Abstract
Background:
The existence of isolated cognitive relapses (ICRs) in persons with MS (PwMS) has been debated.
Objective:
To examine relapses with decline on Symbol Digit Modalities Test (SDMT) but no change on Expanded Disability Status Scale (EDSS).
Methods:
This 3-year prospective cohort study identified PwMS experiencing a relapse with decrease on SDMT. Participants with SDMT decline/stable EDSS were labeled “ICR,” while those with a corresponding decrease on EDSS were classified “Relapse with Cognitive Decline (RCD).” Two definitions of SDMT decline were explored: (1) ⩾ 8 points, and (2) ⩾ 4 points. Logistic regression was used to analyze the relationship between ICR and RCD.
Results:
The full cohort had 592 participants: 83 experienced relapses; 22 (26.5%) had an SDMT decrease of ⩾ 8 points; 14 (63.6%) met ICR criteria. Logistic regression (X2(1) = 5.112, p = 0.024) using demographics and disease characteristics explained 28.4% of the variance in ICR versus RCD. Only the MS Neuropsychological Questionnaire was associated with ICR (odds ratio (OR): 8.6; 95% confidence interval (CI): 1.1–16.4) 40 relapsing participants with SDMT decrease of ⩾ 4 points were identified: 26 (65%) had a stable EDSS (ICR). Logistic regression did not find any variable predictive of ICR.
Conclusion:
This prospective study demonstrates evidence of ICR in PwMS.
Introduction
It is well known that cognitive impairment is common in persons with multiple sclerosis (PwMS), affecting 40%–65% of the population, 1 with information processing speed and episodic memory most commonly impacted. 2 Recently debated in the literature is the existence of isolated cognitive relapse (ICR). An ICR is defined as an acute worsening of cognitive function (cognitive decline) as a result of active disease, which occurs in the absence of physical neurological symptoms.3–5 Traditionally, a change on physical examination or a change on the Expanded Disability Status Scale (EDSS) is used to diagnose a clinical relapse as physical symptoms were considered the hallmark of relapse symptoms. Yet, the EDSS heavily weighted toward physical disability 6 and thus may not reflect other changes noted clinically. Multiple studies have demonstrated that acute cognitive change may occur during an MS relapse.7,8 Indeed, one retrospective analysis documented ⩾ 4 point decline on the Symbol Digit Modalities Test (SDMT), a clinically meaningful change, 7 in conjunction with acute disease activity on magnetic resonance imaging (MRI) in 17 of 99 purportedly “clinically stable” PwMS. 9 Supporting the notion that ICRs are merely a previously unrecognized manifestation of acute MS disease activity, evidence suggests that recovery from ICR is often incomplete and may contribute to progressive cognitive worsening in PwMS, 10 similar to the pattern observed with physical symptoms or changes on the EDSS. 11 However, to date, no prospective or multi-center studies have supported the concept of ICRs. In this study, we aimed to determine the frequency of ICRs in a longitudinal, multi-center database of relapsing PwMS, including statistically reliable decline on cognitive testing, and no documented physical worsening on EDSS, as well any other distinguishing features of an ICR.
Methods
The full methodology of this study has been described elsewhere.12,13 Briefly, this was a prospective cohort study of cognitive change during relapses from three MS centers in the United States and Canada: University at Buffalo, Cleveland Clinic, and London Health Sciences Center. PwMS (n = 592) were enrolled during a period of stability, recruited from September 2014 to August 2017. All subjects completed a baseline neurocognitive assessment and were monitored for clinical relapses or MRI disease activity (MRI and visit frequency determined by the treating physician) over the following 4.5 years, until October 2020.
Participants meeting the following criteria during the monitoring phase of the study were assigned to the relapsing group (RG): A neurologist-confirmed clinical relapse with new or recurrent neurologic symptoms not associated with fever/infection and/or new MRI brain findings of one gadolinium-enhancing lesion > 0.8 cm3 in volume and/or MRI brain findings of two or more gadolinium-enhancing lesions. 14 If the new signs or symptoms or gadolinium-enhancing lesions at the time of relapse implicated pathology in the spinal cord or optic nerve, the subject was excluded, because these areas were not expected to directly affect cognitive function and could confound cognitive assessments. As this study was using standard of care dictated by the center or treating neurologist, gadolinium was not necessarily used for all MRIs performed.
At baseline, all participants underwent a neurological assessment by a neurologist trained as an MS specialist, including the EDSS.6,15 In addition, the standard oral version of the SDMT was administered. 16 RG participants underwent repeat EDSS and SDMT at the time of relapse. An MRI was also performed at this time. Three months post-relapse, the EDSS and SDMT were conducted once again.
Participants in the RG who experienced a worsening on the SDMT were identified. Two objective definitions of acute cognitive decline were explored: (1) a decline of ⩾ 8 raw score points on the SDMT that was recently proposed to mark statistically reliable decline13,17 and (2) a decline of ⩾ 4 raw score points, which is commonly cited as a marker of clinically meaningful change due to its links to relevant anchors such as vocational status.7,8,18,19 Only these subjects were included in the next steps of the analysis. Next, participants within this smaller cohort who had stable EDSS (pre-relapse compared to during-relapse) were identified (see Figure 1). A worsening (non-stable) EDSS was defined as an increase of 1 point from a baseline EDSS of 5.5 or below, and an increase of 0.5 points from a baseline EDSS score of > 5.5 points.
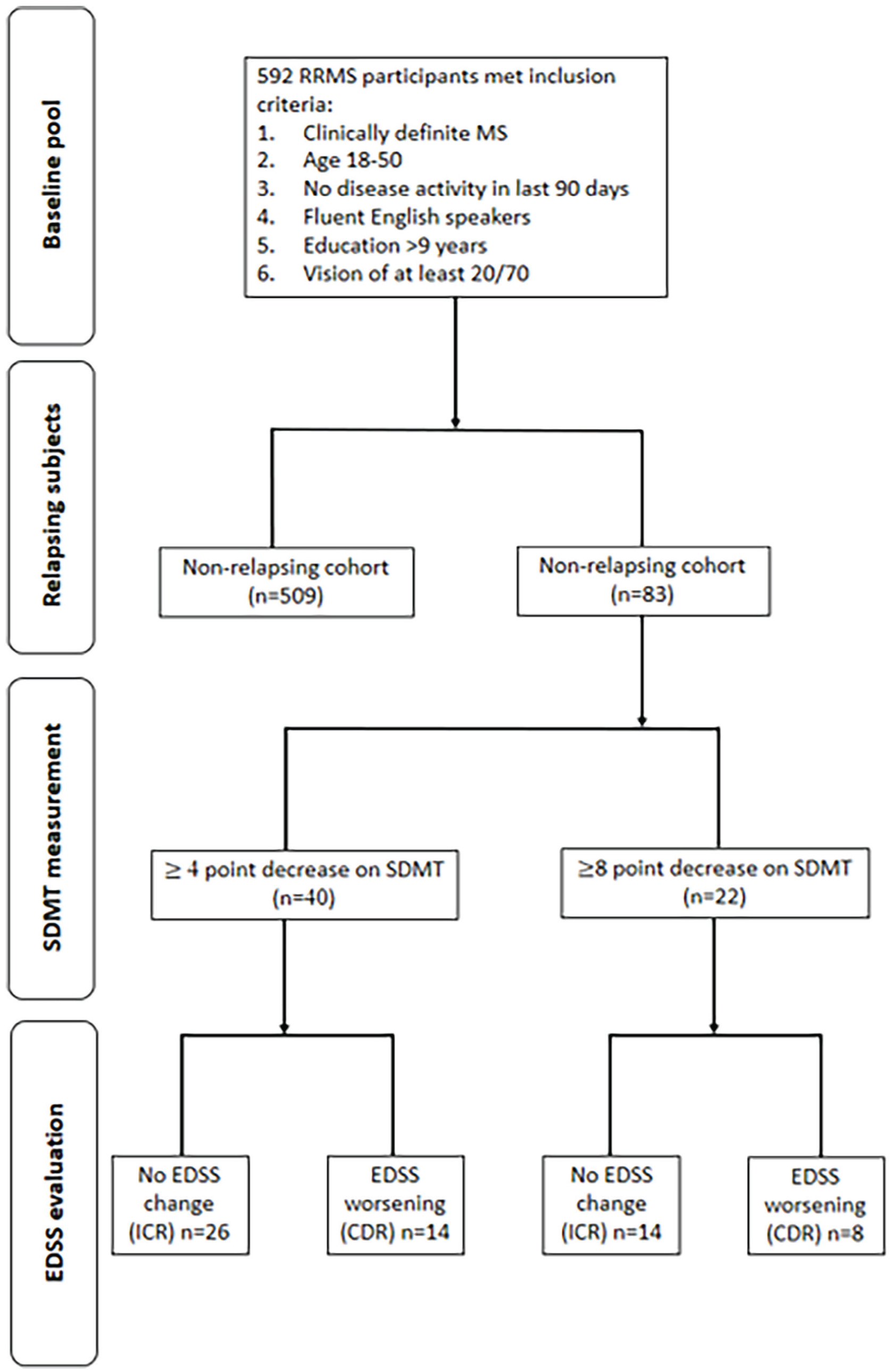
Flow chart of participant selection.
Statistical analysis
RG participants with acute cognitive decline, as defined by either a decrease on SDMT of ⩾ 8 points or ⩾ 4 points, and stable EDSS during relapse were assigned to the ICR group. Participants with acute cognitive decline and EDSS worsening were classified as “Relapse with Cognitive Decline (RCD).” Descriptive statistics were used to examine the change on SDMT in those classified as ICR versus RCD for both definitions of change. Next, the change on SDMT between these two groups was compared using paired sample Student’s t-tests. Sex at birth, baseline SDMT, age, education, FSS, and MSNQ at the time of the relapse assessment were entered in a binary logistic regression model predicting ICR or RCD status among PwMS evidencing acute cognitive decline. For the cohort in which MRI with gadolinium was used, chi-square was used to determine whether the presence of a gadolinium-positive lesion was predictive of an ICR.
Ethics approval and funding
This study was approved by the Institutional Review Board at each participating institution. Written informed consent was obtained. It was supported by a grant from the National MS Society.
Results
For full details of the study cohort, please see the study by Morrow et al. 12 Most participants (n = 509, 85.8%) did not have a relapse, but 83 individuals relapsed and entered the RG. Please see Table 1 for the demographics of this relapsing cohort.
Baseline demographics of the relapsing group (RG).
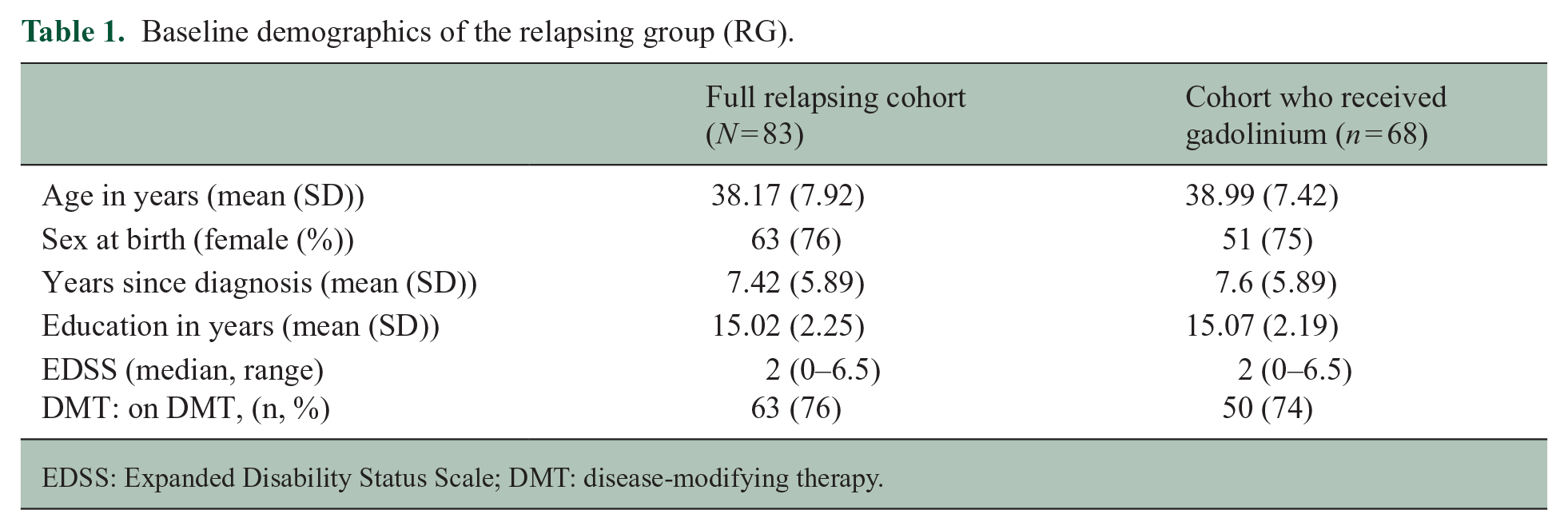
EDSS: Expanded Disability Status Scale; DMT: disease-modifying therapy.
In the RG, 22 (26.5%) participants had a decrease of ⩾ 8 points on the SDMT. Only these participants were included in the next step of the analysis. Of these 22, 14 (63.6%) had no change on their EDSS (baseline EDSS 2.0 (range 1.0–3.5) versus relapse EDSS 2.0 (range 1.0–4.0), p = 0.705) meeting criteria for an ICR. The remaining eight participants were assigned to the RCD group. There was no significant difference between these two groups on baseline demographics. In the ICR group, the SDMT worsened by 14.4 points (SD 7.7), which was not significantly different from the decrease noted in the RCD group (13.6 ± 5.1. p = 0.794; see Figure 2).
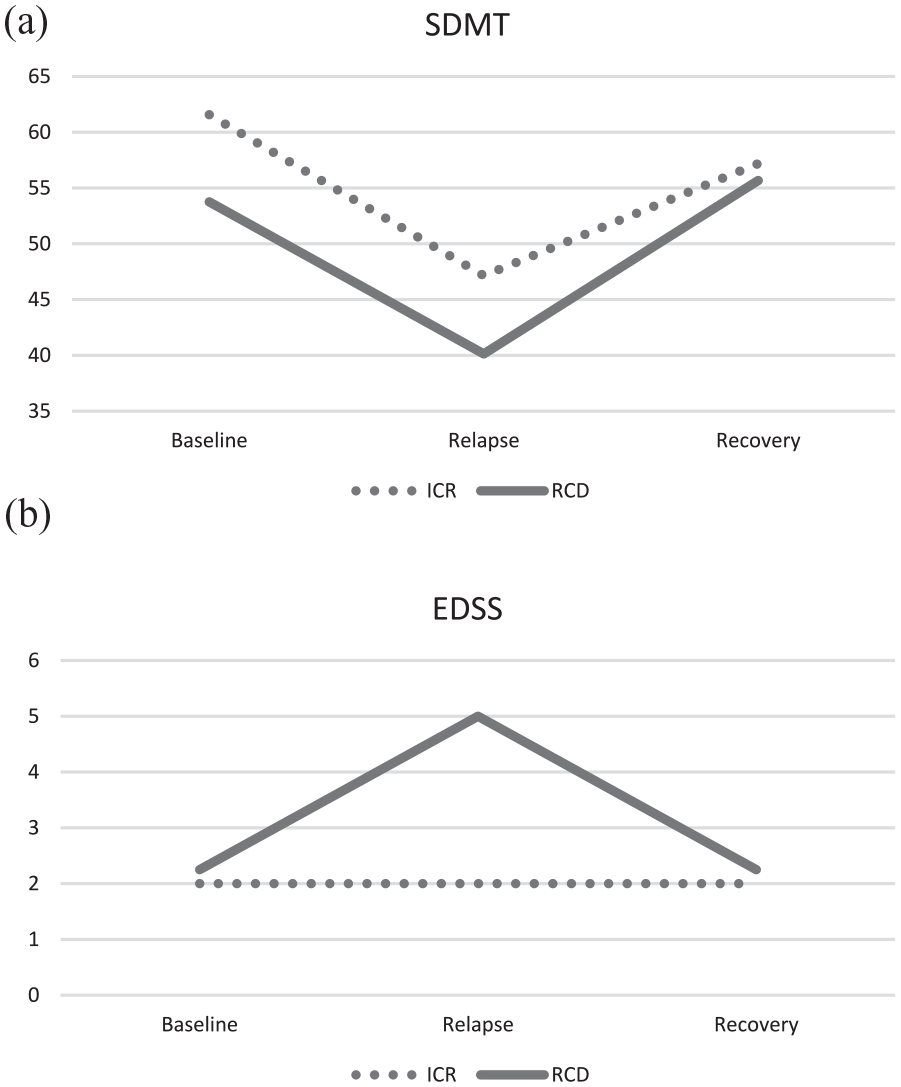
ICR versus RCD using conservative (decrease of ⩾ 8 points on the SDMT) definition. (a) SDMT scores and (b) EDSS scores.
Results of the logistic regression model predicting ICR versus RCD were statistically significant (X2(1) = 5.112, p = 0.024). The model explained 28.4% (Nagelkerke R 2 ) of the variance in ICR versus RCD and correctly classified 68.2% of cases. Of the included variables in the model, only higher MSNQ scores at the time of the relapse was associated with an ICR (odds ratio (OR): 8.6; 95% confidence interval (CI): 1.1–16.4).
Next, we examined the same RG cohort (n = 83) using the second definition of ICR (decrease of ⩾ 4 points on the SDMT). We identified 40 RG participants who met this criterion; only these participants were included in the next step of the analysis. Of these 40, 26 (65%) had a stable EDSS and were therefore assigned to the ICR group. The remaining 14 were assigned to the RCD group. In the ICR group, the SDMT worsened by 10.5 points (SD 7.3), which was not significantly different from the decrease noted in the RCD group (decrease of 9.2 ± 5.2. p = 0.560; see Figure 3). The binary logistic regression did not identify any variables predictive of ICR versus RCD.
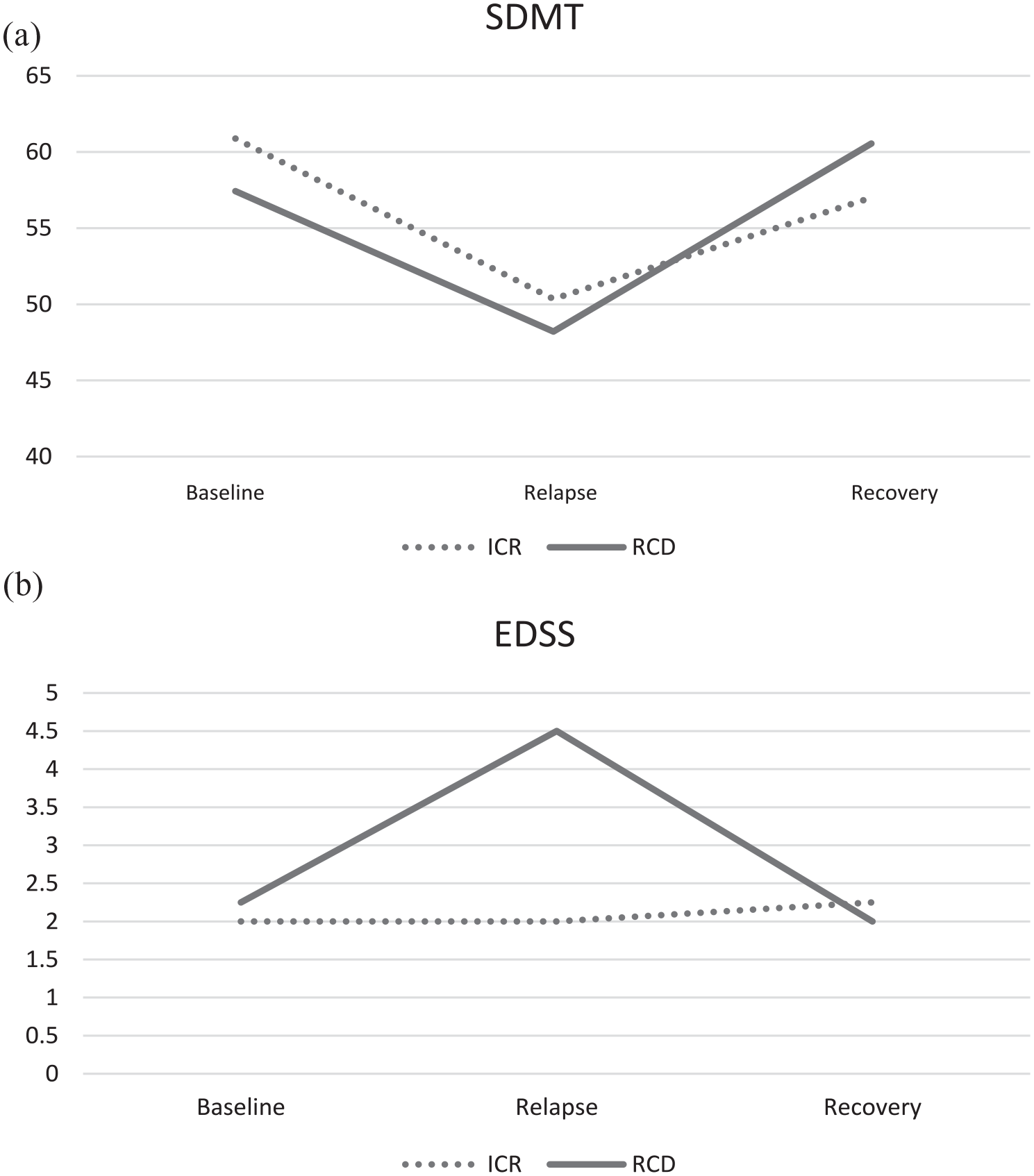
ICR versus RCD using liberal (decrease of ⩾ 4 points on the SDMT) definition: (a) SDMT scores and (b) EDSS scores.
In the second analysis, only the RG cohort that both had a relapse and gadolinium administered were included (n = 68). In this smaller cohort, 17 subjects fit the conservative definition of cognitive change (⩾ 8 point change on SDMT). Of those, 11 (64.7%) fit the definition of an ICR (EDSS stable). There was no significant difference between the ICR and RCD group in terms of presence of gadolinium enhancement (X2 (df 1, N = 17) = 0.235, p = 0.627).
Next, the same analysis was performed for the second definition of ICR (⩾ 4-point change on SDMT). In this smaller cohort, 33 (48.5%) had a clinically meaningful change on SDMT; 21 fit the definition of ICR (EDSS stable). Similarly, there was no statistically significant difference (X2 (df 1, N = 33) = 0.075, p = 0.784).
Discussion
This study provides definitive prospective evidence of the existence of ICRs, in PwMS. It is clearly demonstrated that relapses occurred in which cognitive performance was affected but the EDSS was stable, which followed the same “relapsing-remitting” pattern that is characteristic of MS relapses, in the ICR group.
Cognitive worsening or cognitive decline after relapses was first noted by Foong et al. 20 However, worsening of cognitive performance during an MS relapse was first demonstrated in a retrospective study by Morrow et al. in 2011. 7 This study, using Safety of Tysabri Re-dosing and Treatment (STRATA) data in which SDMT and MSNQ were administered monthly, demonstrated a significant decline in SDMT scores immediately following relapse onset when compared to non-relapsing matched controls. There was also partial recovery of SDMT scores over the next 3 months, similar to a “relapsing-remitting” pattern. This change on SDMT during relapses was reproduced in a prospective study by Benedict et al. 8
The relationship of gadolinium-enhancing lesions to cognitive worsening without any traditional signs/symptoms of a relapse (ICR), was also reported by Pardini et al. 21 Their retrospective study identified subjects with a gadolinium-enhancing lesion on MRI who also demonstrated at least a four-point decline on the SDMT. Similar to our study, these subjects had recovery at 6-month follow-up, although sometimes incomplete, similar to what is observed with MS relapses affecting physical function. However, these data were extracted from a longitudinal cohort study; as a result, it did not provide unmistakable evidence that what occurred was truly a “relapse” or an ICR. In contrast, our study prospectively identified relapses mainly clinically; only 4 were identified only by MRI/gadolinium-enhancing lesion, thus demonstrating that an ICR occurred.
It should be noted that SDMT worsening occurred in both ICR and RCD. Although we did note cognitive decline without a change in EDSS (ICR), cognitive decline also occurred with an EDSS change, and the degree of cognitive change was not significantly different between these two groups. Given this, our data support that both ICRs and relapses with EDSS change can affect both cognitive functioning. Conversely, it is important to note that all subjects in the RG sample, from which our smaller cohort was derived, were diagnosed with a clinical relapse. Although a small proportion (n = 4) were identified based on a new gadolinium-enhancing lesion on MRI alone, it must be presumed that in the majority of cases there were clinical features suggestive of a relapse in order to be included in this (RG) group. Thus, a limitation of our study is that we have not identified exactly how the subjects were identified clinically as experiencing a relapse.
It is interesting to note that the presence of gadolinium enhancement was not a predictor of ICR. Gadolinium-enhancing lesions are correlated with pathologic evidence of blood–brain barrier breakdown, and only appear when there is acute inflammation. Thus, gadolinium-enhancing lesions are strongly correlated with active relapses.22,23 In a study by Bellmann-Strobl et al., 24 worsening on the PASAT was associated with the presence of gadolinium-enhancing brain lesions, which improved after resolution of the enhancement. Furthermore, as noted above, Pardini reported similar findings with the SDMT. 21 In our study, it is possible that the small sample size did not provide the appropriate power to detect this association. It may also be due to the fact that identifying a specific well-circumscribed area that relates to cognitive impairment in PwMS has been difficult. Certainly, white matter lesion load has been associated with a great risk of cognitive impairment, 25 but atrophy and other more specialized MRI measures have been found to be more substantial than any specific white matter lesion location. The lack of predictive relationship may also be due to the fact that both groups did demonstrate a change in cognitive function during the relapse, and thus gadolinium enhancement is simply predictive of a clinical relapse, not necessarily the type (ICR vs RCD). It could be due to the fact that we only used one cognitive screening test (SDMT) that focuses in only one area of cognition in PwMS (information processing speed). The inclusion of other cognitive domains in this analysis may have expanded the definition of cognitive change during relapse and allowed for a more robust exploration of the relationship between gadolinium-enhancing lesions and cognitive changes during relapses.
In contrast, at least for the “statistically reliable decline” definition of ICR, the MSNQ was predictive of an ICR. The MSNQ is a self-report questionnaire designed to screen for cognitive impairment in PwMS and includes 15 statements concerning cognitive problems in daily life that has been found to be reliable and responsive with repeated testing. 26 It is scored from 0 (never, does not occur) to 4 (very often, very disruptive) on each question and a composite score is calculated. 27 Although it has been shown to correlate with cognitive impairment, it is also correlated with depressive symptoms.27,28 This finding supports that an eight-point worsening on the SDMT is not only statistically reliable, but clinically meaningful as it was noted not just objectively, but subjectively by PwMS. However, it also contradicts previous evidence that PwMS are unable to accurately report the presence of cognitive symptoms. 29 Interestingly, the original cognitive relapse study by Morrow et al. (2011) did report that there was a trend toward change in MNSQ. In that study, the MSNQ in the control group improved steadily over time, while the RG had MSNQ worsening prior to the diagnosis of the relapse, followed by a trend toward an improving score after the first visit post-relapse. In the RG in the Morrow et al. 7 (2011) study, the MSNQ score at the second visit post-relapse reached the baseline score two assessments prior to the relapse. It is possible that we, as clinicians, are simply not providing the appropriate language or tools to PwMS to accurately identify or report cognitive changes that occur in a relapsing pattern. This phenomenon requires further study.
Previous studies have supported the idea that a decrease on the SDMT of four or more points is considered a clinically meaningful change. More recent evidence supports that a change of eight or more points is a more appropriate threshold to determine a statistically reliable change at the individual level. 13 In this study, we examined both definitions of clinically meaningful change and statistically reliable change to show that the conclusions reached will not differ across these methodologies. However, it does lead to the question: how do we measure clinically meaningful change in the clinic setting when our tools are not geared toward measuring changes in cognition? The recommendation that a four-point change on the SDMT be considered clinically meaningful was based on deterioration in employment status over time, yet this study focused on group changes (mean difference in change over time between the stable employment vs the deterioration in employment group) as opposed to the individual level. 19 Furthermore, unlike other measures that are used in the clinical setting, the SDMT is considered a screening measure for cognition. Although it is well known that the SDMT is a very sensitive measure of cognition and cognitive changes in PwMS, it does not have the same weight as a marker of change as does a change in strength or ataxia, as noted on the EDSS. Similar to the EDSS, what is considered a clinically meaningful change may also be dependent on the level of disability at baseline; a change from 1.0 to 1.5 is not equivalent to a change of 6.0 to 6.5 in terms of the clinical impact this change has on the PwMS’s daily function. Furthermore, the test–retest variability may differ based on the baseline disability: a change from 1.5 to 2.0 could simply be due to a different examiner or being examined at a different time of day, whereas a change from 6.5 to 7.0 is less likely to be misinterpreted as a non-meaningful change in clinical function. Therefore, as suggested recently, relative (i.e. percent-based) or adjusted (i.e. Z-score-based) scales may me better suited to describe and recognize cognitive changes at the level of the individual. 30 Additional work is needed to determine the best means for defining clinically meaningful cognitive decline in PwMS.
Conclusion
For the first time, we provide tangible evidence of ICR in a prospective study of PwMS. Although questions remain concerning the best means for recognizing meaningful cognitive changes in individual PwMS, our data support an emerging body of literature that suggests cognition is a unique component of the MS clinical picture and is capable of progressing independently of physical dysfunction. The implications of this research are clinically relevant as they force us to consider how we define disease activity and conversely, disease stability or lack of disease activity. 31 Furthermore, if isolated acute cognitive decline is indeed considered sufficient evidence of disease activity, future research will need to determine what sort of interventions (e.g. medicinal or rehabilitative) are indicated. We believe that a heightened sensitivity to acute cognitive changes—regardless of physical worsening—will enhance patient assessment and may provide additional avenues for personalized care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SAM has, in the past 3 years, served on advisory boards for Biogen Idec, EMD Serono, Genzyme Canada, Novartis, Roche; has received Investigator Initiated Grant Funds from Biogen Idec, Novartis, Roche; has acted as site principal investigator (PI) for multi-center trials funded by Novartis, Genzyme, Roche, and AbbVie. DC has received research support paid to his institution by Novartis Pharmaceuticals and the National MS Society. He has received consulting fees from Novartis Pharmaceuticals and speaking fees from Biogen. BW-G has participated in speaker’s bureaus and/or served as a consultant for Biogen, EMD Serono, Novartis, Genentech, Celgene/Bristol Meyers Squibb, Abbvie, Sanofi Genzyme, and Mallinckrodt. She has also received grant/research support from the agencies listed in the previous sentence. She serves in the editorial board for BMJ Neurology, Children, CNS Drugs, MS International and Frontiers Epidemiology. MGD has received consultant fees from Claret Medical and EMD Serono and research grant support from Novartis. RZ has received personal compensation from Bristol Myers Squibb, EMD Serono, Sanofi, and Novartis for speaking and consultant fees. He received financial support for research activities from Sanofi, Novartis, Bristol Myers Squibb, Mapi Pharma, Keystone Heart, Protembis, and V-WAVE Medical. RHBB received honoraria, speaking or consulting fees from Biogen, Celgene, EMD Serono, Genentech, Medday, Novartis, and Roche, and has received research support from Biogen, Genentech, and Novartis. He has received royalties from Psychological Assessment resources, Inc. ZLW, OM, TF, MGJ, and SE have no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the National MS Society for providing funding for this study (NMSS RG 5195A4/1). The NMSS did not participate in the design and conduct of the study; did not have access to the data; and did not assist in manuscript preparation or review.
