Abstract
Background:
Demographic characteristics, social determinants of health (SDoH), health inequities, and health disparities substantially influence the general and disease-specific health outcomes of people with multiple sclerosis (MS). Participants in clinical trials do not represent all people with MS treated in practice.
Objective:
To provide recommendations for enhancing diversity and inclusion in clinical trials in MS.
Methods:
We held an international workshop under the Auspices of the International Advisory Committee on Clinical Trials in MS (the “Committee”) to develop recommendations regarding diversity and inclusivity of participants of clinical trials in MS. Workshop attendees included members of the Committee as well as external participants. External participants were selected based on expertise in trials, SDoH, health equity and regulatory science, and diversity with respect to gender, race, ethnicity, and geography.
Results:
Recommendations include use of diversity plans, community engagement and education, cultural competency training, biologically justified rather than templated eligibility criteria, adaptive designs that allow broadening of eligibility criteria over the course of a trial, and logistical and practical adjustments to reduce study participant burden. Investigators should report demographic and SDoH characteristics of participants.
Conclusion:
These recommendations provide sponsors and investigators with methods of improving diversity and inclusivity of clinical trial populations in MS.
Introduction
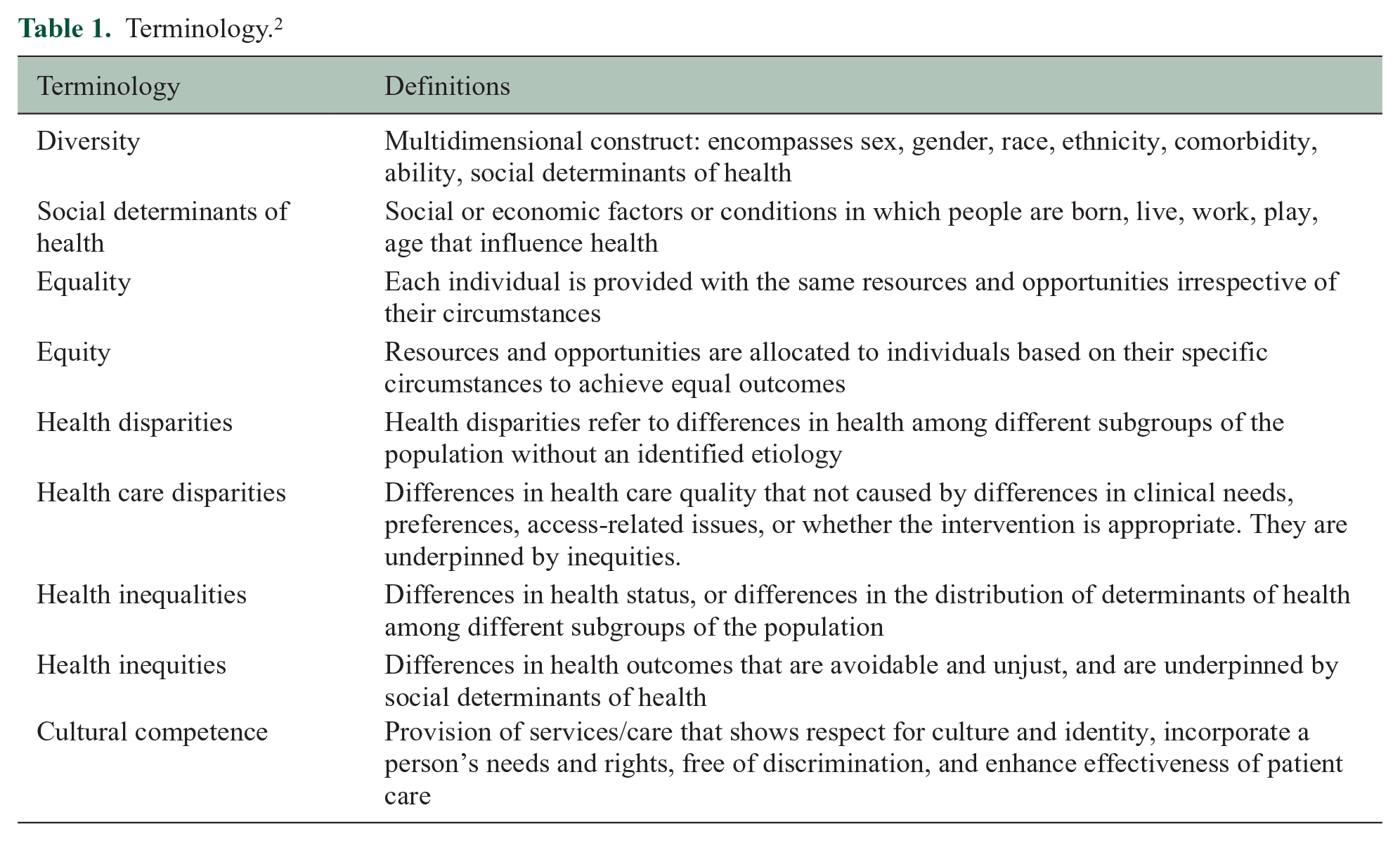
Diversity is multidimensional and encompasses sex, gender, age, race, ethnicity, comorbidity, ability, and social determinants of health (SDoH; Table 1, Figure 1); these dimensions overlap. SDoH are the social or economic factors or conditions in which people are born, develop, live, learn, work, play, and age that influence health. 1 Among these factors are educational access, quality, and achievement, economic stability; food security and stability; neighborhood and built environment; health care access and quality; and social and community contexts, among others. SDoH are not distributed evenly across the population. Health inequities and disparities often disproportionately affect members of the population of particular genders, sexual orientations, racial, ethnic, and religious identities, and abilities.
Terminology. 2
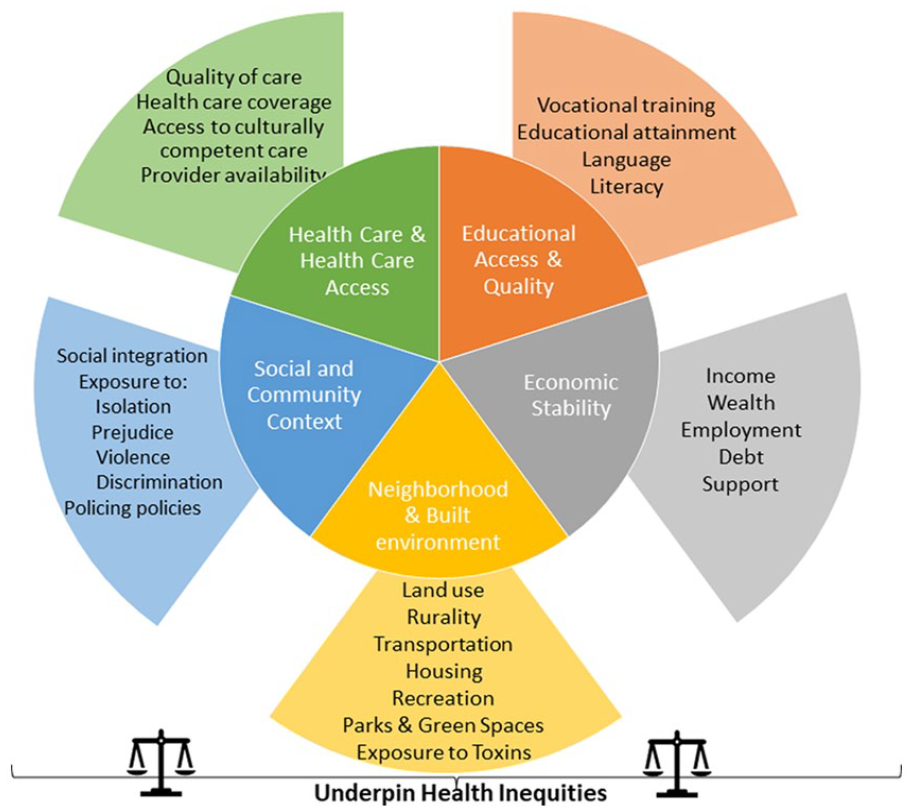
Social determinants of health.
Among people with multiple sclerosis (MS), demographic characteristics such as sex, race, ethnicity, and SDoH importantly influence health outcomes. 3 For example, Hispanic/Latinx American and African American persons with MS attending two American centers had higher MS Severity Scores after adjusting for age and gender than those who were White Americans. 4 Hypertension is more likely to be underdiagnosed among Hispanic/Latinx persons with MS. 5 Immigrants to Canada with MS have more comorbidities 6 and are hospitalized more often in the year of diagnosis than long-term Canadian residents. 7 In one survey, participants with MS who identified as transgender reported lower comfort discussing sexual health with their physician. 8 Another survey found that people with MS who identified as lesbian, gay, bisexual, and/or transgender were more likely to change MS centers, a change attributed to the perception of homophobic behaviors. 9 A sample of African American women with MS reported their diagnoses had been delayed due to misbeliefs by physicians about the risk of MS in their racial group. 10 Less is known about the influence of race and ethnicity outside North America; half of Organisation for Economic Cooperation and Development countries do not routinely collect racial or ethnic identity data, and collection of such data is considered sensitive in Europe. 11 Among people with MS, lower as compared to higher area-level socioeconomic status (SES) is associated with increased hospitalization rates, 12 greater disability progression, 13 and increased mortality. 14
In December 2022, an international group held a workshop, sponsored by the European Committee on Treatment and Research in MS and the US National MS Society. Participants included members of the International Advisory Committee on Clinical Trials in MS whose members are selected to represent different disciplines relevant to MS, geographic regions, and stages of career, while balancing gender and considering race and ethnicity. External participants were selected based on expertise in trials, SDoH, health equity, regulatory science, and diversity with respect to gender, race, ethnicity, and geography.
Herein, we review evidence regarding the diversity of participants enrolled in clinical trials in MS, barriers to more inclusive trial populations, and regulatory perspectives. Finally, we make recommendations to enhance the diversity of clinical trial populations and the inclusiveness of clinical trials going forward.
Health inequities and disparities
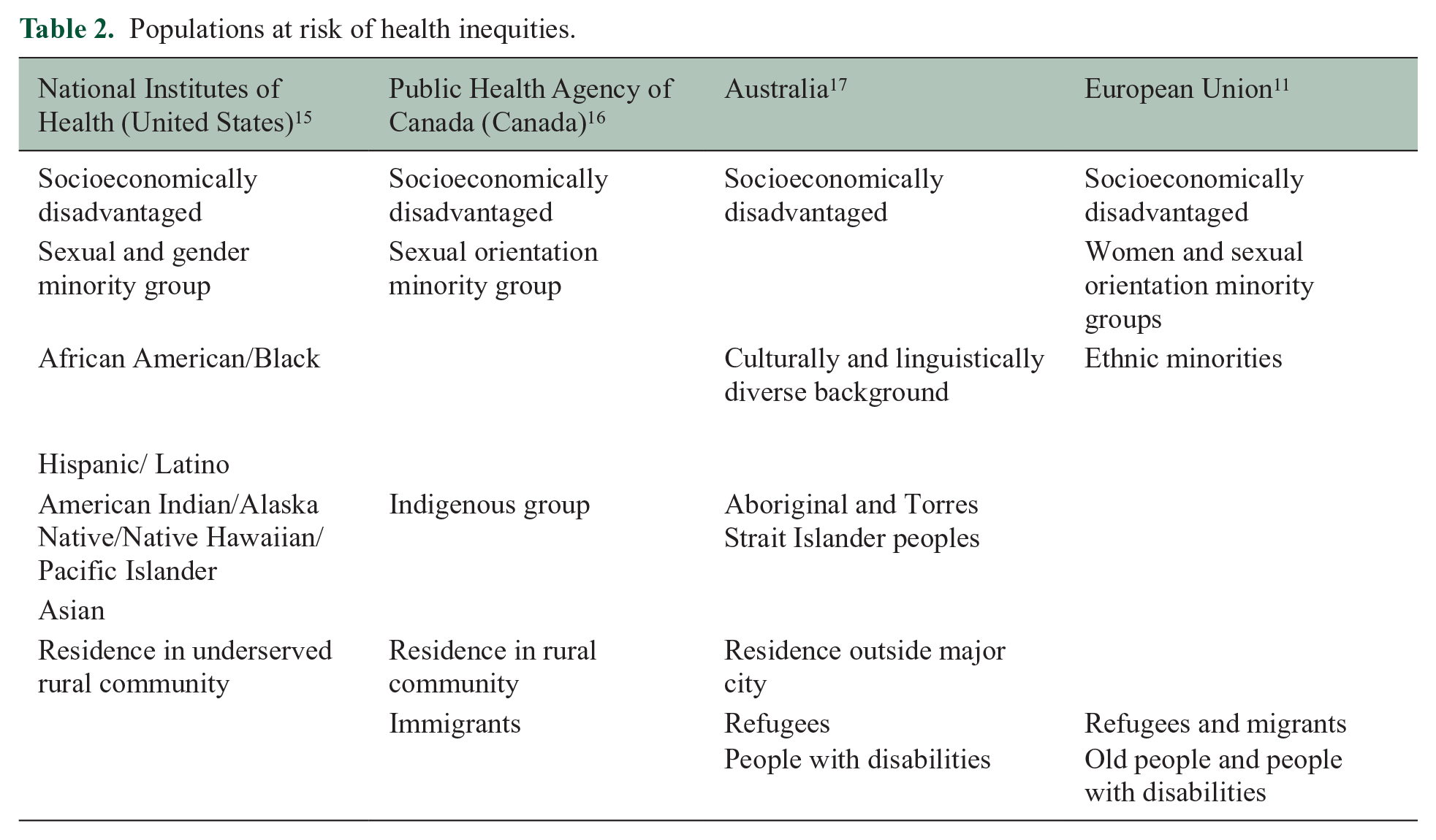
Health and health care disparities refer to differences in health and health care quality among different subgroups of the population. These disparities are underpinned by inequities, that is, differences in health outcomes that are avoidable, and the inequities are underpinned by SDoH. These disparities can be viewed from multiple, often overlapping perspectives commonly related to population type, geography, and risk factors. Population could refer to race, ethnicity, age, gender, sexual orientation, or ability. Geography could refer to urban versus rural, high-income versus low- or middle-income country as defined by the World Bank, or degree of neighborhood privilege versus deprivation. Risk factors could refer to access to care or environmental risks. The formulation of these perspectives may differ across countries because they are shaped by power, wealth, and systemic racism (Table 2).15,16
Populations at risk of health inequities.
Health disparities and inequities affect individuals, health systems, and societies. An actuarial analysis of the relationship between health care disparities secondary to sex, gender, race, SES, and health care spending in the context of diabetes, asthma, cardiovascular disease, and breast and colorectal cancers 18 found that consequent health inequities accounted for $320 billion (USD) in health care spending and $42 billion in lost productivity annually in the United States. In the European Union, the economic burden of socioeconomic-related health inequities was ~10% of the gross domestic product in 2011. 19 The causal pathways between health care disparities and costs are complex, but the high costs emphasize the importance of addressing health inequities related to SDoH, including in the context of clinical trials.
The consequences of failure to recruit and retain diverse, representative populations in clinical trials are manifold. First, the findings may lack generalizability to the entire population of interest. Second, the lack of variation reduces the ability to evaluate the heterogeneity of disease biology and treatment effects. Third, it creates unequal access to the benefits of research and the perpetuation of health inequities, mistrust, lack of uptake of interventions, and harm from the use of ineffective or unsafe therapies. 20 As noted in a report by the Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard, “Race, ethnicity, sex, gender, age, and geographic ancestry do not define distinct genetic or biological groups; yet along with social, cultural, and economic factors, these factors can be associated with important differences in disease susceptibility and manifestation, [and] treatment response . . .” 20 Potential benefits of diversifying trial populations (e.g. for participants at extremes of age, those with comorbidities, and underrepresented racial and ethnic groups) include improvements in scientific credibility, social responsibility, compliance with regulatory guidelines and funder expectations, 20 and improved clinical decision-making and outcomes.
Diversity of clinical trial populations in MS: current state
Clinical trials suffer from a lack of diversity with respect to leadership, 21 stakeholder involvement, and participant characteristics. Generally, women who lead research teams are more likely to consider sex and gender effects in study design and analysis than men who lead research teams. 22 A review of heart failure trials found that those led by women were more likely to report race and ethnicity data and enrolled a higher percentage of participants identifying as Black, Indigenous, or people of color (BIPOC). 23 Yet, women are underrepresented as authors of seminal clinical trials for MS disease-modifying therapies (DMT). 21 Leadership of MS trials by BIPOC individuals has not been described, possibly due to a paucity of data.
Explicit and implicit exclusion criteria for trials are problematic. In trials published in high-impact general medical journals, only 47.2% of the exclusion criteria were strongly justified. 24 A review of trials in cardiology, mental health, and oncology 25 found that trial populations were highly selected, and excluded older adults, those with comorbidities, and those with a lower SES. These exclusions result in the ineligibility of 50%–80% of typical clinical populations. A review of 45 phase 3 clinical trials in MS conducted for DMT approved between 1995 and 2020 found that 17 (37.8%) of trials did not report race or ethnicity, 26 14 (31.1%) trials reported the proportion of the study participants who were White, and the remaining 14 (31.1%) reported ⩾2 ethnicities. At the individual level, the median percentage of participants who did not identify as White was only 6.2% (1.9% Black, 0.5% Asian); the remainder were largely classified as “other” (unspecified). That review did not address reporting of factors such as educational attainment, annual income, or distance lived from the trial site.
A scoping review that examined the extent to which SDoH are considered in the recruitment of participants in MS rehabilitation trials found that exclusions due to implicit and explicit factors were common. 27 Implicit factors included living in rural or remote areas, limited access to an MS clinic, and inability to reach the study site or pay for study-related costs (such as missed time from work). Explicit factors included greater disability, cognitive impairment, physical and mental comorbidities, older age, and language or literacy. The extent of the problem was difficult to evaluate because, other than age and biological sex, most SDoH were not reported. Thus, the lack of diversity in trial populations extends across the full spectrum of clinical trials in MS, and inconsistent reporting hinders understanding of the problem.
Regulatory perspectives
Regulatory agencies have emphasized the need to increase diversity and inclusion of underrepresented populations in clinical trials,28,29 to ensure that participant characteristics reflect clinically relevant populations with respect to age, sex, race, ethnicity, comorbidity, and disease severity. Exclusion criteria must be well-justified based on evidence of lower efficacy or higher risks of adverse events in the excluded group. Trials in many fields have historically excluded participants in whom the risks are perceived to outweigh the benefits of treatment, or in whom the risks are not known. This has resulted in common, templated criteria across clinical trials.30,31 For example, trials in MS have often had common exclusion criteria based on age (<18 or >55 years), level of disability (Expanded Disability Status Scale score > 5.5), and comorbidities (e.g. any other disease or condition that could interfere with participation). 30 This requires clinicians and people with MS who do not meet trial criteria to make decisions about treatment in which benefits and risks are uncertain.
Inclusive practices in trial design that are supported by the US Food and Drug Administration and European Medicines Agency can address challenges related to explicit exclusions on the basis of uncertain risk and benefit. First, eligibility criteria can be modified as the evaluation of an intervention progresses along the clinical trial continuum. For example, age criteria can be broadened in phase 3 trials to include older adults and youth based on accumulated safety and pharmacology data from earlier phase studies, and should not simply be carried forward from earlier phase studies. 32 Second, for individuals with organ dysfunction and other comorbidities, distinctions can be made by severity, so that individuals with mild dysfunction are included, whereas those with moderate or severe dysfunction are excluded. Exclusions for comorbidities should be based on specific rationales, rather than blanket exclusions. For example, excluding individuals with cardiac arrhythmias from trials of fingolimod is justifiable based on the drug’s mechanism of action as a modulator of sphingosine-1-phosphate receptors expressed in cardiac tissue. Third, drug metabolism studies can be conducted earlier in the drug development pipeline to gain a better understanding of the potential for adverse drug reactions in sub-populations such as older adults or those with organ (e.g. liver, kidney) dysfunction that is often relevant to drug safety. Fourth, adaptive clinical trial designs could be used to expand or contract eligibility using pre-specified criteria and interim analyses that rely on safety data accrued during the trial.33,34 Similarly, individuals unlikely to benefit from the intervention could be excluded (discussed further elsewhere). 35 These approaches also address challenges related to conducting clinical trials in pediatric MS, a rare condition which faces difficulties achieving timely adequately powered trials. 36 Specifically, they would allow staggered enrollment of persons with MS aged <18 years, beginning with adolescents followed by younger children as well as older adults. Other strategies to enhance clinical trial design in pediatric MS are discussed in a companion paper. 35 Finally, mixed methods designs may be useful for clinical trials of non-pharmacologic interventions. Qualitative data may provide insight when valid and reliable instruments are lacking for the groups of interest, or when the applicability of concepts or questions to ask are unclear.
Specific action is needed to improve the enrollment of minority racial and ethnic and other disadvantaged populations who are not explicitly excluded. On 29 December 2022, the US Federal Food Drug and Cosmetic Act was amended to require that sponsors submit a race and ethnicity diversity action plan for clinical investigation of a new drug in phase 3 or another pivotal study; prior draft guidance was issued in April 2022. 37 The plan should provide (1) an overview of the disease, including what is known about it in underrepresented populations; (2) the scope of the development program, including planned clinical trials and their design elements, how inclusion of underrepresented populations will be addressed, and summarize data regarding differential treatment response in underrepresented populations; (3) an indication of the underrepresented populations of interest and the justification of specific enrollment targets for these populations; and (4) an operational plan for recruitment and retention that addresses intended site locations, measures to limit participant burden, the community engagement strategy, and the evaluation plan to measure progress.
Barriers to participation of underrepresented/underserved groups
In the context of historical injustices and ongoing experiences of discrimination in research and health care,38,39 multiple potential barriers affect the participation of underserved groups in clinical trials. 40 Broadly, these barriers can be categorized as those related to language and communication, lack of trust, access to trials, eligibility criteria, attitudes and beliefs, lack of knowledge regarding clinical trials, and logistical challenges. 41 Barriers can also be classified by whether they act at the system, individual, or interpersonal levels. 42 System factors, for example, include the availability of trials being limited to tertiary care centers, restrictive inclusion criteria, lack of community engagement, and financial burden related to participation. Individual factors can relate to the patient, such as language, or the provider/investigator, such as implicit bias. Interpersonal factors may include the physician–patient relationship.
A systematic review of 44 studies identified multiple shared barriers to participation across African American, Hispanic/Latinx, Asian American, or Pacific Islander 43 groups. These included mistrust and fear of participation, lack of access to information, competing demands related to time and financial resources, and logistical concerns related to scheduling, childcare, and lack of transportation. Similarly, a recent survey of 2599 persons with MS identifying as African American, White, Hispanic/Latinx, non-Hispanic, non-disclosed, and living in the United States found that all groups supported research. 44 However, research priorities differed as did preferred sources of information about research opportunities and which sources were most trusted. All groups had concerns about potential harms to their health, confusing study information, not being fully informed about a study when participating, and difficulty accessing the study site. Hispanic/Latinx participants and those of undisclosed ethnicity were more concerned about the effects of research participation on employment, legal status, and loss of health insurance compared to non-Hispanic/Latinx participants. As compared to White participants, African American participants were more concerned about privacy, receiving poor quality medical care, and being taken advantage of by the research team, consistent with observations in the general population and stemming from negative historical and sociopolitical perspectives.
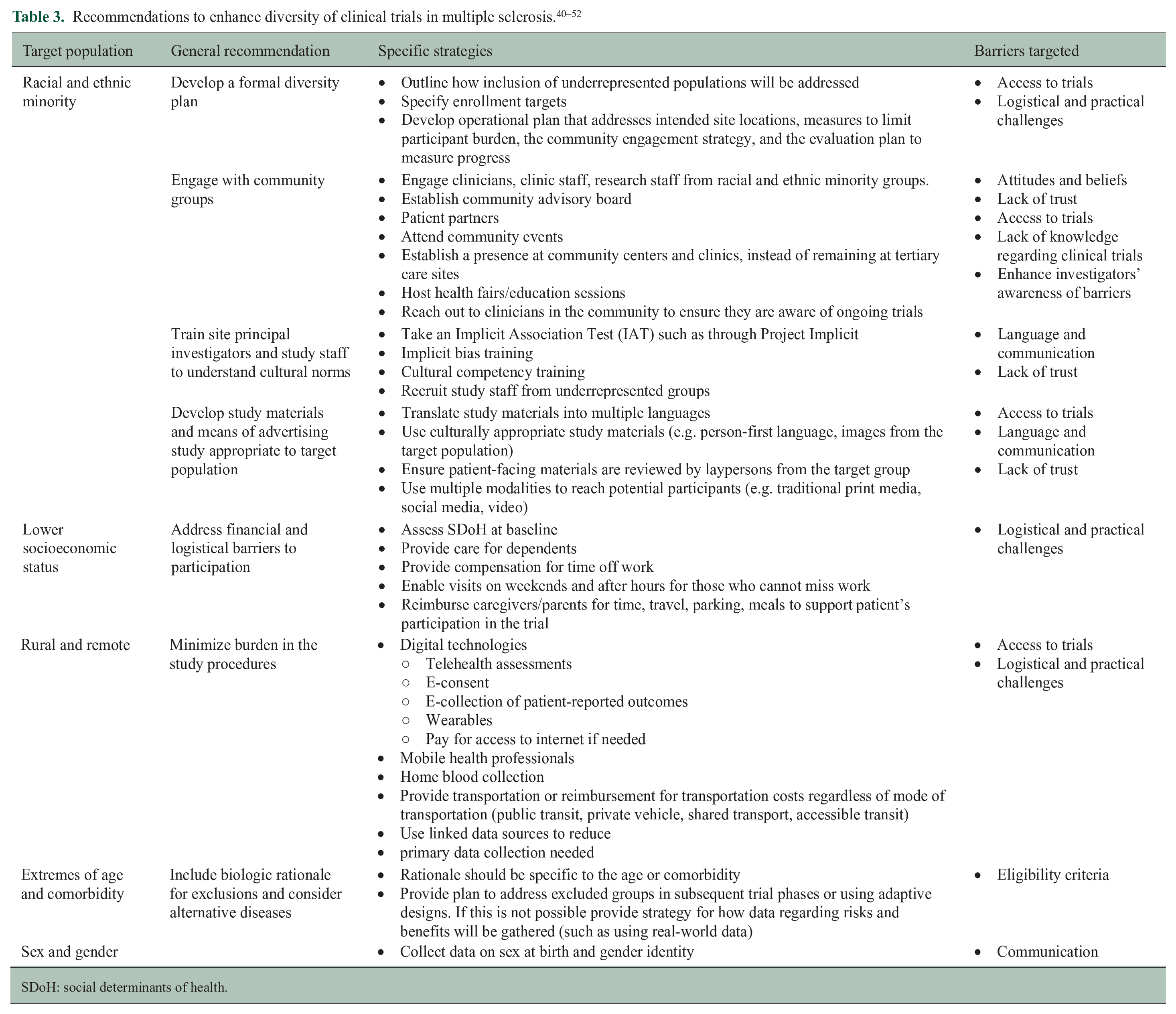
Recommendations
Workshop attendees made recommendations to enhance the diversity and inclusiveness of trials in MS (Table 3). The dimensions of diversity that are relevant may vary with the intervention and by country. 20 For example, the characteristics of people with MS vary by region, and thus the underrepresented groups may also vary. Broadly, sponsors and investigators should develop a formal diversity plan for their trials as described earlier. Sponsors, investigators and their study teams should engage with the community on an ongoing basis, and expand trial sites to underserved communities. Investigators and study staff should undergo cultural competency and implicit bias training, and teams should include people who identify with the underrepresented groups. Explicit eligibility criteria need to be biologically justified and can be modified as the trial progresses using adaptive designs. Sponsors, funders, and investigators must minimize the logistical and practical burdens of study participation to avoid implicit exclusions related to geography and SES. For example, use of digital technology, data collection through linked data sources, conducting evening or weekend study visits, and providing appropriate financial supports may help. 45 Table 3 outlines more recommendations (by population group). Generally, multiple strategies are needed to increase inclusion, and the optimal set of strategies will vary across studies and even across sites (both within and between countries) within multi-site studies.41,46 –51 We also refer the reader to other resources.20,52 Concerns that focusing on enhancing diversity of trial populations is too time-consuming, costly, or may adversely affect trial outcomes due to heterogeneity of treatment effect are not supported by evidence. 53
SDoH: social determinants of health.
Both recruitment methods and the transparent reporting of the characteristics of populations enrolled in clinical trials must improve to ensure trial populations represent those affected by the disease and to measure progress toward the goal of improved diversity and inclusion. Currently, reporting of race and ethnicity information is infrequent and not relevant worldwide, 26 and other SDoH are not reported. 27 The CONSORT-Equity statement, an extension of the CONSORT statement used for reporting of clinical trials, was developed to improve reporting in clinical trials in which health equity is relevant. Health equity was considered to be relevant when the target population was one experiencing social disadvantage, or when heterogeneity of treatment effect between two groups with differing levels of social disadvantage was of interest. The statement used the PROGRESS-Plus framework to define potentially disadvantaged groups. PROGRESS indicates place of residence, race/ethnicity/culture/language, occupation, gender/sex, religion, education, SES and social capital. 54 PROGRESS-Plus adds personal characteristics associated with discrimination (e.g. age), features of relationships between people and their settings, and time-dependent relationships (e.g. recent immigration) that may impose temporary disadvantage. Investigators should report gender, race and ethnicity, education, income, and place of residence for all trials, sex where biologically relevant, and recruitment strategies used. Potential reporting standards are proposed in Table 4.
Participant characteristics relevant to health equity.
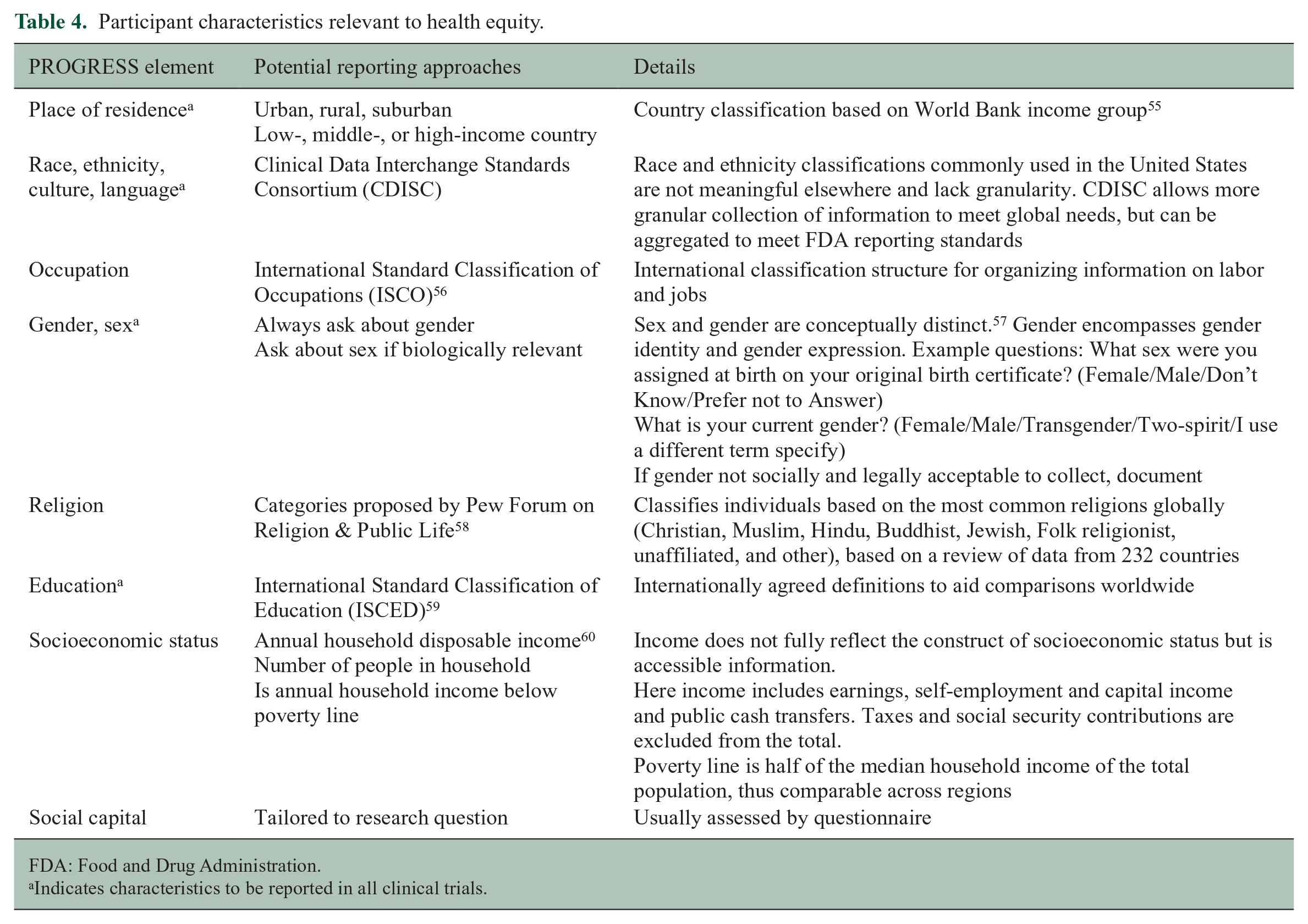
FDA: Food and Drug Administration.
Indicates characteristics to be reported in all clinical trials.
The CHIMES (Prospective Study to Assess Disease Activity and Biomarkers in Minority Participants with Relapsing Multiple Sclerosis After Initiation and During Treatment with Ocrelizumab) trial (NCT04377555) provides a salient example of timely and successful recruitment of underrepresented racial and ethnic minority populations in MS who were not well-represented in the original phase 3 trials. The CHIMES trial is a phase 4 clinical trial study of the safety and efficacy of ocrelizumab in African American and Hispanic/Latinx persons with MS, sponsored by Genentech. 61 The trial was designed collaboratively, engaging people with MS, researchers, and advocacy groups. By design, it addressed historical SDoH that act as barriers to enrollment of underrepresented populations. The trial protocol provides compensation for loss of earnings, transportation to the study site, reimbursement for childcare expenses, reimbursement for travel costs and meals, and greater flexibility with respect to study visits to assist with recruitment and retention of participants.
Conclusion
Clinical trial populations in MS are not adequately diverse to support effective shared decision-making by people with MS and their health care providers. Opportunities exist to refocus and amend trial processes to be more inclusive and equitable. Effective strategies are available to address this deficit, and we recommend concerted action by investigators, funders, advocacy groups, people affected by MS, and ethics committees. More comprehensive reporting of participant characteristics with respect to race, ethnicity, and SDoH will inform these efforts.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231189677 – Supplemental material for Enhancing diversity of clinical trial populations in multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585231189677 for Enhancing diversity of clinical trial populations in multiple sclerosis by Ruth Ann Marrie, Jeremy Chataway, Barbara E Bierer, Marcia Finlayson, Elena H Martinez-Lapiscina, Jennifer Panagoulias, Maria Pia Sormani, Mitzi Joi Williams and Lilyana Amezcua in Multiple Sclerosis Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ruth Ann Marrie receives research funding from: Canadian Institutes of Health Research, Research Manitoba, MS Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, Consortium of MS Centers, the Arthritis Society, U.S. Department of Defense. She is supported by the Waugh Family Chair in Multiple Sclerosis. She is a co-investigator on a study funded in part by Biogen Idec and Roche (no funds to her or her institution). In the last 3 years, Jeremy Chataway has received support from the Efficacy and Evaluation (EME) Programme, a Medical Research Council (MRC), and National Institute for Health Research (NIHR) partnership and the Health Technology Assessment (HTA) Program (NIHR), the UK MS Society, the US National MS Society, and the Rosetrees Trust. He is supported in part by the NIHR University College London Hospitals (UCLH) Biomedical Research Centre, London, UK. He has been a local principal investigator for a trial in MS funded by the MS Canada. He is a local principal investigator for commercial trials funded by Ionis, Novartis, and Roche, and has taken part in advisory boards/consultancy for Azadyne, Biogen, Lucid, Janssen, Merck, NervGen, Novartis, and Roche. Barbara Bierer reports receiving research funding from the National Institutes of Health, US Food and Drug Administration, World Health Organization, Bill & Melinda Gates Foundation, The Greenwall Foundation, and Comprehensive and Integrative Medicine Institute (South Korea). She has served as a consultant on bioethical issues for Merck and Lilly. She serves on the Board of Directors of Clinithink, Edward P. Evans Foundation, Vivli, and North Star Research Board. Marcia Finlayson is a co-investigator on projects funded by the Patient-Centered Outcomes Research Institute, the University Hospitals Kingston Foundation, and the National Multiple Sclerosis Society. She has received consulting/speaker fees from Novartis and Biogen and serves on the editorial board of the IJMSC. Jennifer Panagoulias does not have any disclosures to declare. MP Sormani has received consulting fees from Biogen, Genzyme, GeNeuro, MedDay, Merck, Novartis, Roche, and Teva. Elena Martinez-Lapiscina is an employee of the European Medicines Agency. The views expressed in this article are the personal views of the author(s) and may not be understood or quoted as being made on behalf of or reflecting the position of the European Medicines Agency or one of its committees or working parties. Lilyana Amezcua received personal compensation for consulting, speaking, or serving on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, EMD Serono and research support from the National MS Society, NIH NINDS, Bristol Myer Squibb Foundation, Race to Erase MS and Biogen Idec. She is a local PI for commercial trials funded by Genentech and Sanofi, Genzyme.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The International Advisory Committee on Clinical Trials in Multiple Sclerosis and the International Conference on Innovations in Clinical Trial Design & Enhancing Inclusivity of Clinical Trial Populations were supported by the National Multiple Sclerosis Society and the European Committee for Treatment and Research in Multiple Sclerosis. There was no involvement of the sponsors in the design, collection, analysis, or interpretation of data discussed at the Conference. The opinions expressed are those of the authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.