Abstract
Background:
Ofatumumab has demonstrated superior efficacy and favorable safety for up to 2.5 years versus teriflunomide in relapsing multiple sclerosis (RMS).
Objective:
Further characterize efficacy and safety of ofatumumab in RMS.
Methods:
Efficacy set: patients randomized to ofatumumab/teriflunomide in ASCLEPIOS I/II (core). Safety set: patients who received ⩾ 1 dose of ofatumumab in ASCLEPIOS I/II, APLIOS, APOLITOS (all core), or ALITHIOS (umbrella open-label extension). Patients received continuous ofatumumab or were newly switched from teriflunomide. Data cut-off: 25 September 2021.
Results:
In the efficacy set (n = 1882), the continuous ofatumumab group had a low annualized relapse rate (ARR 0.05 (95% confidence interval: 0.04–0.07)), low numbers of gadolinium-enhancing (Gd+) T1 lesions (0.01 lesions/scan) and fewer new/enlarging T2 lesions (annualized rate 0.08). Overall, 78.8% met three-parameter “no evidence of disease activity” criteria through 4 years. Switching from teriflunomide led to reduced ARR, risk of confirmed disability worsening (CDW), new/enlarging T2 lesions, Gd+ T1 lesions, and serum neurofilament light chain. In the continuous and newly switched ofatumumab groups, cumulative 3- and 6-month CDW rates remained low. In the safety set (n = 1969), the most frequently reported adverse events were infections and infestations (58.35%). No new safety signals were identified.
Conclusion:
Ofatumumab has a favorable longer-term benefit–risk profile in RMS.
Trial registry:
ALITHIOS (NCT03650114): https://clinicaltrials.gov/ct2/show/NCT03650114
Introduction
Ofatumumab is a fully human, anti-CD20 monoclonal antibody (mAb) approved for relapsing multiple sclerosis (RMS) in many countries.1,2 The approved regimen with three weekly and then monthly (Q4W) subcutaneous (s.c.) ofatumumab 20 mg produces a rapid, sustained B-cell depletion, with minimal repletion between doses. 3
Ofatumumab approval was based on the ASCLEPIOS I (NCT02792218) and ASCLEPIOS II (NCT02792231) Phase 3 studies, which demonstrated superior efficacy versus teriflunomide and a favorable benefit–risk profile up to 2.5 years.4,5 Ofatumumab achieved the primary endpoint, a reduction in annualized relapse rate (ARR); 51% in ASCLEPIOS I and 58% in ASCLEPIOS II (both p < 0.001), and demonstrated greater efficacy versus teriflunomide for most secondary clinical and magnetic resonance imaging (MRI) outcomes. 4
To further assess the benefit–risk profile of ofatumumab in RMS and its longer-term tolerability, patients from ofatumumab studies (ASCLEPIOS I/II, 4 APLIOS, 6 and APOLITOS) 7 transitioned to ALITHIOS (NCT03650114), a Phase 3b, open-label, extension, where they continued with ofatumumab or switched from placebo/teriflunomide to ofatumumab. After 3.5 years of follow-up in ALITHIOS, ofatumumab was well tolerated with no new safety signals; rates of adverse events (AEs) and serious AEs (SAEs) were consistent with previous findings. 8
Emerging data indicate that early initiation of high-efficacy therapies for RMS improves longer-term outcomes compared with delayed initiation of, or escalation from lower efficacy therapies.9–11 A subgroup analysis of recently diagnosed, treatment-naïve patients in ASCLEPIOS I/II concluded that ofatumumab has a favorable benefit–risk profile versus teriflunomide, 12 supporting first-line use of ofatumumab in these patients.
We report the efficacy and safety of ofatumumab in patients with RMS, for up to 4 years.
Patients and methods
Trial design and patients
The methods of ASCLEPIOS I/II, APLIOS, and APOLITOS have been reported previously.4,6,7 ALITHIOS is an ongoing, Phase 3b, open-label, umbrella extension study (initiated 22 November 2018) to assess the longer-term safety, tolerability, and effectiveness of ofatumumab (20 mg s.c. Q4W) in patients with RMS. 8 Patients who completed treatment in the core periods of ASCLEPIOS I/II, APLIOS, 6 or APOLITOS 7 could enter ALITHIOS (see Figure 1). For key inclusion/exclusion criteria, refer to Supplementary Materials. Interim analyses presented here are from the core and extension periods (data cut-off (DCO): 25 September 2021).
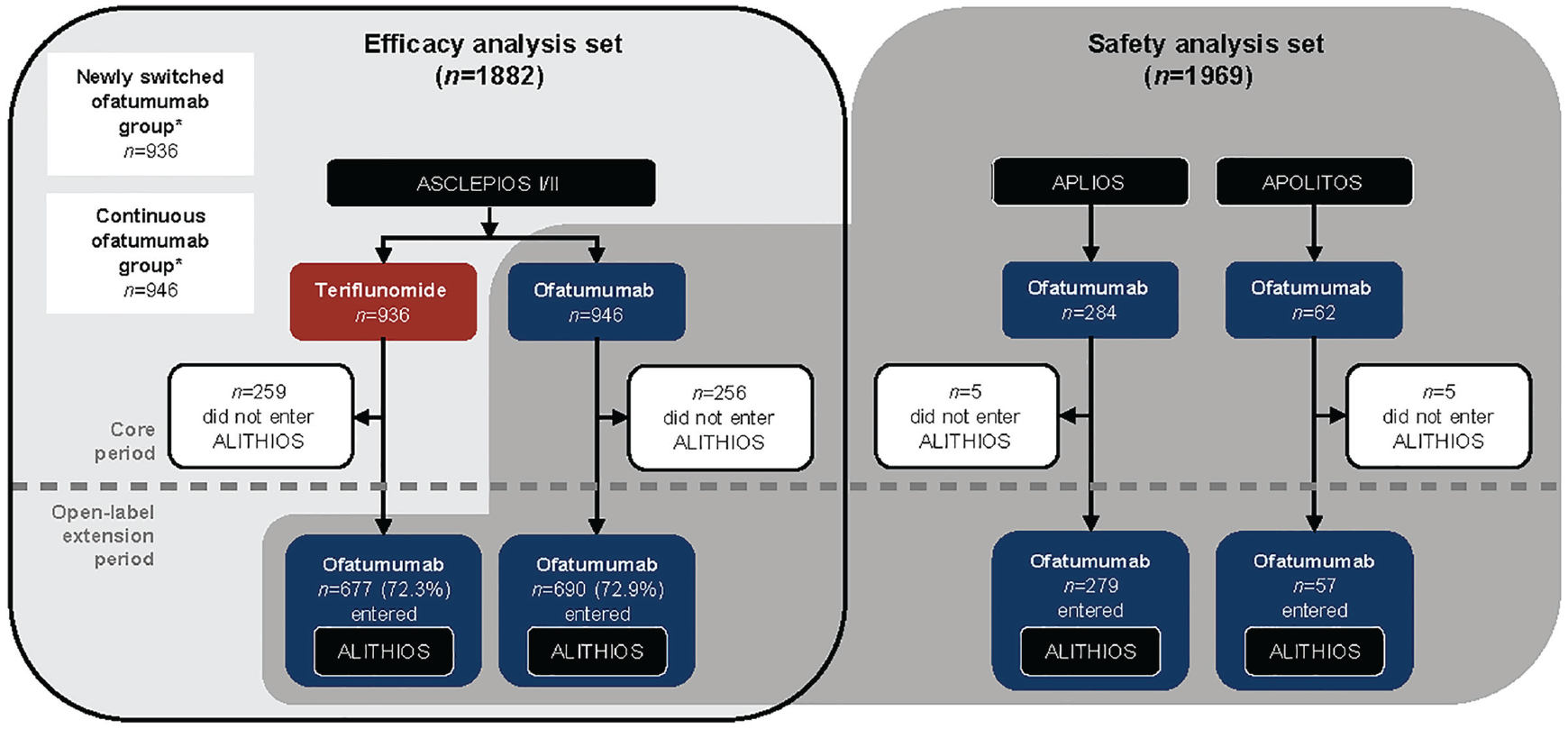
Analysis sets and time periods.
Analysis populations
Efficacy analysis set
Efficacy analyses consisted of data from patients randomized to ofatumumab or teriflunomide in ASCLEPIOS I/II. For three-parameter no evidence of disease activity (NEDA-3), a modified efficacy analysis set was used, excluding patients who discontinued treatment early except due to lack of efficacy or death and had NEDA-3 prior to early discontinuation.
Safety analysis set
Safety analyses consisted of data from patients who received ⩾ 1 dose of ofatumumab in ASCLEPIOS I/II, APLIOS, APOLITOS, or ALITHIOS (see Figure 1).
Analysis of delayed versus early initiation of ofatumumab
Two analysis subgroups were defined: the “continuous ofatumumab group” and the “newly switched ofatumumab group” (see Figure 1). Continuous ofatumumab group: in the efficacy analysis, this comprised patients randomized to ofatumumab in ASCLEPIOS I/II, whereas in the safety analysis, this comprised patients who received ⩾ 1 dose of ofatumumab in ASCLEPIOS I/II, APLIOS, or APOLITOS. Newly switched ofatumumab group: in both the efficacy and safety analyses, this comprised patients who received teriflunomide in ASCLEPIOS I/II and switched to ofatumumab in ALITHIOS.
Efficacy endpoints
The endpoints assessed were: ARR; confirmed disability worsening (CDW) events (increase from baseline Expanded Disability Status Scale score sustained for ⩾ 3/6 months (3/6mCDW) mean number of gadolinium-enhancing (Gd+) T1 lesions per scan; number of new/enlarging T2 (neT2) lesions per year; serum neurofilament light chain (sNfL) concentration at ASCLEPIOS I/II baseline, 3-/12 months post-baseline, and every 6 months thereafter; and NEDA-3 status (no 6mCDW events, no confirmed relapses, and no MRI activity (new Gd+ T1 or neT2 lesions)) in the core and extension periods, in Year 1, beyond Year 1, and overall. For further details, see Supplementary Materials.
Safety and tolerability evaluation
AEs were graded according to the Common Terminology Criteria for Adverse Events version 5.0, 13 with preferred terms per Medical Dictionary for Regulatory Activities version 24.1. An independent expert reviewed cases of opportunistic infection.
Statistical analyses
The ARR for the newly switched and continuous ofatumumab groups was estimated using a piecewise negative binomial model. Cumulative 3/6mCDW were assessed using Kaplan–Meier curves. Lesions per scan and adjusted annualized rates of lesions were estimated using piecewise negative binomial models. Between-group comparisons for number of relapses and number of lesions were analyzed using the Wilcoxon rank sum test. sNfL concentration was analyzed by mixed-effect modeling of repeated measures. NEDA-3 during the core and extension periods, and overall was analyzed separately using logistic regression models fitted to modified efficacy analysis sets, which excluded patients who discontinued early for reasons other than lack of efficacy/death and had NEDA-3 prior to discontinuation.
Results
Patients
Efficacy subgroups: baseline demographics and reasons for discontinuation
The efficacy analysis set included 1882 patients: in ASCLEPIOS I/II, 936 and 946 patients were randomized to teriflunomide and ofatumumab, respectively; 72.6% (n = 677 (teriflunomide) n = 690 (ofatumumab) entered ALITHIOS (see Figure 1). In total, n = 1214/1367 (88.8%) were receiving ofatumumab at DCO. Baseline demographics and disease characteristics were balanced between groups (see Table 1). In ALITHIOS, the most common reasons for ofatumumab discontinuation were AEs and patient/guardian decision (both ~4%, see Supplementary Table 1).
Baseline demographics and clinical characteristics (efficacy analysis set a ).
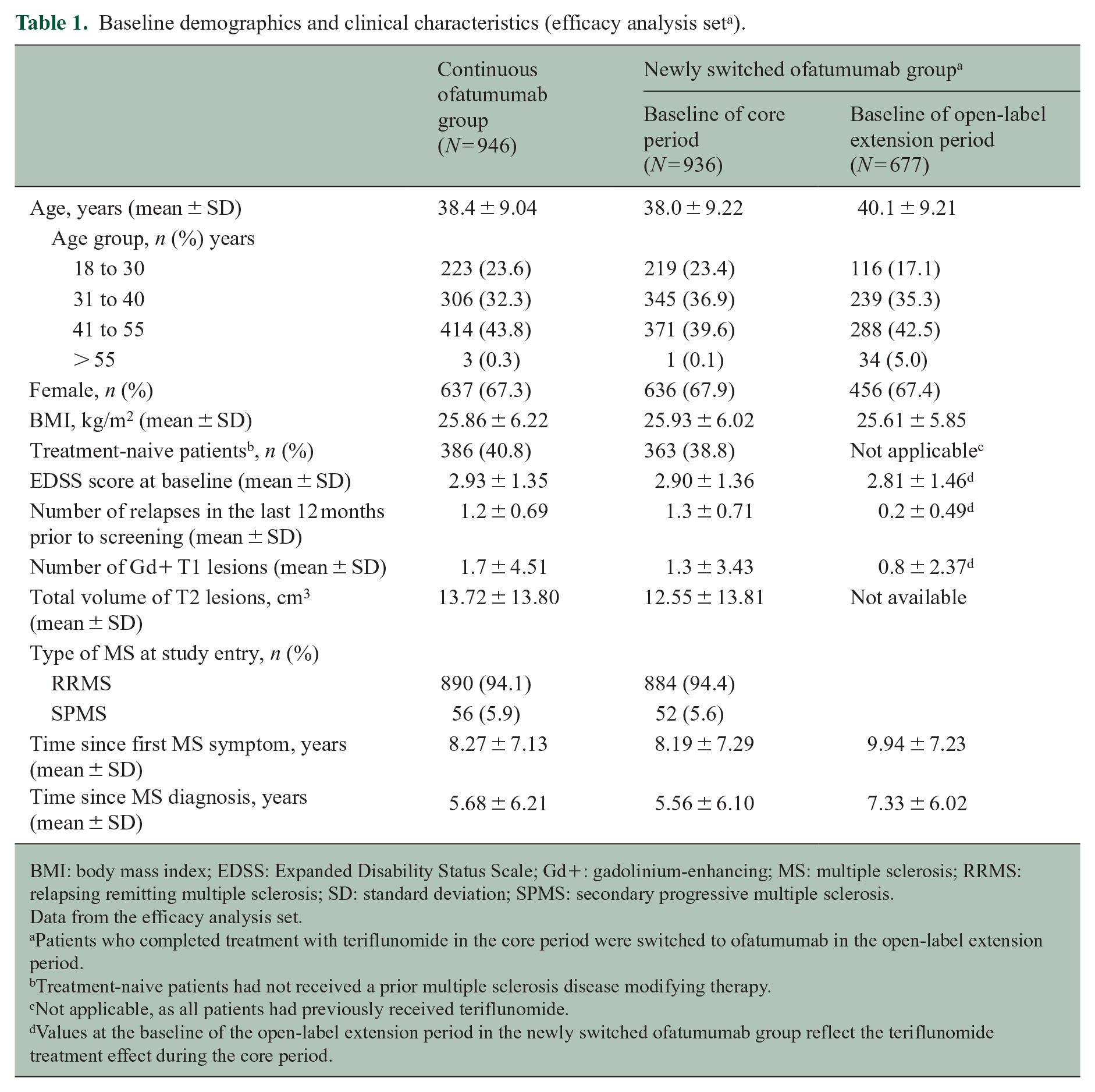
BMI: body mass index; EDSS: Expanded Disability Status Scale; Gd+: gadolinium-enhancing; MS: multiple sclerosis; RRMS: relapsing remitting multiple sclerosis; SD: standard deviation; SPMS: secondary progressive multiple sclerosis.
Data from the efficacy analysis set.
Patients who completed treatment with teriflunomide in the core period were switched to ofatumumab in the open-label extension period.
Treatment-naive patients had not received a prior multiple sclerosis disease modifying therapy.
Not applicable, as all patients had previously received teriflunomide.
Values at the baseline of the open-label extension period in the newly switched ofatumumab group reflect the teriflunomide treatment effect during the core period.
Safety analyses
The safety analysis set included 1969 patients (see Figure 1). Baseline demographics and disease characteristics of the continuous ofatumumab (n = 1292/1969) and newly switched ofatumumab group (n = 677/1969) were comparable (see Supplementary Table 2). Total ofatumumab exposure was 4032.5 patient-years (PYs; continuous ofatumumab 2761.4 versus newly switched ofatumumab 1271.1 PYs), and mean ofatumumab exposure was 2.9 and 1.9 years, respectively (see Supplementary Table 3). Mean adherence to ofatumumab treatment was > 95% (see Supplementary Table 4).
Clinical efficacy assessments
Relapses
The continuous ofatumumab group maintained a low ARR for up to 4 years: adjusted ARRs in the core and extension periods were 0.11 (95% confidence interval (CI): 0.08–0.13) and 0.05 (95% CI: 0.04–0.07), respectively (49.4%; p < 0.001), corresponding to an adjusted rate of one relapse every 20 years during the extension period. For the newly switched ofatumumab group, the ARR was reduced by 71.7% in the extension versus core period (p < 0.001); adjusted ARRs were 0.23 (95% CI: 0.18–0.28) and 0.06 (95% CI: 0.05–0.09), respectively (see Figure 2(a)). See Supplementary Figure 1A for between-group comparison. The cumulative number of confirmed relapses in the continuous ofatumumab group (n = 269; 3123.4 years) was 43.4% lower (between-group analysis; p < 0.001) than in the newly switched ofatumumab group (n = 475; 3042.2 years; see Figure 2(b)).
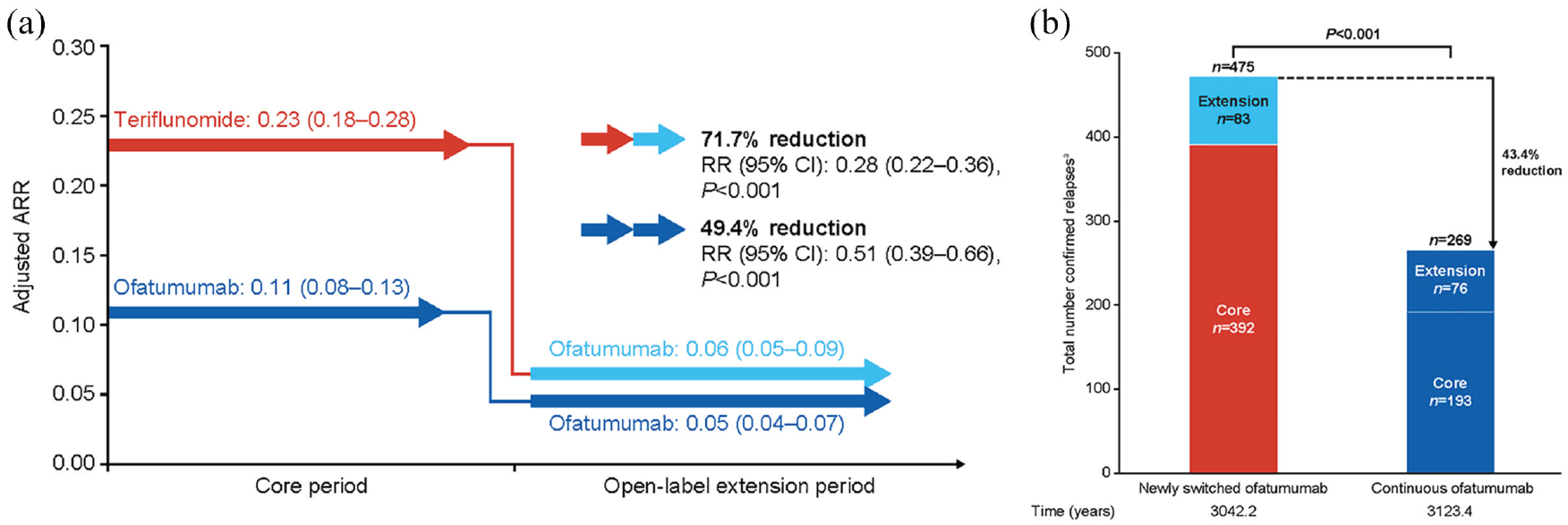
Relapse data: (a) within-group comparison of ARR during the core and open-label extension periods; (b) between-group comparison of total confirmed relapses up to 4 years in the newly switched ofatumumab group and continuous ofatumumab group (efficacy analysis set).
CDW events
Cumulative 3/6mCDW event rates (Kaplan–Meier estimate) remained lower with continuous ofatumumab versus newly switched ofatumumab (3mCDW rate at Month 48: 19.1% and 23.1%, respectively; overall log-rank p = 0.021, see Figure 3(a); 6mCDW rate at Month 48: 15.8% and 18.9%, respectively; overall log-rank p = 0.066, see Figure 3(b)). Cumulative numbers of 3mCDW and 6mCDW events were 17.9% and 16.0% lower in the continuous ofatumumab group versus the newly switched ofatumumab group (3mCDW events: n = 156 versus n = 190, respectively, see Figure 3(a); 6mCDW events: n = 131 versus n = 156, see Figure 3(b)).
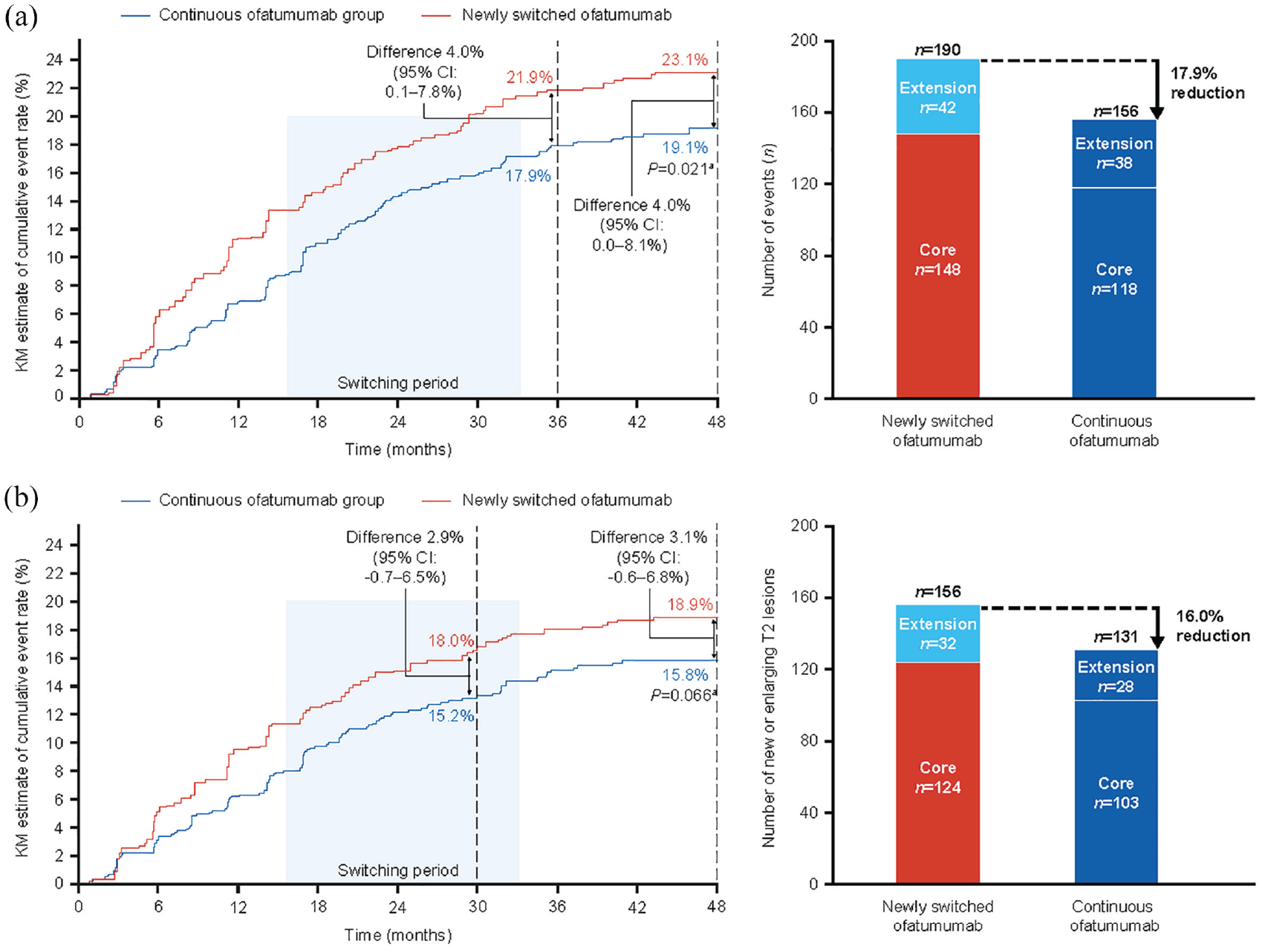
Kaplan–Meier estimates of cumulative event rates (% patients) for confirmed disability worsening (left-hand panel) and cumulative number of confirmed disability worsening events (right-hand panel): (a) 3mCDW and (b) 6mCDW (efficacy analysis set).
MRI assessments
Near-complete suppression of MRI activity was maintained through 4 years in the continuous ofatumumab group. The adjusted mean number of Gd+ T1 lesions per scan in the core and extension periods was: 0.02 (95% CI: 0.02–0.03) and 0.01 (95% CI: 0.00–0.02), respectively; a reduction of 65.0% (p = 0.003). For the newly switched ofatumumab group, the adjusted mean number of Gd+ T1 lesions per scan reduced from 0.55 (95% CI: 0.47–0.65) in the core period, to 0.01 (95% CI: 0.01–0.02) in the extension period; a reduction of 97.4% (p < 0.001; see Figure 4(a)). The cumulative number of Gd+ T1 lesions in the continuous ofatumumab group (n = 66) was 95% lower (p < 0.001) versus the newly switched ofatumumab group (n = 1310; see Figure 4(b)). For between-group comparison, see Supplementary Figure 1B.
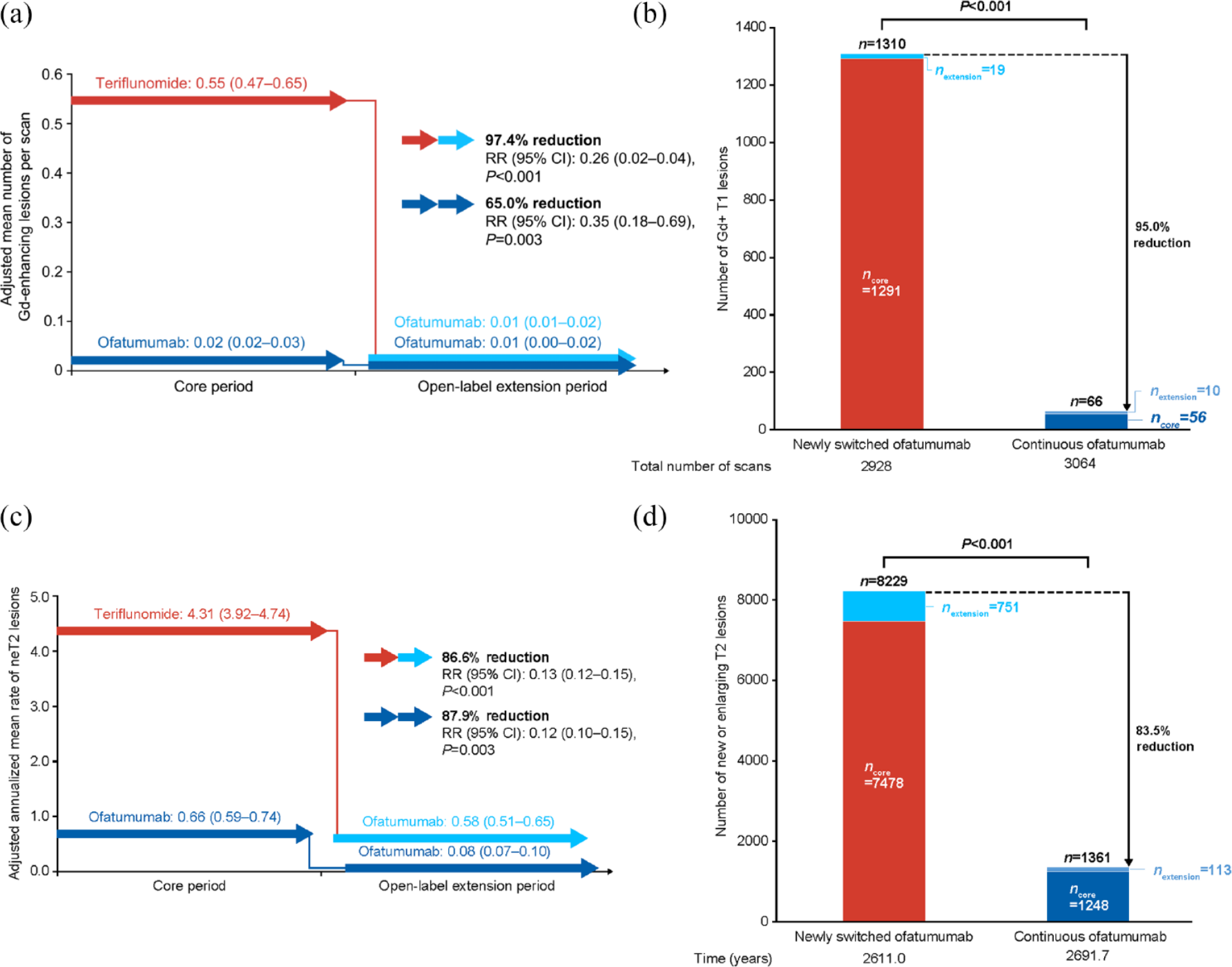
Lesion activity on MRI in the core and extension periods: (a) within-group comparison, mean number of Gd+ T1 lesions/scan; (b) between-group comparison, mean number of Gd+ T1 lesions; (c) within-group comparison, mean annualized rate of neT2 lesions; and (d) between-group comparison, mean number of neT2 lesions (efficacy analysis set).
Continuous ofatumumab treatment led to an 87.9% reduction (p < 0.001) in the adjusted annualized rate of neT2 lesions (core period: 0.66 (95% CI: 0.59–0.74); extension period: 0.08 (95% CI 0.07–0.10)). There was also a reduction with the newly switched ofatumumab group, where the adjusted mean number of neT2 lesions per scan decreased from 4.31 (95% CI: 3.92–4.74) in the core period, to 0.58 (95% CI: 0.51–0.65) in the extension period; an 86.6% per scan rate reduction (p < 0.001; see Figure 4(c)). The cumulative number of neT2 lesions in the continuous ofatumumab group (n = 1361 at DCO) was reduced by 83.5% (p < 0.001) versus the newly switched ofatumumab group (n = 8229; see Figure 4(d)). For between-group comparison of the annualized rate of neT2 lesions, see Supplementary Figure 1C.
sNfL concentration
In the core period, sNfL concentration was lower with ofatumumab versus teriflunomide (concentration at Month 12: 8.03 versus 10.25 pg/mL; at Month 24: 7.96 versus 9.97 pg/mL, respectively; both p < 0.001; Supplementary Figure 2A). sNfL concentration remained low in the extension period with continuous ofatumumab treatment (Month 24: 8.50 pg/mL; Supplementary Figure 2B). Switching from teriflunomide to ofatumumab lowered sNfL levels: in the newly switched ofatumumab group, sNfL concentration remained higher versus the continuous ofatumumab group up to 6 months after switch (9.07 versus 8.31 pg/mL; p < 0.001), but low in both groups at 24 months (8.23 versus 8.50 pg/mL; see Supplementary Figure 2B). The benefits of continuous ofatumumab treatment on sNfL concentration were evident for up to 48 months (see Supplementary Figure 2C).
NEDA-3 status
The likelihood of maintaining NEDA-3 for up to 4 years was over three times higher with early ofatumumab initiation. A greater proportion of the continuous ofatumumab group than the newly switched ofatumumab group maintained NEDA-3 during the core period: 36.7 versus 16.1% (odds ratio (OR) 3.53 (95% CI: 2.76–4.51), p < 0.001); extension period: 78.8 versus 51.0% (OR 3.89 (95% CI: 3.01–5.02), p < 0.001); and overall: 30.5 versus 12.6% (OR 3.51 (95% CI: 2.69–4.57, p < 0.001; see Figure 5(a)). See Supplementary Figures 3A–D for data on each component of NEDA-3.
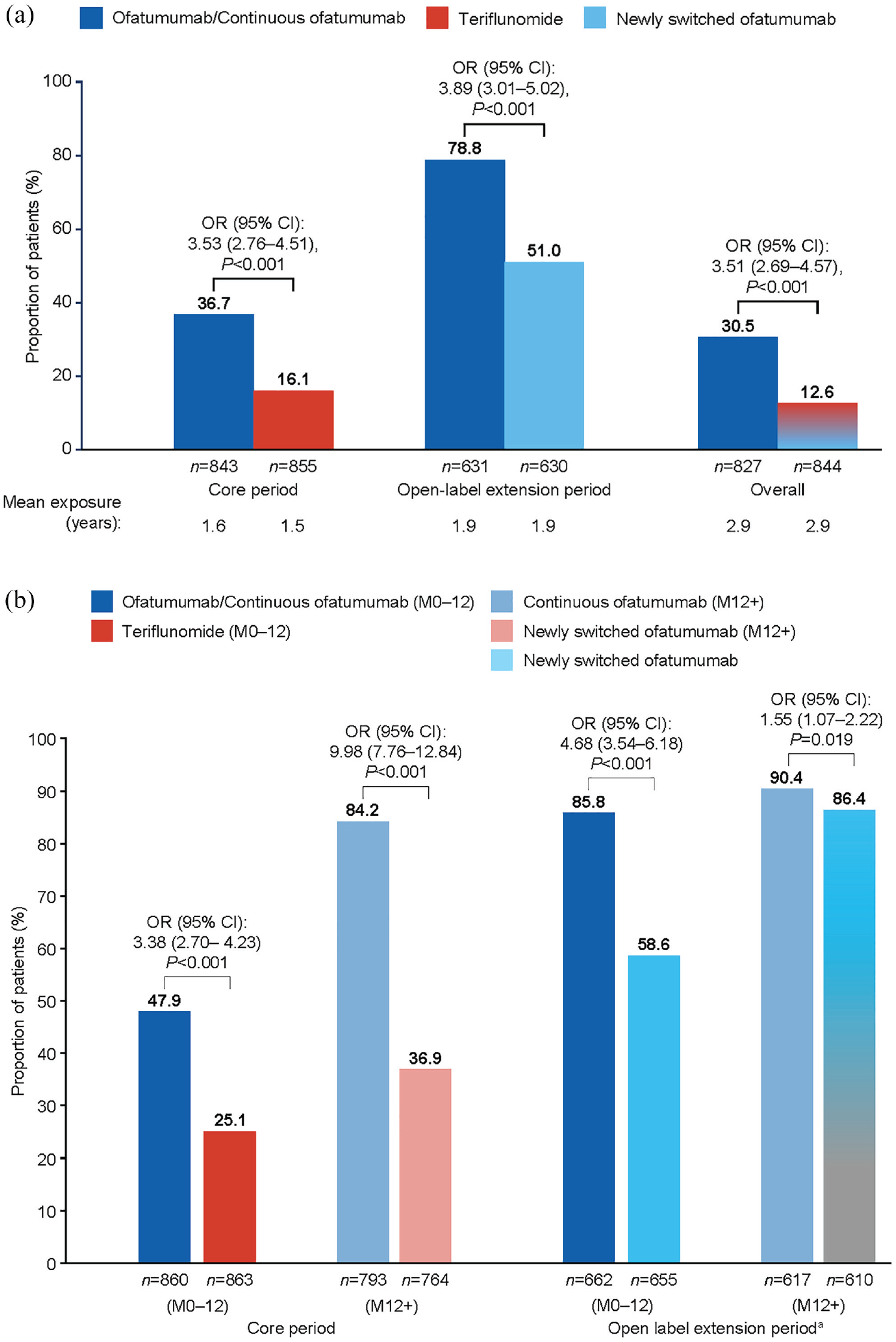
No evidence of disease activity (NEDA-3): (a) core period, open-label extension period and overall; (b) during the first year of treatment and after first year of treatment, by study period (efficacy analysis set).
To explore the influence of rapid suppression of inflammatory activity on MRI with ofatumumab, NEDA-3 status in the first year of treatment was determined (see Figure 5(b)). In the core period, the likelihood of maintaining NEDA-3 during the first year was approximately three-fold higher with ofatumumab versus teriflunomide (47.9 versus 25.1% OR 3.38 (95% CI: 2.70–4.23) p < 0.001), and 10-fold higher beyond that time (84.2 versus 36.9%; OR 9.98 (95% CI: 7.76–12.84), p < 0.001; see Figure 5(b)). In the extension period, 85.8% of the continuous ofatumumab group and 58.6% of the newly switched ofatumumab group maintained NEDA-3 during the first year (OR 4.68 (95% CI: 3.54–6.18), p < 0.001). Subsequently, NEDA-3 increased to 86.4% in the newly switched ofatumumab group and became comparable to the proportion in the continuous ofatumumab group (90.4%; OR 1.55 (95% CI: 1.07–2.22); p = 0.019).
Safety
AE profile
Safety was consistent with previous reports4,8 and no new safety signals emerged. Overall, n = 1698/1969 (86.23%) of the ofatumumab safety analysis set had ⩾ 1 AE; the exposure-adjusted incidence rate (EAIR) was 135.11 per 100 PYs (95% CI: 128.83–141.69; see Table 2). In the ASCLEPIOS I/II core period, n = 791/946 (83.61%) of the ofatumumab group had ⩾ 1 AE and the EAIR was 188.55 (95% CI: 175.86–202.16). The incidence of AEs leading to discontinuation was low (overall ofatumumab safety analysis set: n = 128/1969 (6.50%); ASCLEPIOS I/II core period: n = 54/946 (5.70%)). Serious AEs were reported in n = 242/1969 (12.30%) of the ofatumumab safety analysis set (EAIR 4.96 (95% CI: 4.37–5.63)), and in n = 86/946 (9.10%) of the ofatumumab group in the core period (EAIR 5.39 (95% CI: 4.36–6.65)). The most frequently reported AEs were infections and infestations (overall ofatumumab safety analysis set: n = 1149/1969 (58.35%); ACLEPIOS I/II core period: n = 488/946 (51.58%)) consistent with previous findings up to 2.5 years (n = 488/946 (51.6%).4,5
Safety summary (safety analysis set).
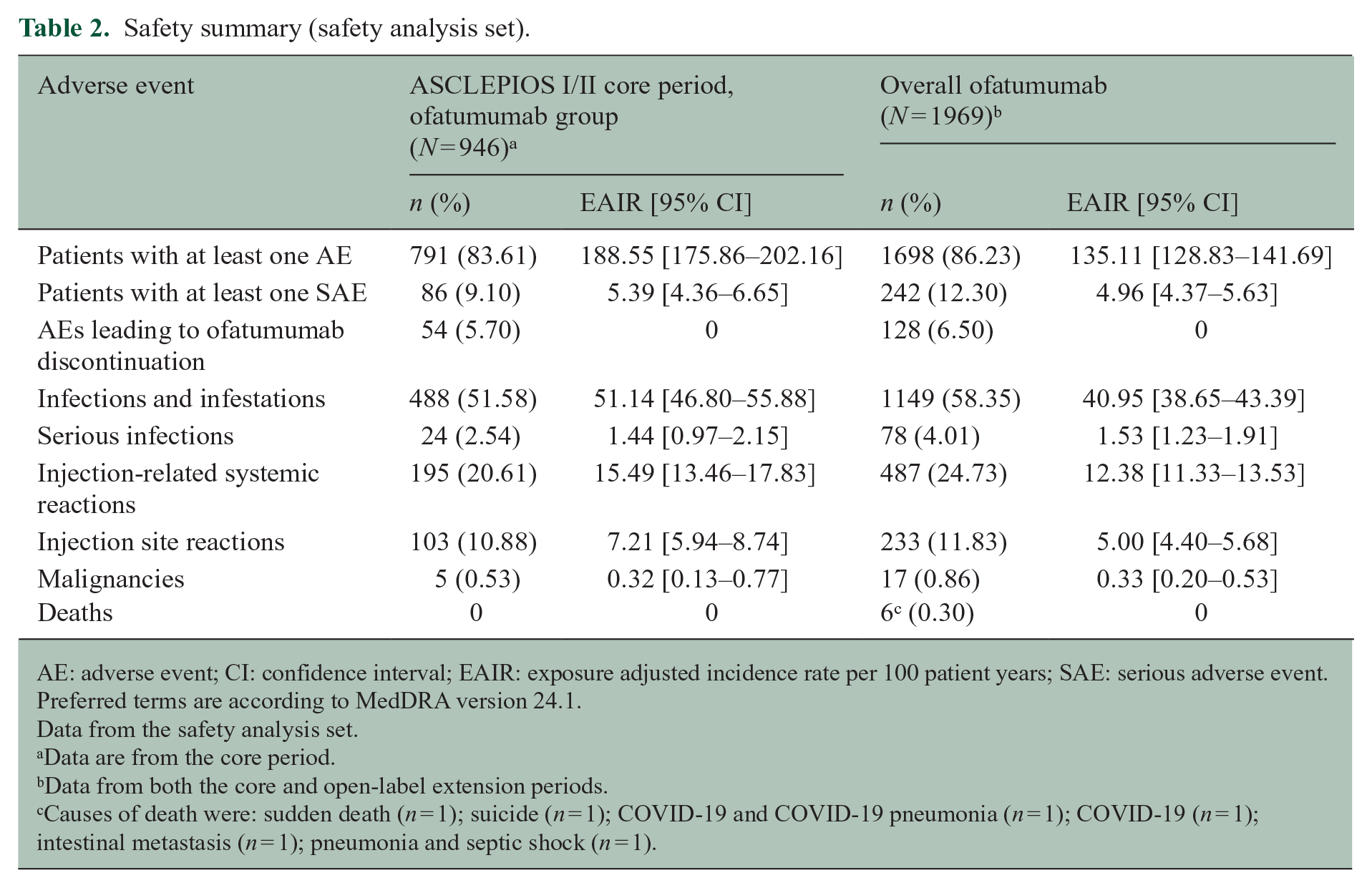
AE: adverse event; CI: confidence interval; EAIR: exposure adjusted incidence rate per 100 patient years; SAE: serious adverse event.
Preferred terms are according to MedDRA version 24.1.
Data from the safety analysis set.
Data are from the core period.
Data from both the core and open-label extension periods.
Causes of death were: sudden death (n = 1); suicide (n = 1); COVID-19 and COVID-19 pneumonia (n = 1); COVID-19 (n = 1); intestinal metastasis (n = 1); pneumonia and septic shock (n = 1).
Serious infections
The EAIR of serious infections remained stable (overall safety analysis set (n = 78/1969) 1.53 (95% CI: 1.23–1.91); ASCLEPIOS I/II core period ofatumumab group (n = 24/946) 1.44 (95% CI: 0.97–2.15); see Table 2). The most frequent were COVID-19 infections (n = 1/1969; 0.05%) and appendicitis (n = 14/1969; 0.7%). Most serious infections (n = 73/1969; 3.7%) resolved without discontinuation. A small proportion of serious infections (n = 6/1969; 0.3%) were Grade 4.
Immunoglobin G and immunoglobin M
Mean serum immunoglobin G (IgG) levels remained stable and above the lower limit of normal (LLN; 5.65 g/L), even in patients with lower IgG levels at baseline (see Supplementary Figures 5A and 5C); mean serum immunoglobin M (IgM) levels decreased but remained above the LLN (0.40 g/L; see Supplementary Figure 5B and D). Of 1969 patients, interruption was reported in 2 (0.1%) and 185 (9.5%) patients due to low IgG and IgM, and discontinuation in 1 (0.1%) and 60 (3.0%) patients, respectively (see Supplementary Tables 6 and 7). Sensitivity analyses confirmed early interruption/discontinuation of ofatumumab due to low IgG/IgM did not impact overall IgG/IgM patterns (see Supplementary Figures 5E–5G). Low levels of IgG or IgM were not associated with an increased incidence of serious infections (see Supplementary Table 8).
Neutropenia and lymphopenia
Mean lymphocyte and neutrophil levels remained stable and were above the LLN; any reductions below the LLN occurred randomly and were not persistent (see Supplementary Figure 6A and 6B). During the core period, although neutrophil levels were lower with teriflunomide, they returned to baseline levels following the switch to ofatumumab (see Supplementary Figure 6B). No serious AEs of lymphopenia or neutropenia were reported. Treatment was interrupted in 2/1969 (0.1%) patients with lymphopenia; neither discontinued treatment. There was no association between decreased lymphocyte/neutrophil counts and risk of serious infection.
Injection-related reactions
Of 1969 patients, 487 (24.7%) and 233 (11.8%) experienced a systemic and injection site-related reaction, respectively. Most systemic and injection site-related reactions (IRRs) were Grade 1/2 (n = 483/487 (99.2%) and n = 232/233 (99.5%); see Supplementary Figure 4A and 4B). No life-threatening IRRs were reported.
Malignancies
The incidence of malignancies was low (overall safety analysis set: 0.86%; EAIR 0.33 (95% CI: 0.20–0.53), see Supplementary Table 5); with an EAIR comparable to the core period (ASCLEPIOS I/II ofatumumab group: 0.53%; EAIR 0.32 (0.13–0.77)).
Deaths
In total, 6/1969 deaths occurred during the extension period and were reported by investigators as unrelated to ofatumumab (see Table 2).
Discussion
This interim analysis provides new insights into the longer-term efficacy and safety of ofatumumab in RMS, supporting a favorable benefit–risk profile. The low rate of relapses with ofatumumab, as identified in ASCLEPIOS I/II, 4 was further reduced in the extension period, and together with the almost complete suppression of MRI lesions and low risk of CDW, demonstrate the sustained efficacy of ofatumumab for up to 4 years.
Early initiation of high-efficacy treatment for RMS improves longer-term outcomes compared with delayed initiation or switching from lower efficacy therapies.9–11 This comparison of continuous ofatumumab versus switching to ofatumumab from teriflunomide demonstrated the cumulative benefit (up to 4 years) of early ofatumumab initiation with regard to cumulative number of relapses, and number of Gd+ T1/neT2 lesions. Moreover, the odds of maintaining NEDA-3 during the extension period were three times greater with early initiation. After the first year in the core period, a very high proportion (84.2%) of the continuous ofatumumab group retained NEDA-3 status; this beneficial effect continued throughout the extension period, supporting the sustained efficacy of ofatumumab. In contrast, only 36.9% of patients randomized to teriflunomide retained NEDA-3 status after a year. Although between-group differences for all components of NEDA-3 favored continuous ofatumumab, the greatest differences were for neT2 and Gd+ T1 lesions. Thus, patients initially randomized to teriflunomide were at higher risk of not maintaining NEDA-3 in the first year of treatment mainly due to MRI disease activity. The legacy of lower efficacy teriflunomide appeared to negatively affect the likelihood of maintaining NEDA-3 during the first year of open-label ofatumumab treatment, but with longer treatment, the odds of maintaining NEDA-3 increased to 9/10 patients and remained high for up to 4 years.
The 3mCDW and 6mCDW rates at 36 and 48 months, and the cumulative number of events, indicate that early ofatumumab treatment leads to superior disability outcomes that cannot be recovered in patients initially randomized to teriflunomide. These findings are consistent with recent studies of the longer-term benefits of early initiation of high-efficacy DMTs and disability outcomes in MS.9–11
The results also illustrate the value of switching from a low efficacy therapy to ofatumumab. In the newly switched ofatumumab group, there was a marked reduction in the ARR, together with almost complete suppression of Gd+ T1 lesion activity, a pronounced reduction of neT2 lesions, sustained reduction of neuroaxonal injury (sNfL), and increased likelihood of maintaining NEDA-3 status.
Treatment with biological drugs may trigger an immune response that leads to the formation of anti-drug antibodies (ADAs). 14 The development of high titers of neutralizing ADAs may lead to suboptimal treatment exposure and thereby might limit efficacy. 14 However, it has been previously reported that very few patients receiving ofatumumab in ASCLEPIOS I/II developed ADAs (0.2%), with no treatment enhancing or neutralizing ADAs reported (0%).1,15
The cumulative safety data indicate that extended ofatumumab treatment is well tolerated in patients with RMS, with no new risks identified. Ofatumumab tolerability was reflected by a high level of adherence and a low rate of discontinuation throughout the core and extension periods. The EAIRs of AEs and SAEs were consistent with those previously reported in ASCLEPIOS I/II. 4 During extended exposure, the incidence of malignancies remained low, and both serious infections (EAIRs) and IgG levels remained stable. Although IgM levels declined, average IgM levels remained above the LLN. Neither low IgG nor low IgM were associated with increased incidence of serious infection. IgG levels fell below the LLN in only 1.6% of patients, and in most cases, did not lead to treatment interruption/discontinuation. Additional sensitivity analyses confirmed that treatment interruption/discontinuation due to low IgG/IgM did not impact overall Ig trends confirming the robustness of IgG stabilization. With longer-term use of ofatumumab, lymphocyte and neutrophil levels remained stable and above the LLN. The incidence of lymphopenia and neutropenia were comparable with previous studies and remained low, with no serious events reported.4,6,7 Overall, safety was in line with a previous interim analysis with up to 3.5 years of treatment. 8
The analyses of longer-term efficacy data presented here for the open-label ALITHIOS extension study (including data from the start of the blinded ASCLEPIOS I/II studies) are subject to the limitations of any open-label study, including lack of a comparator arm beyond the core studies. As such, the conclusions related to longer-term clinical efficacy outcomes may be limited due to the potential influence of regression to the mean. Also, as blinding is not maintained during an open-label extension study, the risk of rater assessment bias may increase.
In addition, upon completion of the core studies, participation in ALITHIOS was voluntary and patients were free to discontinue due to any reason, creating the potential for selection bias. This seems unlikely, however, as an equal proportion of patients from the ASCLEPIOS I/II teriflunomide (72.3%) and ofatumumab (72.9%) arms enrolled into ALITHIOS; 88.8% of whom were still receiving ofatumumab at data cut-off. Furthermore, in ALITHIOS, the rate of discontinuations was identical for both the ofatumumab and teriflunomide arms (11.2%) and reasons for discontinuation were similar (for example, discontinuations due to AEs: 4.2% vs 3.8%, respectively; patient/guardian decision: 3.6% vs 4.3%, respectively), with low rates of discontinuations due to lack of efficacy (0.6% vs 0.9%, respectively; see Supplementary Table 1). Thus, there was no evidence for an impact of selection bias on these data.
The ongoing ALITHIOS study coincided with the global outbreak of COVID-19. People with MS are at increased risk of serious infection,16–19 and anti-CD20 mAbs may compromise the immune response 20 and attenuate humoral responses to COVID-19 vaccination.21–23 In this interim analysis, most reported COVID-19 cases were non-serious, and 241/245 (98.4%) of patients recovered. 24 Two COVID-19-related deaths (2/245 (0.8%) occurred in unvaccinated ofatumumab-treated patients (one with additional risk factors); both were deemed unrelated to study treatment. Fatalities occurred less frequently than reported for the general population. 25
Our findings of sustained reductions in cumulative number of relapses, MRI lesion activity, and risk of CDW with ofatumumab treatment add to a growing body of evidence that supports the value of early initiation of high-efficacy therapies in RMS,4,10 and highlight the favorable benefit–risk profile of long-term (up to 4 years) ofatumumab treatment for patients with RMS. Early access to highly efficacious treatments such as ofatumumab can help to reduce disease burden, risk of RMS progression, and contribute to longer-term improvements in quality of life. 26
Supplemental Material
sj-docx-1-msj-10.1177_13524585231195346 – Supplemental material for Efficacy and safety of four-year ofatumumab treatment in relapsing multiple sclerosis: The ALITHIOS open-label extension
Supplemental material, sj-docx-1-msj-10.1177_13524585231195346 for Efficacy and safety of four-year ofatumumab treatment in relapsing multiple sclerosis: The ALITHIOS open-label extension by Stephen L Hauser, Ronald Zielman, Ayan Das Gupta, Jing Xi, Dee Stoneman, Goeril Karlsson, Derrick Robertson, Jeffrey A Cohen and Ludwig Kappos in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors thank the patients of the trials included in this publication, the clinical study team for the conduct of the study, Roseanne Sullivan of Novartis for assistance with review and interpretation of ofatumumab safety data, Xixi Hu and Alex Ocampo of Novartis for assistance with the statistical analyses. The studies were sponsored by Novartis Pharma AG, Basel, Switzerland. Medical writing support for the development of this publication, under the direction of the authors, was provided by James Currie (BSc, Hons; PhD), Laura Crocker (BMedSci, Hons) and Philippa Lloyd (BSc, Hons) of Ashfield MedComms, an Inizio company, and was funded by Novartis Pharma AG.
Author contributions
Conception or design of the work was done by Ayan Das Gupta, Jing Xi, and Ronald Zielman. Data collection was done by Ronald Zielman and Jing Xi. All authors contributed to data analysis and interpretation, drafting/revision of the article, and final approval of the version to be published.
Data availability statement
Trial data are available on reasonable request, provided the reason for the request is in line with current ethical and intellectual property requirements surrounding the use of data. Requests should be directed through ClinicalStudyDataRequest.com.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All support for the present manuscript (e.g. funding, provision of study materials, medical writing, article processing charges) and the studies were sponsored and funded by Novartis Pharma AG, Basel, Switzerland. Novartis Pharma AG supported the development of this manuscript, provided data analyses according to the direction of the authors, and provided funding for medical writing support. Open access fee was paid by Novartis. Medical writing support for the development of this publication, under the direction of the authors, was provided by James Currie (BSc, Hons; PhD.), Laura Crocker (BMedSci, Hons), and Philippa Lloyd (BSc, Hons) of Ashfield MedComms, an Inizio company, and was funded by Novartis Pharma AG. Stephen L Hauser currently serves on the scientific advisory board of ACCURE, Alector, Annexon, board of directors of Neurona, and has previously consulted for BD, Moderna, and NGM Bio. Dr Hauser also has received travel reimbursement and writing support from F. Hoffmann-La Roche and Novartis AG for anti-CD20-therapy-related meetings and presentations. Grants: NIH/NINDS (R35NS111644) and Valhalla Foundation; within the past 36 months, no longer active: National Multiple Sclerosis Society (RR 2005-A-13). Ronald Zielman is a full-time employee of Novartis. Ayan Das Gupta is a paid and permanent employee of Novartis. Jing Xi is an employee of Novartis. Dee Stoneman is a full-time employee of Novartis. Goeril Karlsson is a full-time salaried employee of Novartis. Derrick Robertson declares grants or contracts from Anokion, Atara Biotherapeutics, Biogen, GW Pharmaceuticals, Novartis, PRIME CME, TG Therapeutics, CorEvitas, EMD Serono, Genentech, Janssen, PCORI and Sanofi; consulting fees from Biogen, Genentech, EMD Serono, Janssen, Bristol Myers Squibb, Horizon, Novartis, Sanofi, TG Therapeutics, Alexion, Greenwich Biosciences and Mallinckrodt; payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Biogen, EMD Serono, Genentech, TG Therapeutics, Bristol Myers Squibb, Janssen, PRIME CME, Sanofi, Alexion and Horizon. Jeffrey A Cohen declares consulting fees from Biogen, Convelo, EMD Serono, Gaossamer Gio, Mylan and PSA; Leadership or fiduciary role in other board, society, committee or advocacy group, paid or unpaid for ACTRIMS; other financial or non-financial interests for Sage—serving as an Editor of Mult Scler J. Ludwig Kappos declares grants or contracts from any entity (payments made to institution) from Novartis, Roche, and Innosuisse; consulting fees (payments made to institution) from AurigaVision AG, Bayer AG, df-mp Molnia & Pohlman, Genentech, Glaxo Smith Kline, Janssen LLC, Japan Tobacco Inc., Merck, Novartis, Roche, Senda Biosciences Inc., Shionogi BV, Wellmera AG; payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (payments made to institution) from BMS, Celgene, Janssen Pharmaceuticals, Merck, Novartis and Roche; support for attending meetings and/or travel (payments made to institution) from MH Consulting, Österreichische Gesellschaft für Neurologie, Novartis Biocieˆncias S.A. and Eli Lilly; participation on a Data Safety Monitoring Board or Advisory Board (AB, payments made to institution) from Actelion, Merck Healthcare KGaA, Novartis, Roche, Sanofi and TG Therapeutics and (DSMB, payments made to institution) from Minoryx Therapeutics S.L. and Santhera Pharmaceuticals; leadership or fiduciary role in other board, society, committee or advocacy group paid or unpaid from Neurostatus-UHB AG—Supervisory Board (unpaid), payment to institution; (charitable) Foundation Clinical Neuroimmunology and Neuroscience Basel (“RC2NB”)—CEO (employment by University Hospital Basel); MAGNBIMS Steering Committee, EUROPEAN CHARCOT FOUNDATION (Board membership)—no payment.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies were sponsored and funded by Novartis Pharma AG, Basel, Switzerland. Novartis Pharma AG supported the development of this manuscript, provided data analyses according to the direction of the authors, and provided funding for medical writing support. Open access fee was paid by Novartis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
