Abstract
Background:
There is limited and inconsistent information on the prevalence of cognitive impairment in neuromyelitis optica spectrum disorders (NMOSD).
Objective:
To assess cognitive performance and changes over time in NMOSD.
Methods:
This study included data from 217 aquaporin-4-IgG-seropositive (80%) and double-seronegative NMOSD patients. Cognitive functions measured by Symbol Digit Modalities Test (SDMT), Paced Auditory Serial-Addition Task (PASAT), and/or Multiple Sclerosis Inventory Cognition (MuSIC) were standardized against normative data (N = 157). Intraindividual cognitive performance at 1- and 2-year follow-up was analyzed. Cognitive test scores were correlated with demographic and clinical variables and assessed with a multiple linear regression model.
Results:
NMOSD patients were impaired in SDMT (p = 0.007), MuSIC semantic fluency (p < 0.001), and MuSIC congruent speed (p < 0.001). No significant cognitive deterioration was found at follow-up. SDMT scores were related to motor and visual disability (pBon < 0.05). No differences were found between aquaporin-4-IgG-seropositive and double-seronegative NMOSD.
Conclusions:
A subset of NMOSD patients shows impairment in visual processing speed and in semantic fluency regardless of serostatus, without noticeable changes during a 2-year observation period. Neuropsychological measurements should be adapted to physical and visual disabilities.
Introduction
Neuromyelitis optica spectrum disorders (NMOSD) are inflammatory autoimmune diseases of the central nervous system that characteristically affect the optic nerve, spinal cord, or area postrema. 1 In 2008, a study of 30 NMOSD patients reported for the first time negative effects of the diseases on cognition: More than half of the patients performed worse than healthy controls in at least one cognitive score. 2 Subsequent studies showed that NMOSD patients were most frequently impaired in the domains of attention, information processing speed, and memory.2–14 The reported prevalence of cognitive impairment in NMOSD varied between 29% 10 and 67% (Table 1). 15
Overview of previous studies about cognitive impairment in patients with NMOSD.
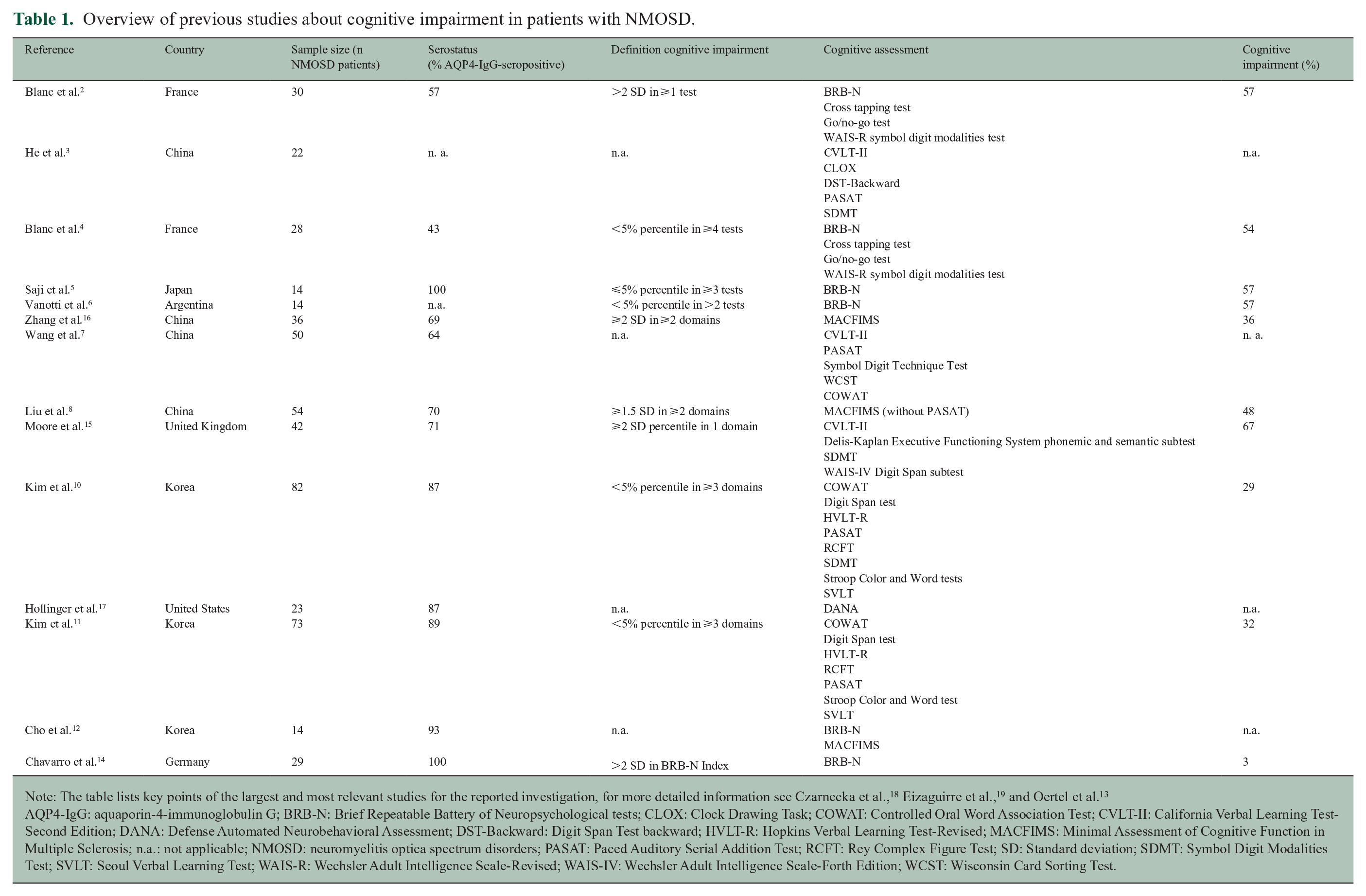
Note: The table lists key points of the largest and most relevant studies for the reported investigation, for more detailed information see Czarnecka et al., 18 Eizaguirre et al., 19 and Oertel et al. 13
AQP4-IgG: aquaporin-4-immunoglobulin G; BRB-N: Brief Repeatable Battery of Neuropsychological tests; CLOX: Clock Drawing Task; COWAT: Controlled Oral Word Association Test; CVLT-II: California Verbal Learning Test- Second Edition; DANA: Defense Automated Neurobehavioral Assessment; DST-Backward: Digit Span Test backward; HVLT-R: Hopkins Verbal Learning Test-Revised; MACFIMS: Minimal Assessment of Cognitive Function in Multiple Sclerosis; n.a.: not applicable; NMOSD: neuromyelitis optica spectrum disorders; PASAT: Paced Auditory Serial Addition Test; RCFT: Rey Complex Figure Test; SD: Standard deviation; SDMT: Symbol Digit Modalities Test; SVLT: Seoul Verbal Learning Test; WAIS-R: Wechsler Adult Intelligence Scale-Revised; WAIS-IV: Wechsler Adult Intelligence Scale-Forth Edition; WCST: Wisconsin Card Sorting Test.
Previous studies reported heterogeneous results regarding the effect of disease severity on cognitive impairment.2,3,5,6,8,10,12,15,16 Moreover, studies disagree about the association between depression and cognitive impairment in NMOSD: Correlations between depression severity and overall cognitive impairment, or isolated neuropsychological test scores, were found in various studies.3,6,10,15 In other studies, however, such correlations were absent.2,12,17
A major shortcoming of numerous studies is the lack of consideration of antibody status in the study analysis. To date, no study has found a link between the presence of aquaporin-4-immunoglobulin G (AQP4-IgG) and cognitive impairment, but patient samples were small.2,4,8,11,15,16
Inconsistent results might be due to high heterogeneity in terms of demographic and clinical characteristics, cognitive measurement methods, and the definition of cognitive impairment. In most former studies, cognitive impairment was defined as scoring below the fifth percentile in two or more subtests. However, there is no consistent definition of cognitive impairment across the studies (Table 1). In addition, small sample sizes (ranging from 14 to 82 participants) and predominantly single-center studies result in low statistical power (Table 1).13,18,19
Cognitive impairment seems to occur independently of disease duration.2,4,5,8,10–12,15–17 Furthermore, it was shown that cognitive impairment exists even in the limited form of NMOSD (i.e. AQP4-IgG-positivity with isolated optic neuritis or myelitis), suggesting that deficits affect patients from the early disease stage. 5 However, so far, no study has investigated cognitive abilities in NMOSD in a longitudinal study design.
The aim of this study was to investigate cognitive performance in a large sample of NMOSD patients with well-established antibody status in a multicenter study design and to gain initial insight into longitudinal cognitive changes in NMOSD patients.
Methods
Study participants
In this prospective, longitudinal, observational study patients (aged 18 years or older) were recruited at 17 German Neuromyelitis Optica Study Group (NEMOS) centers (www.nemos-net.de) during a 5-year-period (September 2015–April 2021). Only patients diagnosed with NMOSD according to the International Panel for NMO Diagnosis (IPND) 2015 criteria were included in the study.1,20 Exclusion criteria were predominance of a disease other than NMOSD and lack of informed consent. Moreover, myelin oligodendrocyte glycoprotein-antibody-associated diseases (MOG-AD) were excluded. Two follow-up assessments were analyzed at 11 to 16 months (follow-up one) and 23 to 28 months (follow-up two). Follow-up data surveyed at a different time interval were excluded.
The assessment included sociodemographic data, data on disability (assessed using the Expanded Disability Status Scale (EDSS) by trained physicians), 21 depressive symptoms (German version of the revised Beck Depression Inventory, BDI-II), 22 fatigue screening (Fatigue Scale for Motor and Cognitive Functions, FSMC), 23 and data from neuropsychological tests. To assess visual and motor disability the EDSS visual and motor function system score was used. Visual impairment was defined as any visual acuity below 1.0 and therefore an EDSS visual function system above zero. BDI-II scores were interpreted according to the following cutoff values: no depressive symptoms 0–8; minimal depressive symptoms 9–13; mild depressive symptoms 14–19; moderate depressive symptoms 20–28; severe depressive symptoms 29–63. 22 Fatigue was defined as FSMC score ⩾ 43 for mild fatigue; ⩾53 for moderate fatigue; ⩾63 for severe fatigue. 23
Well-established cell-based assays were used to test for serum antibodies to AQP4- and MOG-IgG.24,25
Standard protocol approvals, registrations, and patient consents
The study was part of the German Ministry of Education and Research (BMBF)-funded German Competence Network Multiple Sclerosis (KKNMS). The NEMOS cohort/KKNMS NationNMO-Study was approved by the ethics boards of the Hannover Medical School (no. 2009-5220) and other participating centers. All participants provided written informed consent before enrollment.
Neuropsychological test procedures
For neuropsychological testing, the Paced Auditory Serial-Addition Task (PASAT), 26 Symbol Digit Modalities Test (SDMT), 27 and Multiple Sclerosis Inventory Cognition (MuSIC) 28 were administered to the participants. The PASAT is a neuropsychological measure of processing speed in the auditory modality. 29 SDMT is used to examine processing speed in the visual modality. MuSIC, a short test battery used in German-speaking countries, evaluates immediate recall (recall of two orally presented lists of 10 words), semantic fluency including category switching (naming words from two different semantic categories in alternating sequence within one minute), visual processing speed (congruent speed) and inhibition score (incongruent speed minus congruent speed) from a modified Stroop task (correctly naming 30 visual presented silhouettes of animals with congruent/incongruent written names, respectively), and delayed recall (recall of the first list of words). The different tests were carried out by trained study staff, and each patient completed these cognitive tests in a separate quiet room.
For each patient, the test scores were z standardized based on normative data from 241 German-speaking healthy controls for the SDMT and PASAT, and 158 German-speaking healthy controls for the MuSIC.28,30 The normative data include healthy controls with a range of age from 19 to 60 and 62 years, respectively. Therefore, patients 60 years and older (n = 60) were excluded for baseline analysis to ensure comparability to the normative data. Among patients under 60 years, 81 patients completed the PASAT, 135 the SDMT, and 76 the MuSIC at baseline assessment. Test performance was considered to be impaired if individual z scores fell below the fifth percentile of the normative distribution. For baseline analysis, we hypothesized that z scores from the NMOSD sample significantly differ from the normative average (i.e. z = 0).
Statistical analyses
Statistical analyses were carried out with SPSS Version 27. Descriptive data are presented as medians with interquartile range (IQR) or range. For baseline analysis, a one-sample t test with the z standardized test scores and the test value 0 was performed. For follow-up analysis paired t test for normally distributed variables (follow-up one) and the Wilcoxon–Mann–Whitney test for variables with non-normal distribution (follow-up two) was used. We compared AQP4-IgG-seropositive with double-seronegative NMOSD patients, SDMT and MuSIC congruent speed between patients with and without visual impairment, and patients in an acute disease state (attack within the last 30 days of cognitive assessment) with patients in a chronic disease state using a two-sample t test. The Welch test and Wilcoxon–Mann–Whitney test were used if the assumptions were not given. Furthermore, we evaluated whether baseline neuropsychological test scores correlated with demographic or clinical parameters. The analyzed variables were age, sex, education, disease duration, EDSS motor and visual functional system scores, BDI-II depressive symptoms, and FSMC scores. Correlations were calculated using Spearman’s ρ. For categorical variables, eta coefficient (η) was used and tested for significance by one-way ANOVA. A multiple linear regression model was conducted to assess the relationship between the different baseline cognitive test values and demographic (age and education) and clinical parameters (serostatus, disease duration, EDSS, BDI-II, and FSMC score). Before analysis, all continuous variables were verified for normal distribution, and if necessary, logarithmic transformation was applied. Extreme values (>3 SD IQR distance) were excluded. Bootstrapping was performed. Dichotomous variables (i.e. education and antibody status) were dummy-coded. A two-sided p-value < 0.05 was considered statistically significant. Bonferroni correction was applied for correlation analysis and regression model. Effect sizes were calculated as Cohen’s d. Missing data were addressed by pairwise deletion to use all information observed. Sum scores (i.e. BDI-II, FSMC) were calculated only if all subscores were available. Except for baseline analysis with the z standardized test scores, all patients with available cognitive data were included.
Data availability
Anonymized data not published within this article will be made available on reasonable request from any qualified investigator.
Results
Patient’s characteristics
Datasets from 174 AQP4-IgG-seropositive patients and 43 double-seronegative patients with a median disease duration of 6 years were included. Double-seronegative patients were negative for both, AQP4-IgG and MOG-IgG. The selection procedure is illustrated in Figure 1. For the follow-up assessment data from 79 (follow-up one with a range of 11 to 16 months to follow-up) and 52 patients (follow-up two with a range of 23 to 28 months to follow-up) were available. Twenty-four percent (n = 19/79) and 22% (n = 11/51) had at least one attack between baseline and follow-up one and follow-up two, respectively. Median BDI-II score was 8 (i.e. no depressive symptoms) and median FSMC score was 56 (i.e. moderate fatigue). Demographic and clinical sample characteristics are shown in Table 2.
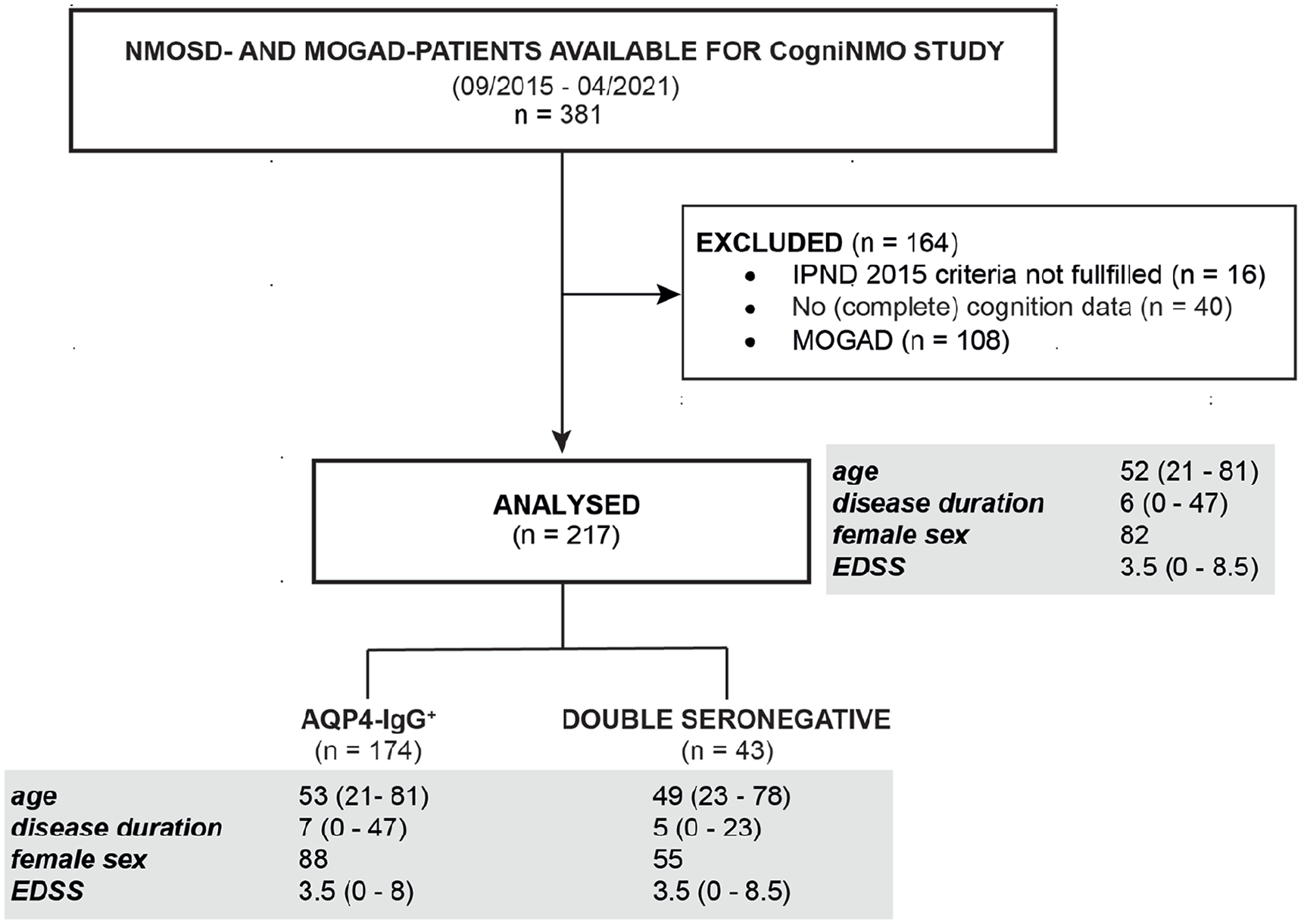
CogniNMO-Study—Selection procedure and cohort characterization stratified by serostatus.
Sample characteristics.
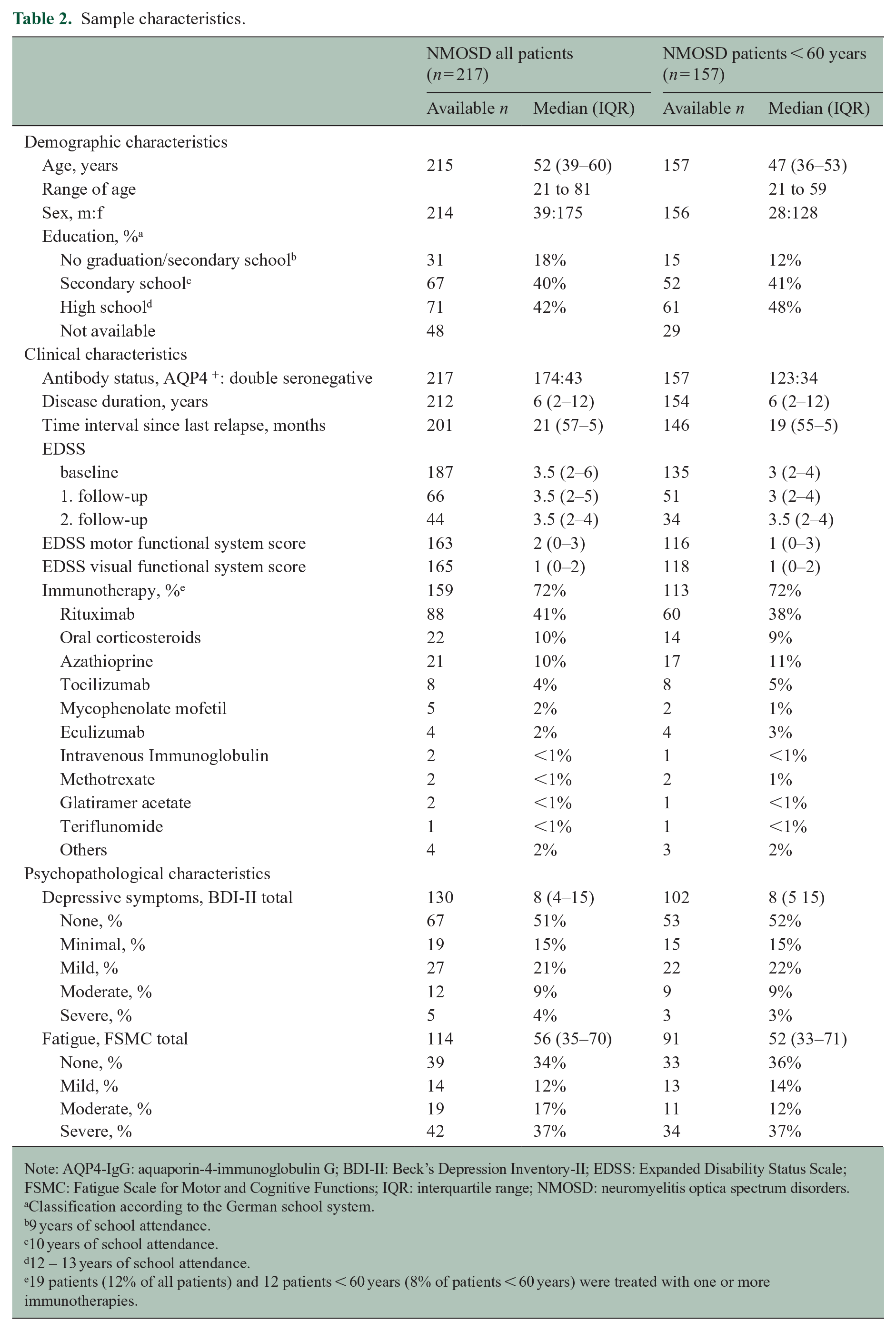
Note: AQP4-IgG: aquaporin-4-immunoglobulin G; BDI-II: Beck’s Depression Inventory-II; EDSS: Expanded Disability Status Scale; FSMC: Fatigue Scale for Motor and Cognitive Functions; IQR: interquartile range; NMOSD: neuromyelitis optica spectrum disorders.
Classification according to the German school system.
9 years of school attendance.
10 years of school attendance.
12 – 13 years of school attendance.
19 patients (12% of all patients) and 12 patients < 60 years (8% of patients < 60 years) were treated with one or more immunotherapies.
Baseline analysis
At baseline, we considered only patients younger than 60 years (n = 157) to ensure strict comparability with normative data. Patients performed worse than the average population norm on the SDMT (mean = −0.37, 95% confidence interval (CI) (−0.63, −0.10)), on MuSIC semantic fluency (mean = −0.42, 95% CI (−0.62, −0.22)), and on MuSIC congruent speed (mean = −1.71, 95% CI (−2.36, −1.06)). Patients performed better than the population norm on MuSIC immediate recall list B (mean = 0.28, 95% CI (0.06, 0.51)). The means with 95% CI are provided in Figure 2 and Table 3.
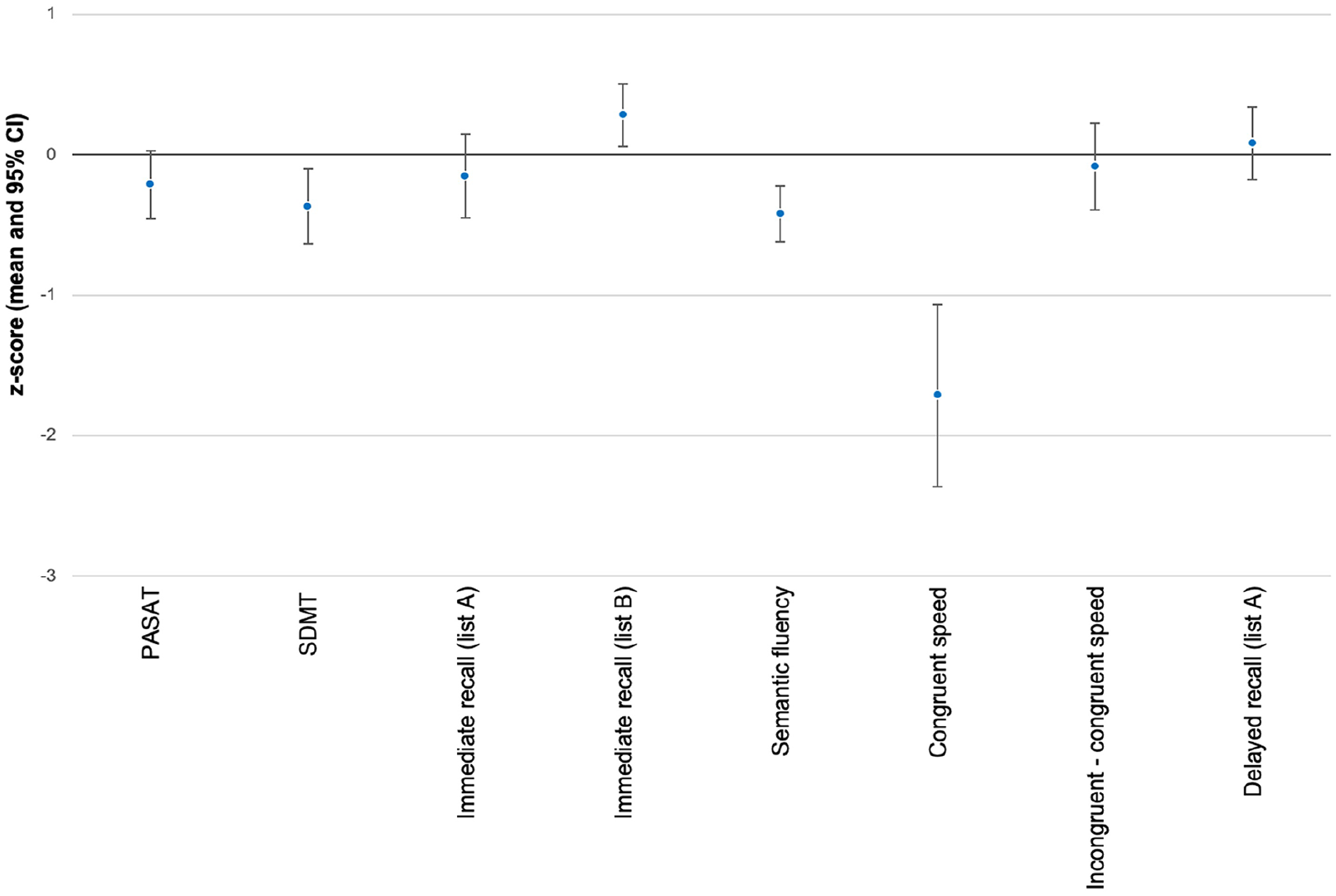
Mean z scores of cognitive tests at baseline.
Mean normative z scores of cognitive tests and t test at baseline.
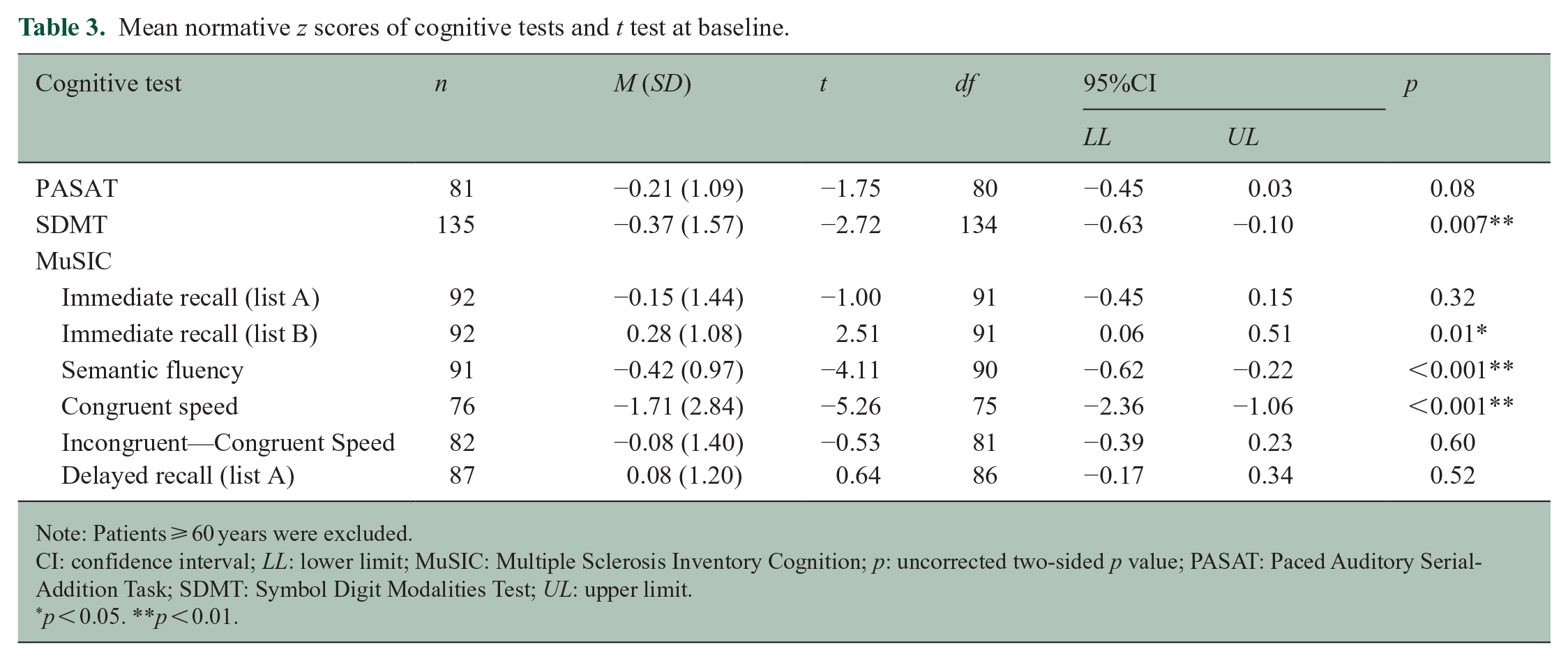
Note: Patients ⩾ 60 years were excluded.
CI: confidence interval; LL: lower limit; MuSIC: Multiple Sclerosis Inventory Cognition; p: uncorrected two-sided p value; PASAT: Paced Auditory Serial-Addition Task; SDMT: Symbol Digit Modalities Test; UL: upper limit.
p < 0.05. **p < 0.01.
On at least one test score 40% (n = 62/157) of NMOSD patients performed below the fifth percentile. Among patients who completed two or more tests, 19% (n = 23/123) showed evidence of impairment on at least two cognitive test scores (Table 4). The highest proportion of impairment in cognitive tests was reached for MuSIC congruent speed (n = 31/76, 41%).
Frequencies of patients with impairment in cognitive tests at baseline, 1- and 2-year follow-up.
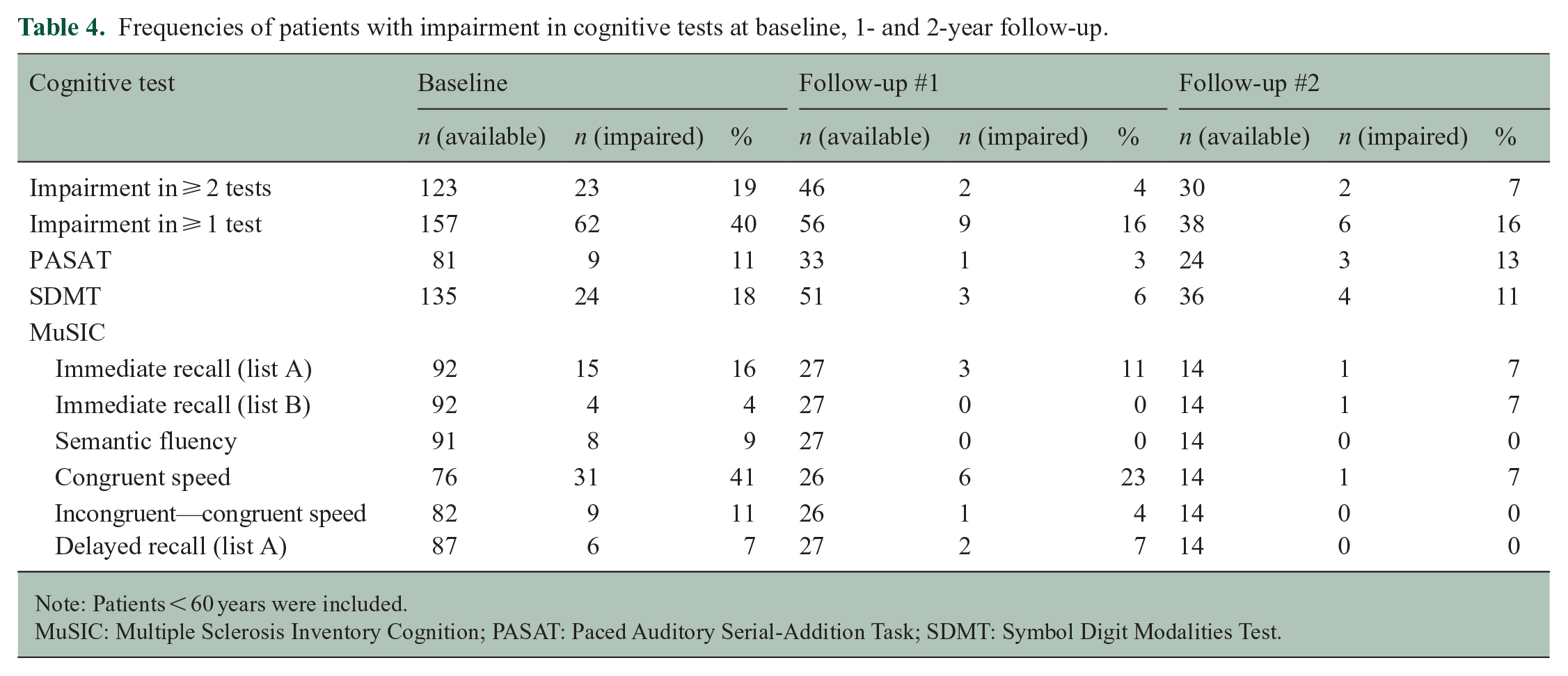
Note: Patients < 60 years were included.
MuSIC: Multiple Sclerosis Inventory Cognition; PASAT: Paced Auditory Serial-Addition Task; SDMT: Symbol Digit Modalities Test.
Twenty-three patients (11 % of all patients), and 15 patients < 60 years (10 % of patients < 60 years) had an attack within the last 30 days of cognitive assessment. Importantly, there were no significant differences in the cognitive tests between patients in an acute disease state and patients in a chronic disease state (Supplemental eTable 1).
Across all cognitive tests, no significant differences were found between AQP4-IgG-seropositive and double-seronegative NMOSD patients (Supplemental eTable 2). Both groups were comparable in terms of demographic and clinical variables (Supplemental eTable 3).
Follow-up analysis
Among all NMOSD patients, there was no significant change in performance across all cognitive test scores at 1- and 2-year follow-up. Regarding MuSIC congruent speed patients performed better at first (p = 0.04) and at the second follow-up (p = 0.007) compared to baseline. Frequencies of impairment in cognitive tests and mean normative z scores of cognitive tests at baseline and follow-up are given in Table 4 and Supplemental eTable 4, respectively.
Association between cognitive test scores and clinical and demographic variables
SDMT performance correlated with EDSS visual function score (rs = −0.31, 95% CI (−0.46, −0.16)) and with EDSS motor function score (rs = −0.42, 95% CI (−0.55, −0.27)). BDI-II score was associated with SDMT and MuSIC congruent speed, but both correlations did not remain significant after Bonferroni correction. FSMC score was related to SDMT test score (rs = −0.35, 95% CI (−0.52, −0.15)). There was no substantial difference in the results when correlation analysis excluded data from patients older than 60 years.
Interestingly, disease duration was not associated with cognitive performance. Correlations between test scores and demographic and clinical parameters are shown in Table 5.
Spearman’s correlation coefficients at baseline.
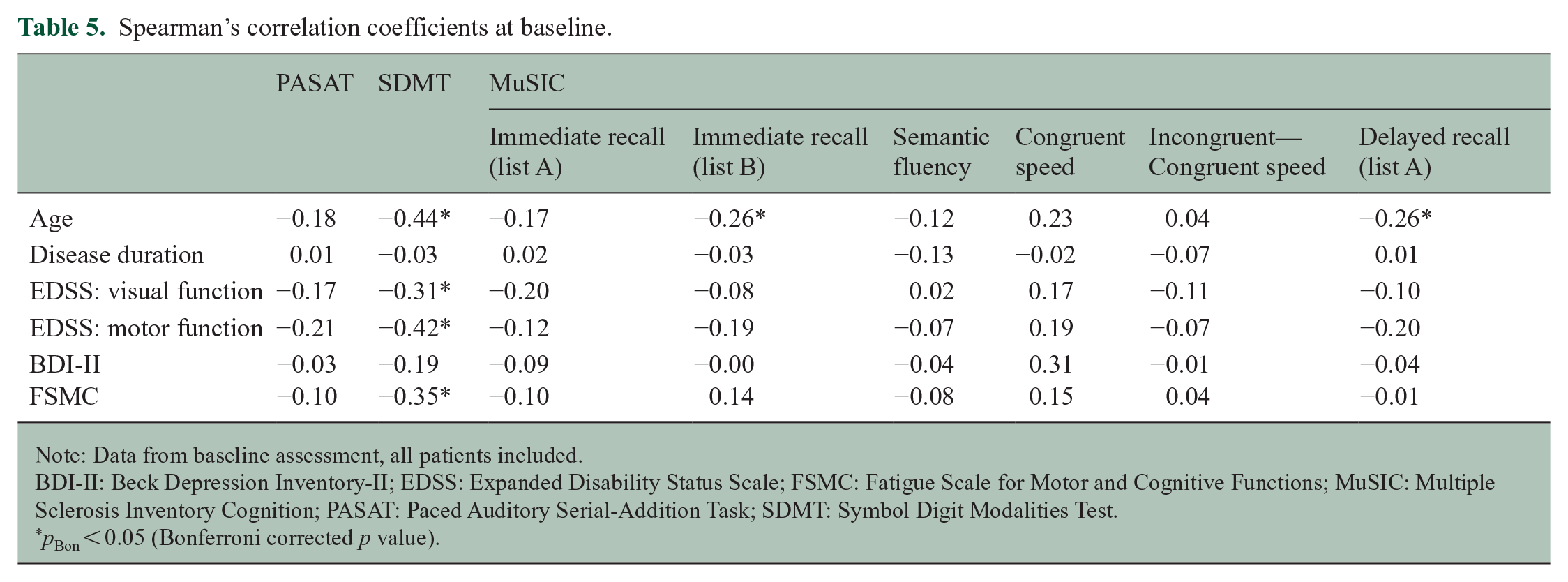
Note: Data from baseline assessment, all patients included.
BDI-II: Beck Depression Inventory-II; EDSS: Expanded Disability Status Scale; FSMC: Fatigue Scale for Motor and Cognitive Functions; MuSIC: Multiple Sclerosis Inventory Cognition; PASAT: Paced Auditory Serial-Addition Task; SDMT: Symbol Digit Modalities Test.
pBon < 0.05 (Bonferroni corrected p value).
The education level correlated with PASAT (η = 0.30, p = 0.03), SDMT (η = 0.26, p = 0.008), MuSIC immediate recall list A (η = 0.33, p = 0.005), MuSIC congruent speed (η = 0.27, p = 0.04), and MuSIC delayed recall (η = 0.31, p = 0.01). Only MuSIC immediate recall list A remained significant after Bonferroni correction (pBon < 0.05). There was no association between sex and cognitive performance across all cognitive tests (Supplemental eTables 5 and 6).
Results of multiple linear regression showed that clinical and demographic variables explain a significant amount of the variance in SDMT score (R2 = 0.45, adjusted R2 = 0.39, p < 0.001) (Supplemental eTable 7). Only age remained as a significant variable after Bonferroni correction (higher age associated with lower cognitive test performance, p = 0.02). The model could not explain variance in PASAT score and MuSIC subtest scores (Supplemental eTables 8–14).
Comparing SDMT test scores of visually impaired patients (EDSS visual function system score > 0) with those of patients without visual impairment (EDSS visual function system score = 0) revealed a significant difference (mean difference = 5.71, CI 95% (0.83, 10.59)). No difference between these patient groups was observed at MuSIC congruent speed (U = 612.50, Z = −1.17, p = 0.24).
Discussion
By examining data from 217 NMOSD patients, this study aimed to characterize the frequency and the type of impairment as well as the intra-individual changes in cognitive performance over a two-year period. We further investigated if clinical or demographic parameters are associated with cognitive performance. We made a clear distinction to MOG-IgG-seropositive patients by including only AQP4-IgG-seropositive or double-seronegative patients in the analysis.
Compared to normative data of healthy controls, NMOSD patients showed below-average visual processing speed and semantic fluency. These results are in line with previous studies.2–11,14,16,17 In contrast to what has been suggested in some studies, however, we found no impairment with regard to immediate and delayed recall of verbal material presented in the auditory modality.2–5,7,11,16
We refer to low performance in cognitive tests as ‚impairment in cognitive tests’ rather than as “cognitive impairment” in an attempt to avoid over-interpretation of the data. As an example, poor performance on tests that involve visuomotor abilities may reflect visual, motor, or cognitive impairment, or any conceivable combination of these. Nineteen percent of patients were impaired in at least two cognitive subtests and 40% of patients exhibited impairment in at least one cognitive subtest. Earlier studies reported a prevalence of 29% to 67%, partly reflecting differences in the definition of impairment (i.e. impairment in one or two or more subtests, see Table 1). Our results from a large prospective multicenter cohort indicate that the prevalence of impairment in cognitive tests in NMOSD seems to be lower than previously assumed. However, previous studies were based on small sample sizes, potentially leading to an overestimation of the true prevalence due to selection bias.
Remarkably, our study reveals that NMOSD patients differed from healthy controls in cognitive performance measured by the SDMT but not by the PASAT. Both tests examine similar cognitive domains (i.e. processing speed) but differ with regard to stimulus modalities (visual vs auditive). Most previous studies reported cognitive impairment measured by the PASAT as well.2–6,8,9,11,14,16 Nevertheless, our results are in line with two previous studies of larger NMOSD samples.7,10 Prior findings indicated that SDMT and PASAT test performance were independent of visual impairment. 14 In our study, the SDMT score was moderately to strongly linked to physical and visual disability, unlike PASAT test performance. Other studies also showed that SDMT test performance can be influenced by visual ability.31,32 This might be a possible explanation for the discrepancy between SDMT and PASAT results.
As an interim conclusion, our data suggest below-average visual processing speed (as evidenced by the SDMT and MuSIC congruent speed results), but intact auditory processing speed in NMOSD patients. Furthermore, episodic memory for auditory verbal material was preserved in NMOSD patients. The result for MuSIC semantic fluency constituted the sole exception from the rule that visual processing was affected, while nonvisual processing speed was left intact in NMOSD patients. It remains to be seen whether or not this finding reflects the fact that the MuSIC semantic fluency task requires switching between two different semantic categories, i.e., that it places heavy demands on cognitive flexibility.
Results of the correlation analysis indicate that especially the SDMT might not be suitable to assess cognitive abilities in NMOSD patients. EDSS visual and motor function scores along with FSMC scores were significantly related to SDMT test performance even after conservative correction for multiple testing.
No difference in cognitive performance was found between AQP4-IgG-seropositive and double-seronegative patients in direct comparison. A recent study showed that astrocytopathy (as the underlying NMOSD pathology) was not restricted to affected brain areas implying a whole-brain involvement in NMOSD patients. The astrocytic reactions occurred independently of IgG-antibodies against AQP4. 33 This may explain a whole-brain involvement with resulting impairment in visual processing speed and semantic fluency in both, AQP4-IgG-seropositive and double-seronegative NMOSD patients.
No significant cognitive deterioration was observed at 1- and 2-year follow-up. However, retesting effects and/or attrition bias might be responsible for this outcome. In line with other studies, cognitive performance was not linked to disease duration.2,4,5,8,10–12,15–17
Considering the rarity of the disease, the strength of this study lies in the large patient sample. In addition, all cognitive tests were performed by experienced and trained study investigators. Furthermore, all clinical data were derived from a cohort database in which, for instance, EDSS data were assessed by trained physicians. Serological results were crosschecked within the NEMOS network via a central laboratory, allowing valid differentiation between AQP4-IgG-seropositive and double-seronegative NMOSD. Notably, no differences were found.
We examined cognitive functions by well-established neuropsychological tests, but as other studies partly used different test batteries with different subtests, a comparison of the frequency of cognitive deficits across studies is limited and should be made with caution.
Another limitation of our study consists of a potentially too short follow-up interval, possibly explaining why we were unable to detect relevant changes in cognitive performance over time. Furthermore, cognitive deterioration might be masked by learning effects. Indeed, this is emphasized by the fact that patients performed significantly better in most cognitive tests at follow-up. This is a frequently reported phenomenon when testing cognitive performance in longitudinal study designs.29,34,35 Even though different test versions were used for the PASAT, patients seemed to become familiar with the type of testing. This implies that different stimulus does not prevent practice effects.34,35 In future studies, a longer observation period should be considered. Another limitation is that data at follow-up are restricted compared to baseline analysis. However, as the neuropsychological testing is part of an ongoing study, some follow-up assessments are still pending.
In the current study, the cognitive performance of NMOSD patients over 60 years of age could not be compared to healthy controls. Thus, normative cognitive data of healthy controls over 60 years should be provided to examine elderly NMOSD patients.
Conclusion
In summary, this study assessed the extent of impairment in neuropsychological tests in the largest NMOSD sample reported to date and analysed cognition data at follow-up. The data illustrate that NMOSD patients displayed below-average visual processing speed and semantic fluency. The prevalence of impairment in cognitive tests was lower than previously reported. Cognitive performance was independent of disease duration and stayed constant for two years. Furthermore, there was no difference in cognitive performance between AQP4-IgG-seropositive and double-seronegative NMOSD patients. We propose to develop a standard change-sensitive test battery adapted to NMOSD patients for use in future longitudinal studies to examine cognitive performance independently of visual and motor disabilities.
Supplemental Material
sj-docx-1-msj-10.1177_13524585231151212 – Supplemental material for Cognition in patients with neuromyelitis optica spectrum disorders: A prospective multicentre study of 217 patients (CogniNMO-Study)
Supplemental material, sj-docx-1-msj-10.1177_13524585231151212 for Cognition in patients with neuromyelitis optica spectrum disorders: A prospective multicentre study of 217 patients (CogniNMO-Study) by Martin W Hümmert, Carlotta Stern, Friedemann Paul, Ankelien Duchow, Judith Bellmann-Strobl, Ilya Ayzenberg, Carolin Schwake, Ingo Kleiter, Kerstin Hellwig, Sven Jarius, Brigitte Wildemann, Makbule Senel, Achim Berthele, Katrin Giglhuber, Felix Luessi, Matthias Grothe, Luisa Klotz, Rasmus Schülke, Stefan Gingele, Jürgen H Faiss, Annette Walter, Clemens Warnke, Florian Then Bergh, Orhan Aktas, Marius Ringelstein, Jan-Patrick Stellmann, Vivien Häußler, Joachim Havla, Hannah Pellkofer, Tania Kümpfel, Bruno Kopp and Corinna Trebst in Multiple Sclerosis Journal
Footnotes
Acknowledgements
We would like to thank all patients for participating in the study, their family members, and all contributors of the Neuromyelitis Optica Study Group (NEMOS) for their support. Special thanks also go to Sikinika Hache, Karin Fricke, Kathrin Scheiwe, Ilona Cierpka-Leja, Heike Miethke, Monika Höveler, and Carolin Risau for excellent practical support. A sincere thank you to Peter Scherer (Neurozentrum Zehlendorf, Berlin) for providing the raw data on PASAT and SDMT of healthy subjects for comparison in our study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MWH reports no disclosures relevant to the manuscript. CSt reports no disclosures relevant to the manuscript. FP receives honoraria for lecturing, and travel expenses for attending meetings from Guthy Jackson Foundation, Sanofi Genzyme, Novartis, Alexion, Viela Bio, Roche, UCB, Mitsubishi Tanabe, and Celgene. His research is funded by the German Ministry for Education and Research (BMBF), Deutsche Forschungsgemeinschaft (DFG), Einstein Foundation, Guthy Jackson Charitable Foundation, EU FP7 Framework Program, Biogen, Genzyme, Merck Serono, Novartis, Bayer, Teva, Alexion, Roche, Parexel, Viela Bio, and Almirall. FP serves on advisory boards and steering committees for Novartis and Viela Bio and is Associate Editor of Neurology, Neuroimmunology & Neuroinflammation and Academic Editor for PLoS ONE. AD reports no disclosures relevant to the manuscript. JBS has received travel grants and speaking honoraria from Bayer Healthcare, Biogen Idec, Merck Serono, Sanofi Genzyme, Teva Pharmaceuticals, Roche, and Novartis all unrelated to this work. IA received personal fees from Roche, Alexion, and Merck and received research support from Diamed, none related to this manuscript. CSc reports no disclosures relevant to the manuscript. IK has received personal compensation for consulting, serving on a scientific advisory board, speaking, or other activities with Alexion, Biogen, Celgene, Hexal, Horizon, Merck, and Roche/Chugai. KH received consultant and speaker honoraria from Bayer, Biogen, Merck, Novartis, Sanofi Genzyme, Roche, and Teva. SJ reports no disclosures relevant to the manuscript. BW received grants from the German Ministry of Education and Research, Deutsche Forschungsgemeinschaft, Dietmar Hopp Foundation, and Klaus Tschira Foundation, grants and personal fees from Merck, Novartis, and personal fees from Roche; none related to this work. MS has received consulting and/or speaker honoraria from Alexion, Bayer, Biogen, Merck, Roche, and Sanofi Genzyme. She has received research funding from the Hertha-Nathorff-Program. None of this interfered with the current report. AB received speaker and consulting honoraria from Alexion, Biogen, Bayer Healthcare, Celgene, Merck, Novartis Pharma, and Roche; all outside the submitted work. KG reports no disclosures relevant to the manuscript. FL received consultancy fees from Roche and support with travel cost from Teva Pharma. MG received honoraria and travel reimbursements for attending meetings from Biogen, Celgene, Merck Serono, Novartis, Roche, Sanofi-Genzyme, and Teva and research grants from the German Ministry for Education and Research (BMBF), Merck Serono, and Novartis. None of this interfered with the current report. LK received compensation for serving on Scientific Advisory Boards for Alexion, Biogen, Celgene GmbH, Genzyme, Horizon, Janssen, Merck Serono, Novartis, and Roche. She received speaker honoraria and travel support from Bayer, Biogen, Celgene GmbH, Genzyme, Grifols, Merck Serono, Novartis, Roche, Santhera, and Teva. She receives research support from the German Research Foundation, the IZKF Münster, IMF Münster, Biogen, Immunic AG, Novartis, and Merck Serono. RS reports no disclosures relevant to the manuscript. SG has received speaker honoraria from Alnylam, not related to this manuscript. JHF reports no disclosures relevant to the manuscript. AW received speaker honoraria and meeting expenses from Novartis, Bayer, Biogen, Sanofi Genzyme, Teva, Roche, and Merck. CW has received institutional honoraria and/or grant support from Novartis, Sanofi-Genzyme, Alexion, Janssen, Merck, Biogen, and Roche. FTB has received honoraria for speaking and advisory board consultation from Alexion, Roche, and Horizon Therapeutics; none of these had an impact on this manuscript. OA has received personal fees from Alexion, Bayer Healthcare, Biogen, Celgene, Merck Serono, MedImmune, Novartis, Roche, Teva, and Zambon, outside of the submitted work. MR received speaker honoraria from Novartis, Bayer Vital GmbH, Roche, Alexion, and Ipsen and travel reimbursement from Bayer Schering, Biogen Idec, Merz, Genzyme, Teva, Roche, and Merck, none related to this study. JPS reports no disclosures relevant to the manuscript. VH reports no disclosures relevant to the manuscript. JH reports personal fees, research grants and non-financial support from Merck, Novartis, Roche, Santhera, Biogen, Alexion, Celgene, Janssen; and non-financial support of the Guthy-Jackson Charitable Foundation, all outside the submitted work. J.H. is (partially) funded by the German Federal Ministry of Education and Research [Grant Numbers 01ZZ1603[A-D] and 01ZZ1804[A-H] (DIFUTURE)]. HP received honoraria for lectures from Bayer Health Care, Biogen Idec, and Teva Pharma and travel reimbursement from Novartis. TK has received speaker honoraria and/or personal fees for advisory boards from Bayer Healthcare, Teva Pharma, Merck, Novartis Pharma, Sanofi-Aventis/Genzyme, Roche Pharma, Alexion, and Biogen as well as grant support from Novartis and Chugai Pharma in the past. None of this interfered with the current report. BK reports no disclosures relevant to the manuscript. CT has received honoraria for consultation and expert testimony from Alexion Pharma Germany GmbH, Biogen Idec/GmbH, Chugai Pharma Germany GmbH, MERCK, Novartis Pharma GmbH and Roche Pharma GmbH. None of this interfered with the current report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The NEMOS cohort/NationNMO is supported by the German Ministry for Education and Research (BMBF) as part of the German Competence Network Multiple Sclerosis (KKNMS; for NEMOS NationNMO-DAB FKZ 01GI1602 C to J.-P.S., NationNMO-PAT FKZ 01GI1602B to O.A., and NationNMO-LAB to B.W.)
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
