Abstract
Background:
Risk factors for aquaporin-4 (AQP4+) antibody neuromyelitis optica spectrum disorder (NMOSD) are not well-established.
Objective:
To investigate demographic and environmental factors associated with NMOSD using a validated questionnaire and case–control design.
Methods:
We enrolled patients with AQP4 + NMOSD through six Canadian Multiple Sclerosis Clinics. Participants completed the validated Environmental Risk Factors in Multiple Sclerosis Study (EnvIMS) questionnaire. Their responses were compared to those of 956 unaffected controls from the Canadian arm of EnvIMS. We calculated odds ratios (ORs) for the association between each variable and NMOSD using logistic regression and Firth’s procedure for rare events.
Results:
In 122 participants (87.7% female) with NMOSD, odds of NMOSD in East Asian and Black participants were ⩾8 times that observed in White participants. Birthplace outside Canada was associated with an increased risk of NMOSD (OR = 5.5, 95% confidence interval (CI) = 3.6–8.3) as were concomitant autoimmune diseases (OR = 2.7, 95% CI = 1.4–5.0). No association was observed with reproductive history or age at menarche.
Conclusion:
In this case–control study, risk of NMOSD in East Asian and Black versus White individuals was greater than that observed in many previous studies. Despite the preponderance of affected women, we did not observe any association with hormonal factors such as reproductive history or age at menarche.
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an autoimmune disease of the central nervous system with an estimated prevalence ranging from 0.7 to 10 per 100,000 population.1–3 AQP4-antibody seropositive (AQP4+) NMOSD occurs predominantly among women at a ratio of up to 9:1 of women to men. 2 Other demographic and environmental risk factors for AQP4 + NMOSD are not well-established. Previous research has suggested ethnic or racial background,2–4 other autoimmune conditions, 5 smoking,6,7 and infections 8 as putative risk factors. Hormonal exposures, including menstrual and pregnancy history, are of interest given the overwhelming female preponderance. 9 However, many past investigations into exposures associated with NMOSD have been limited by lack of controls, small samples and inclusion of both AQP4+ and seronegative cases. It can be challenging to study etiology and risk factors that contribute to rare diseases. 10 It is unlikely that a sufficiently large cohort could be followed for enough time to allow for a well-powered prospective study. Case–control studies are thus the preferred method to investigate etiology of rare diseases like NMOSD, given the opportunity to leverage a large control population.
Many of the proposed risk factors for NMOSD overlap with those observed in multiple sclerosis (MS). Hence, in this multi-centre Canadian study, we aimed to determine risk factors for AQP4 + NMOSD using a case–control design and the validated Environmental Risk Factors in Multiple Sclerosis Study (EnvIMS) questionnaire.11,12 For this study, we utilized an innovative design that incorporated a large number of unaffected controls from the Canadian arm of EnvIMS to optimize statistical power.
Methods
This was a multi-centre unmatched case–control study of demographic and environmental risk factors for AQP4 + NMOSD in Canada. Canada has a universal public health insurance system without a parallel private service, and therefore, all NMOSD patients receive care through the public system.
Research Ethics Board approval was obtained at each participating site for recruitment of NMOSD participants. As in the original EnvIMS project, return of the questionnaire was taken as consent to participate. For inclusion of controls from EnvIMS, Research Ethics Board Approval was obtained from McGill University, the lead Canadian site of EnvIMS, and from the international EnvIMS Steering Committee.
Cases
Almost all NMOSD patients are followed at demyelinating disease clinics in Canada due to the specialized care required and approval process for recommended immunotherapies. Participants were enrolled at the following six MS centres: University of British Columbia Hospital (Vancouver, British Columbia), University of Manitoba/Health Sciences Centre Winnipeg (Winnipeg, Manitoba), London Health Sciences Centre (London, Ontario), St. Michael’s Hospital (Toronto, Ontario), Sunnybrook Health Sciences Centre (Toronto, Ontario) and the Ottawa Hospital (Ottawa, Ontario). NMOSD patients were identified through clinic databases, and then eligible participants were approached in clinic or by mail to participate. Recruitment began on 20 December 2018 and closed on 11 February 2021. Cases were required to meet 2015 diagnostic criteria for NMOSD and have a positive serum AQP4 test by either the enzyme-linked immunosorbent assay (ELISA) or cell-based antibody assay. 1 Adequate comprehension of English was required for participants to answer the questionnaire. To minimize selection bias, participants who were blind were allowed to have questions read to them by a family member, friend, or study coordinator. Participants who were unable to handle a writing implement were permitted to verbally report answers to a family member, friend, or study coordinator to record on the paper form.
Controls
Controls from the Canadian arm of EnvIMS were recruited in 2012–2013 using random digit dialling in the greater Montreal area, greater Toronto area and Manitoba, mainly in Winnipeg. 11 As described previously, random digit dialling was employed because there is no population-based register in Canada from which a sample of controls can be identified. 11 Controls were excluded if they self-reported a previous diagnosis of MS or optic neuritis.
Questionnaire
EnvIMS-Q (see Supplement Material) is a detailed self-administered, paper questionnaire. For this study, we added a question concerning history of NMOSD to the section on past medical history in EnvIMS-Q to establish face validity. Other questions were kept the same as EnvIMS-Q to ensure valid comparison of NMOSD cases to controls from EnvIMS. EnvIMS-Q was first developed in English, the version used for this study, and has high acceptability and reliability in people of different cultural and national backgrounds. 12 The questionnaire was designed based on a review of the literature concerning MS etiology. Part of the intent was to explore early life exposures, prevalence across geographic regions and association with MS in different populations, and to facilitate future studies. 11 For this study, we focused analysis on items for which there was prior evidence suggesting an association with NMOSD: sex, age, ethnicity, birthplace, comorbid autoimmune conditions, history of infectious mononucleosis, body mass index (BMI), exercise habits during adolescence, history of cigarette smoking, history and number of past pregnancies and age at menarche.3,5–9,13
Place of birth was collected as town/city and country of birth, and then categorized as Canada versus outside of Canada for analysis. Participants reported the ethnicity of each of their birth parents, as was done in EnvIMS, and is in keeping with the United Nations’ framework for assessing ethnicity. Response options included White, Chinese, Latin American, Arab, Aboriginal, West Asian, Black, Japanese, Southeast Asian, Korean, South Asian, Filipino and Other. For simplicity of analysis, ethnicity was categorized as White, Black, East Asian (Chinese, Japanese, or Korean) if both parents were from the relevant group. If both parents were of the same ethnicity, but neither White, Black nor East Asian, they were categorized as ‘both other and the same’. If the parents were of different ethnic backgrounds, they were categorized as ‘Mixed’. The final two groups were considered together as ‘Other’ due to small numbers.
Past medical history included questions on history of common autoimmune diseases, MS, NMOSD (questionnaire given to cases only) and optic neuritis. Age at first diagnosis was requested when a condition was reported. Age at NMOSD onset was recorded as the age given for the first demyelinating attack. History of any of lupus, rheumatoid arthritis (RA), Celiac disease, or psoriasis was categorized as presence of another autoimmune disease. The questionnaire queried history of hypothyroidism and hyperthyroidism but we did not include these conditions with autoimmune disease due to lack of specificity.
Respondents reported their exercise habits during adolescence (between 13 and 19 years old) in terms of participation in vigorous physical activities such as jogging, running, a stair machine, or sports; heavy exercise was considered where vigorous physical activities were reported ⩾3 times weekly. BMI was calculated from self-reported current weight and height at the time of answering the questionnaire. ‘Ever smoking’ was defined by regular smoking of ⩾1 cigarette per day for ⩾6 months. Hormonal exposures were queried only in women and captured age at menarche and past pregnancies, including instances of pregnancy loss.
Analysis
Data were recorded from all participants, with missing data noted for each item; missing data were not imputed. We calculated crude odds ratios (ORs) for the association between each variable and NMOSD using separate models for each variable and Firth’s procedure for rare events. 14 We then calculated adjusted ORs and 95% confidence intervals (CI), using logistic regression and Firth’s procedure, and controlling for age at the time of the study as a continuous variable, sex (reference: male) and place of birth (reference: born in Canada), with the following exceptions. For ethnicity and place of birth, we calculated adjusted ORs controlling only for age and sex because of co-linearity between place of birth and ethnicity. For analyses that included number of past pregnancies history and age at menarche, only female participants were included, and we controlled for age and place of birth. We conducted a sensitivity analysis, limiting the analysis to cases and controls residing in Ontario. Ontario is the most populous Canadian province, and its population is more ethnically diverse and more likely to dwell in urban centres compared to other provinces, suggesting that demographic and environmental characteristics should be explored separately in this region. 15
For variables that were consistently found to be significantly associated with NMOSD, we investigated age at onset of NMOSD, comparing age at onset across categories using analysis of variance (ANOVA) and two-tailed t-tests as appropriate. Statistical analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Out of 180 eligible participants, 142 (78.9%) initially agreed to receive the questionnaire, and it was returned by 122 participants (85.9% response). NMOSD participants were predominantly female, ethnically diverse, immigrants and residents of the province of Ontario (Table 1). There were 956 unaffected controls from the Canadian arm of EnvIMS. Controls were more likely to be male, slightly younger, White, living outside of Ontario and born in Canada compared to NMOSD participants.
Descriptive characteristics of NMOSD cases and EnvIMS controls.
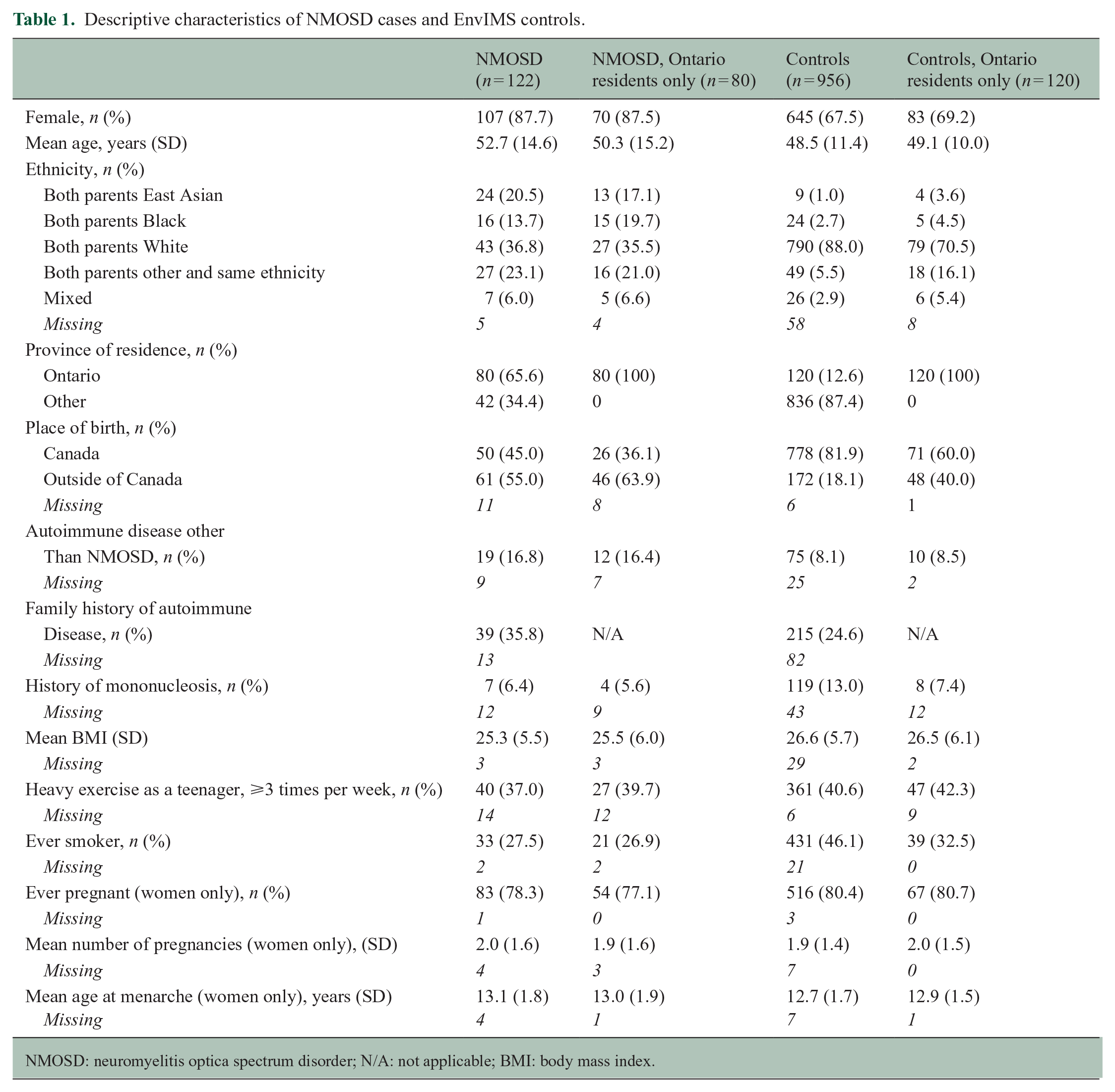
NMOSD: neuromyelitis optica spectrum disorder; N/A: not applicable; BMI: body mass index.
We observed an association between ethnicity and risk of NMOSD (p < 0.0001), where ORs exceeded 8 for East Asian and Black participants compared to White participants across all analyses. We also observed an increased risk of NMOSD with place of birth outside of Canada (p < 0.0001) and other concomitant autoimmune diseases (p = 0.002) in the crude and adjusted analyses (Table 2).
Odds ratios for NMOSD cases versus controls. a
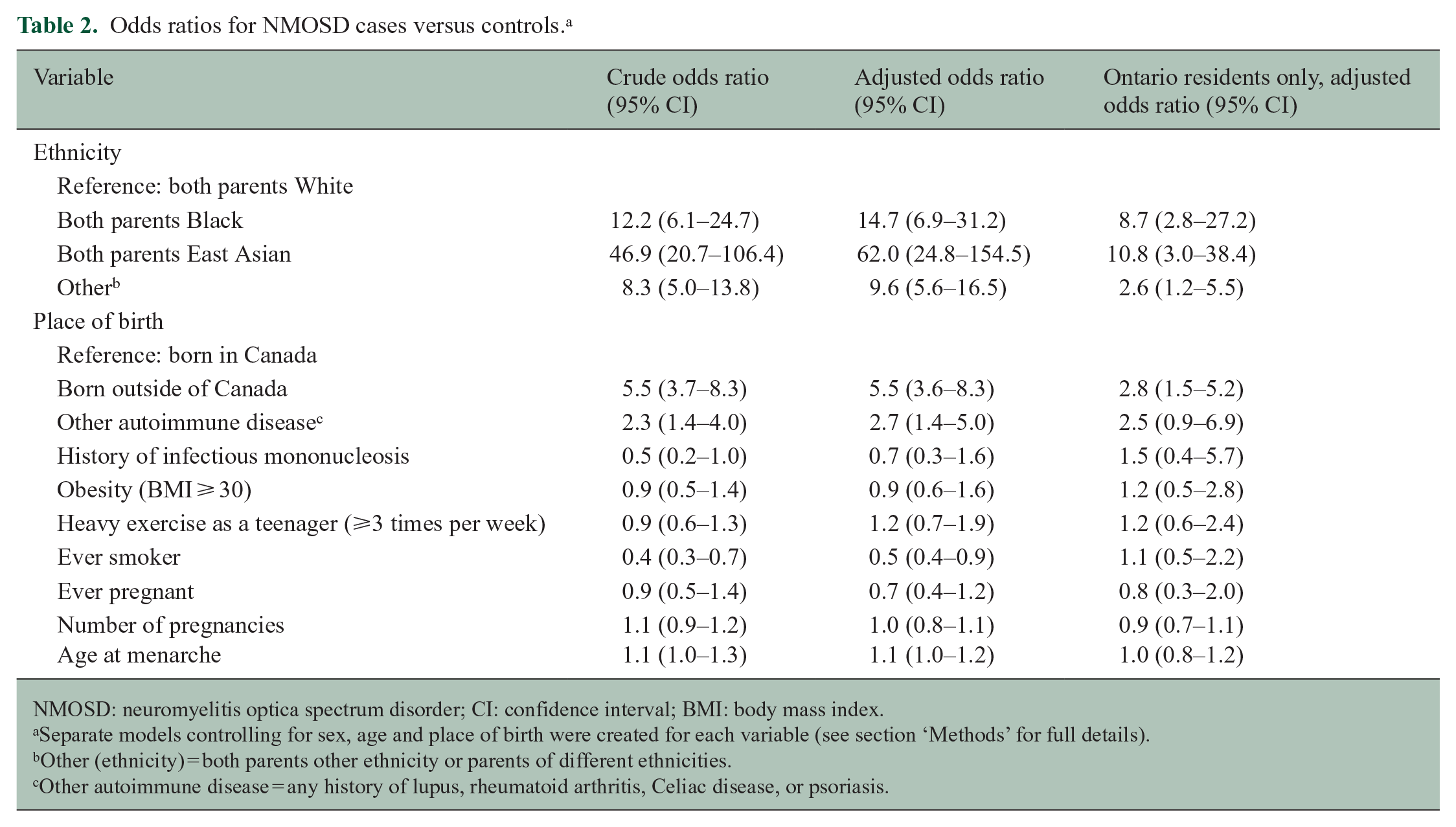
NMOSD: neuromyelitis optica spectrum disorder; CI: confidence interval; BMI: body mass index.
Separate models controlling for sex, age and place of birth were created for each variable (see section ‘Methods’ for full details).
Other (ethnicity) = both parents other ethnicity or parents of different ethnicities.
Other autoimmune disease = any history of lupus, rheumatoid arthritis, Celiac disease, or psoriasis.
There was a decreased risk of NMOSD observed with ever smoking in the crude and adjusted analyses. In the sensitivity analysis limited to Ontario residents, we observed consistent associations between the variables above and NMOSD risk, except for smoking. Ontario controls were considerably less likely to report ever smoking (32.5%) than controls in the general Canadian cohort (46.1%). We did not observe any association of NMOSD with ever having been pregnant, number of past pregnancies, or age at menarche in the overall or sensitivity analyses.
We did not observe any statistically significant differences in age of NMOSD onset by sex, ethnicity, place of birth and other autoimmune diseases (Table 3); however, onset age was generally older in Whites, women and those with other autoimmune diseases.
Age at NMOSD onset by variables associated with NMOSD risk.
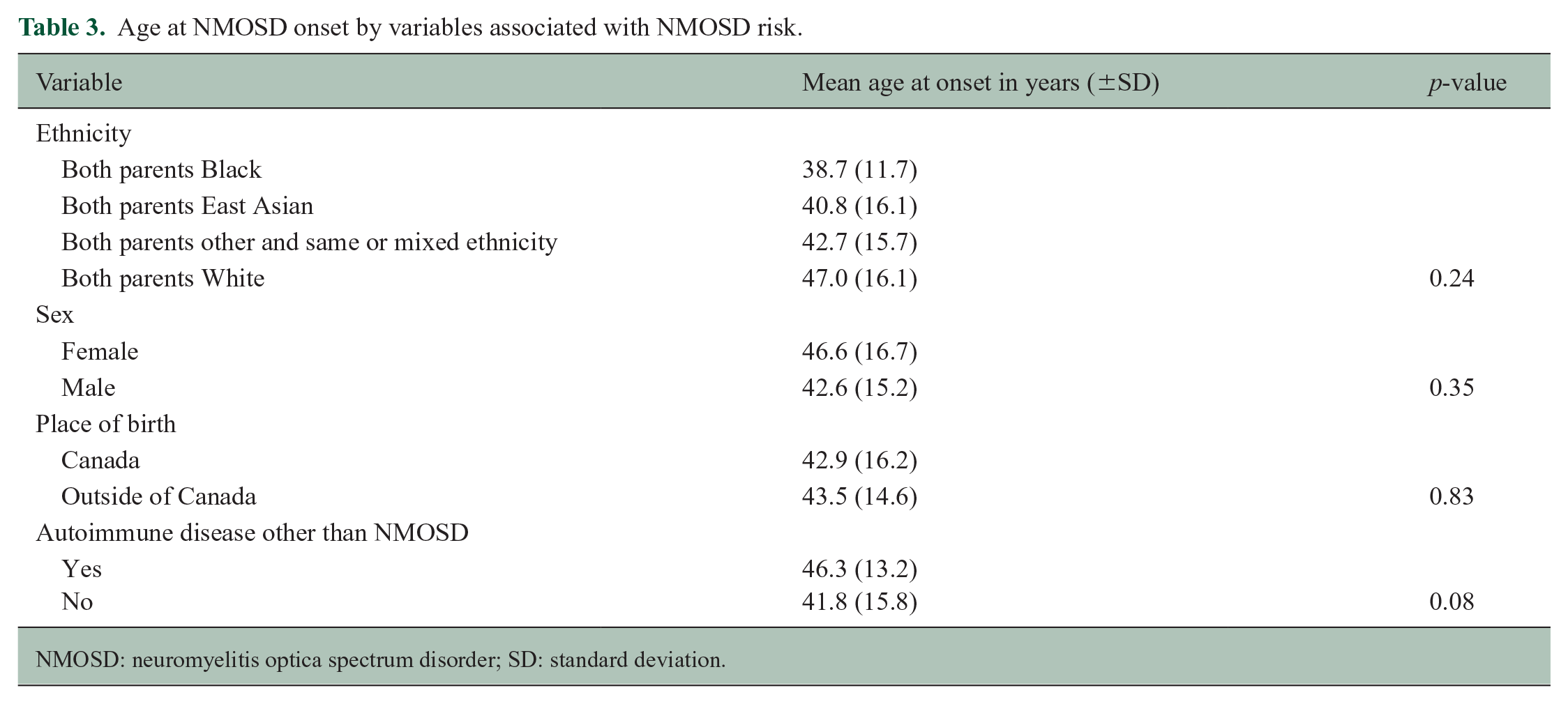
NMOSD: neuromyelitis optica spectrum disorder; SD: standard deviation.
Discussion
In this Canadian multi-centre case–control study, we identified consistent associations between East Asian, Black and other non-White ethnic background; birthplace outside of Canada; and concomitant autoimmune disease with an increased risk of NMOSD. We did not find evidence of an association of endogenous hormonal exposures such as age at menarche and reproductive history with NMOSD which is consistent with findings from a previous questionnaire study which did not include comparison to a control population. 9 Our results do not support a relationship between history of infectious mononucleosis, obesity or physical activity with NMOSD, despite some conflicting reports in the past literature.7,16–18 This study illustrates the importance of controlled studies to investigate environmental risk factors for NMOSD, and consideration of potential confounders such as sex, age, ethnicity and place of birth in study design and analysis.
We found that the odds of NMOSD in East Asian and Black individuals compared to White individuals were greater than previously reported.2,3,19–21 Due to our ethnically diverse participant population and controlled study design, we were able to compare multiple discrete ethnic groups relative to the ethnic distribution of controls within a single geographic region. Past studies have been limited by lack of controls or adjustment for underlying ethnic composition of the population, comparisons across multiple regions, small numbers and inclusion of seronegative or myelin oligodendrocyte glycoprotein (MOG) + cases.3,20–23 In a study from Australia and New Zealand, NMOSD was three times more common in Asian individuals than in the rest of the population, but the authors did not distinguish East Asian from other Asian individuals, and some cases were seronegative or lacked laboratory data. 21 In Olmsted County in the United States, estimated prevalence of NMOSD was about three times greater in Black (13/100,000) versus White individuals (4.0/100,000), with a total of six reported cases and five in White individuals. 20 A recent review of worldwide prevalence of NMOSD reported the lowest prevalence of NMOSD (0.7 to 1.9/100,000) in countries with predominantly White populations, prevalence ranging from 1.6 to 4.9/100,000 in Asian countries and higher prevalence estimates (~10/100,000) in two studies from small Caribbean island nations.3,20,24 Knowledge of the distribution of NMOSD cases across different ethnic groups is important to avoid under-recognition of NMOSD among non-White groups in the future.
The basis for increased risk of NMOSD in certain ethnic groups is not fully understood but may be due to both genetic and environmental factors. Certain human leukocyte antigen (HLA) variants predominate, in particular, ancestral groups and may potentiate risk of NMOSD, such as HLA-DQB1 and HLDA-DRB1 alleles in Native Americans with NMOSD in Mexico 4 and HLA-DRB1 alleles in Muslim Arabs in Israel 25 and in Japan. 26 The increased risk of NMOSD observed in immigrants may derive primarily from waves of immigrants to Canada from higher risk ethnic groups (e.g. East Asian, Black Caribbean) in the last decades 27 or could indicate geographic differences in unrecognized environmental risk factors for NMOSD.
We observed in this study very high odds of NMOSD in East Asians compared to Whites, although CIs were wide due to the small sample size. There may have been overestimation of magnitude of risk particularly in the national analyses due to regional differences in ethnic composition and possible under-representation of non-Whites among EnvIMS controls. In the 2016 national census, 6.2% of the Canadian population was identified as East Asian and 3.4% as Black, and about 22% of the population was born outside of Canada. 15 Under-representation of minorities is a known and widespread phenomenon in medical studies, and an important limitation to address in future controlled studies of NMOSD. 28 However, even if one were to compare NMOSD cases directly against the general distribution of ethnic groups from the 2016 census, the OR for NMOSD would remain higher than in many previous studies: 7.0 in East Asians and 8.5 in Blacks.
Two international studies have identified younger age of onset in Black compared to White individuals who develop NMOSD.13,19 In one multi-national study of 603 AQP4 + NMOSD patients, Blacks had a mean onset age of 33 years, Asians 36 years and Whites 44 years. 13 We observed a similar pattern across ethnic groups within the same geographic region, although these differences were not statistically significant. Understanding how age of onset may differ across ethnic groups may abet earlier diagnosis, prognostication and institution of tailored therapeutic approaches.
We did not observe any association between risk of NMOSD and age at menarche or reproductive history. An earlier study investigating age of NMOSD onset relative to age at menarche and reproductive history similarly did not find evidence of association. 9 Earlier age of menarche is thought to be a risk factor for MS and may partially explain why MS onset is rare before puberty. In contrast, NMOSD onset is relatively evenly distributed across the lifespan, although the postpartum phase appears to be a higher risk time for disease onset. 29 Other authors have postulated that female sex bias in certain autoimmune diseases may be influenced more by specific genes on the X-chromosome and environmental factors, such as differences in the gut microbiota, than by hormonal exposures.30,31
We identified an increased risk of NMOSD with other concomitant autoimmune diseases as previously reported.5,32 We observed an inverse association between ever smoking and NMOSD in the overall analysis, but no association in the Ontario-only sensitivity analysis. We believe that the lower NMOSD risk observed with ever smoking was a spurious finding related to high concentration of NMOSD cases and lower proportion of control participants from Ontario. There are known regional differences in smoking habits across Canada, with smoking being less common in Ontario, and in particular in Ontario women, and we observed the same tendency in this study. 33 One of the main challenges of epidemiological studies of rare diseases like NMOSD is to identify a control population which is sufficiently large and diverse, in terms of sex, age, ethnicity, immigrant status and geography to adequately represent the base population from which the cases are drawn.
Strengths of this study included our ability to use a validated, detailed questionnaire and a large population of unaffected controls from EnvIMS, a recent MS study. In this multi-centre study, we selected only AQP4 + NMOSD cases, thus minimizing the possibility of diversity in underlying biologic mechanisms of disease. We were able to separately assess effects of place of birth and ethnicity, an advantage over previous studies. Limitations include the potential for recall and misclassification bias which are inherent to retrospective questionnaire studies, the use of historical control data from EnvIMS, and the possibility of selection bias for NMOSD cases. Control responses from the original EnvIMS study were collected about 7–9 years before the NMOSD participant responses. We do not believe that the environmental exposures studied changed substantially during this period in the general population, but cannot exclude this possibility. Common biases in questionnaire studies include selection of cases with lower morbidity and higher socioeconomic status, 34 although we endeavoured to minimize this by multiple reminders to participants, as reflected in our high response rate, and by allowing help with questionnaire completion (although not response content) where disability may have interfered. We may have missed some cases particularly among immigrants, because the adapted EnvIMS questionnaire for this study was only available in English. While we conducted a sensitivity analysis in participants residing in Ontario, modelling of variables such as ethnicity and geography was limited by relatively small numbers, and thus, we may not fully have accounted for effects of these factors. Assessing risk factors in paediatric (<18 years) versus adult NMOSD onset cases would be of interest, but in this study, the subset with paediatric onset NMOSD (n = 6) was too small to allow for such a comparison. We utilized a questionnaire that was originally designed for the MS population. This approach may have restricted our ability to identify possible risk factors for NMOSD that do not overlap with those for MS. Notably, the high risk of NMOSD that we identified in immigrants versus those born in Canada sharply contrasts with the much lower risk of MS observed in immigrants versus non-immigrants in Canada, and may suggest critical differences in the environmental factors contributing to these disorders. 35
Conclusion
In conclusion, we found a higher risk of AQP4 + NMOSD in East Asian and Black compared to White individuals in Canada than that identified in many previous international studies. Risk of NMOSD was also increased in other non-White participants, those born outside of Canada, and those with other concomitant autoimmune disease. No association was observed with reproductive history or age at menarche despite the preponderance of women affected. This study illustrates the need for research exploring environmental risk factors for NMOSD distinct from those identified for MS. We recommend that future studies employ a case–control study design with careful consideration of unique demographic characteristics of the NMOSD population, including female predominance, immigrant composition and ethnic distribution.
Supplemental Material
sj-pdf-1-msj-10.1177_13524585231151953 – Supplemental material for A national case–control study investigating demographic and environmental factors associated with NMOSD
Supplemental material, sj-pdf-1-msj-10.1177_13524585231151953 for A national case–control study investigating demographic and environmental factors associated with NMOSD by Dalia L Rotstein, Christina Wolfson, Robert Carruthers, Mark S Freedman, Sarah A Morrow, Liesly Lee, Jodie M Burton, Rosane Nisenbaum, Andrea Konig, Sandra Magalhaes and Ruth Ann Marrie in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors acknowledge Shared Health/Health Sciences Centre Winnipeg.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: D.L.R. has received research support from the MS Society of Canada, Consortium of Multiple Sclerosis Centers (CMSC) and Roche Canada. She has received speaker or consultant fees from Alexion, Biogen, EMD Serono, Novartis, Roche and Sanofi Aventis. C.W. receives research funding from CIHR, Multiple Sclerosis Society of Canada and the Canada Foundation for Innovation. R.C. is the site investigator for studies funded by Roche, Novartis, Vielo Bio and Serono, and receives research support from Teva Innovation Canada, Roche Canada and Vancouver Coastal Health Research Institute. R.C. has received honoraria from Roche, EMD Serono, Sanofi, Biogen, Novartis and Alexion. M.S.F. has received research or educational grants from Sanofi Genzyme Canada. He has received honoraria or consultation fees from Alexion, Atara Biotherapeutics, Bayer Healthcare, BeiGene, BMS (Celgene), EMD Inc., F. Hoffmann-La Roche, Janssen (J&J), Merck Serono, Novartis, Sanofi Genzyme and Teva Canada Innovation. He has served as a member of an advisory board or board of directors for Alexion, Atara Biotherapeutics, Bayer Healthcare, BeiGene, BMS (Celgene), Celestra, F. Hoffmann-La Roche, Janssen (J&J), McKesson, Merck Serono, Novartis and Sanofi Genzyme. He has participated in a speaker’s bureau for Sanofi Genzyme and EMD Serono. S.A.M. has served as an advisory board member or received consulting fees from Biogen Idec, Bristol Myers Squibb/Celgene, EMD Serono, Novartis, Roche, Sanofi Genzyme and Teva Neuroscience. She has participated in a speaker’s bureau for Biogen Idec, Bristol Myers Squibb/Celgene, EMD Serono, Novartis, Roche and Sanofi Genzyme. She has received research support from Biogen Idec, Novartis, Roche and Sanofi Genzyme. She has participated as a site investigator in clinical trials sponsored by AbbVie, Bristol Myers Squibb/Celgene, EMD Serono, Novartis, Genzyme, Roche and Sanofi Genzyme. L.L. is a site investigator for studies funded by Roche, Novartis and Sanofi Aventis. He has received consultation fees from Alexion, Biogen, Bristol Myers Squibb, EMD Serono, Novartis, Roche and Sanofi Aventis. J.M.B. has received honoraria for speaking engagements and education activities and consultations from Roche, Biogen, Novartis and Prime. She has served as an advisor to CADTH. R.N. has nothing to disclose. A.K. has nothing to disclose. S.M. has nothing to disclose. R.A.M. receives research funding from Canadian Institutes of Health Research (CIHR), Research Manitoba, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, CMSC and US Department of Defense and the Arthritis Society. She is supported by the Waugh Family Chair in Multiple Sclerosis. She is a co-investigator on a study funded, in part, by Biogen Idec and Roche (no funds to her/her institution).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the Consortium of Multiple Sclerosis Centers (CMSC) and the University of Toronto Division of Neurology.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
