reported safety outcomes, there was no higher risk of relapse, AE, adverse effects or SAE for exercise training than the comparator. Exercise training may be promoted as safe and beneficial to persons with MS.
Background
Exercise training represents a rehabilitation-based approach for reversing multiple sclerosis (MS) dysfunction and managing symptoms and should be promoted among persons with MS throughout the disease trajectory.1,2 Systematic reviews and meta-analyses report exercise training can improve physical fitness and walking mobility,3,4 balance, 5 cognition, 6 fatigue, 7 depressive symptoms8–10 and quality of life 7 in MS. Exercise has effects on the hippocampus and other brain structures,11,12 sleep quality13,14 and cardiovascular and metabolic comorbidity.15,16 Upwards of 80% of people with MS do not engage in sufficient amounts of exercise necessary for health-related benefits. 17
The clinical guidelines for exercise prescription in MS7,18,19 are endorsed internationally.1,20 Yet, we know very little about the safety profile of exercise among persons with MS.
Relapse-risk, adverse events (AEs) and serious adverse events (SAEs)21,22 represent metrics for gauging the safety profile of exercise in MS clinical studies, and we described those terms in Table 1. Three reviews have considered the safety of exercise training for MS.23–25 The first and most analytical review included 26 randomised controlled trials (RCTs) published until November 2013. 25 That review reported that persons with MS who received exercise training conditions had lower rates of relapse than control conditions (27% lower relapse rate). Notably, the rate of other AEs was higher for exercise, as represented by a 67% higher risk of AE from exercise than control. Those rates were not based on meta-analytic procedures.
Items for extraction and definition of terms used in this systematic review.

RCT: randomised controlled trial; MS: multiple sclerosis; RR: relative risk; EDSS: Expanded Disability Status Scale; PDDS: Patient-Determined Disease Steps; RPE: rate of perceived exertion; VO2R or HRR–VO2 Reserve or Heart Rate Reserve; AE: adverse event; SAE: serious adverse event; PEDro: Physiotherapy Evidence Database.
Considered for subgroup analyses.
To date, the focus on intervention characteristics, such as exercise type, delivery style (e.g. supervised, independent or remotely supervised), participant disability level or the prescription of exercise consistent with minimal exercise guidelines for persons with MS,7,18 has not been considered in terms of safety.
This systematic review builds on our previous review. 25 By considering publications from 2013 onward, we identify the number of relapses, AE, adverse effects, SAE and serious adverse effects in exercise training studies in MS and quantify the relative risk (RR) of relapses, AE and SAE for exercise conditions compared with non-exercise and non-pharmacological comparison conditions. Secondary aims include exploration of potential sources of variability in overall RR, including (1) exercise types, (2) exercise delivery styles, (3) disability levels (based on established disability cut-points 37 ) and (4) prescription of exercise training based on guidelines for persons with mild-to-moderate MS.7,18
Methods
Our review adhered to the Preferred Reporting Items for Systematic review and Meta-Analysis Protocol statement. 38 An original review by the research team 25 was a basis for this study. The protocol 39 for the current review was registered with the international prospective register of systematic reviews (PROSPERO 2020 CRD42020190544). The Participants, Interventions, Comparisons and Outcomes (PICO) strategy 40 was adapted to suit the scope of the review as follows: the terms were persons with Multiple Sclerosis (participants) and Exercise training (intervention) (refer to search strategy). The comparison was a non-exercise, non-pharmacological comparator, and the outcomes included relapse, AE, adverse effects, SAE and serious adverse effects.
Eligibility criteria
The inclusion criteria were as follows: RCT reporting safety outcomes overall and per condition during the study; adults (aged 18 years or above) with MS and an intervention type which meets the definition of exercise training. 33 We consider the definition of exercise as a subset of physical activity. Exercise is physical activity that is planned, structured, repetitive and purposive towards the improvement or maintenance of physical fitness. 33 All non-active/non-exercise/non-pharmacological control conditions were considered, for example, ‘usual activity’.
The following exclusion criteria were applied: studies not written in English, non-human participants, physical interventions which are primarily sedentary (e.g. manual therapy, breathing exercises, pelvic floor exercises), physical intervention requiring external support to facilitate movement (e.g. hippotherapy or robot assisted exercise), studies where safety outcomes were not reported or unclearly reported (in cases of unclear reporting we contacted study authors a minimum of two times to collect safety data) and studies which were previously included in the 2014 publication. 25
Search strategy and information sources
We searched for publications of RCTs of exercise training in persons with MS using eight databases: Ovid MEDLINE All; Ovid Embase; ProQuest PsycINFO; Cochrane Central Register of Controlled Trials (CENTRAL), Ebsco CINAHL, Scopus, Web of Science Core Collection (CC) and PEDro – Physiotherapy Evidence. We conducted our first search on 17 March 2020 and a final search on 27 April 2022. The search was completed from 2013 to the date of the search. This aligns with our original review search, which ended in November 2013. Supplemental Material 1 comprises the search strategy per database. We also searched the reference lists of included studies and relevant systematic reviews for studies meeting our inclusion criteria, searched clinical trial registries and performed forward citation searches on our included studies.
Data management and extraction
Screening and data extraction were undertaken using Covidence (https://www.covidence.org) and Microsoft Excel, respectively. Articles were screened, and data were extracted per our protocol. 39 Briefly, two authors independently screened studies at the title/abstract and full-text stages, and any disagreement resolved through third author discussion. Data were also extracted independently by two authors, with disagreement resolved through discussion. Where data were not reported in manuscripts, authors were contacted a minimum of two times. Where data could not be sought, data were recorded as not reported. Details of data extracted from each study are available in Table 1.
The risk of bias was determined via the Physiotherapy Evidence Database (PEDro) scale. 34 The PEDro scale is an 11-item scale that relates the external validity or generalisability of the sample (item 1) and the internal validity (items 2–11) of a study. We focused on internal validity (range = 0–10). Thus, a score of 10 indicates the study satisfies the maximum items for internal validity. We considered a score above six representative of high internal validity 25 and hence high methodological quality.
Outcomes
The primary outcomes of interest were (1) Relapse, (2) Adverse Events (AEs), (3) Adverse Effects, (4) Serious Adverse Events (SAEs) and (5) Serious Adverse Effects. Definitions of each outcome are provided in Table 1. Where data were not reported and could not be sought through author contact, data were recorded as not reported.
Analysis
Analyses were conducted using SPSS (IBM SPSS v26; MeanES and MetaReg macros) and Stata (Stata Corp v14; meta).
Calculation of RR
Overall, RR was calculated for each of our primary outcomes (for exercise compared with the comparator). The unit of analysis for each outcome was the total number of relapses, AEs or SAEs experienced in each group of each study.
For example, the overall rate of relapse, defined as the total number of reported relapses in the exercise and comparator (i.e. number of relapses in each condition by the total participants in each condition), was pooled across all studies, respectively. We calculated the overall RR of relapse using standard risk procedures (i.e. ratio of rates) defined as the ratio of participants in a treatment group who experience an illness, condition or event (i.e. relapse) to those in a comparator who experience the same illness, condition or event. 41 We calculated RR by dividing the overall rate of relapse, AEs, adverse effects or SAEs for exercise by the overall rate of relapse, AEs or SAEs, respectively, in the comparator. RR > 1.0 indicated no difference in risk between exercise and comparator; RR > 1.0 indicated higher risk for exercise training and RR < 1.0 indicated a lower risk for exercise training. We only reported data from studies that directly report on the presence or absence of relapse or events. RR, its standard error and 95% confidence intervals (CIs) were calculated according to standard procedures. 42 Where zeros created problems with the computation of RR or standard errors, 0.5 was added to all cells (e.g. exercise relapse, exercise non-relapse, control relapse and control non-relapse).43,44
Data synthesis and analysis
Using an SPSS MeanES macro, 45 a random-effects model was used to aggregate the mean RR for each outcome. Based on established methods, 46 Cochrane Q and I 2 were calculated to evaluate heterogeneity and consistency, respectively. Sampling error was calculated using Cochrane’s Q according to established methods; heterogeneity was indicated if sampling error accounted for <75% of the observed variance and when p < 0.05 for Q 47 . I 2 values of 25%, 50% and 75% indicate small, moderate and large amounts of heterogeneity, respectively. 48
Results
Study selection
We identified 39 eligible reports via the database search and one other via forward citation search (Figure 1). Of those 40 RCTs, 46 exercise interventions were included in the analysis. Sufficient data were included to perform meta-analysis and report on the primary outcome of (1) relapse (k = 46); (2) AEs (k = 40) and (3) SAEs (k = 39). We identified 34 interventions with adverse effects. As adverse effects are AE where a causal relationship is related to the intervention we cannot undertake meta-analyses comparison, as our comparator was usual care (i.e. not an intervention). Serious adverse effects were not reported in any study.
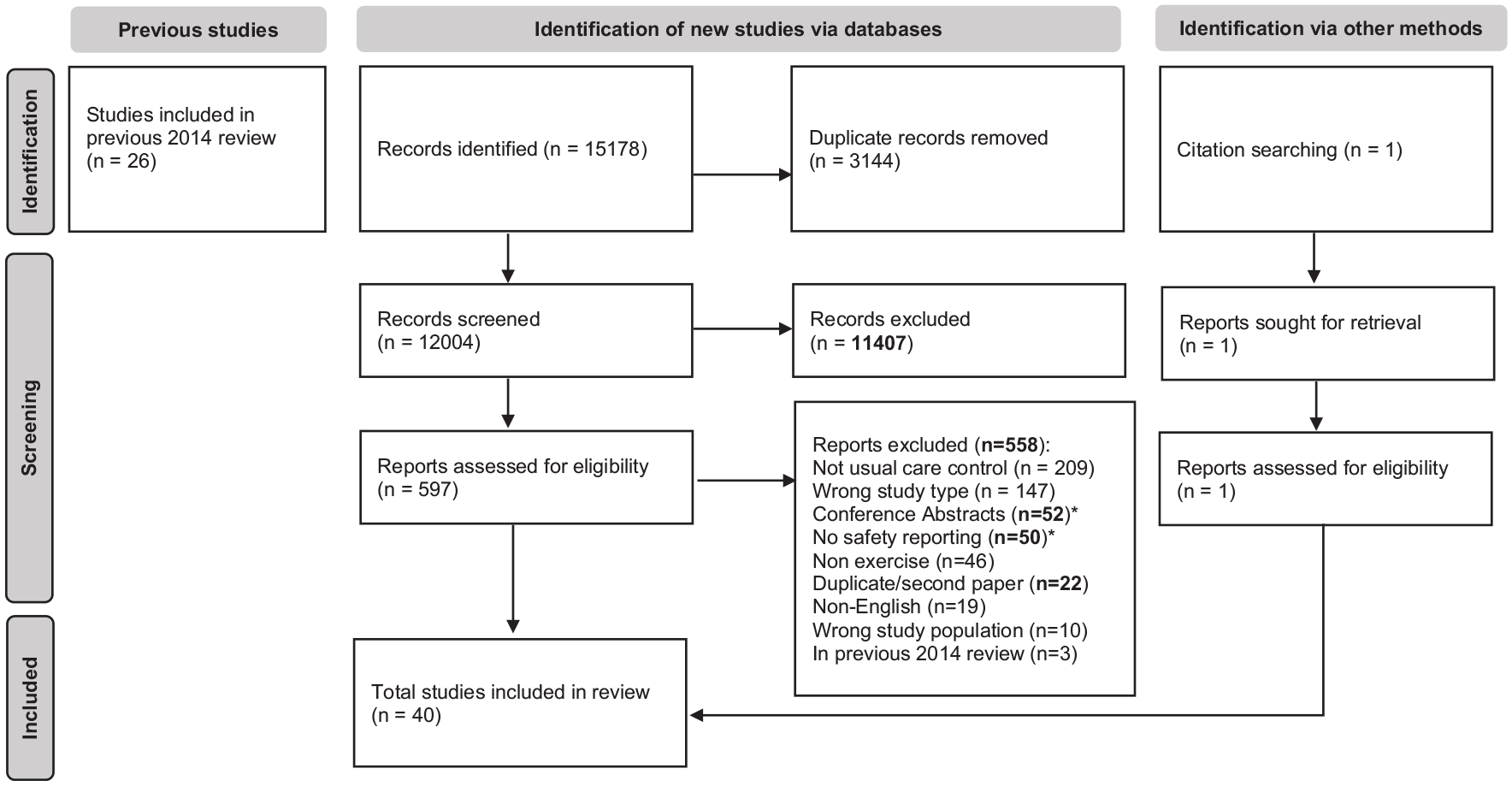
PRISMA flow diagram of updated review.
Study characteristics
Table 2 provides characteristics of included RCTs. The studies were published between 2013 and 2022. The majority of the studies were conducted in Iran (n = 8, 20%), followed by the United States (n = 7, 18%), Belgium (n = 4, 10%), Turkey and Italy (n = 3 per country), Australia, Denmark and the United Kingdom (n = 2 per country), and Brazil, Ireland, the Netherlands, Norway, Slovenia and Spain (n = 1 per country). In 23 (56%) studies, the first author was female. Funding was disclosed for 27 (68%) studies.
All included studies.
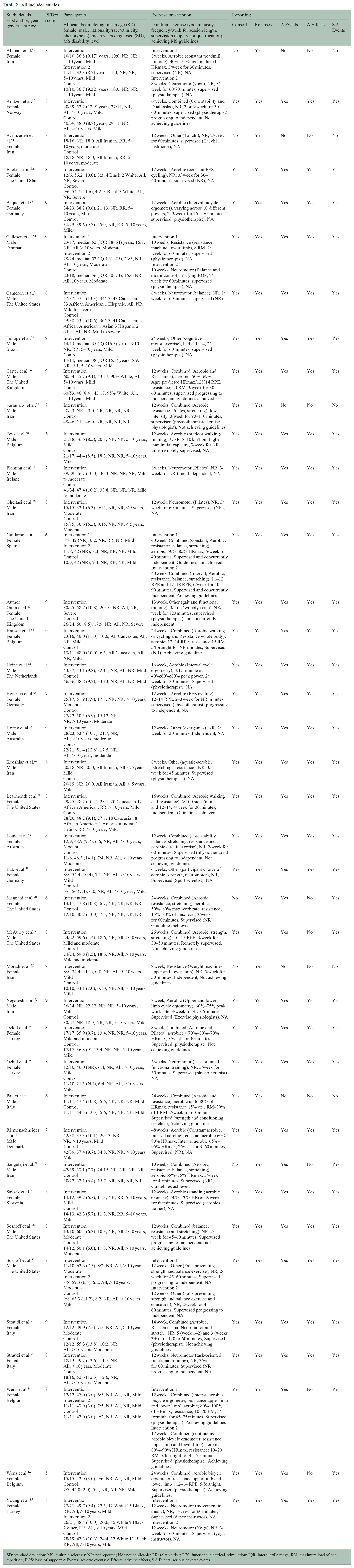
SD: standard deviation; MS: multiple sclerosis; NR: not reported; NA: not applicable; RR: relative risk; FES: functional electrical, stimulation; IQR: interquartile range; RM: maximum load of one repetition; BOS: base of support; A Events: adverse events; A Effects: adverse effects, S A Events: serious adverse events.
Methodological quality
Methodological quality judgement identified PEDro scores ranging between 5 and 9 (mean score 7.7 ± 1.0) on the 10-point internal validity scale (refer to Supplemental Tables). Almost all studies (n = 39, 98%) achieved a score of at least 6 out of 10, indicating high quality (Table 2). No study secured participant blinding, and all but one study failed to secure therapist blinding. 62
Participant characteristics
The mean number of participants per control group or exercise training group was 21 (range 6–60) and 22 (range 8–60), respectively. Over half (n = 21, 53%) of studies reported a mean age over 45 years. The mean age of participants in the studies ranged between 32 and 63 years. There were 679 females and 279 males in the exercise interventions and 597 females and 225 males in the control conditions. Data on participant nationality, race or ethnicity were provided in eight studies.
Participants had primarily mild MS disability in 22 (55%) studies,16,36,48–50,53,56,58,61,63,64,68,72–77,79,84,85 primarily moderate MS disability in nine (23%) studies51,54,60,65,66,69,81–83 and primarily severe MS disability in 3 (8%) studies.52,55,62 Participants had mild and moderate MS disability in three (8%) studies,59,70,71 and two studies did not report disability level.57,78 Participants primarily had a relapsing remitting MS phenotype in 6 (15%) studies36,51,53,61,68,79 and all MS phenotypes in 20 (50%) studies;48,50,52,54,56,62–64,66,67,69,71,72,74,80–84 MS phenotype was not reported in 13 (33%) studies.16,49,57–60,65,70,73,75,77,78,85 Participants had been diagnosed with MS for less than 5 years in two studies,60,67 between 5 and 10 years in nine (23%) studies36,49,51,53,56,58,74,79,81 and over 10 years in 14 (35%) studies;48,50,54,65,66,68,69,71,75,77,80–82,85 13 (33%) studies did not report disease duration.16,52,55,57,59,61–64,70,76,78,84
Exercise intervention characteristics
Exercise intervention length ranged between 2 and 48 weeks (mean 14 weeks), with the number of sessions per week between one and seven (mean 3 sessions per week). Prescribed intensity varied across studies. Nine aerobic,49,52,53,58,64,65,73,77,79 2 resistance,54,72 16 combined16,50,56,57,61,63,68–71,74,76,78,80,82,84 and 7 neuromotor54,55,59,60,75,83,85 exercise interventions were included. Eight other exercise interventions did not meet these criteria types of exercise and were classified as ‘other’.36,48,49,51,62,66,67,81 Details on each intervention are listed in Table 2. Eight of the combined exercise interventions16,56,61,63,68,70,76,78,84 met the 2013 MS exercise guidelines7,18 (see Table 1). The exercise sessions lasted between 30 and 100 minutes. Twenty-five interventions were supervised in-person,16,36,48,49,51–55,57,60,63,64,67,70,73–79,82,84,85 three interventions were completed independently by participants, with minimal researcher input,59,66,72 three interventions included remote supervision,58,68,71 three interventions included both supervised and independent exercise throughout,56,61,62 and seven interventions began supervised and progressed to independent exercise.50,61,65,69,80,81,83
Safety criteria
Rate of events
Our systematic search for relevant studies indicated that many (n = 50) studies were excluded for unclear or missing safety reporting (Supplemental reference list). Among the included studies, the number and description of relapses, AE, adverse effects and SAE are presented in Table 3. Across all studies, 40 (100%), 35 (88%), 30 (75%) and 34 (85%) reported relapses, AE, adverse effects and SAE, respectively. The number of relapses per study in the exercise and control conditions ranged from 0 to 6 and 0 to 9, respectively, across the studies, with an overall prevalence rate of 14% and 43%, respectively. The number of AEs in the exercise and control conditions ranged from 0 to 7 and 0 to 5 per study, respectively, and yielded overall prevalence rates of 38% and 22%, respectively. The number of SAEs in the exercise and control conditions was 0 to 4 and 0 to 5, respectively, and yielded prevalence rates of 9% and 10%, respectively. Adverse effects were only applicable to the exercise condition (i.e. not quantifiable against control for effect size and RR) and the number of adverse effects ranged from 0 to 6, with a prevalence rate of 29%.
Number and rate of relapses, adverse events and effects and serious adverse events.
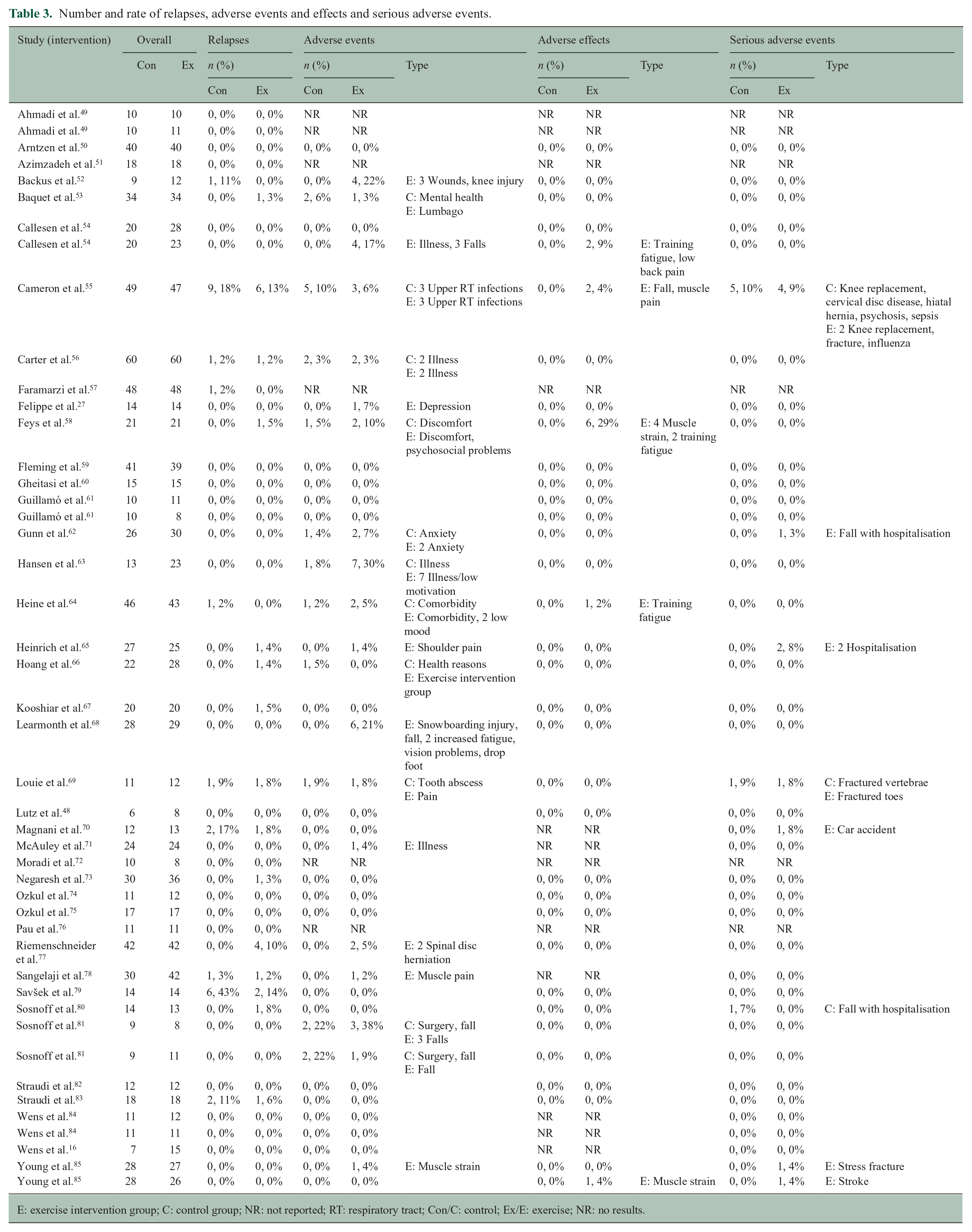
E: exercise intervention group; C: control group; NR: not reported; RT: respiratory tract; Con/C: control; Ex/E: exercise; NR: no results.
Meta-analyses of RRs
Overall, 125 effects were derived from 40 RCTs; 46, 40 and 39 effects were derived for relapse, AEs, adverse effects and SAEs, respectively. Forest plots for relapse, AE and SAE are illustrated in Figures 2–4, respectively. The number of relapses, AE, adverse effects and SAE per clinical and exercise delivery moderator is shown in Supplemental Tables 4–6, respectively.
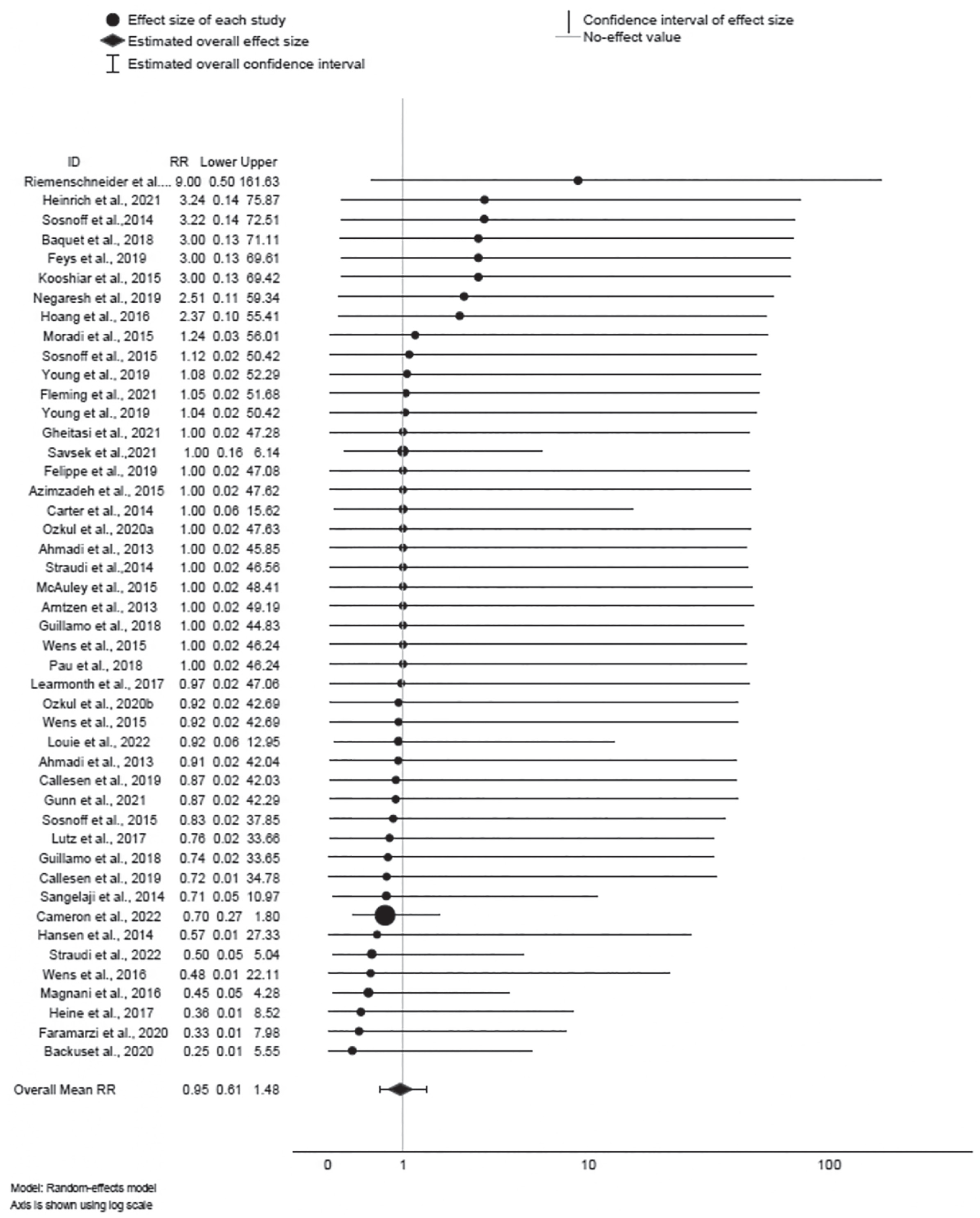
Forest plot of relapse.
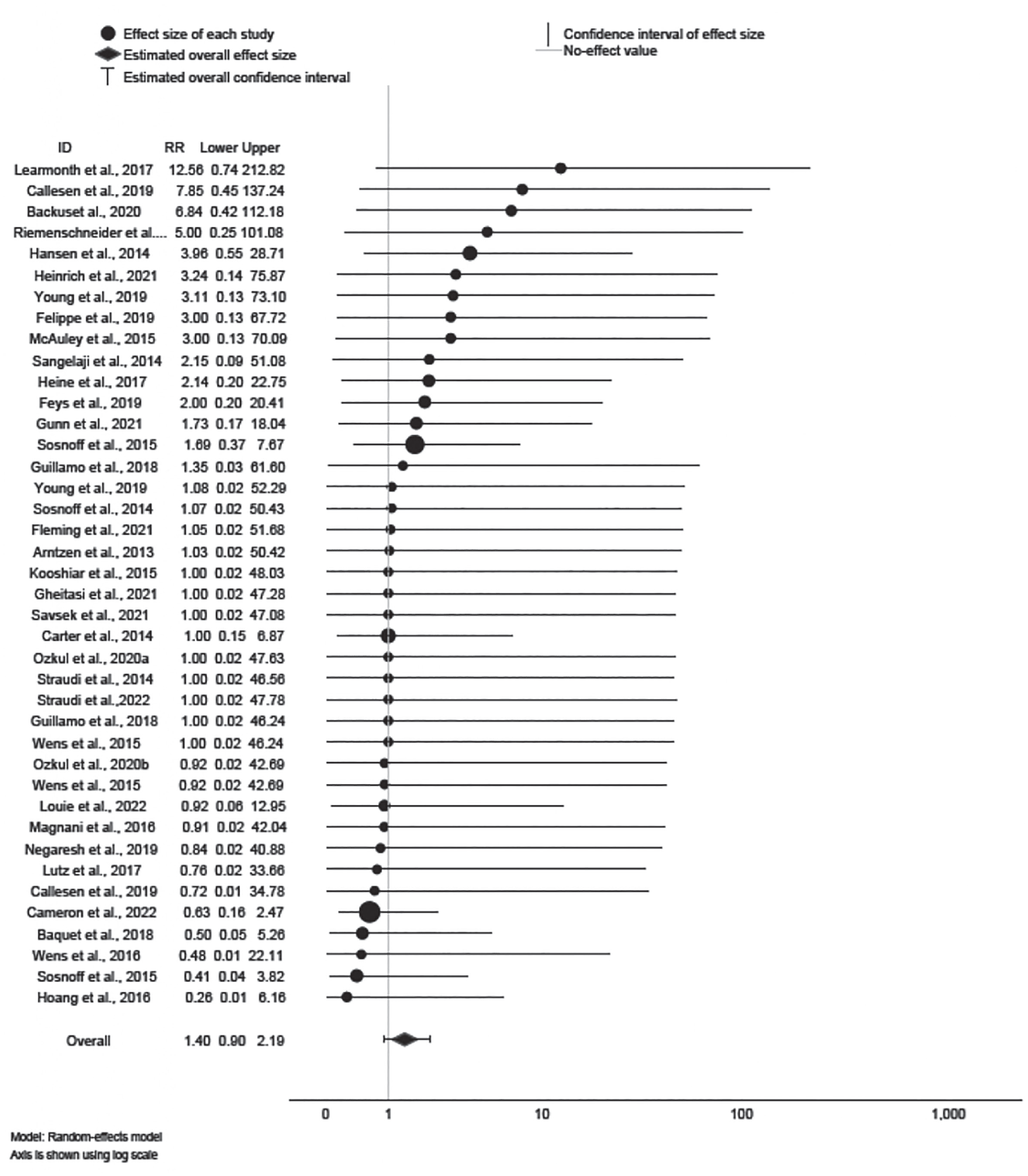
Forest plot of adverse events.
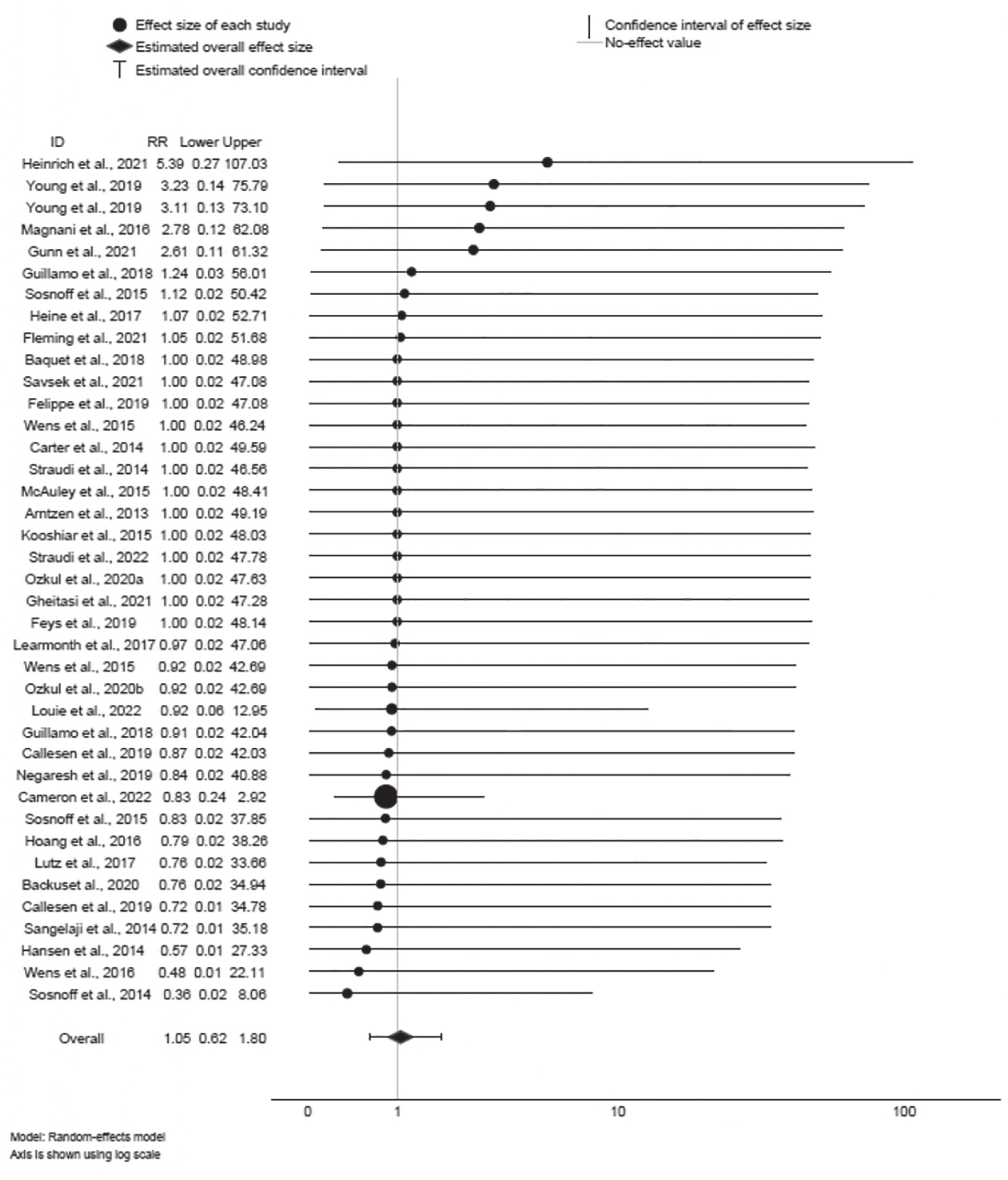
Forest plot of serious adverse events.
Of 46 effects provided for relapse, 20 RR were < 1, suggesting lower risk of relapse for exercise training; 13 RR were > 1, suggesting higher risk of relapse for exercise training. However, the 95% CIs for all 46 effects encompassed 1; therefore, statistical significance cannot be inferred. The mean effect ∆ = 0.95 was not statistically significant (95% CI: 0.61, 1.48; z = −0.22, p > 0.82); the effect was not heterogeneous (Q45 = 8.66, p = 1.00; I 2 = 0%), and sampling error accounted for 100% of observed variance.
Of the 40 effects provided for AE, 12 RR were < 1, suggesting lower risk of AE for exercise training; 19 RR were > 1, suggesting higher risk of AE for exercise training. However, the 95% CIs for all 40 effects encompassed 1; therefore, statistical significance cannot be inferred. The mean effect ∆ = 1.40 was not statistically significant (95% CI: 0.90, 2.19; z = 1.48, p > 0.14); the effect was not heterogeneous (Q39 = 13.58, p = 1.00; I 2 = 0%), and sampling error accounted for 100% of observed variance.
Of 39 effects provided for SAE, 17 RR were < 1, suggesting lower risk of SAE for exercise training; 9 RR were > 1, suggesting higher risk of SAE for exercise training. However, the 95% CIs for all 39 effects encompassed 1; therefore, statistical significance cannot be inferred. The mean effect ∆ = 1.05 was not statistically significant (95% CI: 0.62, 1.80; z = 0.19, p > 0.85); the effect was not heterogeneous (Q38 = 3.85, p = 1.00; I 2 = 0%), and sampling error accounted for 100% of observed variance.
Discussion
This study quantified the rate of relapse, AE, adverse effects and SAE in RCTs of exercise training in persons with MS published between November 2013 and April 2022; since our original review. 25 The exercise training in this review comprised studies of aerobic, strength or neuromotor exercise and included both supervised and unsupervised exercise. We identified that in studies where safety data are reported that exercise was safe for persons with MS based on the following: (1) there were no serious adverse effects reported from exercise training and (2) there were no higher risks of relapse, AEs or SAEs in participants who engaged in exercise training compared with participants in control conditions.
The results of our meta-analyses did not reveal any significant variability in risk of exercise training across the potentially important factors of exercise type, delivery style (e.g. supervised, independent or remotely supervised), participant disability level or the prescription of exercise consistent with minimal exercise guidelines for persons with MS.7,18 This finding is important and supports the current evidence-based guidelines of exercise for all persons with MS across the disease trajectory. 1 Together, the confirmed safety of exercise and available guidelines should provide confidence for clinicians, health advocacy organisations and governments interested in promoting exercise among all persons with MS.
When reviewing articles for inclusion in our study, many were excluded for not clearly reporting safety data. Based on the unclear safety reporting, our study might not capture all safety events occurring in RCTs of exercise training in MS. Our past systematic review 25 and studies providing a narrative description of relevant safety events in MS exercise studies23,24 have all advocated that researchers include transparent and consistent reporting of safety outcomes. Here, we direct the field towards clarity in the definitions and reporting of relapse, AE, adverse effects SAE and serious adverse effects and would encourage consideration of the applied definitions as described in Table 1.21,22,86 We recommend that researchers enable strategies to address the dearth of safety-related outcomes in MS research.25,87 Researchers should define these events in study protocols, report all events in study reporting, even if unanticipated or deemed unrelated to the exercise itself, and clearly report if no events occurred. 87 To facilitate this, we recommend researchers follow a plan to monitor, record and vet all relapses and AE and SAE during exercise studies. Methodological quality was high for almost all included RCTs, indicating a low risk of study bias. Our inclusion criteria for reporting on safety outcomes may be associated with studies of higher methodological quality. The identified high study quality brings confidence to our results, and we acknowledge our recommendation advocating for exercise promotion as safe is associated with exercise interventions delivered with high-quality methodology.
One limitation of our study was the lack of heterogeneity across effects that did not permit an analysis of the influence of clinical characteristics or exercise prescription characteristics. Another limitation is that the RCTs often included screening for risk factors for AE with exercise training and those presenting with elevated risk profiles were excluded from the RCTs. This might obfuscate the actual safety profile and make our results only applicable under conditions wherein participants have low risk of contraindications. Since our search was completed we are aware of only two new articles reporting study results of exercise: telehealth supervised pilates or yoga training 88 and in-person supervised strength-training, 89 respectively, in MS using a randomised controlled design and reporting on safety outcomes.89,90 Three AEs, three respiratory infections were reported in the intervention groups of the pilates or yoga-based study, with no events in the control group, while no AEs were reported in the strength-training study group. These results would not change our overall conclusions.
Conclusion
In exercise studies involving persons with MS where safety outcomes were reported, we identified that aerobic, strength or neuromotor exercise performed under both supervised and unsupervised settings was safe for persons with MS based on the following: (1) there were no serious adverse effects reported from exercise training and (2) there were no higher risks of relapse, AE or SAE in participants who took part in the exercise training interventions compared with participants who took part as control participants.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231204459 – Supplemental material for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis
Supplemental material, sj-docx-1-msj-10.1177_13524585231204459 for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis by Yvonne C Learmonth, Matthew P Herring, Daniel I Russell, Lara A Pilutti, Sandra Day, Claudia H Marck, Bryan Chan, Alexandra P Metse and Robert W Motl in Multiple Sclerosis Journal
Supplemental Material
sj-docx-2-msj-10.1177_13524585231204459 – Supplemental material for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis
Supplemental material, sj-docx-2-msj-10.1177_13524585231204459 for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis by Yvonne C Learmonth, Matthew P Herring, Daniel I Russell, Lara A Pilutti, Sandra Day, Claudia H Marck, Bryan Chan, Alexandra P Metse and Robert W Motl in Multiple Sclerosis Journal
Supplemental Material
sj-docx-3-msj-10.1177_13524585231204459 – Supplemental material for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis
Supplemental material, sj-docx-3-msj-10.1177_13524585231204459 for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis by Yvonne C Learmonth, Matthew P Herring, Daniel I Russell, Lara A Pilutti, Sandra Day, Claudia H Marck, Bryan Chan, Alexandra P Metse and Robert W Motl in Multiple Sclerosis Journal
Supplemental Material
sj-docx-4-msj-10.1177_13524585231204459 – Supplemental material for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis
Supplemental material, sj-docx-4-msj-10.1177_13524585231204459 for Safety of exercise training in multiple sclerosis: An updated systematic review and meta-analysis by Yvonne C Learmonth, Matthew P Herring, Daniel I Russell, Lara A Pilutti, Sandra Day, Claudia H Marck, Bryan Chan, Alexandra P Metse and Robert W Motl in Multiple Sclerosis Journal
Footnotes
Author contributions
Y.C.L., A.P.M., L.A.P. and R.W.M. conceived the idea and designed the study. B.C. conducted the literature search. Y.C.L., D.I.R., S.D., L.A.P., A.P.M. and C.H.M. reviewed the studies and extracted data. M.P.H. conducted the analyses. Y.C.L., A.P.M., L.A.P., M.P.H. and R.W.M. drafted the manuscript. Y.C.L., D.R., S.D., L.A.P., M.P.H., R.W.M., B.C., C.H.M. and A.P.M. reviewed the manuscript. All authors read and approved the final draft of the protocol. The authors thank Dr Brook Galna, Murdoch University for recommendations on table layouts.
Availability of supporting data
Included studies are cited.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CHM and YCL are MS Australia Post Doctoral Research Fellows.
Systematic review registration
This study was registered with PROSPERO 2020 CRD42020190544.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
