Abstract
Background:
Suboptimal performance during neuropsychological testing frequently occurs in multiple sclerosis (MS), leading to unreliable cognitive outcomes. Neurophysiological alterations correlate with MS-related cognitive impairment, but studies have not yet considered performance validity.
Objectives:
To investigate neurophysiological markers of cognitive impairment in MS, while explicitly addressing performance validity.
Methods:
Magnetoencephalography recordings, neuropsychological assessments, and performance validity testing were obtained from 90 MS outpatients with cognitive complaints. Spectral and resting-state functional connectivity (rsFC) properties were compared between cognitively impaired (CI), cognitively preserved (CP), and suboptimally performing (SUB) patients using regression models and permutation testing.
Results:
CI had higher power in low-frequency bands and lower power in high bands compared to CP, indicating neuronal slowing. CI also showed lower beta power compared to SUB. Overall power spectra visually differed between CI and CP, and SUB showed overlap with both groups. CI had lower rsFC than CP and SUB patients. CP and SUB patients showed no differences.
Conclusion:
Neuronal slowing and altered rsFC can be considered cognitive markers in MS. Patients who performed suboptimally showed resemblance with patients with and without cognitive impairments, and although their overall neurophysiological profile was more similar to patients without impairments, it suggests heterogeneity regarding their pathophysiology.
Keywords
Introduction
Cognitive impairment is a frequent (43%–70%) and disabling symptom in multiple sclerosis (MS). 1 Previous studies highlight the value of functional neuroimaging measures in explaining and predicting MS-related cognitive impairment.2 –5 Both changes in oscillatory brain activity4,6 –8 and resting-state functional connectivity (rsFC),9,10 as measured with electroencephalography (EEG) or magnetoencephalography (MEG), correlate with cognitive impairment. Previous EEG and MEG studies found associations between worse cognitive function and slower brain oscillations, as expressed by higher alpha14,6,8 and theta6,7 relative power, as well as a higher theta/beta ratio. 4 Moreover, both higher and lower rsFC have been related to cognitive impairment.9,10 Regarding both oscillatory brain activity and rsFC, regional alterations may provide important insights into MS-related cognitive impairment, as particularly functional alterations within the deep gray matter (DGM) structures, such as the thalamus, seem to relate to cognitive impairment in MS.6,11
However, one of the concerns when investigating MS-related cognitive impairment is the validity of patients’ cognitive test results (i.e. performance validity).12 –15 Recent studies have shown that up to 20% of the MS patients do not perform to the best of their abilities during cognitive testing, which can be measured with performance validity tests.12 –14 These tests are presented to patients as cognitive tests, but are relatively insensitive to actual cognitive impairments and easy to perform. A low score on these tests indicate suboptimal performance, irrespective of whether it is intentional or unintentional. The cognitive scores of patients who perform suboptimally can thereby not be validly interpreted, which may consequently lead to incorrect categorizations of cognitive impairment in the clinic and may also affect the validity of research data. No uniform explanations for suboptimal performance in MS have been found, although psychological, financial and demographic aspects may play a role.12 –14 Notably, a previous study reported that MS patients who performed suboptimally were similarly affected as cognitively impaired (CI) patients in terms of lesion load and brain volumes. 13 As these structural measures correlate with alpha1 power, 16 it could be that these patients also show similar functional alterations as CI patients. However, as these structural brain alterations could not differentiate between CI patients and suboptimal performers, it may also be expected that there are in fact functional detectable differences. Taken together, studying functional brain alterations in patients who perform suboptimally is an important step toward understanding the pathophysiology of patients within this unique group. Also, for the diagnosis and study of cognitive impairment in MS it is essential to have clinical diagnostic tools that are not influenced by suboptimal performance.
This study therefore investigated global and regional MEG-derived oscillatory brain activity (i.e. spectral measures) and rsFC in MS patients with and without cognitive impairment. In addition, we explicitly addressed the issue of performance validity by comparing patients who performed suboptimally to CI and cognitively preserved (CP) patients.
Methods
Participants
This study analyzed existing data from 90 MS patients from the Second Opinion MS and COGnition (SOMSCOG) outpatient clinic of the MS Center Amsterdam. 13 Patients were referred to this tertiary outpatient clinic by a primary care physician or medical specialist because of cognitive complaints. Patients were included in this study if they had undergone routine neuropsychological assessment, including a performance validity test, and a MEG recording. The Medical Ethics Research Committee of Amsterdam UMC concluded that the Medical Research Involving Human Subjects Act (WMO) did not apply to this study, as the data collection was part of clinical care (METC #2016.395). All patients gave written informed consent for use of their data.
Data acquisition
Neuropsychological evaluation and performance validity
Cognitive function was assessed by a test battery based on the minimal assessment of cognitive function in multiple sclerosis (MACFIMS), 17 as described previously, 13 assessing five (sub-)domains: (1) verbal memory (Dutch version of the California Verbal learning Test), (2) visuospatial memory (Brief Visuospatial Memory Test-Revised), (3) processing speed (Symbol Digit Modalities Test and Stroop Color-Word Test cards I and II), (4) executive function—verbal fluency (Controlled Oral Word Association Test), and (5) executive function—response inhibition (Stroop Color-Word Test interference score). Based on a normative sample of Dutch healthy controls (HCs), scores were corrected for age, sex and education when applicable, transformed into test-specific z-scores and accordingly averaged into domain-specific z-scores. 13 Based on test-specific z-scores, patients were characterized as CI (i.e. ⩾1.5 standard deviations below means of HCs on ⩾20% of the test outcomes, 18 corresponding to ⩾ 3/11 test scores) or CP.
In addition, performance validity was assessed with the Amsterdam Short Term Memory test (ASTM), which is a forced-choice verbal recognition test designed to detect suboptimal performance, with a maximal score of 90. 19 Patients who scored ⩽82 had indications of suboptimal performance on their neuropsychological evaluation and were thereby characterized as suboptimal performers (SUB), independently of their cognitive characterization. 13 A detailed description of these assessments can be found elsewhere. 13
Magnetic resonance imaging (MRI)
Patients were scanned on a 3-Tesla whole-body MRI (General Electric Signa HDxt), as described previously. 20 Indicators of cerebral damage were lesion load and whole-brain, gray matter, and white matter volumes. Lesion load was calculated after automatically segmenting Fluid-attenuated inversion recovery (FLAIR) images. Brain volumes were calculated using FMRIB Software Library (FSL) after filling 3DT1 images (using LEAP). All measures were normalized using the V-scaling factor (see Supplementary Information). 20
MEG recordings and pre-processing
MEG recordings were performed with a 306-channel whole-head system (Elekta Neuromag, Oy, Helsinki, Finland) while the patients were in task-free resting-state with eyes closed. Supplementary Information provides the standardized processing procedure. In short, a maximum of 12 malfunctioning channels were removed after visual inspection (ES) and artifacts were removed using the temporal extension of Signal Space Separation (tSSS). 21 Source-localized MEG data was then constructed using an atlas-based beamforming approach: 22 78 cortical and 12 deep gray-matter (DGM) regions of interest (ROIs)23,24 of the automated anatomical labeling (AAL). For each patient, 10 non-overlapping, artifact-free epochs of 16384 samples (13.11 seconds) were selected by visual inspection and filtered into six classical frequency bands: delta (0.5–4 Hz), theta (4–8 Hz), alpha1 (8–10 Hz), alpha2 (10–13 Hz), beta (13–30 Hz), and gamma (30–48 Hz).
MEG measures
Spectral and rsFC measures were computed using the in-house developed software package Brainwave (version 0.9.152.12.26; available at https://home.kpn.nl/stam7883/brainwave.html).
Regarding the spectral measures, five epochs that did not contain any indication of drowsiness 25 (Supplementary Information) were used out of the ten selected and downsampled by a factor of 4. For every ROI of each patient, the relative powers in the six frequency bands and the peak frequency (i.e. the frequency with most power in the 4–13 Hz range) were calculated and averaged over epochs.
Regarding rsFC, a phase-based (i.e. phase lag index (PLI)) 26 and an amplitude-based rsFC measures (i.e. corrected amplitude envelope correlation (AECc)) were calculated for each pair of ROIs of each patient. 27 The PLI was estimated using ten epochs split into 40 epochs (3.28 seconds) for the six frequency bands, and the AECc was estimated with the ten epochs downsampled by a factor of 4 for theta, alpha1, alpha2, and beta frequency bands (the other frequency bands seem less reliable for the AECc). 27 A detailed description of the AECc approach can be found elsewhere. 28 For each patient and each frequency band, averaged matrices of pairwise rsFC (90 × 90 ROIs) of the PLI and AECc were obtained by averaging over all epochs. These matrices were subsequently averaged per ROI, resulting in (90 × 1) vectors with the average connectivity of each ROI with all other ROIs.
Both spectral and rsFC measures were further averaged and analyzed on a global (i.e. cortical and DGM separately), regional (i.e. frontal, temporal, parietal, occipital, hippocampus, thalamus), and ROI level. Supplementary Table 1 describes the ROIs in each region.
Statistical analysis
Analyses were performed using R (version 3.6.3) and in-house-developed MATLAB routines (permutation testing in AnalyseBrainwaveOutput (version 4.23)). Normality was checked by visual inspection.
Depending on normality and data type, demographic and clinical characteristics were pairwise compared between groups (i.e. CI-CP, CI-SUB, and CP-SUB) using independent samples t-tests, Wilcoxon rank-sum tests, Chi-square tests, or Fisher exact tests. The alpha level was set at .05.
Global and regional spectral and rsFC measures were pairwise compared between groups in each frequency band, using linear regression models while controlling for age and gender. In the absence of normality, linear regression of log-transformed outcomes or the non-parametric Wilcoxon rank-sum test were performed. For the Wilcoxon rank-sum test, age and gender could not be controlled for. The alpha level was Bonferroni corrected and set to 0.007 for the spectral analyses (i.e. peak frequency and relative powers of six frequency bands; α = .05/7), 0.008 for the PLI (six bands; α = 0.05/6), and .0125 for the AECc (four bands; α = 0.05/4).
Additional pairwise comparisons at the ROI level for both spectral and rsFC measures were performed by permutation testing. For every spectral and rsFC measure in each ROI, age and gender were regressed out from every two groups before comparing the groups with permutation testing (inspired by Sjøgård et al. 9 ). The values from both groups for every ROI were then randomly shuffled to generate the unpaired permutation distribution of t vectors. A total of 5000 permutations were performed, with an alpha level of 0.05. The significance level of these permutations was determined using the two-tailed maximum-statistic testing to correct for multiple testing. 29
Results
The total sample (N = 90, 66.7% women, age 47.3 ± 9.6 years, median disease duration 12.3 ± 8.9 years, 66.7% relapsing-remitting MS) consisted of 42 CI, 30 CP, and 18 SUB MS patients. Table 1 presents the group characteristics. Group differences were found for gender and disability level: the CI group consisted of relatively more men (43%) than the CP group (20%) (p = 0.042), and the CP group had lower disability levels (median EDSS = 3.25) than the SUB group (median EDSS = 4.00) (p = 0.045).
Demographics, disease-related characteristics, and cognitive function per group.
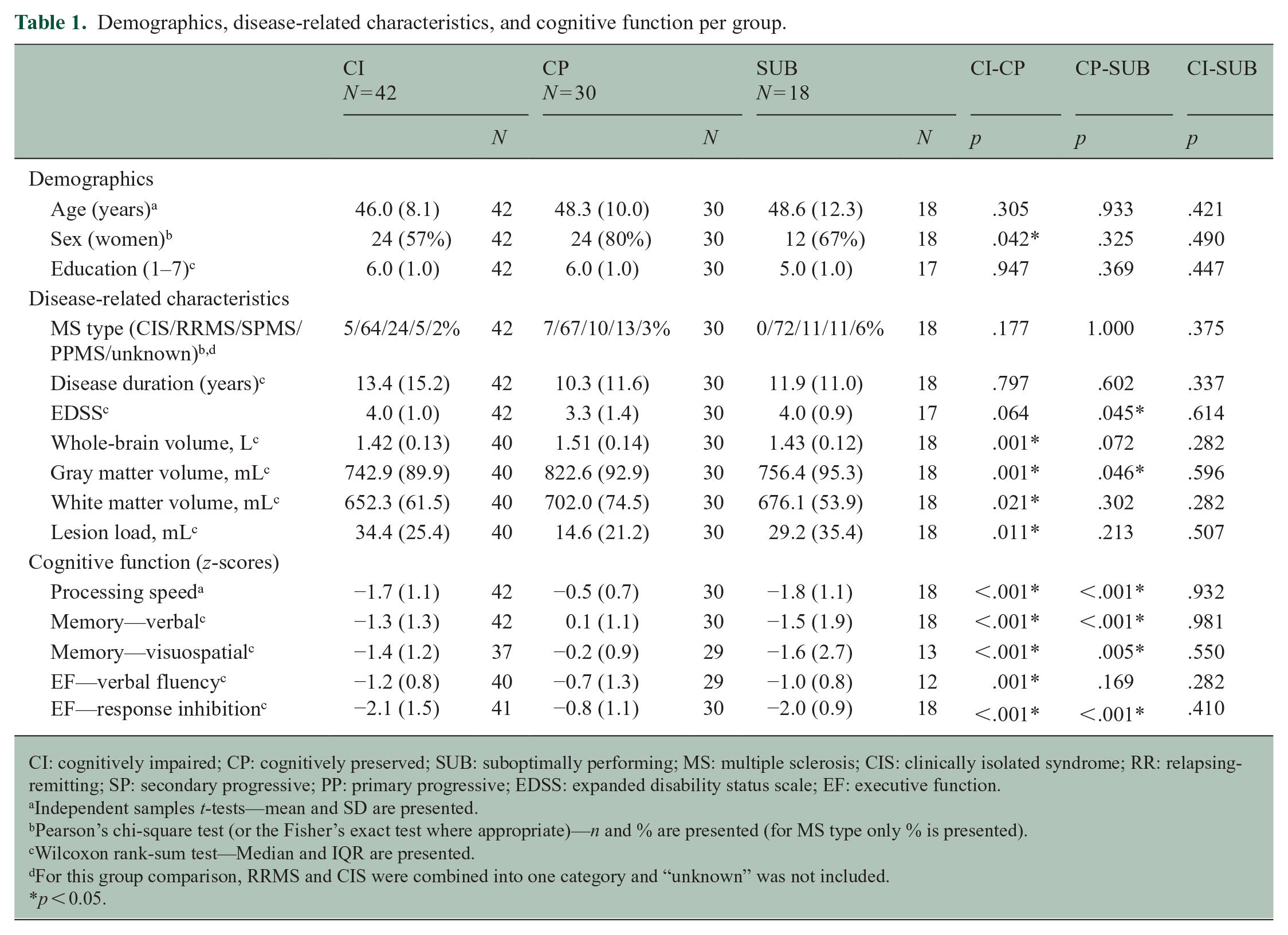
CI: cognitively impaired; CP: cognitively preserved; SUB: suboptimally performing; MS: multiple sclerosis; CIS: clinically isolated syndrome; RR: relapsing-remitting; SP: secondary progressive; PP: primary progressive; EDSS: expanded disability status scale; EF: executive function.
Independent samples t-tests—mean and SD are presented.
Pearson’s chi-square test (or the Fisher’s exact test where appropriate)—n and % are presented (for MS type only % is presented).
Wilcoxon rank-sum test—Median and IQR are presented.
For this group comparison, RRMS and CIS were combined into one category and “unknown” was not included.
p < 0.05.
Furthermore, group differences were found for cerebral damage: CI patients had a higher lesion load and smaller brain volumes than CP patients (p < 0.05), and SUB patients had smaller gray matter volumes than CP patients (p = 0.046). Regarding cognition, CI and SUB patients had worse domain scores than CP patients (p < 0.05), although SUB patients’ scores cannot be considered reliable. If not accounting for performance validity, 15 out of 18 (83%) SUB MS patients would be categorized as CI.
Spectral analyses
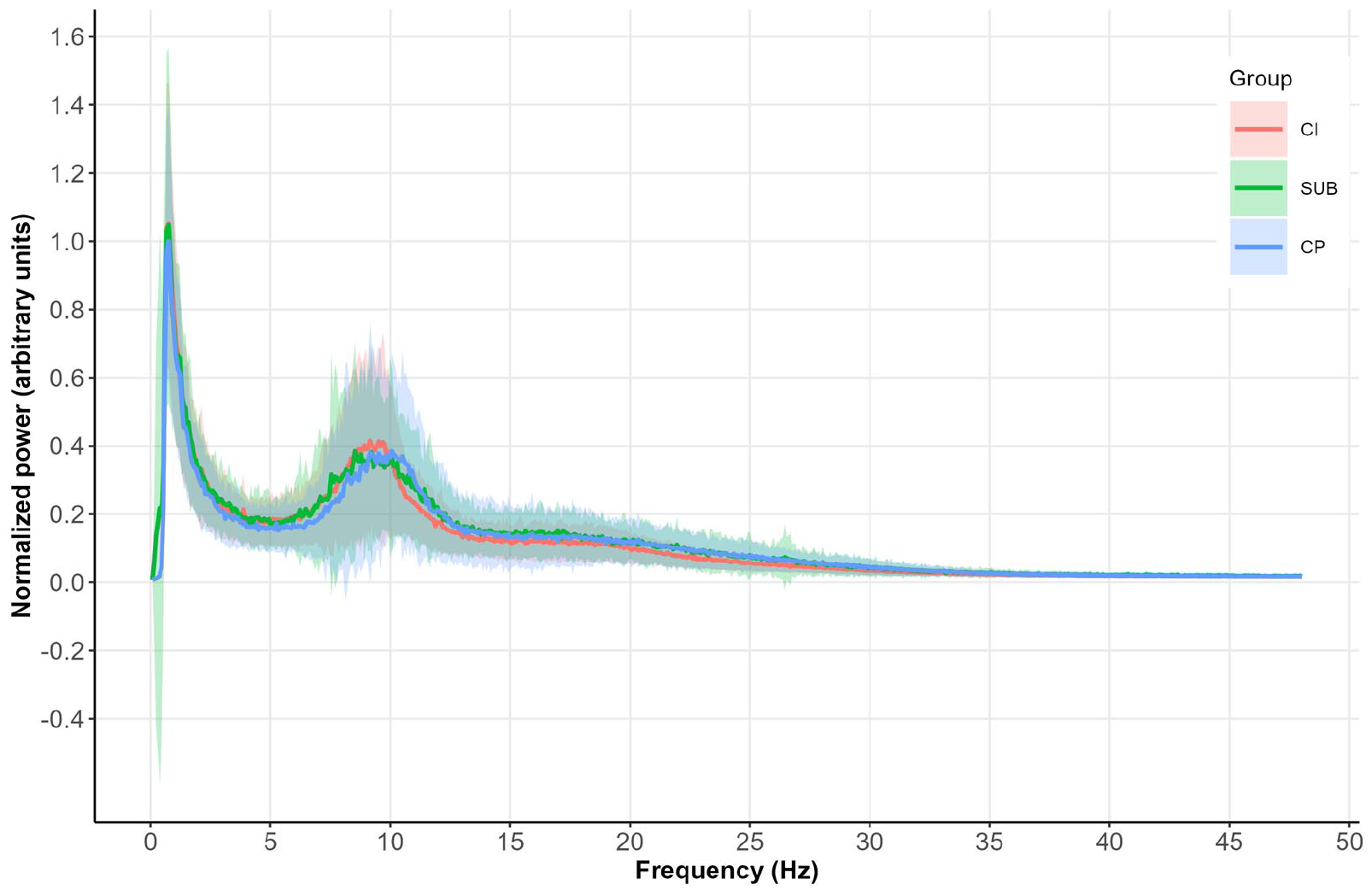
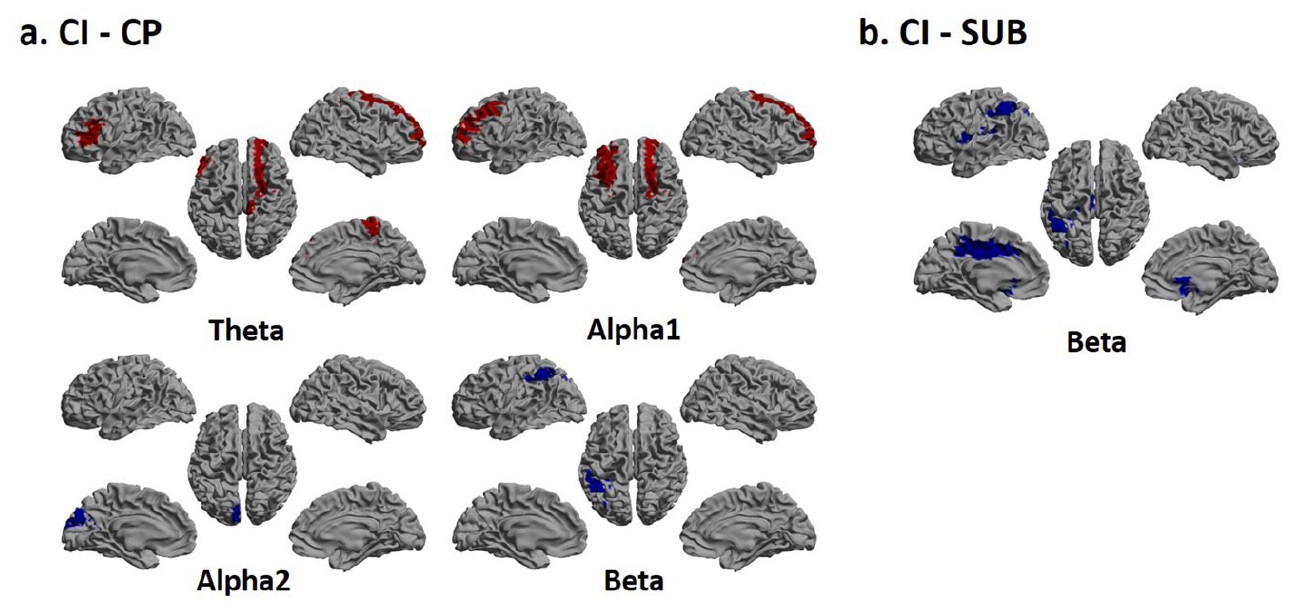
Global average power spectra are plotted in Figure 1. On visual inspection, CI and CP patients showed differences at multiple frequencies. The curve of the SUB group showed similarities with both the CP and CI patients. Formal testing within each frequency band only showed lower beta power in the DGM in CI patients compared to SUB patients (p = 0.004) (Table 2), no other global differences between the groups were found (p > 0.007).
Differences in global spectral and connectivity measures between groups.
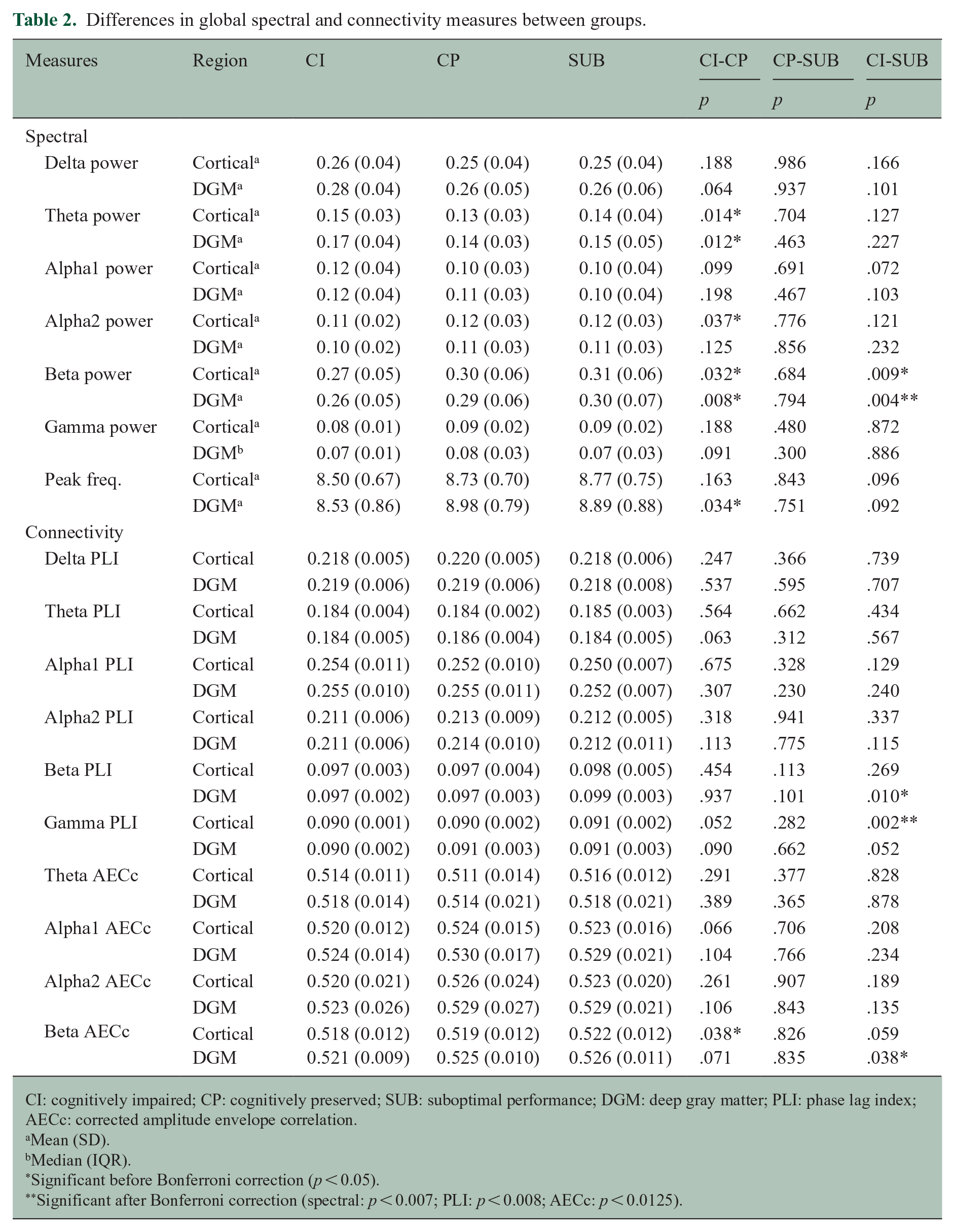
CI: cognitively impaired; CP: cognitively preserved; SUB: suboptimal performance; DGM: deep gray matter; PLI: phase lag index; AECc: corrected amplitude envelope correlation.
Mean (SD).
Median (IQR).
Significant before Bonferroni correction (p < 0.05).
Significant after Bonferroni correction (spectral: p < 0.007; PLI: p < 0.008; AECc: p < 0.0125).
In the analysis per brain region, CI patients had higher theta power in the frontal lobe and thalamus (p < 0.007), and lower alpha2 occipital power (p = 0.002) (Table 3) compared to CP patients. In addition, CI patients had lower beta power in the thalamus compared to CP (p = 0.007) and SUB patients (p = 0.003), as well as in the frontal lobe (p = 0.005) and hippocampus (p = 0.007) compared to SUB patients. Supplementary Table 2 presents the non-significant regional spectral results before Bonferroni correction (p > 0.05).
Regional spectral and connectivity measures that significantly differed between groups (before and after correction).
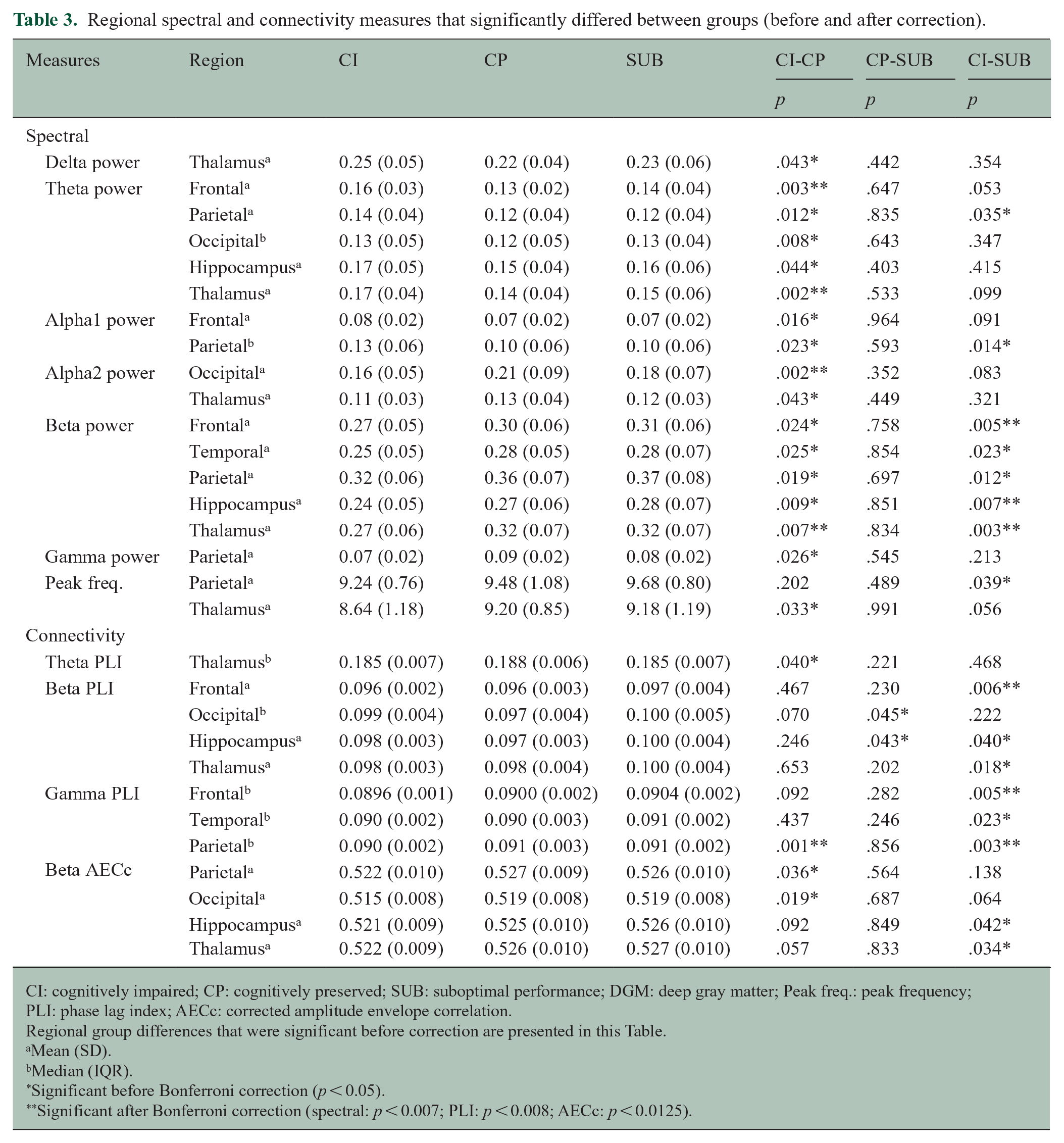
CI: cognitively impaired; CP: cognitively preserved; SUB: suboptimal performance; DGM: deep gray matter; Peak freq.: peak frequency; PLI: phase lag index; AECc: corrected amplitude envelope correlation.
Regional group differences that were significant before correction are presented in this Table.
Mean (SD).
Median (IQR).
Significant before Bonferroni correction (p < 0.05).
Significant after Bonferroni correction (spectral: p < 0.007; PLI: p < 0.008; AECc: p < 0.0125).
The ROI level group differences are presented in Figure 2. In short, CI patients had higher theta and alpha1 power and lower alpha2 and beta power in several ROIs compared to CP patients (Figure 2(a)). CI patients also showed reduced beta power in several ROIs compared to the SUB patients (Figure 2(b)). No differences were found between CP and SUB patients.
Connectivity analyses
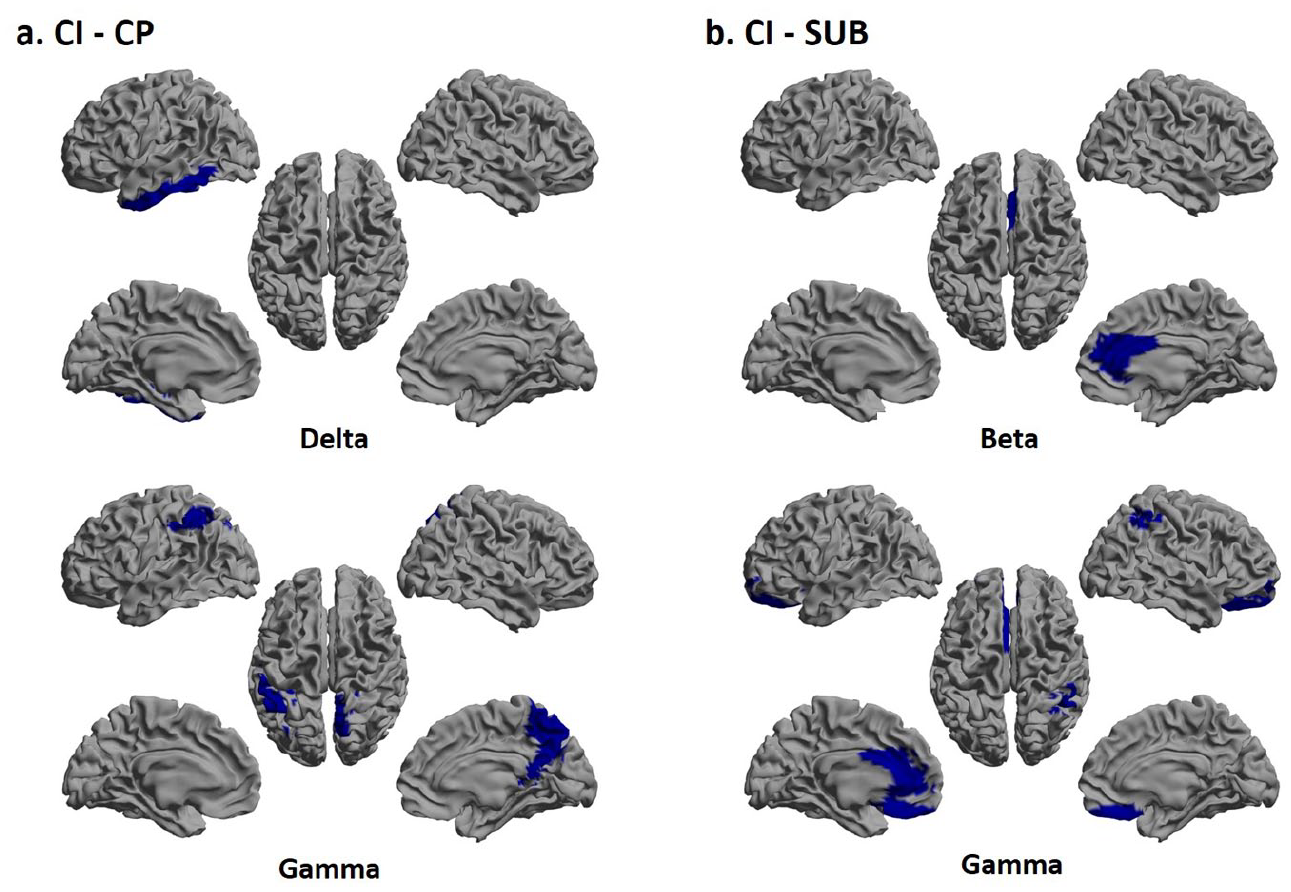
Regarding the global results, CI patients had lower cortical gamma PLI-rsFC than SUB patients (p = 0.002) (Table 2). No global differences regarding the AECc-rsFC were found (p > 0.0125).
Regionally, CI patients had lower gamma PLI-rsFC in the parietal regions compared to CP (p = 0.001) and SUB patients (p = 0.003), and in the frontal regions compared to SUB patients (p = 0.005) (Table 3). Also, CI patients had lower beta PLI-rsFC in the frontal regions compared to SUB patients (p = 0.006). No regional differences regarding the AECc-rsFC were found (p > 0.0125). Supplementary Table 3 presents the non-significant regional rsFC results before Bonferroni correction (p > 0.05).
The ROI level group differences are presented in Figure 3. In short, CI patients had lower delta and gamma PLI-rsFC than CP patients (Figure 3(a)), and lower beta and gamma PLI-rsFC than SUB patients in several ROIs (Figure 3(b)). No differences were found between CP and SUB patients, nor regarding the AECc-rsFC.
Discussion
This study investigated neurophysiological characteristics of cognitive impairment in a clinical cohort of MS patients with cognitive complaints, while explicitly addressing performance validity. Our results showed that neuronal slowing and altered rsFC related to cognitive impairment, both at global and regional levels. Interestingly, the neurophysiological characteristics of patients who performed suboptimally differed from CI patients, but not from CP patients, even though most of these patients would be categorized as CI if performance validity was not taken into account. These results emphasize the importance of performance validity consideration when investigating underlying mechanisms of cognitive impairment in MS as well as during the clinical diagnosis of cognitive impairment.
The neuronal slowing found in CI patients relative to CP patients (excluding patients who performed suboptimally) was indicated by higher relative power in the lower frequency bands (i.e. theta and alpha1) and lower relative power in the higher frequency bands (i.e. alpha2 and beta). It was also expressed in a non-significant trend of lower peak frequency in CI patients. Previous studies also reported neuronal slowing among MS patients compared to healthy controls, and similarly observed higher relative theta6,7 and alpha1 power4,6 as correlates of cognitive impairment. Regionally, alterations in relative power were most pronounced in the frontal cortex and the thalamus, but were also found in the parietal and occipital lobes and in additional DGM regions. Frontal4,7 and thalamic involvement was also highlighted by previous studies, including a study from our group on another patient sample, 6 where power differences in these areas were found between CI patients and healthy controls. Moreover, particularly the thalamus is known to play a central role in MS-related cognitive impairment, both at the structural and functional levels. 11 Taken together, neuronal slowing seems a consistent correlate of cognitive impairment in MS. As cognitive complaints themselves are not a good indicator of actual cognitive impairment, 30 neuronal slowing may serve as a clinically applicable marker of cognitive impairment.
In addition to these spectral differences, altered rsFC was also associated with cognitive impairment. More specifically, CI patients had lower delta and gamma PLI than CP patients. At a regional level, these rsFC differences were found in the temporal and parietal cortex. Previous studies reported mixed findings regarding the direction of rsFC in relation to MS-related cognitive impairment: both lower and higher values have been related to worse cognitive function, 3 as any deviation away from a normal network can be maladaptive. Notably, no rsFC group differences were found using the AECc. Since the AECc and PLI are amplitude- and phase-based measures respectively, they presumably differ in their sensitivity to detect brain alternations. Overall, taken together with the inconsistency of rsFC results in Tewarie et al. 31 and only a few group differences found in our study, functional connectivity values alone, at least the ones obtained using current metrics, may be difficult to utilize as direct biomarkers of cognitive impairment in MS.
The spectral and rsFC alterations indicative of cognitive impairment were not found among patients who performed suboptimally during neuropsychological testing, even though most of these patients would have been classified as CI if their performance validity was not taken into account (83%). More specifically, suboptimal performers differed from CI patients and not from CP patients regarding spectral and connectivity measures. These results indicate that patients who performed suboptimally may be more comparable to CP patients than CI patients on a functional brain level. On a structural level, suboptimal performers seemed more affected than CP patients (i.e. smaller gray matter volume), but not to the same extent as CI patients. Therefore, it could be that their structural pathology is not severe enough for their functional networks to be affected in a similar way as the CI patients. 32 However, suboptimal performers may also be heterogeneous in terms of underlying structural and functional pathology, as they likely include patients with and without cognitive impairments, as is also suggested by the overlap in averaged power spectra with both cognitive groups. Possibly, a uniform neurophysiological pattern in the suboptimal performers may not exist and rather two distinct groups exist with substantial overlap. This underscores the need for a reanalysis in a large MS sample, given that this is the first study to look at brain function in suboptimally performing MS patients. Nonetheless, considering suboptimally performing individuals as CI patients may induce noise in research data, preventing the discovery of underlying mechanisms of cognitive impairment, and in the clinic, it could lead to inappropriate treatments.
This study has some limitations. First, we investigated a large variety of regions and neurophysiological measures, and therefore used strict corrections for multiple comparisons. We could thereby have missed certain markers of cognitive impairment. Still, there were clear differences between CI and CP patients, particularly regarding neuronal slowing. An additional limitation that could have led to false negatives, was the relatively small group of suboptimal performers. Moreover, we categorized patients as suboptimal performers based on one performance validity test. We did apply a stricter criterion than the recommended criterion to classify patients as suboptimal performers to reduce the chance of incorrect classifications, but two performance validity tests are generally preferred. 33 It is also important to mention that MEG’s spatial resolution is lower for deep sources. Furthermore, previous studies (e.g. Pizzo et al. 34 ) only provided evidence that intracerebral thalamic activity in some subjects correlates with independent components obtained from cortical MEG activity. Source reconstructed MEG data at DGM locations should, therefore, be interpreted with caution. In addition, administration of benzodiazepine drugs, which is common in MS, could have potentially influenced our neurophysiological measures, as it was shown to affect the spectral power distribution. 35 This information was not available in our study, and it should be taken into account in future studies. Finally, previous studies indicate that functional brain alterations in MS differ between men and women. 36 We therefore performed the group comparisons while controlling for gender. However, in studies with a larger sample size, it would be relevant to investigate whether neurophysiological markers of cognitive impairment in MS are different for men and women.
To conclude, this study is the first to analyze MEG data in an outpatient clinic specifically dedicated to cognition. Also in this population, we consistently found neuronal slowing, and to a lesser extend also altered rsFC, to relate to cognitive impairment, confirming previous results. Since patients who performed suboptimally were on a neurophysiological level more comparable to CP patients than CI patients, neuronal slowing may serve as a clinically applicable marker for cognitive performance in MS. A next step would be to investigate the ability of these markers to individually predict cognitive impairment in MS.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231159817 – Supplemental material for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585231159817 for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis by Shira Simon, Ilse M Nauta, Arjan Hillebrand, Menno M Schoonheim, Bernard MJ Uitdehaag, Maureen van Dam, Hanneke E Hulst, Martin Klein, Cornelis J Stam, Brigit A de Jong and Eva MM Strijbis in Multiple Sclerosis Journal
Supplemental Material
sj-docx-2-msj-10.1177_13524585231159817 – Supplemental material for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis
Supplemental material, sj-docx-2-msj-10.1177_13524585231159817 for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis by Shira Simon, Ilse M Nauta, Arjan Hillebrand, Menno M Schoonheim, Bernard MJ Uitdehaag, Maureen van Dam, Hanneke E Hulst, Martin Klein, Cornelis J Stam, Brigit A de Jong and Eva MM Strijbis in Multiple Sclerosis Journal
Supplemental Material
sj-docx-3-msj-10.1177_13524585231159817 – Supplemental material for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis
Supplemental material, sj-docx-3-msj-10.1177_13524585231159817 for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis by Shira Simon, Ilse M Nauta, Arjan Hillebrand, Menno M Schoonheim, Bernard MJ Uitdehaag, Maureen van Dam, Hanneke E Hulst, Martin Klein, Cornelis J Stam, Brigit A de Jong and Eva MM Strijbis in Multiple Sclerosis Journal
Supplemental Material
sj-docx-4-msj-10.1177_13524585231159817 – Supplemental material for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis
Supplemental material, sj-docx-4-msj-10.1177_13524585231159817 for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis by Shira Simon, Ilse M Nauta, Arjan Hillebrand, Menno M Schoonheim, Bernard MJ Uitdehaag, Maureen van Dam, Hanneke E Hulst, Martin Klein, Cornelis J Stam, Brigit A de Jong and Eva MM Strijbis in Multiple Sclerosis Journal
Supplemental Material
sj-pdf-5-msj-10.1177_13524585231159817 – Supplemental material for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis
Supplemental material, sj-pdf-5-msj-10.1177_13524585231159817 for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis by Shira Simon, Ilse M Nauta, Arjan Hillebrand, Menno M Schoonheim, Bernard MJ Uitdehaag, Maureen van Dam, Hanneke E Hulst, Martin Klein, Cornelis J Stam, Brigit A de Jong and Eva MM Strijbis in Multiple Sclerosis Journal
Supplemental Material
sj-pdf-6-msj-10.1177_13524585231159817 – Supplemental material for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis
Supplemental material, sj-pdf-6-msj-10.1177_13524585231159817 for Neurophysiological MEG markers of cognitive impairment and performance validity in multiple sclerosis by Shira Simon, Ilse M Nauta, Arjan Hillebrand, Menno M Schoonheim, Bernard MJ Uitdehaag, Maureen van Dam, Hanneke E Hulst, Martin Klein, Cornelis J Stam, Brigit A de Jong and Eva MM Strijbis in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors thank the psychologists of the Amsterdam UMC, Department of Medical Psychology, and the EEG/MEG technicians of the Amsterdam UMC, Department of Clinical Neurophysiology and MEG Center, for the data acquisition. The authors also thank all patients for their participation.
Data Availability Statement
The anonymized data, not published in the article, will be shared upon reasonable request from a qualified investigator.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Dutch MS Research Foundation, grant #15-911.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
