Abstract
Background:
The N-MOmentum trial investigated safety and efficacy of inebilizumab in participants with neuromyelitis optica spectrum disorder (NMOSD).
Objective:
Evaluate the attack identification process and adjudication committee (AC) performance in N-MOmentum.
Methods:
Adults (n = 230) with NMOSD and Expanded Disability Status Scale score ⩽8 were randomized (3:1) to inebilizumab 300 mg or placebo. The randomized controlled period was 28 weeks or until adjudicated attack. Attacks were adjudicated according to 18 predefined criteria. Magnetic resonance imaging (MRI) and biomarker (serum glial fibrillary acidic protein [sGFAP]) analyses were performed.
Results:
A total of 64 participant-reported neurological events occurred; 51 (80%) were investigator-determined to be attacks. The AC confirmed 43 of the investigator-determined attacks (84%). There was high inter- and intra-AC-member agreement. In 25/64 events (39%) and 14/43 AC-adjudicated attacks (33%), MRI was reviewed during adjudication. Retrospective analysis revealed new domain-specific T1 and T2 MRI lesions in 90% of adjudicated attacks. Increased mean sGFAP concentrations (>2-fold change) from baseline were observed in 56% of adjudicated attacks versus 14% of investigator-determined attacks rejected by the AC and 31% of participant-reported events determined not to be attacks.
Conclusion:
AC adjudication of NMOSD attacks according to predefined criteria appears robust. MRI lesion correlates and sGFAP elevations were found in most adjudicated attacks.
Keywords
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an autoimmune central nervous system disease characterized by recurrent disabling attacks of optic neuritis, transverse myelitis, and, less commonly, brain or brainstem inflammation.1–3 Up to 90% of patients with NMOSD have pathogenic autoantibodies to the water channel protein aquaporin-4 (AQP4) found on astrocytic foot processes.1,4,5 NMOSD attacks result in destruction of astrocytes and surrounding tissue, are often severe, and recovery is frequently incomplete despite treatment. Attack diagnosis is imperative to monitor treatment response, yet well-defined, universally accepted criteria for accurate and objective diagnosis of NMOSD attacks are lacking. 1
N-MOmentum was a randomized, placebo-controlled, double-blind, phase 2/3 study assessing the efficacy and safety of inebilizumab, an anti-CD19, B-cell depleting antibody, in participants with NMOSD that met the primary endpoint on delaying time to attack. 6 In the absence of consensus criteria for NMOSD attack diagnoses at the time of study initiation, specific criteria were defined to enhance uniform diagnosis of attacks. Eighteen attack criteria were defined by the study steering committee and revised following the US Food and Drug Administration feedback. 1
The aim of the present analysis was to assess the process of attack diagnosis in N-MOmentum. In addition, adjudication committee (AC) performance was evaluated in the context of the potential disease activity biomarker serum glial fibrillary acidic protein (sGFAP), an intermediate filament protein expressed by astrocytes that is released into the parenchymal interstitial fluid following astroglial injury or blood–brain barrier breakdown, and domain-specific MRI findings.7–11
Patients and methods
Trial design
Full details of the N-MOmentum study were previously published. 6 Eligible participants were adults with a diagnosis of NMOSD, an Expanded Disability Status Scale (EDSS) score ⩽8, and a history of either ⩾1 attack requiring rescue therapy in the year before screening or ⩾2 attacks requiring rescue therapy in the 2 years before screening. A total of 230 participants were randomized 3:1 to intravenous inebilizumab 300 mg or placebo on days 1 and 15; the randomized controlled period (RCP) was 197 days or until an AC-adjudicated attack occurred.
Standard protocol approvals, registrations, and participant consents
Participants were screened at 99 outpatient specialty clinics or hospitals in 25 countries. Institutional review boards or ethics committees at each study site approved the protocol. The study is registered with ClinicalTrials.gov (NCT02200770). The study was conducted in accordance with the provisions of the International Council for Harmonisation Guidelines for Good Clinical Practice and the principles of the Declaration of Helsinki. All participants provided written informed consent.
Assessment of NMOSD attack
Predefined attack criteria were used to diagnose each on-study attack (Supplementary Table 1). The 18 criteria covered optic neuritis, myelitis, and brain/brainstem domains.1,6
The attack assessment process has been described previously and is defined in Figure 1(a).1,6 Briefly, potential attacks were evaluated by an on-site investigator, who also decided whether rescue therapy was needed independently of the adjudication status. In addition, the AC independently determined whether the event met the definition of an NMOSD attack in all cases.
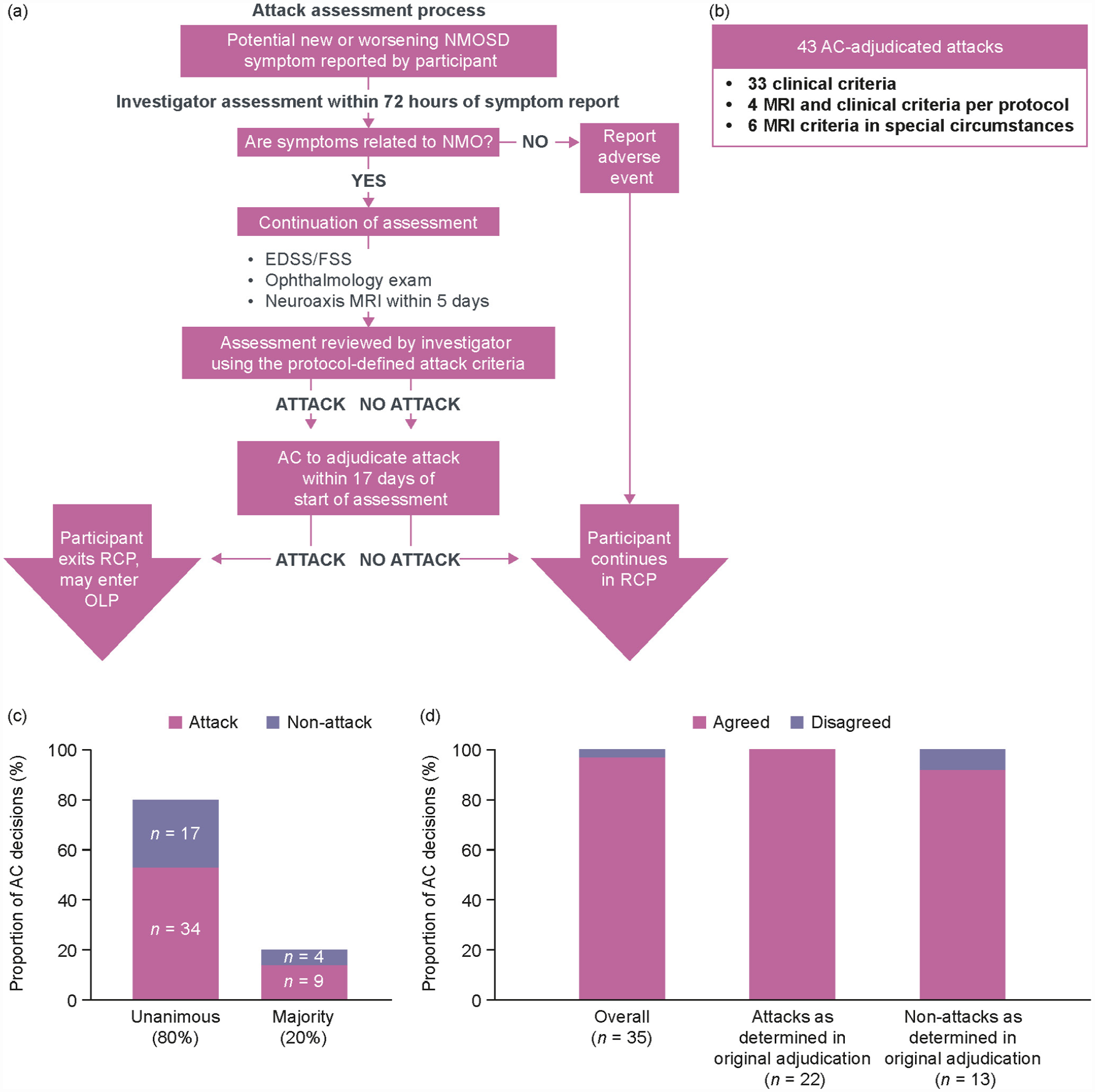
(a) Attack evaluation and adjudication process, and breakdown of (b) attack assessment, (c) inter-member, and (d) intra-member AC decisions.
The AC comprised three physicians (two neurologists and one neuro-ophthalmologist) not involved in the study. For data analysis, cases in which the participant had reported a potential attack that was evaluated as a non-attack by the investigator and AC were labeled “participant-reported symptoms.” Similarly, cases determined to be an attack by the investigator but not by the AC were labeled “investigator-determined attack.” Cases determined to be attacks by the investigator and AC were labeled “AC-adjudicated attack.”
Further information has been included in the Supplementary methods.
MRI assessment
Participants received a full neuroaxis MRI at screening, at the end of the RCP, and at attack assessment visits. All MRI images were sent to the central imaging vendor (NeuroRx, Montreal, QC, Canada) and read by two independent neuroradiologists. In the event of a discrepancy between the readers, a consensus read was performed and reported. Of note, images of the optic nerve were obtained during acquisition of brain scans.
During attack adjudication, the investigator and AC reviewed MRI data of only the relevant neuroanatomic region when the selected attack criteria required MRI review (criteria 9–11, 14–17). In addition, MRI review could be requested by AC members when clinical data were deemed inconclusive or potentially inconsistent. An independent request for MRI review by a majority of AC members resulted in MRI images of the relevant domain being provided to all AC members. An MRI lesion was deemed domain-specific if it correlated with reported attack-related symptoms.
All domain-specific MRI findings were retrospectively reviewed for all potential events. Further information has been included in the Supplementary methods.
Assessment of sGFAP
sGFAP levels were not used for diagnosis of NMOSD attacks in this study. Blood samples were collected from participants during study visits at baseline (day 1) and during any attack assessment visit. sGFAP concentrations were determined by single-molecule arrays (SIMOA; Quanterix, Lexington, MA, USA). Elevated sGFAP was defined as ⩾2 standard deviations above the healthy donor mean concentration (⩾170 pg/mL) according to established laboratory procedures.7,12 To account for variations in baseline levels, changes in sGFAP from baseline were also assessed, with elevations defined as >2-fold change (FC) from baseline.
Statistical methods
Statistical analysis of AC inter-rater reliability was conducted by calculation of Krippendorf’s α (rating of ⩾0.7 denotes good reliability, ⩾0.5 to <0.7 fair reliability, and <0.5 poor reliability). All other data were summarized descriptively.
Data availability
Access to anonymized, individual, and trial-level data may be granted upon reasonable request to qualified researchers for independent scientific research (see the Supplementary methods for further information).
Results
Attack diagnosis
In total, 64 potential attacks were reported by participants during the RCP. Attack criteria were met for 51/64 attacks (80%) as determined by site investigators. The AC reviewed all 64 potential attacks and confirmed 43/51 investigator-determined attacks (84%) (Figure 1(b)). Of the 43 AC-adjudicated attacks, 20 were optic neuritis, 27 myelitis, and one a brainstem attack. Five attacks affected >1 domain (Supplementary Table 2). MRI was reviewed by the AC in 25/64 reported events (39%): 5/13 patient-reported symptoms (38%), 6/8 non-adjudicated attacks (75%), and 14/43 AC-adjudicated attacks (33%).
Reliability of attack identification and adjudication
There was a high degree of inter-AC-member agreement. Of AC decisions, 51/64 were unanimous (80%) and 13/64 were by majority (20%) (Figure 1(c)). A high degree of intra-AC-member reliability was also observed (Figure 1(d)). Pre-planned re-adjudication to assess intra-rater agreement concurred with the initial decision in 34/35 (97%) of cases. In 3/35 cases re-adjudicated (9%), a single adjudicator’s decision was different in the re-review compared with the initial review; however, the AC majority decision was unchanged.
There was good agreement between investigator and AC decisions regarding all potential attacks (56/64, 88%). Eight cases were considered to meet predefined attack criteria by the investigator but not by the AC; 7/8 rejected attacks (88%) were by unanimous AC decision, and for 6/8 rejected attacks (75%), MRI scans were reviewed by all three adjudicators (Supplementary Table 3). No case was adjudicated as an attack by the AC when the site investigator determined that attack criteria were not met. Agreement between AC members indicated good inter-rater reliability (Krippendorff’s α = 0.801), whereas agreement between the AC and investigator was moderate (Krippendorff’s α = 0.686).
Attack incidence and MRI findings
Overall, MRI lesion correlates were found in most adjudicated attacks (37/41, 90%) for which MRI data were available, including those adjudicated without MRI data (23/27, 85%). Of the four adjudicated attacks not found to have MRI lesion correlates, two were adjudicated as optic neuritis and two as myelitis (3/4 in the inebilizumab treatment group).
For cases in which MRI lesion correlates were observed, gadolinium (Gd+) T1 lesions were detected more often than T2 lesions in AC-adjudicated events (Table 1). By contrast, domain-specific MRI findings were identified retrospectively in only 1/8 investigator-determined attacks not confirmed by the AC (a Gd + T1 lesion in the optic nerve from the affected eye in an inebilizumab-treated AQP4-seronegative participant). This participant presented with new blurred vision but had no visual acuity loss and therefore did not meet the attack criteria.
Breakdown of Gd + T1 and T2 domain-specific MRI lesions in participants with adjudicated attacks.

AC: adjudication committee; Gd+: gadolinium; MRI: magnetic resonance imaging.
Excluding four participants without domain-specific MRI findings.
The investigators determined that 13 participant-reported events were not attacks, all confirmed as non-attacks by the AC. A retrospective analysis found that six had new domain-specific MRI lesions. Four participants who complained of sensory symptoms, subjective weakness, and/or ambulatory dysfunction without sufficient findings on neurological exam were found to have spinal cord lesions (one Gd + T1 only, one T2 only, and two with both Gd + T1 and T2 lesions). Two individuals who complained of blurred vision without findings on ophthalmologic exam (high- and low-contrast visual acuity tests and assessment for relative afferent pupillary defect) were found to have Gd + T1 lesions of the affected optic nerve (Figure 2). Of these six individuals with domain-specific MRI lesions, four were in the inebilizumab treatment group and one was AQP4 seronegative.
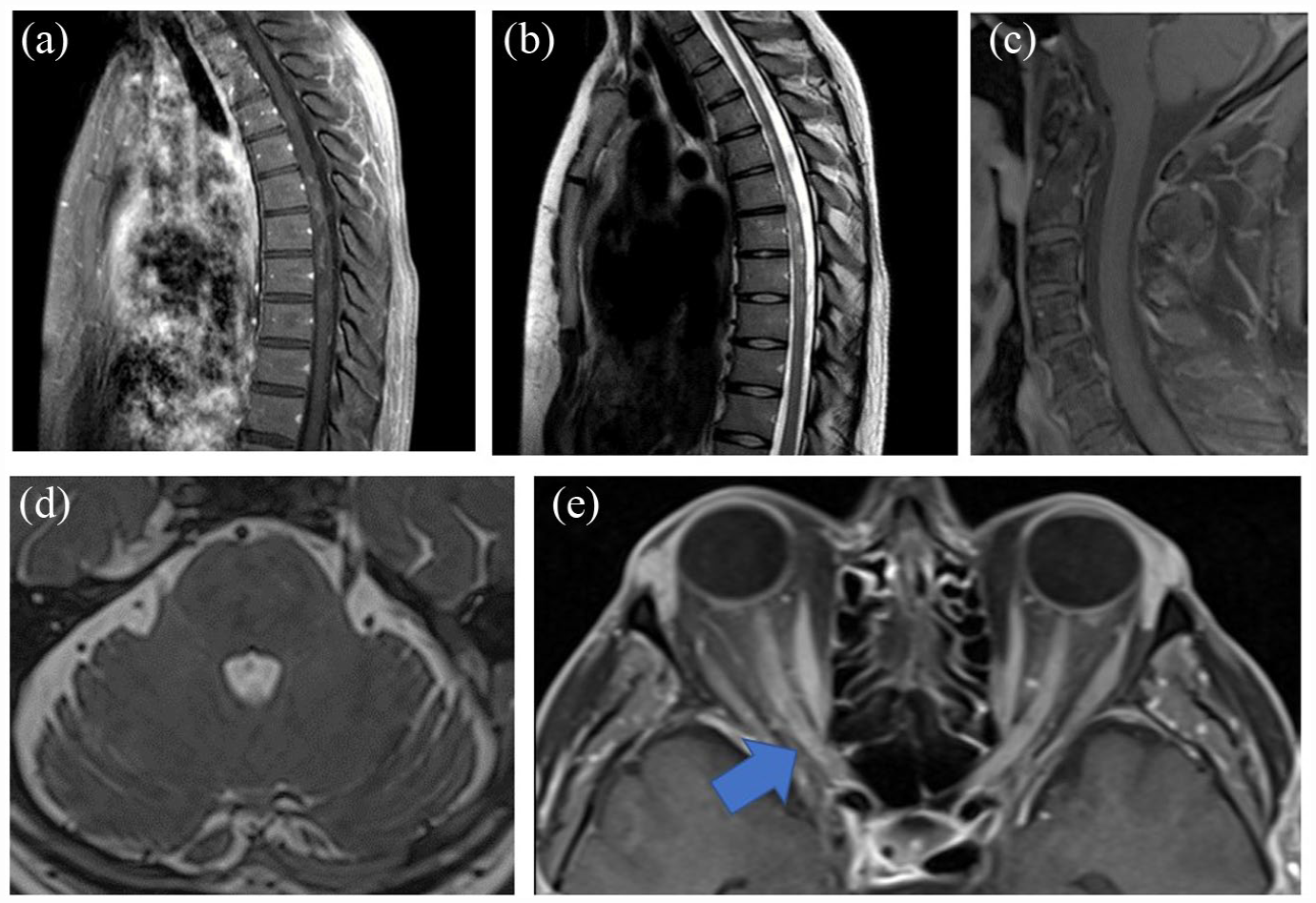
MRI lesions in selected participants with subjective symptoms with insufficient clinical exam findings to satisfy attack criteria. (a) and (b) Gd + T1 and T2 thoracic cord lesion in a participant experiencing abdominal numbness and tightness with no weakness. Exam-notable sensory findings in right trunk. (c)–(e) Participant experiencing nausea (48 h) and worsening of blurred vision in the right eye without change in visual acuity. Gd + T1 sagittal image of cervical spinal cord with partial view of the brain stem (c) and T2 axial of brain stem (d) without clear evidence of lesion. (e) Enhancing lesion of right optic nerve (arrow). MRI of the brain stem was reviewed at the time of adjudication, but not of the optic nerves.
Attack incidence and sGFAP concentrations
The concentration of sGFAP measured during adjudicated attacks was assessed as a potential marker of attack events 7 (Figure 3(a) and Table 2). Levels of sGFAP were elevated (>170 pg/mL) 7 in more AC-adjudicated attacks (29/37, 78%) than in non-AC-adjudicated events (4/7 (57%) investigator-determined but non-AC-adjudicated attacks; 6/13 (46%) participant-reported symptoms). sGFAP concentrations at end of RCP were elevated in 9/26 participants (35%) on placebo and 19/117 (16%) on inebilizumab who did not report new NMOSD symptoms (Figure 3(b)). However, among AC-adjudicated attacks, elevated sGFAP was less common in the inebilizumab than in the placebo group (65% [13/20] vs 94% [16/17], p = 0.048).
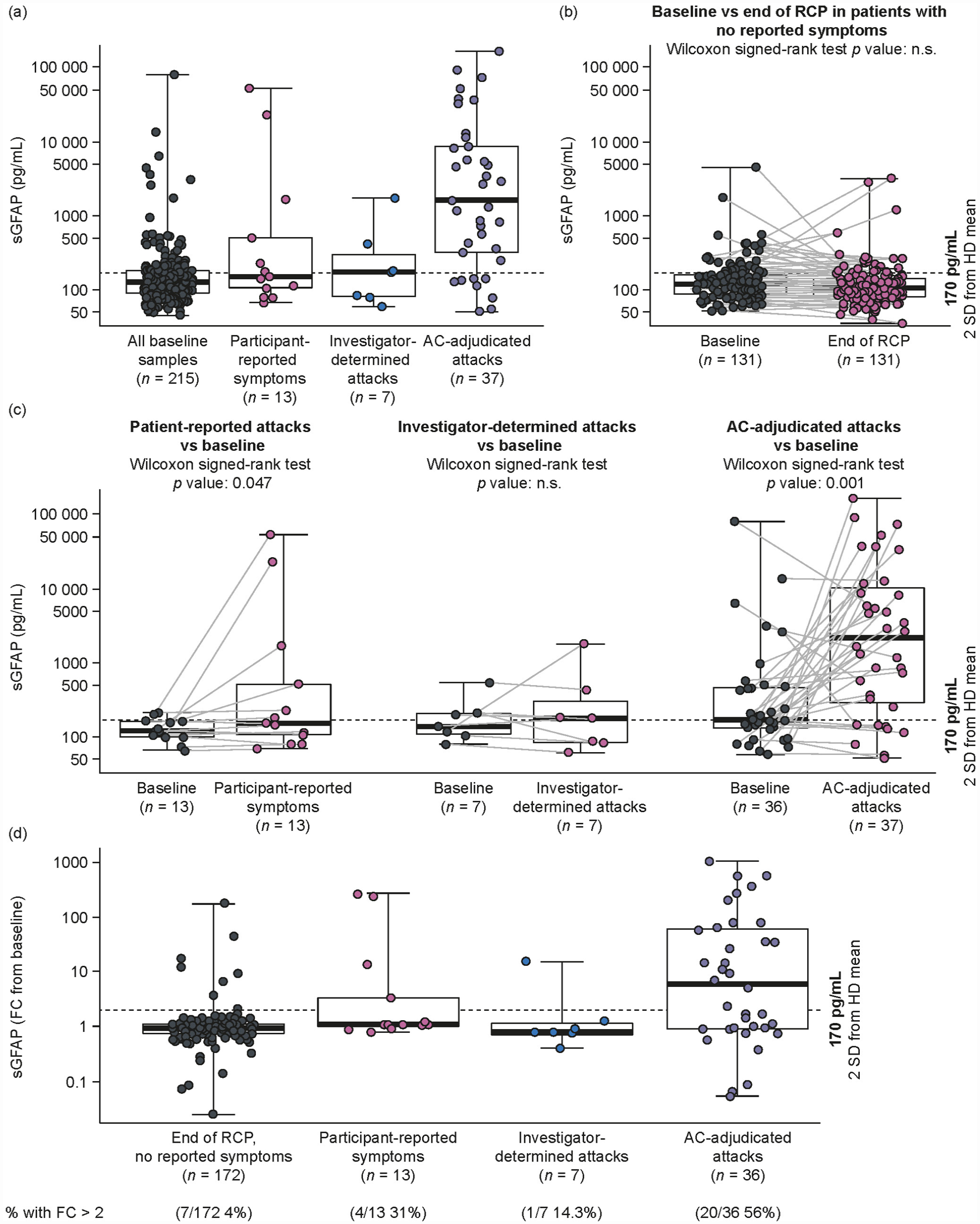
sGFAP concentration versus stages of attack adjudication. (a) Samples drawn during different stages of attack adjudication. (b) Paired baseline and end-of-RCP samples in participants who did not report new NMOSD symptoms. (c) Paired baseline and attack assessment samples at different stages of attack adjudication process. (d) Fold change from baseline in sGFAP in samples drawn during different stages of attack adjudication.a
Domain-specific MRI lesions and elevated sGFAP levels by attack status and treatment group.
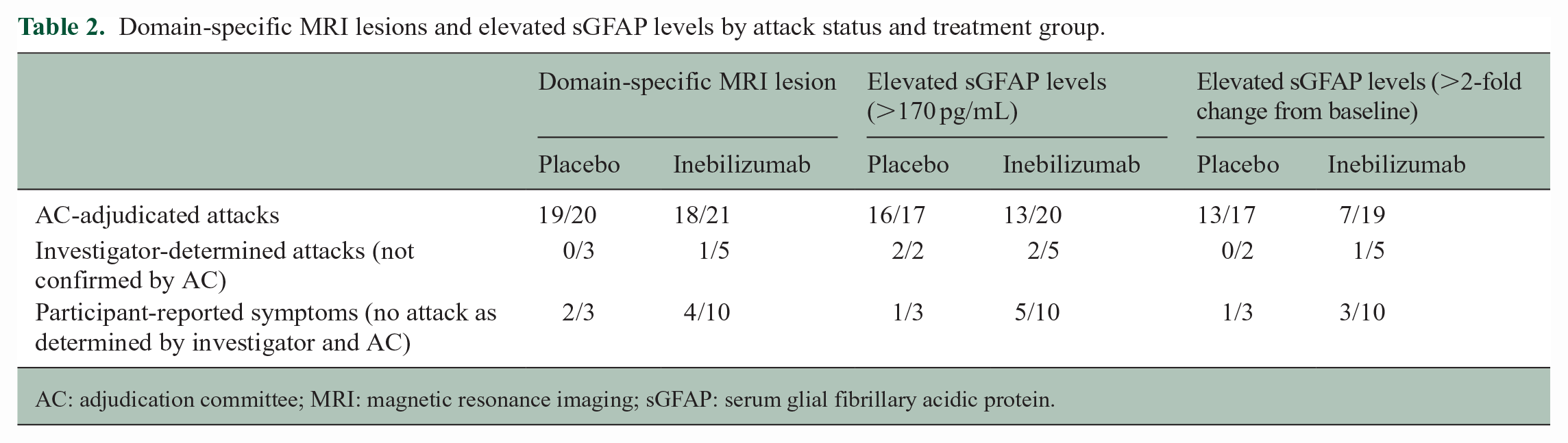
AC: adjudication committee; MRI: magnetic resonance imaging; sGFAP: serum glial fibrillary acidic protein.
Several participants had elevated sGFAP at baseline; thus, sGFAP data were also analyzed relative to baseline concentration (Figure 3(c)). A higher proportion of participants with AC-adjudicated attacks had increased sGFAP (>2-FC from baseline) at the time of event (20/36, 56%) than participants with non-AC-adjudicated events (1/7 [14%] investigator-determined attacks; 4/13 [31%] participant-reported symptoms; 7/172 [4%] participants with no new reported symptoms; Figure 3(d)). When compared with participants with no new symptoms at end of RCP, sGFAP was significantly increased in participants with AC-adjudicated attacks or participant-reported symptoms, but not those with investigator-determined attacks (Table 3 and Figure 3(d)). For AC-adjudicated events, elevated sGFAP was notably less frequent with inebilizumab than with placebo: 37% of participants on inebilizumab had sGFAP > 2-FC from baseline compared with 76% on placebo. 7
Comparison of number of participants with elevated sGFAP levels by attack status.
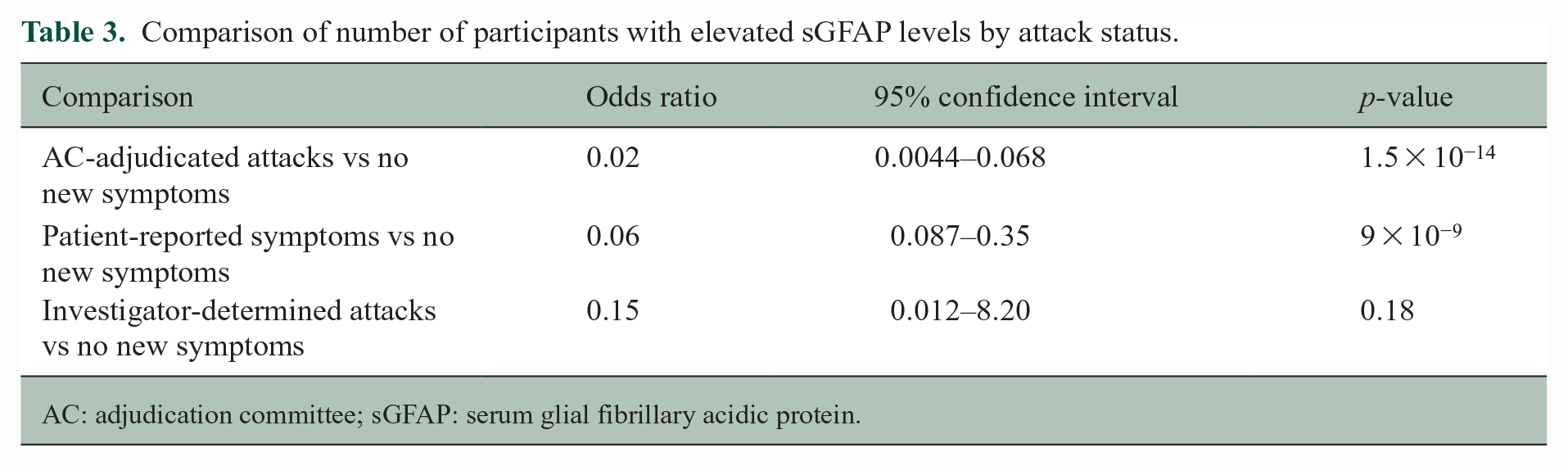
AC: adjudication committee; sGFAP: serum glial fibrillary acidic protein.
The proportion of AC-adjudicated attacks with MRI lesions and sGFAP elevation is shown in Figure 4.
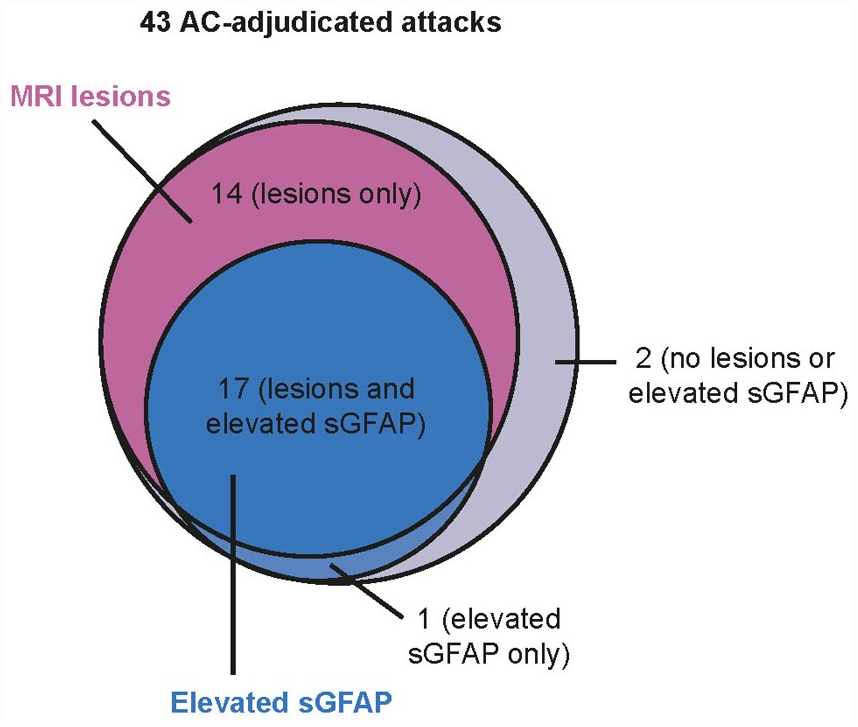
Proportion of investigator-reported and AC-adjudicated attacks with sGFAP elevation (>2-fold change) and MRI lesions.
Of eight investigator-determined attacks rejected by the AC, one had a domain-specific MRI lesion, and one had a 2-FC elevation from baseline in sGFAP. None had both domain-specific MRI finding and sGFAP elevation. One participant had missing sGFAP data. Of the 13 participant-reported symptoms that were rejected by both the investigator and the AC, four had domain-specific MRI lesions and sGFAP elevation from baseline, two had domain-specific MRI findings only, and none had isolated sGFAP elevations from baseline; none had missing data (Supplementary Appendix 1).
Discussion
When the N-MOmentum study was designed, there were no standardized clinical criteria for diagnosing NMOSD attacks. 1 The need for a reproducible, formal process to diagnose NMOSD attacks emerged during the PREVENT trial when lack of standardized criteria led to the formation of an AC after 88 patients were already enrolled. 13 In contrast, the N-MOmentum investigators developed predefined criteria for diagnosing an NMOSD attack, and an AC was constituted to standardize assessments.1,6 To date, N-MOmentum remains the only pivotal phase 3 NMOSD trial to integrate MRI criteria into attack assessments, allowing inclusion of attacks for which exam findings were inconclusive.
Across the three pivotal NMOSD trials, investigating eculizumab, satralizumab, and inebilizumab, there was variation in protocol definitions of NMOSD attack, the process of attack adjudication, inclusion criteria related to prior treatment, participant relapse history, and allowance for concomitant immunotherapy.6,13–15 Thus, while reductions in attack risk were evident across all three trials, direct comparison may be misleading. Indeed, the differences in attack diagnosis across these trials highlight inherent difficulties in diagnosing attacks. 16 The definition of NMOSD attack is essential for the accurate and consistent assessment of treatment effects, and the provision of detailed, predefined attack criteria helps to reduce this variability. In the N-MOmentum trial, the combination of predefined diagnostic criteria and the adjudication process proved critical in providing robust determination of attacks. Only 16% of investigator-determined attacks were rejected following AC review, and no investigator-rejected attack decisions were reversed following adjudication. The high degree of agreement between on-site investigators and AC underscores the importance of detailed, predefined criteria in reliably and reproducibly diagnosing attacks.
Expert adjudication of on-study attacks was also applied in the pivotal trials of eculizumab and satralizumab.13–15 As with N-MOmentum, potential attacks were adjudicated centrally according to objective changes in neurological examinations; however, attack definitions in the eculizumab study were not defined. In the eculizumab study, 21/45 investigator-determined attacks (47%) (11/14 [79%] in the eculizumab arm and 10/31 [32%] in the placebo arm) were rejected by the AC compared with only 16% in N-MOmentum. Most of these investigator-determined attacks (18/21, 86%) were rejected by the AC owing to inconclusive exam findings. Sensitivity analyses demonstrated that the adjudication process significantly influenced the observed hazard ratio (HR) versus placebo (HR = 0.18 for investigator-determined attacks vs 0.06 for adjudicated attacks). 13 Similarly, sensitivity analysis of the trial of satralizumab monotherapy demonstrated that the adjudication process significantly influenced the observed HR versus placebo. 14 When satralizumab was investigated with background immunotherapy, there was a considerable difference in the determination of attacks by central adjudication compared with initial clinical interpretation; the percentage of treated participants remaining attack-free at 48 weeks was 89% versus 69%, respectively. 15 By comparison, in N-MOmentum, sensitivity analyses revealed similar HRs for inebilizumab versus placebo for investigator-determined attacks (0.323) and AC-adjudicated attacks (0.272) and the difference between AC adjudication and investigators in the determined proportion of inebilizumab-treated participants remaining attack-free at the end of the N-MOmentum RCP differed by only 2.5%.6,17
The evidence of the robust and internally consistent nature of the attack criteria and adjudication process developed for N-MOmentum suggests that a set of unified and widely accepted criteria could promote consistency of attack monitoring across future studies and support clinical management. However, our results suggest that including MRI findings could potentially improve the sensitivity of attack diagnosis. In N-MOmentum, MRI was needed for confirmation of 33% of adjudicated attacks for which clinical findings were inconclusive. Without MRI, these attacks may not have been included. However, the use of MRI in the setting of suggestive symptoms, even in the absence of objective findings, may have improved the sensitivity and objectivity of attack diagnosis further given that retrospective analysis of MRI findings revealed four adjudicated attacks that did not have lesion correlates. It would be worthwhile investigating whether instances of attacks without lesion correlates are associated with T2 lesions that are identifiable on spinal cord MRI at subsequent time points. There was also evidence of a domain-specific MRI lesion in 1/8 investigator-determined attacks rejected by the AC and 6/13 potential attacks rejected by the investigator and AC for which participants had minimal symptoms with no or minimal changes in functional and EDSS scores. Concerns of regulators about the use of a non-clinical measure limited the incorporation of MRI in this study to situations in which MRI was supportive of objective findings that were considered equivocal, but it was not permitted as a “standalone” indicator of an attack. The outcomes of the trial are designed to optimize specificity over sensitivity, minimizing false positive results. However, incorporation of MRI assessments might be considered for future studies and in clinical management of patients with new or worsening symptoms. Nonetheless, owing to the limited availability of MRI in worldwide clinical settings, 18 establishing a set of robust and specific clinical criteria for the diagnosis of NMOSD attacks could improve the sensitivity of attack adjudications in routine clinical care.
The results from this analysis may also support a potential role for sGFAP, a biomarker of astrocytic damage, in the evaluation of potential NMOSD attacks. Elevations in sGFAP correlate with attack risk, 7 and were detected in the majority of participants with adjudicated attacks, suggesting that sGFAP concentrations correlate well with AC-adjudicated attacks. Among these, elevations in sGFAP were notably less common in the inebilizumab than in the placebo group. This is consistent with previous demonstration of a reduction in sGFAP with inebilizumab in participants without an adjudicated attack, and fewer inebilizumab-treated participants with raised sGFAP compared with participants receiving placebo. 7 Increased sGFAP (>2-FC from baseline) was also observed in 1/7 participants with investigator-determined attacks (non-AC-confirmed) and 4/13 participants with reported symptoms that were deemed not attacks by both investigators and AC. Given the previously documented favorable effects of inebilizumab on sGFAP levels and the significant reduction in risk of attack with inebilizumab versus placebo, it is possible that these observed cases in the inebilizumab group may be relatively mild.6,7 It should be noted that, although a modest but clear correlation of sGFAP level with age has been observed in both patients with NMOSD and healthy individuals, 7 no age-dependent cut-off was applied in this study. However, the healthy donor reference population was age- and sex-matched. Overall, the evidence of elevated sGFAP in attacks suggests that this biomarker could potentially play a role in future attack assessment with the caveat that elevations in sGFAP were not routinely observed in inebilizumab-treated participants. It is clear that sGFAP represents a clinical signal in NMOSD, but its utility as a biomarker needs further exploration.
The attack criteria used in this study were novel, albeit based on commonly used procedures in clinical practice, and a formal validation had not taken place at the time of the study. The criteria were developed by a group of experts in the field, and regulatory authorities, participant organizations, and the NMOSD medical/scientific community were engaged in review of the study design, including the attack adjudication process. 1 Although the relationship between MRI lesions and clinical events is incompletely understood and validation in follow-up studies and other data sets is warranted, the current results suggest that incorporation of MRI lesion activity, together with introducing biomarker assessments such as sGFAP, may help to improve the sensitivity of attack-diagnosis criteria, but would need to be carefully assessed to be sure that such incorporation did not compromise specificity. Even so, the criteria applied in the N-MOmentum study appear to have provided relatively robust and consistent assessment of attacks in the hands of the study investigators and especially the AC.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231172145 – Supplemental material for Attack adjudication in neuromyelitis optica spectrum disorder: Substantiation of criteria by magnetic resonance imaging and biomarkers in N-MOmentum
Supplemental material, sj-docx-1-msj-10.1177_13524585231172145 for Attack adjudication in neuromyelitis optica spectrum disorder: Substantiation of criteria by magnetic resonance imaging and biomarkers in N-MOmentum by Brian G Weinshenker, Dean M Wingerchuk, Ari J Green, Jeffrey L Bennett, Ho Jin Kim, Sean J Pittock, Kazuo Fujihara, Friedemann Paul, Gary Cutter, Romain Marignier, Orhan Aktas, Hans-Peter Hartung, Dewei She, Michael Smith, William Rees, Kristina Patterson, Daniel Cimbora, Eliezer Katz and Bruce AC Cree in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors thank all the participants of the N-MOmentum trial. This manuscript was submitted on behalf of the principal investigators of the N-MOmentum study group, 6 who administered the clinical trial. Writing and editing assistance were provided by Ester Baixauli PhD and Jenny Thorp of Oxford PharmaGenesis.
Author Contributions
On behalf of the N-MOmentum study investigators (![]() ),
6
B.G.W., D.M.W., A.J.G., J.L.B., H.J.K., S.J.P., K.F., F.P., G.C., R.M., O.A., H.-P.H., D.S., M.S., W.R., K.P., D.C., E.K., and B.A.C.C. were involved in design/conceptualization of the study; acquisition, analysis/interpretation of data; and drafting/revising the manuscript for intellectual content.
),
6
B.G.W., D.M.W., A.J.G., J.L.B., H.J.K., S.J.P., K.F., F.P., G.C., R.M., O.A., H.-P.H., D.S., M.S., W.R., K.P., D.C., E.K., and B.A.C.C. were involved in design/conceptualization of the study; acquisition, analysis/interpretation of data; and drafting/revising the manuscript for intellectual content.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.G.W. receives payments for serving as chair of attack adjudication committees for clinical trials in NMOSD for Alexion, MedImmune, UCB Biosciences, and Viela Bio/Horizon Therapeutics; has consulted with Chugai, Genentech, Mitsubishi Tanabe Pharma, and Roche Pharmaceuticals regarding clinical trial design for NMOSD; has received speaking fees from Genentech, Horizon Therapeutics, and Roche; and has a patent for NMO-IgG for diagnosis of neuromyelitis optica, with royalties paid by Hospices Civils de Lyon, MVZ Labor PD Dr. Volkmann und Kollegen GbR, Oxford University, and RSR. D.M.W. reports personal fees from Arcus Medica, Biogen, Celgene, Genentech, MedImmune, Novartis, Reistone, TG Therapeutics, and Third Rock Ventures; research support paid to Mayo Clinic by Alexion Pharmaceuticals and Terumo BCT; and serves on a clinical trial adjudication committee for MedImmune and Viela Bio. A.J.G. reports grants from Conrad N. Hilton Foundation and Tom Sherak MS Hope Foundation; other financial relationships (for activities as expert witness, associate editor, advisory board/steering committee participation, and endpoint adjudication) with Bionure, Inception Sciences, JAMA Neurology, MedImmune/Viela Bio, Mylan, Synthon, and Trims Pharma; and personal fees from and other financial relationships with Pipeline Therapeutics. J.L.B. reports payment for study design/consultation from MedImmune/Viela Bio; personal fees from AbbVie, Alexion, Antigenomycs, BeiGene, Chugai, Clene Nanomedicine, EMD Serono, Genentech, Genzyme, Mitsubishi Tanabe Pharma, Novartis, Reistone Bio, Roche, and TG Therapeutics; grants from the National Institutes of Health, Novartis, and Mallinckrodt; and has a patent for Aquaporumab issued. H.J.K. has received a grant from the National Research Foundation of Korea; and consultancy/speaker fees from Alexion, Celltrion, Eisai, HanAll BioPharma, Merck Serono, Novartis, Sanofi Genzyme, Teva-Handok, and Viela Bio; serves on a steering committee for MedImmune/Viela Bio; and is a co-editor for the Multiple Sclerosis Journal and an associate editor for the Journal of Clinical Neurology. S.J.P. reports grants, personal fees, and non-financial support from Alexion Pharmaceuticals, Inc.; grants from Autoimmune Encephalitis Alliance, Grifols; grants, personal fees, non-financial support, and other from MedImmune and Viela Bio; consulting support from Astellas; personal fees for consulting services from UCB; and has a patent # 9,891,219 (Application#12-573942) “Methods for Treating Neuromyelitis Optica (NMO) by Administration of Eculizumab to an individual that is Aquaporin-4 (AQP4)-IgG Autoantibody positive.” K.F. serves on scientific advisory boards for Alexion, Bayer Schering, Biogen Idec, Chugai, MedImmune, Merck Serono, Mitsubishi Tanabe Pharma, Nihon Pharmaceutical, Novartis, Ono, and Viela Bio; has received funding for travel and speaker honoraria from Asahi Kasei Medical, Astellas, Bayer Schering, Biogen Idec, Daiichi Sankyo, Dainippon Sumitomo, Eisai, Mitsubishi Tanabe Pharma, Nihon Pharmaceutical, Novartis, and Takeda; and research support from Asahi Kasei Medical, Bayer Schering, Biogen Idec, Chemo-Sero-Therapeutic Research Institute, Chugai, Genzyme Japan, the Ministry of Education, Culture, Sports, Science and Technology of Japan, the Ministry of Health, Welfare and Labor of Japan, Mitsubishi Tanabe Pharma, Nihon Pharmaceutical, Ono, Teijin, and Teva. F.P. has received research support, speaker honoraria, and travel reimbursement from Bayer, Biogen Idec, Merck Serono, Novartis, Sanofi Genzyme, and Teva; is supported by the German Competence Network for Multiple Sclerosis and the German Research Council (DFG Exc 257); received travel reimbursement from the Guthy–Jackson Charitable Foundation; and serves on the steering committee of the OCTIMS study sponsored by Novartis. G.C. has received personal fees for participation on Data and Safety Monitoring Boards from AI therapeutics, AMO Pharma, Applied Therapeutics, AstraZeneca, AveXis Pharmaceuticals, BioLineRx, Brainstorm Cell Therapeutics, Bristol Myers Squibb/Celgene, CSL Behring, Galmed Pharmaceuticals, Green Valley Pharma, Horizon Therapeutics, Immunic, Karuna Therapeutics, Mapi Pharma Ltd., Merck, Mitsubishi Tanabe Pharma Holdings, NHLBI (Protocol Review Committee), Novartis, Opko Biologics, Prothena Biosciences, Reata Pharmaceuticals, Regeneron, Sanofi-Aventis, Teva Pharmaceuticals, University of Pennsylvania, University of Texas Southwestern, and Visioneering Technologies, Inc.; personal fees for consulting or advisory board participation from Alexion, Antisense Therapeutics, Biogen, Clinical Trial Solutions LLC, Entelexo Biotherapeutics, Inc., Genentech, Genzyme, GW Pharmaceuticals, Immunosis Pty Ltd, Immunic, Klein-Buendel Incorporated, Merck/Serono, Novartis, Perception Neurosciences, Protalix Biotherapeutics, Regeneron, Roche, and SAB Biotherapeutics; is employed by the University of Alabama at Birmingham; and is President of Pythagoras, Inc., AL, USA. R.M. serves on scientific advisory boards for MedImmune and Viela Bio; and has received funding for travel and honoraria from Biogen Idec, Merck Serono, Novartis, Roche, Sanofi Genzyme, Teva, and Viela Bio. O.A. reports grants from the German Ministry of Education and Research (BMBF) and the German Research Foundation (DFG); grants and personal fees from Biogen, Genzyme, Novartis, and Teva; and personal fees from Alexion, Almirall, Horizon Therapeutics, Merck Serono, and Roche, and is a member of the European Reference Network for Rare Eye Diseases (ERN-EYE), co-funded by the Health Program of the European Union under the Framework Partnership Agreement No 739534 `ERN-EYE’. H.-P.H. has received fees for consulting, speaking, and serving on steering committees from Bayer Healthcare, Biogen Idec, Celgene Receptos, CSL Behring, GeNeuro, Genzyme, Horizon Therapeutics (formerly Viela Bio) MedDay, MedImmune, Merck Serono, Novartis, Roche, Sanofi, and TG Therapeutics, with approval by the Rector of Heinrich Heine University Düsseldorf. D.S., M.S., W.R., K.P., and D.C. are employees of Horizon Therapeutics (formerly Viela Bio) and own stock. E.K. is a former employee of Horizon Therapeutics and owns stock. B.A.C.C. reports personal fees for consulting from Alexion, Atara, Autobahn, Avotres, Biogen, Boston Pharma, EMD Serono, Gossamer Bio, Hexal/Sandoz, Horizon, Neuron23, Novartis, Sanofi, Siemens, TG Therapeutics, and Therini, and received research support from Genentech.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The N-MOmentum trial was funded by MedImmune and Viela Bio (now Horizon Therapeutics plc, Gaithersburg, MD, USA). Horizon Therapeutics supported the development of this manuscript, provided data analyses according to the direction of the authors, and paid for medical writing support, provided by Oxford PharmaGenesis.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
