Abstract
Background:
Mumps-Measles-Rubella (MMR) and Varicella zoster vaccines (VAR) are live attenuated vaccines, usually administered in a two-dose scheme at least 4 weeks apart. However, single-dose immunization schemes may also be effective and can reduce delays in immunosuppressive treatment initiation in patients with multiple sclerosis (pwMS) who need to be immunized.
Objectives:
To evaluate the immunogenicity of a single-dose attempt (SDA) versus the standard immunization scheme (SIS) with VAR and/or MMR in pwMS.
Methods:
Retrospective observational study in pwMS vaccinated against VAR and/or MMR. We compared seroprotection rates and antibody geometric mean titers (GMTs) between the two strategies.
Results:
Ninety-six patients were included. Thirty-one patients received VAR and 67 MMR. In the SDA group, the seroprotection rate was 66.7% (95% confidence interval (CI): 53.3–78.3) versus 97.2% (95% CI: 85.5–99.9) in the SIS (p < 0.001). For the seroprotected patients, GMTs were similar for both schemes.
Conclusion:
An SDA of VAR and/or MMR vaccines could be sufficient to protect almost two-thirds of patients. Testing immunogenicity after a single dose of VZ and/or MMR could be included in routine clinical practice to achieve rapid immunization.
Introduction
People with multiple sclerosis (pwMS) exposed to highly effective disease-modifying therapies (DMTs) are at higher risk of infections and complications, some of which are preventable through vaccination, such as varicella and/or measles.1,2 Live attenuated vaccines, such as varicella (VAR) and measles-mumps-rubella (MMR), are contraindicated in patients receiving immunosuppressive therapies. Current guidelines recommend confirmation of the immune status against varicella and measles, and if susceptible, immunization must be completed at least 4 weeks before treatment onset.3,4
Given that the standard immunization scheme (SIS) for VAR and MMR vaccines consists of two doses at least 4 weeks apart, patients could face a delay in their treatment initiation of over 2 months. However, a single-dose immunization scheme may also be effective, 5 and could reduce delays in immunosuppressive treatment initiation, especially in highly active MS patients.
This study aimed to evaluate the immunogenicity of a single-dose attempt (SDA) versus the two-dose SIS of the VAR and MMR vaccines in pwMS.
Materials and methods
This study is based on two ongoing open, prospective cohorts that started in 1995: the CIS inception cohort 6 and the Treatment cohort 7 at the Multiple Sclerosis Centre of Catalonia (Cemcat), Vall d’Hebron University Hospital (VHUH), Barcelona. The CIS cohort includes patients aged < 50 years who exhibited a clinically isolated syndrome (CIS) within 3 months of the first clinical evaluation. The Treatment cohort includes patients followed since treatment onset receiving any DMT. Clinical, demographic, radiologic, and biologic data within both cohorts are prospectively collected using prespecified protocols. Patients are seen regularly for clinical evaluation, which includes Expanded Disability Status Scale (EDSS) evaluation and relapses. These two cohorts have been recently merged into a large Cemcat cohort. 8
Since 2015, patients from the Cemcat cohort have been routinely referred to the Preventive Medicine Department at the VHUH for their evaluation and immunization as part of the risk minimization strategy. Data about vaccination (type of vaccine received, dose, and date) and serological information (pre and post-vaccination) were prospectively collected.
For this study, we included consecutive measles and/or VAR seronegative MS patients according to the baseline evaluation, who had received at least one dose of VAR (lyophilized, live attenuated varicella virus Oka strain Varivax (MSD) or Varilrix (GSK)) and/or MMR (Priorix (GSK)) vaccines between July 2016 and April 2022 and with an available post-vaccination serology (performed 4–6 weeks after the last dose). Patients who received MMR due to seronegative rubella and/or mumps were excluded. Patients were divided according to the vaccination scheme received (assigned under physicians’ criteria based on disease activity and the urge to start treatment; see Figure 1). The study outcomes were: seroprotection rates (SRs; proportion of patients with protective titers based on the accepted cut-off levels: over 165 mIU/mL for varicella and 16.5 AU/mL for measles) and the IgG antibody geometric mean titers (GMTs) for varicella and/or measles in post-vaccination serology. Geometric mean is preferred over the arithmetic mean when the data are log-normally distributed. The serology was performed in the Liaison XL® instrument (DiaSorin, Italy) and analysed with two specific chemiluminescence immunoassays (CLIA): LIAISON® VZV IgG (DiaSorin, Italy) and LIAISON® Measles IgG (DiaSorin, Italy) for the determination of the IgG anti-varicella virus and IgG anti-measles virus, respectively. Among these patients who did not achieve seroprotective titers after SDA, an additional dose was administered 4 weeks after the initial dose.
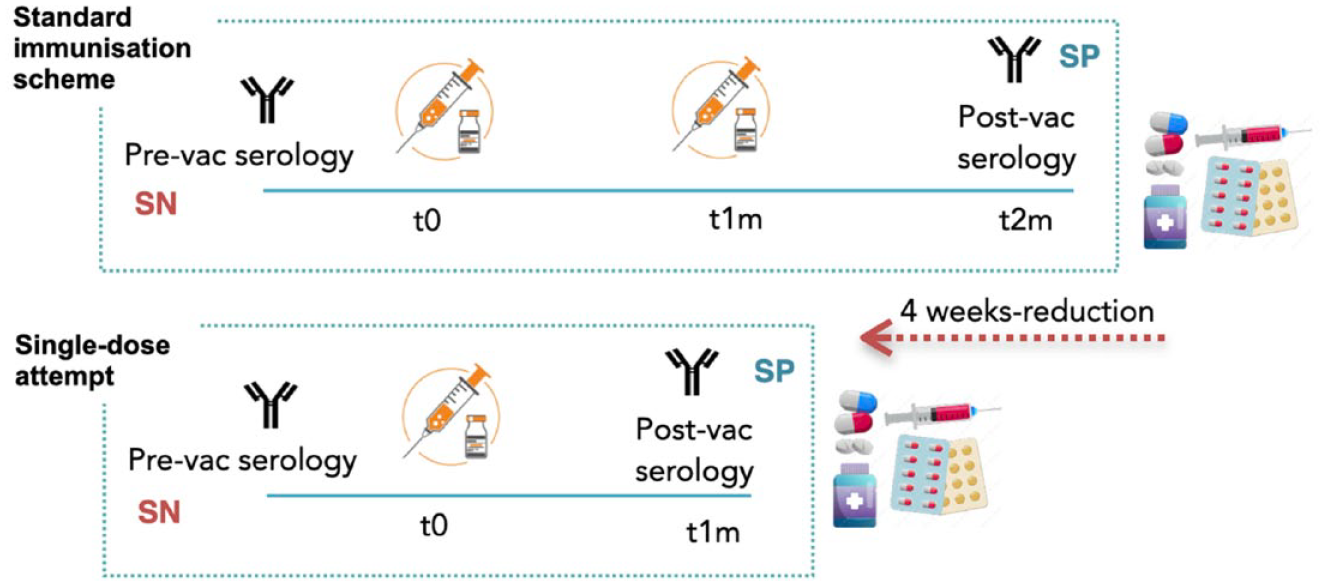
Standard immunization schedule versus single-dose attempt.
The study was approved by the Clinical Research Ethics Committee at Vall d’Hebron University Hospital (EPA (AG) PR(AG)431/2019). All patients signed a written informed consent according to the Declaration of Helsinki.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Statistical analysis
We used parametric or non-parametric tests to compare clinical and demographic characteristics between groups. Seroprotection rates, baseline and post-vaccination GMTs, and their corresponding 95% confidence interval (CI) were determined for each vaccine and compared according to the immunization scheme. Statistical analyses were performed with R Core team, version 4.2.2 (R Foundation Statistical Computing).
Results
A total of 96 patients were included. Sixty-eight (70.8%) were women, with a mean age of 36.7 years (SD 8.9) and a mean disease duration of 6.8 (SD 7.3) years. Nineteen patients (19.7%) were receiving DMTs at the time of vaccination: interferon (N = 10), glatiramer acetate (n = 6), teriflunomide (n = 1), and dimethyl fumarate (n = 2). Thirty-one patients received VAR and 67 MMR. An SDA was done in 60 (62.5%) patients, and 36 (37.5%) received SIS. No differences were observed with respect to the type of vaccine or the demographic and clinical characteristics, except for a higher annualized relapse rate (ARR) in the SDA group (ARR 0.84 (SD 0.8) versus 0.52 (SD 0.6); p = 0.002). In addition, the number of patients receiving high-dose steroids within 30 days prior of the vaccination was higher in the SDA group (7 (11.6%) versus 1 (2.8%); p = 0.05; see Table 1).
Demographical and clinical characteristics at the time of vaccination of measles and/or VAR seronegative MS patients.
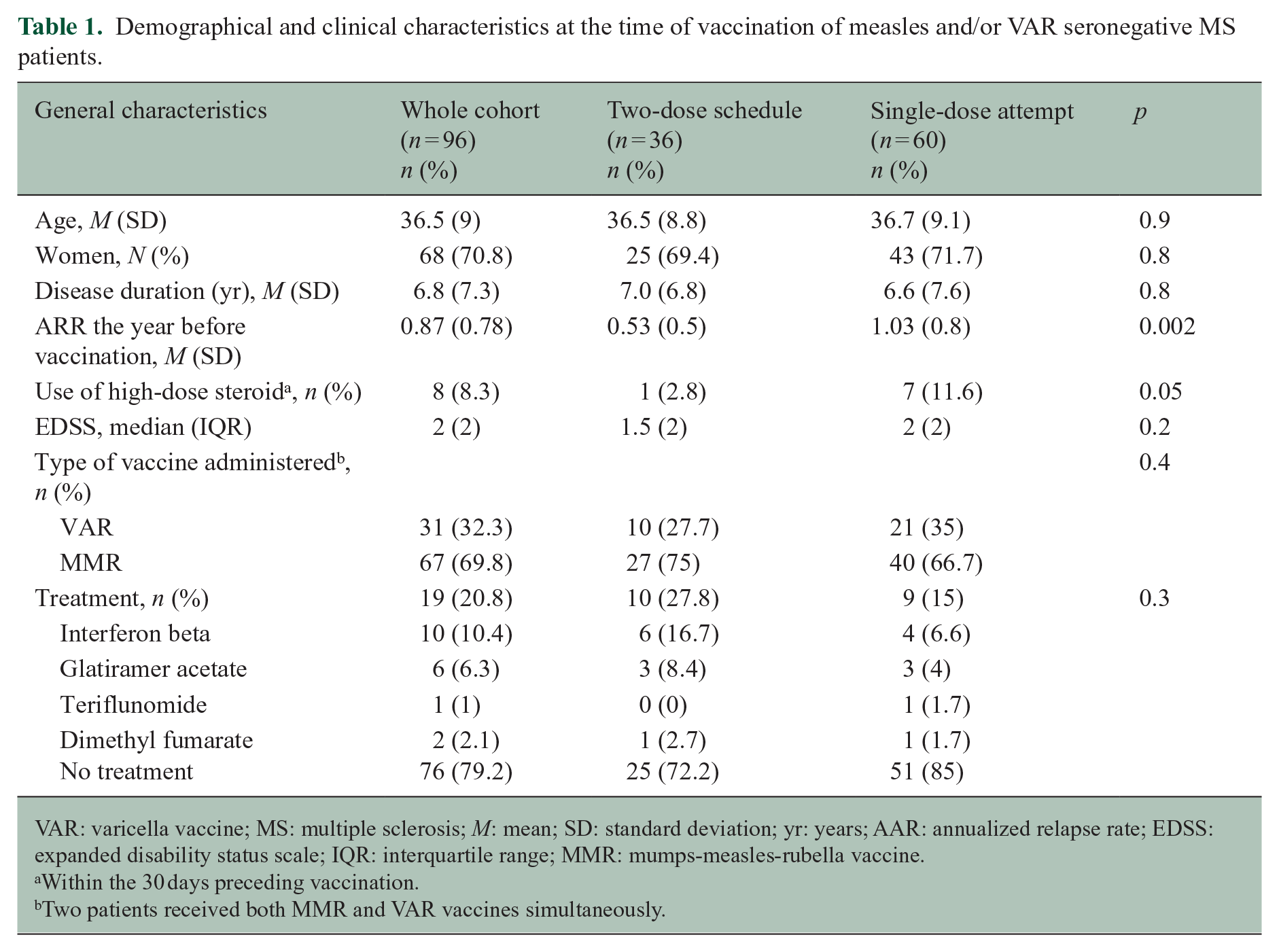
VAR: varicella vaccine; MS: multiple sclerosis; M: mean; SD: standard deviation; yr: years; AAR: annualized relapse rate; EDSS: expanded disability status scale; IQR: interquartile range; MMR: mumps-measles-rubella vaccine.
Within the 30 days preceding vaccination.
Two patients received both MMR and VAR vaccines simultaneously.
In the SDA group (n = 60), 40 patients had protective antibodies, resulting in a global seroprotection rate of 66.7% (95% CI: 53.3–78.3) as compared to 97.2% (95% CI: 85.5–99.9) in the SIS scheme (n = 38), with no differences between treated and untreated patients (66.7% vs 64.7; respectively; p = 0.9). For those immunized against measles (n = 67), the seroprotection was 70% (95% CI: 53.4–83.4) in the SDA group as compared to 96.3 (95% CI: 81–99.9) in the SIS group (p 0.04). For VZ (n = 31), the seroprotection was 57.2% (95% CI: 34–78.2) in the SDA group and 100% in the SIS group (p < 0.001). In the seroprotected patients, GMTs were similar for both schemes (187.1 vs 196.9 AU/mL; p = 0.7 for measles and 1455.7 vs 1465.6 mUI/mL; p = 0.9 for VZ; see Figure 2). An additional dose was administered in those 20 patients not seroprotected after the SDA resulting in a 95% (95% CI: 86.1–99) seroprotection rate.
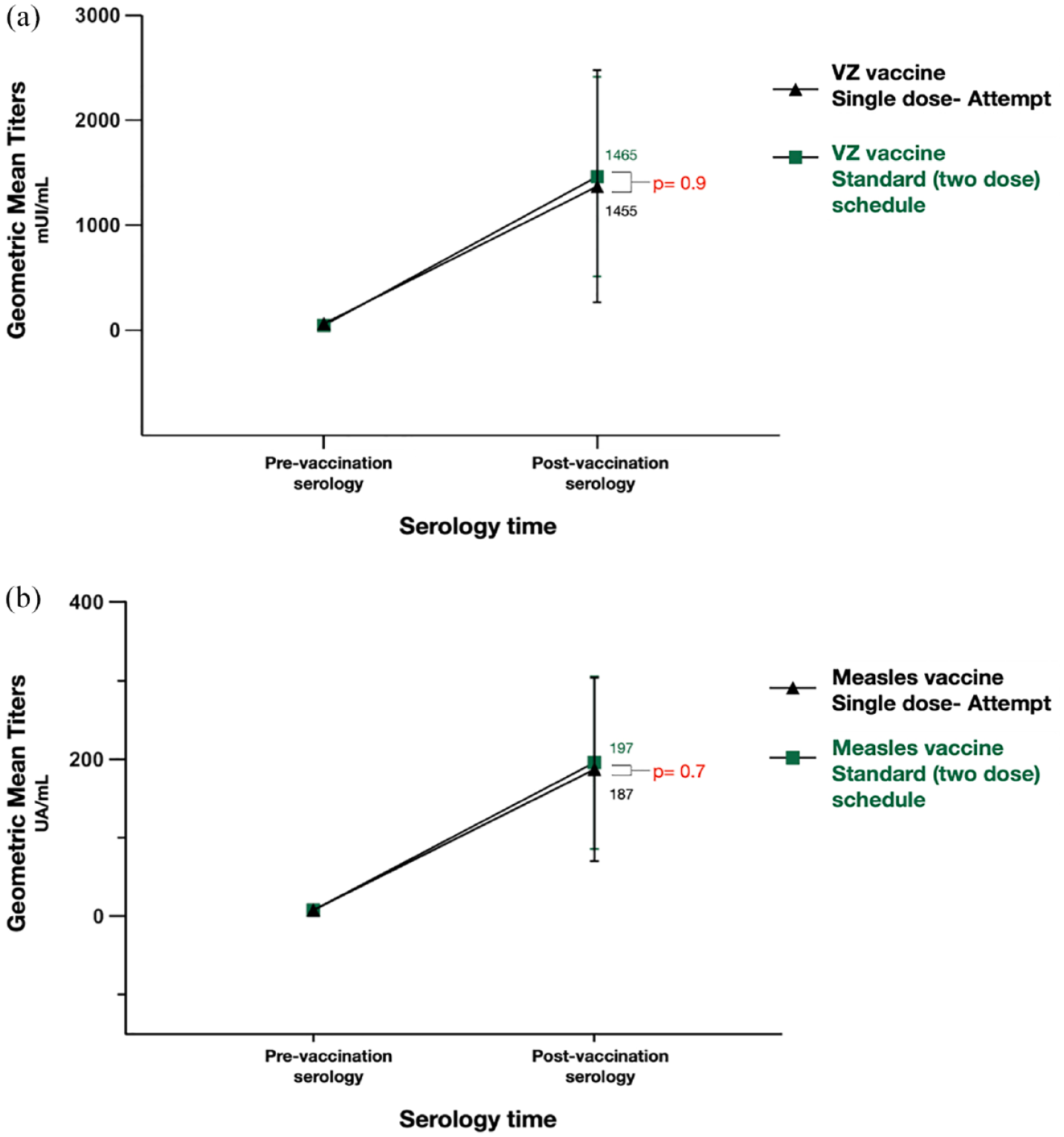
Comparison between geometric mean titers of VZ (a) and measles (b) in sero-responsive patients between single-dose attempt and standard (two-dose) schedule.
Discussion
To our knowledge, this is the first available study reporting data on the immunogenicity of VAR and MMR vaccines in pwMS. Our study shows that a single-dose attempt for immunization with VAR and/or MMR vaccines could be sufficient to protect almost two-thirds of patients. This strategy may reduce the time to treatment initiation by at least 1 month in susceptible (seronegative) MS patients. This is particularly relevant in highly active MS patients in need of a prompt treatment initiation that has been shown to reduce disease progression. 9 Ensuring a correct immunization against vaccine-preventable infections, such as varicella and measles, is highly recommended and specifically required for some DMTs,3,4 and will not be possible once immunosuppressive therapy has started.
Our study evaluated the vaccine immunogenicity by using the accepted cut-off level of IgG specific antibody responses for VAR (over 165 mIU/mL) and for measles (over 16.5 AU/mL).10–12 Both varicella and measles antibody responses have proven to be a good surrogate of protection, inversely correlated with the risk of breakthrough disease.10–12 Although the incidence of measles infection has been reduced significantly after the introduction of the systematic immunization with MMR vaccine in children in many countries after 1974, 13 the number of reported cases worldwide has increased by 79% in the first two months of 2022 compared to the same time the previous year. 14 In addition, the level of IgG measles antibodies measured in young adults belonging to the MMR-vaccinated population in childhood is below the protection threshold in nearly half of the cases. 15 On the contrary, varicella infection can be a problem in immunocompromised patients, such as pwMS treated with immunosuppressive DMTs, for which severe varicella infections have been described. 16 Thus, ensuring adequate immunization against VZ and measles before starting an immunosuppressive therapy is mandatory and recommended in current guidelines.3,4
With respect to the potential effectiveness of a single dose of live attenuated vaccine, previous studies performed in non-MS patients have shown that one dose of VAR provided a moderate protection (82%–85%) against varicella of any severity and a high protection (100%) against severe varicella. 17 In the same line, one dose of MMR vaccine was 95% effective against measles in a healthy children population. 18 This could be explained by the high immunological capacity of live attenuated vaccines that mimic natural infection with resulting antibody levels after a single dose that can provide lifelong immunity. 19
Our data show a global seroprotection rate of 70% against measles after a single dose of MMR, similar to those reported in the literature. 20 In Catalonia, the measles vaccination was introduced in 1981 as part of the immunization schedule for children but this information is only available in the electronic healthcare records starting from 2003. Hence, we assumed that most of the MMR vaccinated patients younger than 40 years at the moment of this study (N = 47) have probably received at least one dose of measles vaccine. The reported coverage rates exceed 80% in 1986 and has remained at 95% since 1999. 21 In this regard, the single dose administered could be considered as a booster dose. Information about previous measles infection was not systematically collected in the study. Epidemiological data in Spain indicates a significant decrease in virus circulation after the vaccine’s introduction in 1981, with reported cases reaching 10 per 100,000 inhabitants by 1996. 22 For those born before 1981, there is a higher likelihood of having a background of natural infection, which we captured through the baseline serological test, given the long-term persistence of measles antibodies after infection. 20
The global seroprotection after SDA for VVZ (50%) was lower than that reported in previous studies. 23 Systematic immunization in childhood was not introduced in Catalonia until 2016 at the age of 15 months. 24 A selective strategy for those without a history of natural infection at age 12 was implemented in 2005. Consequently, only 6.5% of the patients who received VAR in the study had received VAR in the past, and therefore, the dose administered during the study was, in most cases, part of a primo-vaccination scheme. It is also worth noting that commercially available serologic IgG tests might not be sufficiently sensitive to detect low levels of antibody against VZ following vaccination, 25 thus underestimating the seroprotection of the single dose of varicella.
For those 20 patients in which an SDA was not successful, a second dose was recommended and it was protective in all but 3 patients. These susceptible patients should be advised to seek medical care for post-exposure prophylaxis with immunoglobulins in case of a risk contact.3,4
The persistence of protective antibody titers over time in MS patients remain unknown. Previous work in the general population evidence a varicella antibody persistence rate close to 100% throughout a 9-year follow-up after single dose of VAR. 26 Less is known about the measles antibody titers in the long term after a single dose, especially in adults, and its maintenance over time in pwMS receiving DMT is still unknown.
An important aspect to highlight is the use of high-dose steroids’ close proximity to vaccination. Current guidelines recommend postponing vaccination with live attenuated vaccines for 1 month after a short-term pulse of high-dose steroid treatment.3,4 However, in our real-world cohort, we observed that eight patients received high-dose steroids within 30 days prior to vaccination, particularly in the SDA group, due to highly active disease and the urgency for initiating disease-modifying treatment. Despite this, no reactivation of live-attenuated virus or related complications were observed. In terms of immune response, out of these eight patients, five achieved good seroprotective antibody levels, while three of them (all in the single-attempt group) did not.
Our study has some limitations. First, the retrospective nature of the design could entail some differences between the SDA versus SIS groups (indication bias), being the patients included in the SDA more active and with a more aggressive disease. However, all the clinical information has been recorded prospectively under a prespecified protocol with high-quality data, and none of these differences have a priori the potential to modify the post-vaccination immune response. Another consideration refers to the limited sample size that could compromise the precision of the estimates in the sub-group analysis. This is a first exploratory study and further data are needed to confirm our findings. Finally, the description of safety-related adverse events, a critical aspect when considering live attenuated vaccines, was beyond the scope of this study and warrants further research.
The results of our work support that the single-dose immunization with VAR/MMR vaccines could be sufficient to protect almost two-thirds of susceptible pwMS. In this regard, we advocate that a serological confirmation after a single dose of these vaccines to confirm protection should be implemented in clinical practice, particularly in highly active pwMS requiring prompt DMT initiation.
Footnotes
Acknowledgements
Acknowledge the ECTRIMS Clinical Training Fellowship Programme for its support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R. Carvajal is currently being funded by ‘Vall d’Hebron Institut de Recerca’ grand, this project was supported by ECTRIMS Fellowship training performed during 2021–2022, he has also received speaking honoraria and personal compensation for participating on Advisory Boards and from Roche, Novartis, BIIB-Colombia, Merck, and Sanofi. C. Tur is currently being funded by a Junior Leader La Caixa Fellowship (fellowship code is LCF/BQ/PI20/11760008), awarded by ‘la Caixa’ Foundation (ID 100010434), she has also received the 2021 Merck’s Award for the Investigation in MS, awarded by Fundación Merck Salud (Spain) and a grant awarded by the Instituto de Salud Carlos III (ISCIII), Ministerio de Ciencia e Innovación de España (PI21/01860); in 2015, she received an ECTRIMS Post-doctoral Research Fellowship and has received funding from the UK MS Society, she is a member of the Editorial Board of Neurology and Multiple Sclerosis Journal, she has also received honoraria from Roche and Novartis and is a steering committee member of the O’HAND trial and of the Consensus group. X. Martínez-Gómez has received research support fees from GlaxoSmithKline, Sanofi Pasteur MSD, Statens Serum Institut & Janssen Vaccine, as well as travel expenses fees from GlaxoSmithKline and Sanofi Pasteur MS. L. Bollo is supported by a 1-year stipend endowed by the NMSS/AAN John Dystel Prize for Multiple Sclerosis Research awarded to Prof. Xavier Montalban in 2022. J. Esperalba reports no disclosures. M. Rodríguez reports no disclosures. A. Pappolla has received funding travel from Roche and speaking honoraria from Novartis. He developed this project during a 2021 ECTRIMS Clinical Training Fellowship programme, and is currently performing an MSIF-ARSEP Fellowship programme. A. Cobo-Calvo has received a grant from Instituto de Salud Carlos III, Spain; JR19/00007. P. Carbonell yearly salary is supported by a grant from Biogen to Fundació privada Cemcat for statistical analysis. B. Borras-Bermejo has received travel expenses for scientific meetings from GlaxoSmithKline J. Rio has received speaking honoraria and personal compensation for participating on Advisory Boards from Biogen-Idec, Genzyme, Janssen, Merck-Serono, Novartis, Teva, Roche, and Sanofi-Aventis. J. Castilló reports no disclosures. N. Braga has received travel expenses for scientific meetings and speaking honoraria from Roche, Novartis, Biogen, Merck and was funded by ECTRIMS Fellowship in 2022–2023. N. Mongay-Ochoa has a predoctoral grant Rio Hortega, from the Instituto de Salud Carlos III (CM21/00018), she also has received speaking honoraria and travel expenses from Merck and Roche. J.A. Rodigo-Pendás has received research support fees from GlaxoSmithKline, Sanofi Pasteur MSD, Statens Serum Institut, Janssen Vaccines & Prevention B.V. and Spanish Clinical Research Network—SCReN and travel expenses fees from Sanofi Pasteur MSD. A. Vidal-Jordana has engaged in consulting and/or participated as speaker in events organized by Roche, Novartis, Merck, and Sanofi. G. Arrambide has received speaking honoraria and consulting services or participation in advisory boards from Sanofi, Merck, Roche, and Horizon Therapeutics; travel expenses for scientific meetings from Novartis, Roche, and ECTRIMS. I. Galán reports no disclosures. B. Rodríguez-Acevedo has received speaking honoraria from Merck and honoraria for consulting services from Novartis. A. Zabalza has a predoctoral grant Rio Hortega, from the Instituto de Salud Carlos III, Spain (CM22/00237), received travel expenses for scientific meetings from Biogen-Idec, Merck Serono and Novartis, speaking honoraria from Eisai and a study grant from Novartis. L. Midaglia reports no disclosures. M. Comabella has received compensation for consulting services and speaking honoraria from Bayer Schering Pharma, Merck Serono, Biogen-Idec, Teva Pharmaceuticals, Sanofi-Aventis, Genzyme, and Novartis. J. Sastre-Garriga serves as co-Editor for Europe on the editorial board of Multiple Sclerosis Journal and as Editor-in-Chief in Revista de Neurología, receives research support from Fondo de Investigaciones Sanitarias (19/950) and has served as a consultant/speaker for Biogen, Celgene/Bristol Meyers Squibb, Sanofi, Novartis and Merck. X. Montalbán has received speaking honoraria and travel expenses for participation in scientific meetings, has been a steering committee member of clinical trials or participated in advisory boards of clinical trials in the past years with Abbvie, Actelion, Alexion, Biogen, Bristol-Myers Squibb/Celgene, EMD Serono, Genzyme, Hoffmann-La Roche, Immunic, Janssen Pharmaceuticals, Medday, Merck, Mylan, Nervgen, Novartis, Sandoz, Sanofi-Genzyme, Teva Pharmaceutical, TG Therapeutics, Excemed, MSIF, and NMSS. M. Tintore has received compensation for consulting services, speaking honoraria and research support from Almirall, Bayer Schering Pharma, Biogen-Idec, Genzyme, Janssen, Merck-Serono, Novartis, Roche, Sanofi-Aventis, Viela Bio and Teva Pharmaceuticals. Data Safety Monitoring Board for Parexel and UCB Biopharma. S. Otero-Romero has received speaking and consulting honoraria from Genzyme, Biogen-Idec, Novartis, Roche, Excemed, and MSD; as well as research support from Novartis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been funded by Instituto de Salud Carlos III (ISCIII) through the project PI19/01606 and co-funded by the European Union and ECTRIMS clinical fellowship awarded to René Carvajal from 2021 to 2022.
