Abstract
Background:
Over one-third of multiple sclerosis (MS) patients are post-menopausal women, the primary demographic affected by breast cancer. After breast cancer diagnosis, there is little information about patients’ clinical experiences with both diseases.
Objective:
Utilize a case series of MS patients diagnosed with breast cancer to characterize oncologic and MS trajectories, and generate novel insights about clinical considerations using qualitative analysis.
Methods:
A single-center retrospective review was performed on medical record data of patients with MS and breast cancer. Thematic analysis was used to characterize experiences with the concurrent diagnoses.
Results:
For the 43 patients identified, mean age was 56.7 years at cancer diagnosis and MS duration was 16.5 years. Approximately half were treated with MS disease modifying therapy at cancer diagnosis, and half of these subsequently discontinued or changed therapy. Altogether 14% experienced MS relapse(s) during follow-up (with 2 relapses in the first 2 years), with mean annualized relapse rate of 0.03. Cohort Expanded Disability Status Scale (EDSS) scores remained stable during follow-up. Qualitative insights unique to this population were identified regarding immunosuppression use and neurologic symptoms.
Conclusions:
MS relapses were infrequent, and there was modest progression during breast cancer treatment. Oncologic outcomes were comparable to non-MS patients with similarly staged cancer.
Keywords
Background
Women are disproportionately affected by multiple sclerosis (MS) and breast cancer, and the intersection between these two diseases presents many challenging unanswered questions about clinical outcomes, disease trajectory, and appropriate medical management. While commonly diagnosed in young adulthood, 1 over one-third of people living with MS in the United States are post-menopausal women, 2 the same population that is predominantly affected by breast cancer. 3
Epidemiological studies have yielded concerning observations. While it is unclear if breast cancer incidence is greater in women with MS,4–8 they face a worse prognosis with a 28% increased hazard for all-cause mortality than the general population. 9 This raises the question of whether use of immunomodulatory disease modifying therapy (DMT) for MS increases cancer risk or worsens prognosis.10,11 Reassuringly, long-term data for one DMT of concern, ocrelizumab, do not show elevated cancer risk. 12 Breast cancer screening could play a role; breast cancers in MS are less likely to be detected through screening, but delayed detection might not translate to higher cancer stage at diagnosis. 13 Furthermore, the effects of hormonal changes, such as discontinuing menopausal hormone therapy or starting anti-estrogen therapy for breast cancer treatment, on MS trajectory are not understood.
To date, once breast cancer is diagnosed, little is known about MS trajectories or treatment considerations. Here, a case series can provide a complementary approach to large epidemiological studies, by uncovering clinical considerations not typically accounted for. The current study aimed to investigate the experience of women with MS after breast cancer diagnosis by characterizing cancer phenotype and outcomes, MS disability trajectory and treatment during the oncologic treatment period, and by exploring qualitative aspects of the patient experience during oncologic care.
Methods
Study design
This is a single-center retrospective review of data from patients with both breast cancer and MS by 2017 McDonald Criteria cared for in an academic urban center, with local and referral populations. The patient cohort was identified from the Center’s electronic health record (EHR) via a text-based search algorithm modified from prior 14 that includes but is not exclusive to ICD9 codes. Keywords were associated with diagnoses of MS and breast cancer. EHR data regarding oncologic course were entered by an oncologist, and regarding MS course by a neurologist. The initial EHR data extraction yielded 79 patients identified between January 2003 and March 2022 (Figure 1). On manual review, of these, 3 did not have MS and 12 did not have breast cancer. Of the remaining 64, 24 did not have detailed oncologic records (e.g. “breast cancer” listed in medical history without information about type, course, or treatment); most were diagnosed and treated prior to the advent of the EHR or in an outside facility whose EHR was not shared through the “Care Everywhere” interoperability platform (characteristics detailed in Supplemental Table 1). Three patients with MS had breast cancer diagnosed after March 2022 and were subsequently added to this cohort (final n = 43).
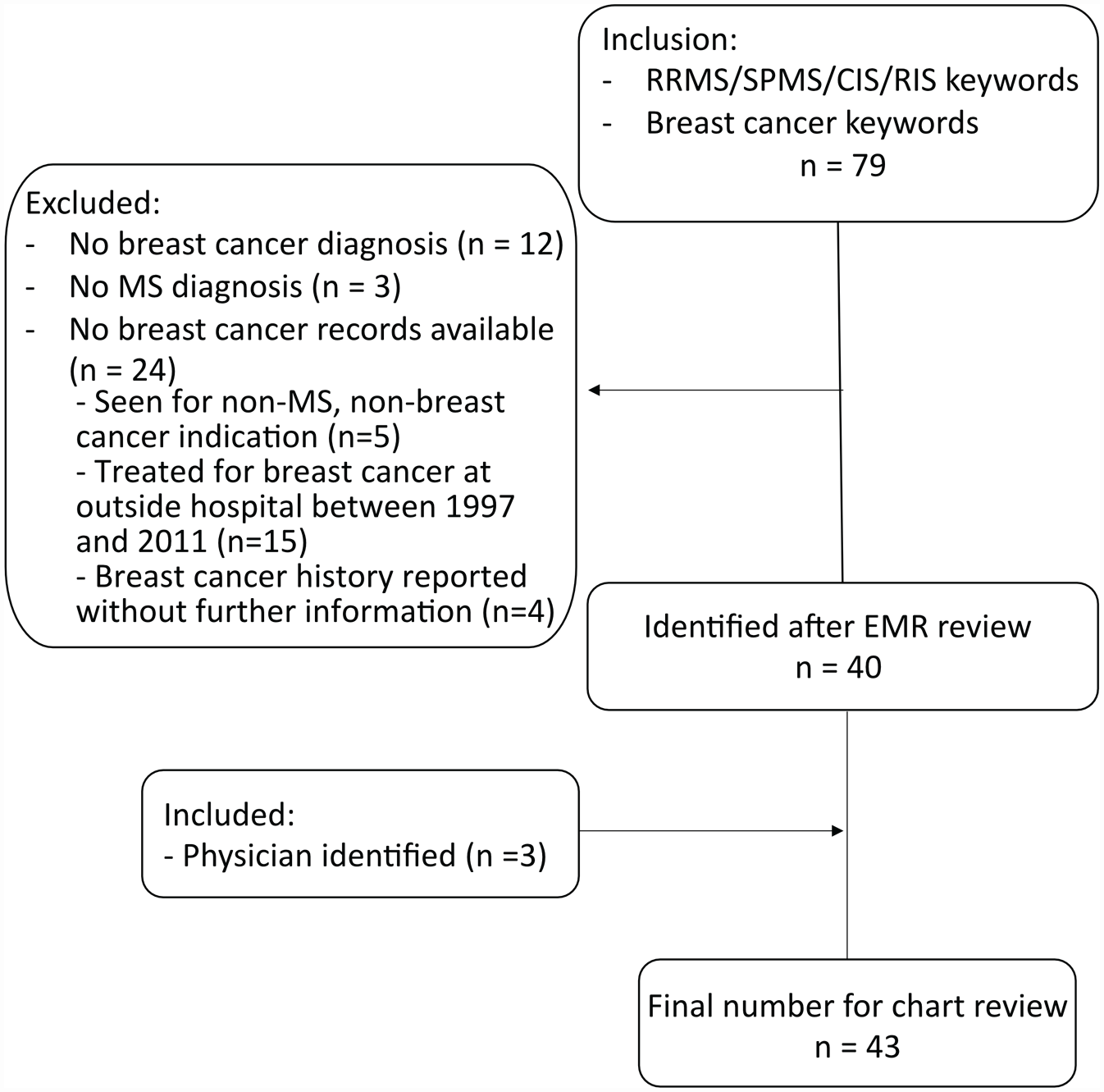
Retrospective review data extraction flow diagram.
Data extraction
Patient records in EPIC, both internal to UCSF and accessible via “Care Everywhere,” were manually reviewed. Data were collected by two investigators using a standardized data collection form.
Qualitative analysis of patient experiences
Qualitative information about patients’ experiences was assessed using thematic analysis, a widely used approach to qualitative research. 16 Clinical notes/communications related to neurology or oncology were read by the first author, who generated preliminary codes and used an inductive approach to identify potential themes. The first and last authors reviewed these codes to define novel or clinically pertinent themes. Records were reviewed in a recursive, iterative process and coded for inclusion of these themes, with ongoing refinement of theme names and definitions. Themes were ranked by frequency of observation in patient charts. Illustrative quotes from patients and physicians were selected.
Statistical analyses
Demographic data and discrete values for breast cancer and MS treatment types are presented descriptively. Individual annualized relapse rates (ARRs) were calculated by dividing each patient’s number of relapses by the time-period of observation in years. EDSS worsening was defined as increase by 1.5 if the EDSS was 0, increase by 1.0 if the EDSS was 1.0–5.0, and increase by 0.5 if the EDSS was 5.5 or higher. 17 Data from a subgroup of female participants in the UCSF EPIC study with age greater than 56 years, at least 5 years of follow-up, and no breast cancer, were extracted and analyzed to provide center-specific context for progression. 17 Statistical analyses of changes in relapses, pseudo-exacerbations, and EDSS scores were analyzed in GraphPad Prism 9.4.0 using Chi-square test for trend or Fisher’s exact test. Intersecting sets of breast cancer treatments were visualized by UpSet plot. A time-to-event survival analysis was conducted for development of metastatic disease using the Kaplan-Meier method with 95% confidence intervals and right censoring.
Ethics and data sharing
The UCSF Committee of Human Research approved the study protocol for retrospective analysis of EHR–derived MS data with no patient contact (Ref #13-11686).
Results
Demographics
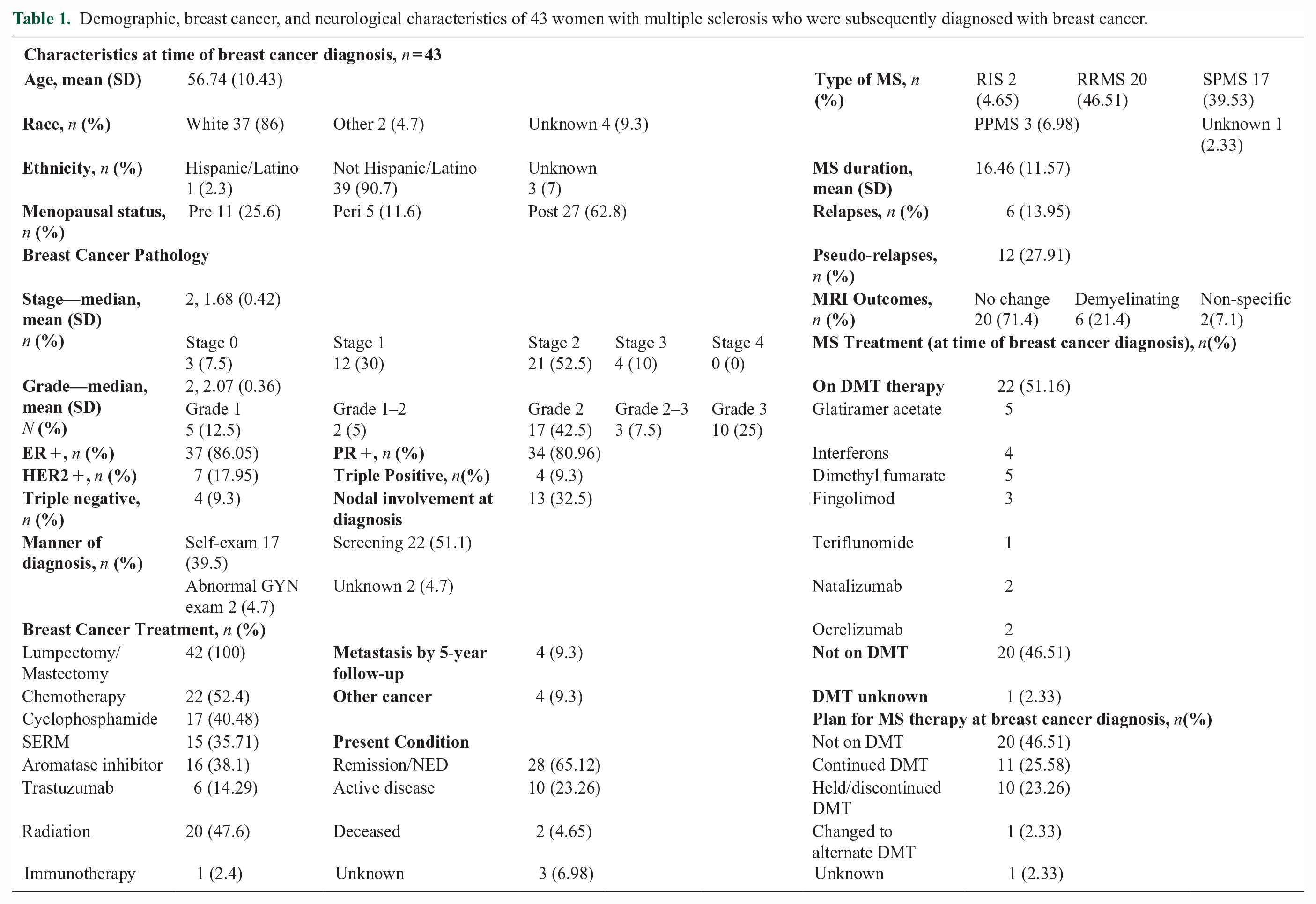
All 43 MS patients identified who developed breast cancer between June 2003 and March 2022 were female. Average age at breast cancer diagnosis was 56.7 (SD = 10.4) years (Table 1); most patients were post-menopausal (62.8%). Most patients were White (86%); only 2.3% were Hispanic/Latino. Patients had a median breast cancer follow-up period of 4.48 years after diagnosis (range = 1–16 years).
Demographic, breast cancer, and neurological characteristics of 43 women with multiple sclerosis who were subsequently diagnosed with breast cancer.
Breast cancer trajectories
Breast cancers were primarily identified by screening mammograms/imaging and self-breast exams (Table 1). Most were stage 2 and grade 2. Notably, 88.3% cancers were hormone receptor (HR)-positive. Only 4 patients (9.3%) had triple-negative disease (HR/HER2-negative).
Oncologic treatment included surgical resection, endocrine therapy, chemotherapy, radiation, and immunotherapy (Table 1, Figure 2(a)). Tumor-targeted therapies were common: 38.1% received aromatase inhibitors (AIs), 35.7% received selective estrogen receptor modulator (SERM) tamoxifen, and 14.3% received trastuzumab for HER2-positive disease. One patient with triple-negative disease received immunotherapy (pembrolizumab).
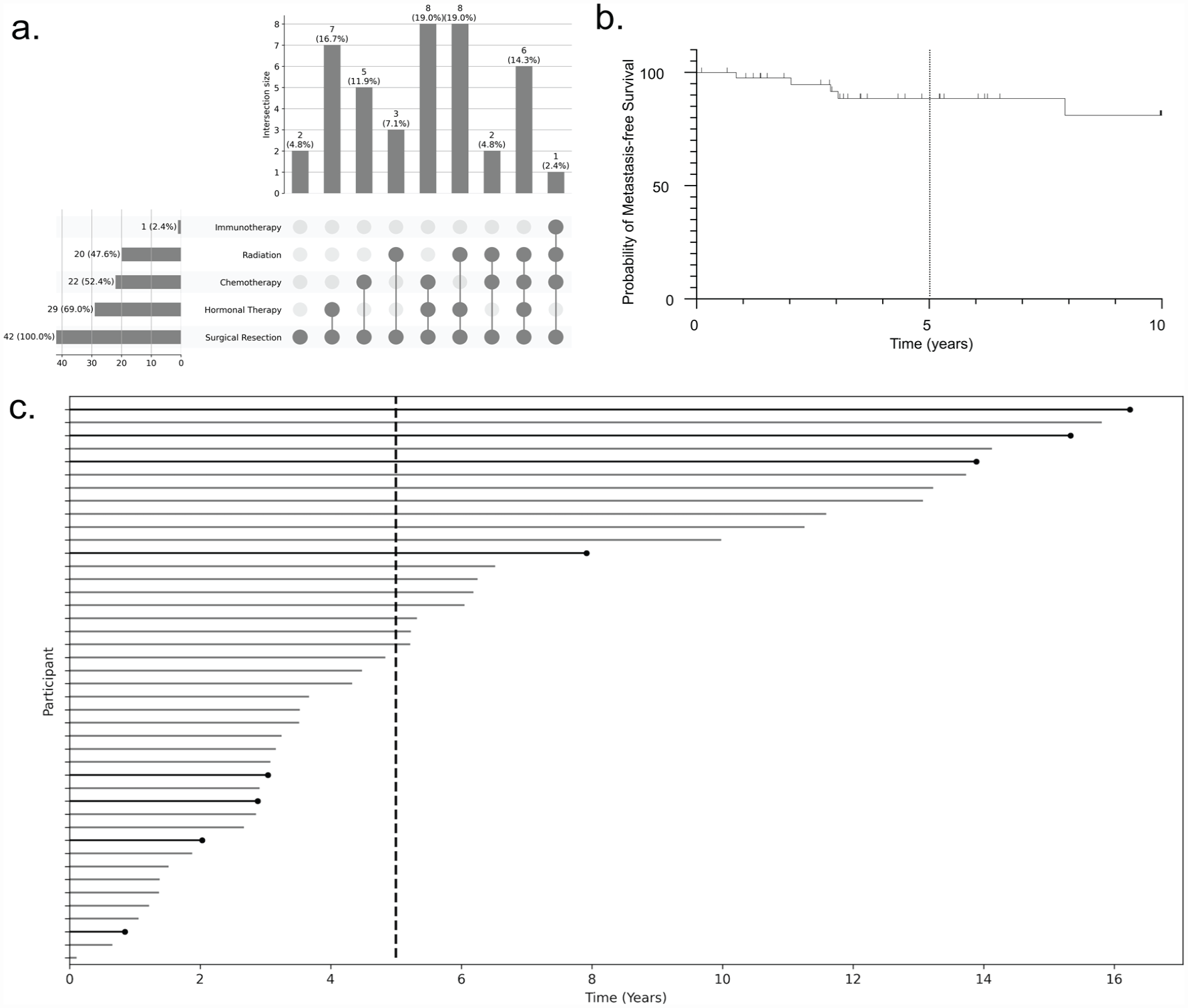
Breast cancer treatments were variable and few patients developed metastatic disease by 5-year follow-up. (a) Overview of breast cancer treatment. Intersecting sets of breast cancer treatments are shown by UpSet plot in 43 women with MS and concurrent breast cancer, (b) time to event plot demonstrating probability of metastasis-free survival, with tick marks representing censored data, and (c) lifetime plot demonstrating variable follow-up lengths and time to development of metastatic disease (line capped with black circle), other lines censored. Dotted line represents 5-year follow-up.
Most patients had no evidence of oncologic disease at most recent follow-up (65.1%), but a substantial minority had active disease (23.3%), two (4.6%) were deceased (mean survival time from diagnosis of 2.83 years); 7.0% were lost to follow-up. Four patients (9.3%) developed metastatic disease by 5-year follow-up; and 8 developed metastases over the full period of review (mean = 6.3 years, SD = 4.9, range = 1–16 years). Of the 4 patients with metastases at 5 years, 3/4 had nodal disease at diagnosis. Given the heterogeneity in follow-up intervals, a time-to-event analysis was conducted: at 5 years the probability of metastasis-free survival was 88.5% (95% CI: 72.0%–95.5%) (Figure 2(b) and (c)).
Four (9.3%) patients subsequently developed a second type of cancer: 2 basal cell carcinomas and 2 melanomas. Of these 4 patients with a second cancer, three were on DMT at breast cancer diagnosis (teriflunomide, dimethyl fumarate, and fingolimod), and 2 continued DMT (teriflunomide and dimethyl fumarate) after diagnosis; one later had an MS relapse, and 2 had EDSS worsening. Regarding breast cancer treatment in this sub-group, all received surgical resection, 50% endocrine therapy, 50% radiation, 25% chemotherapy, and 25% trastuzumab.
MS characteristics
Average MS duration at cancer diagnosis was 17.4 years (SD = 11.5); 46.5% had RRMS and 39.5% SPMS (Table 1). MS outcomes were followed over a median 5 years (mean = 5.1, SD = 3.4, range = 1–19 years); this timeframe slightly differs from breast cancer follow-up due to heterogeneity in oncologic and neurologic follow-up visits.
At cancer diagnosis, 51.2% were taking MS DMT, most commonly glatiramer acetate or dimethyl fumarate (Figure 3(a)), and many remained on the same plan (i.e. DMT or no DMT) after diagnosis (Figure 3(b)). Of the 22 patients on DMT, 11 (50%) changed DMT management at cancer diagnosis: 10 (90.9%) discontinued/held DMT and 1 (9.1%) switched DMT. Notably, 40.5% of all patients received cyclophosphamide for cancer treatment, with at least two treating physicians noting that cyclophosphamide may additionally provide incidental MS treatment.
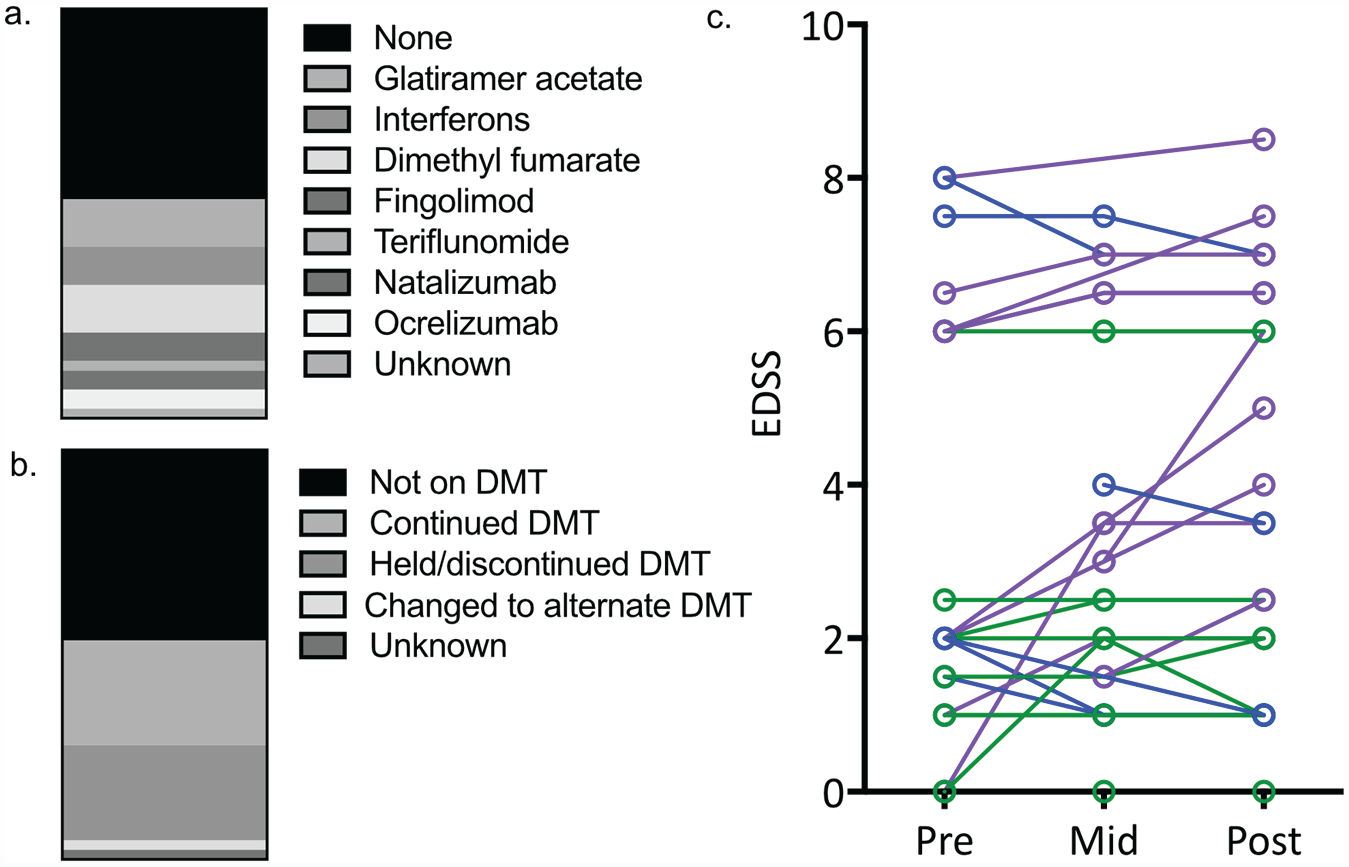
Overview of MS disease modifying therapy (DMT) at time of breast cancer diagnosis and changes in EDSS over breast cancer treatment. (a) Distribution of DMTs at time of breast cancer diagnosis are shown as a proportion of a whole; (b) change in DMT therapy at time of breast cancer diagnosis are shown as a proportion of a whole; and (c) changes in MS-related disability as assessed by the Expanded Disability Status Scale, EDSS), over the course of treatment for breast cancer in 29 women with at least 2 EDSS scores available in the medical records. For visualization purposes, the mid-point of cancer treatment was selected as the neurological visit between 1 to 2 years after initiation of breast cancer treatment. As defined in the manuscript, patients with EDSS worsening are coded in purple, EDSS stability are in green, and EDSS improvement are in blue.
MS relapses and pseudo-exacerbations
Altogether, over the MS follow-up period (mean = 5.1 years), 6 patients (14%) experienced MS relapse(s); mean ARR was 0.03 (SD = 0.07): 0.05 for relapsing patients and 0.01 for progressive patients. Relapses occurred only in patients not on DMT at cancer diagnosis (n = 2) or for whom DMT had been held/discontinued (n = 4, Supplemental Figure 1). Of these 4, 2 discontinued natalizumab (one later started alemtuzumab), 1 discontinued fingolimod (and later started teriflunomide), and 1 discontinued glatiramer acetate. There was no difference in relapses between patients who received cyclophosphamide as part of their breast cancer treatment versus those who did not receive cyclophosphamide. A further 3 patients not on DMT prior to cancer diagnosis later started glatiramer acetate after completing cancer treatment (except ongoing endocrine therapy); 2 of them had experienced an MS relapse.
In the first 2 years post-cancer diagnosis, only 2 patients experienced an MS relapse; both had RRMS; mean MS disease duration was 8.3 years; and mean ARR in these first 2 years after cancer diagnosis was 0.02 (SD = 0.11).
Pseudo-exacerbations occurred in 12 (27.9%) patients, with no significant difference between patients where DMTs were held versus continued. In addition, there was no difference in relapses or pseudo-exacerbations in patients who received hormone treatment versus those who did not.
MRI outcomes
Twenty-eight patients had baseline and follow-up MRIs spanning the breast cancer diagnosis and treatment interval (mean = 4.3 years, SD = 2.3). Most (20/28, 71.4%) had no change noted between baseline and follow-up MRI; 2 had non-specific white matter lesions that were not suspicious for metastatic disease, and 6/28 (21.4%) had likely demyelinating lesions (5 relapsing, 1 progressive).
EDSS scores
For the entire cohort, median EDSS scores remained stable across pre-treatment (2.25, interquartile range [IQR] = 1.5–6), mid-course (3, IQR = 2–6), and follow-up (3, IQR = 2–6) visits, with a median EDSS change of 0.75 over a median follow-up of 5 years (Table 2). Twenty-nine patients had at least 2 EDSS scores available (Figure 3(c)): 12 (41.4%) experienced EDSS worsening 17 independent of relapses experienced, 12 had EDSS stability, and 5 had mild improvement in EDSS scores relative to baseline.
Trajectory of MS-related disability as assessed by changes in EDSS scores during breast cancer treatment.

For the 6 patients with 5 or more years of follow-up, median EDSS change was 0.75 and 3/6 (50%) had EDSS worsening. For context, in the UCSF-based prospective EPIC study, a sub-population of 16 women with mean age of 65.7 years (SD = 7.2) had median EDSS that similarly remained stable from baseline (2, IQR = 1–3) to 5-year follow-up (2.5, IQR = 2–3.5). Median EDSS change was 0.5; 43.8% experienced EDSS worsening.
Qualitative data on patient experience
Thematic analysis performed on medical charts identified 10 themes pertinent to the experiences of the individuals in this case series. These included multiple novel aspects of the experience of patients with MS during breast cancer treatment, clinically complex questions pertaining to the dual diagnoses, as well as echoes of existing oncologic themes. Representative examples are depicted in Figure 4; these are detailed further in Supplemental Table 2.
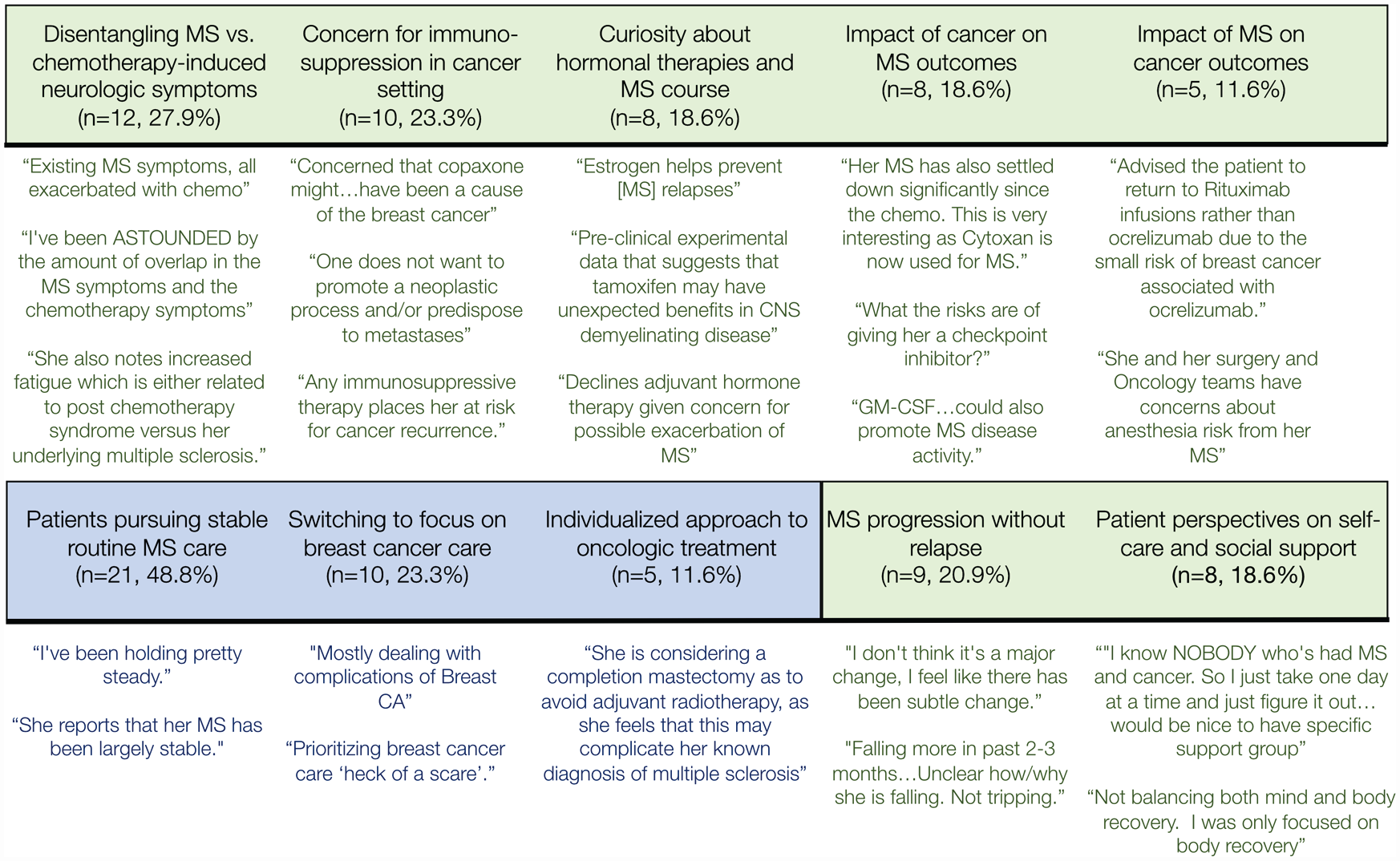
There are multiple novel qualitative themes related to the patient experience with both multiple sclerosis and breast cancer. The green banner and text represents themes novel to the experience of women with neuroinflammatory disease and breast cancer, while the blue banner and text represents themes common to the oncologic literature.
Common oncologic themes
As with other oncologic patients, many patients maintained their usual care of chronic conditions (theme—Patients pursuing stable routine MS care), and a subset reprioritized their interactions with their oncology team, reducing MS visits, but increasing overall contact with the medical system (theme—Switching to focus on breast cancer care).
Oncologic care was not uniform, but tailored to patients’ cancer, health status, and preferences (theme—Individualized approach to oncologic treatment), particularly around risks of worsening MS outcomes with hormone therapy and radiation.
Concerns unique to neuroinflammatory conditions
The typical approach to neurologic symptom management in cancer care was complicated by whether these changes reflected MS relapses, MS progression, or cancer therapy side effects (theme—Disentangling MS vs chemotherapy-induced neurologic symptoms).
Some experienced MS progression independent of relapse (theme—MS progression without relapse). Continuation and selection of MS DMT were additional considerations relating to the intersection of these diseases (theme—Concern for immunosuppression in cancer setting), particularly worries about insufficient anti-tumor response for patients receiving MS DMT. Concerns about impact of cancer-related hormonal treatments on MS course were also identified, including discontinuing menopausal hormone therapies after cancer diagnosis and using anti-estrogen therapies for HR-positive breast cancers (theme—Curiosity about hormonal therapies and MS course).
Finally, with respect to self-care and social support, varying approaches to manage stress and mood changes were identified, with several patients emphasizing difficulties in finding other people with similar experiences (theme—Patient perspectives on self-care and social support).
From these observations, a discussion guide was compiled to prompt discussion between patients and clinicians regarding topics pertinent to breast cancer diagnosis in MS (Table 3).
Conversation guide for patients with MS who are diagnosed with breast cancer and their treating clinicians.

Discussion
To date, epidemiological studies have yielded many observations about breast cancer risk in patients with MS, but once these patients are diagnosed, there is little literature to guide patient and clinician expectations regarding MS course, DMT choices, or qualitative experiences. Reassuringly, in the current case series, the 5-year probability of metastasis-free survival was 88.5%, few MS relapses were reported, and marked disability progression was not observed. Initial qualitative insights were generated pertaining to the experiences and treatment considerations for women with MS after breast cancer diagnosis which can inform individual patient counseling and further research.
At 5 years of follow-up, rates of metastatic breast cancer were similar to previously published rates in this population. 18 The natural history of HR-positive disease is long, with recurrences occurring 20 or more years after diagnosis, 19 and a few patients in this series with longer follow-up times did experience metastases. The reassuring lack of apparent increase in early recurrence could be biased by the mostly low stage, HR-positive disease at onset. Few patients in this series had triple-negative disease, and most treatment was given prior to approval of checkpoint inhibitors for the treatment of triple-negative breast cancer, immunotherapies that could exacerbate existing MS disease or incite new inflammatory demyelination;20–22 further study is recommended.
With respect to MS inflammatory activity, relapses occurred in patients not on DMT at cancer diagnosis or with recent DMT discontinuation. The risk of rebound MS disease when discontinuing fingolimod or natalizumab 23 should be a part of the treatment discussion between oncologists, neurologists, and patients. In other situations, such as pre-conception, a bridging therapy can help reduce risk of relapse after discontinuing DMTs with a high risk of rebound activity. Cyclophosphamide has been used in treatment-refractory MS to reduce relapses, 24 and, if appropriate as part of a chemotherapy plan, may incidentally reduce relapse risk if discontinuing DMTs.
The cohort EDSS was fairly similar before and after cancer diagnosis, and fairly similar to the EPIC subpopulation. Some changes could be spurious—as EDSS scores were not collected prospectively or systemically—but could reflect changes in MS course related to cancer or cancer therapies. 25 Alternatively, EDSS worsening could reflect worsening not specific to MS, but instead symptoms from chemotherapy, radiation treatments, or changes in other physiological domains. Another possible confounder relates to the predominantly post-menopausal status of women in this cohort, where slight acceleration in disability progression has been reported. 26
The effects of hormonal therapies and estrogen modulation on the risk of MS relapses and progression during breast cancer treatment are not well understood. Pre-clinical data suggest that SERMs could also enhance OPC differentiation and promote myelin repair, 27 with theoretical benefits on MS trajectory. Some breast cancer patients discontinue hormone therapies, but clinical data suggest that estriol could reduce MS inflammatory parameters. 28 With few data clearly demonstrating a clinical benefit of hormonal therapies on MS course, treatment decisions should be guided by oncologic indications.
This study has several limitations that limit generalizability of the findings, including modest size, retrospective design, and lack of systemically recorded clinical outcomes. In addition, these data from a quaternary referral center could be biased toward more severe cancers and more active MS. The 86% White population, higher than for our Center overall, 29 could reflect differences in breast cancer risk, differences in access to local vs quaternary health systems for care, or evolving MS demographics. 30 These patients had mild baseline disability, which may not represent the experiences of a higher-risk, more disabled population. Finally, it is unknown whether patients excluded for lack of records in this study may have differed in access to care, socioeconomic background, or disease severity, as most received cancer treatment prior to widespread EHR use.
Overall, the data suggest that MS relapses are infrequent during breast cancer treatment, disability progression is modest, and that while MS symptoms may be complicated by pseudo-exacerbations and sequelae of chemotherapy, many symptoms normalize after treatment is completed. In addition, oncologic outcomes appear to be comparable to non-MS patients with similarly staged cancer. Therefore, while patients would benefit from an individualized approach to their MS DMT management, breast cancer treatment should not be limited due to an MS diagnosis and should be optimized for oncological outcomes. Future research should investigate the use of specific hormonal therapies and comprehensive symptomatic rehabilitation in MS patients after cancer treatment.
Thematic analysis, particularly using an inductive approach, can uncover novel insights regarding treatment and support for patients. Some concerns were common to the oncologic population,31–33 but others were specific to patients with neuroinflammatory disease. For example, many clinicians—both neurologists and oncologists—raised concerns about concurrent immunosuppressive and anti-cancer therapies. Furthermore, when neurologic symptoms arose during breast cancer treatment, expertise was needed to discern between MS relapses, MS pseudo-exacerbations, and new symptoms related to chemotherapy. 34 One important clinical implication of these observations is the need for “warm hand-offs” between oncologists and neurologists to align impressions regarding immune suppression and provide anticipatory guidance regarding a risk of neurologic symptom exacerbation, despite new MS inflammatory activity being typically limited. Experientially, patients articulated a need for emotional and psychological support while navigating treatment for two concurrent illnesses. As telehealth availability increases, telehealth support groups may provide an avenue for patients with comorbid disease processes, disability, or intersecting marginalized identities to access support resources. 35 Future work should focus on identifying specific counseling needs and gaps in social services, as well as the utility of televideo-enabled support groups, to better connect patients to resources during vulnerable periods.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231175975 – Supplemental material for Clinical course of multiple sclerosis and patient experiences during breast cancer treatment
Supplemental material, sj-docx-1-msj-10.1177_13524585231175975 for Clinical course of multiple sclerosis and patient experiences during breast cancer treatment by Alyssa N Nylander, Jessica Singh, Shane Poole, Annika Anderson, Ruth Ann Marrie, Hope Rugo and Riley Bove in Multiple Sclerosis Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AN: AN is funded by a NMSS Clinician Scientist fellowship.
J.S., S.P., A.A.: none.
R.A.M.: RAM receives research funding from: Canadian Institutes of Health Research, Research Manitoba, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, Consortium of MS Centers, the Arthritis Society, US Department of Defense. She is supported by the Waugh Family Chair in Multiple Sclerosis. She is a co-investigator on a study funded in part by Biogen Idec and Roche (no funds to her or her institution).
H.R.: Research support for clinical trials through the University of California, paid to UCSF: Pfizer, Merck, Novartis, Lilly, Roche, Daiichi, Seattle Genetics, Macrogenics, Sermonix, Polyphor, AstraZeneca, Astellas and Gilead. Honoraria from: Puma, Samsung, Chugai, Blueprint and NAPO.
R.B.: R.B. has received research support from NIH, NSF, DOD, NMSS, as well as Biogen, Novartis, and Roche Genentech. She has received consulting or advisory board fees from Alexion, Biogen, EMD Serono, Genzyme Sanofi, Jansen, Novartis, Roche Genentech, TG Therapeutics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by an NMSS Clinician Scientsist Fellowship award, Harry Weaver Scholar Award, and funding from the NIH.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
