Abstract
Background:
The impact of multiple sclerosis (MS) disease-modifying therapies (DMTs) on SARS-CoV-2 vaccination response is uncertain.
Methods:
Post-SARS-CoV-2 vaccination blood samples across multiple DMTs were tested for SARS-CoV-2 immunoglobulin G (IgG) response.
Results:
Three hundred twenty-two people with MS were included; 91.9% received an mRNA vaccine. Post-vaccination reactive IgG rates (IgG index > 1) were 40% for anti-CD20 (32/80 patients); 41% for sphingosine 1-phosphate receptor modulators (S1PRM, 16/39); and 100% for all other classes, including the no DMT group.
Conclusion:
Anti-CD20 therapies and S1PRMs reduce IgG response to SARS-CoV-2 vaccination; IgG response is preserved with other DMTs.
Keywords
Introduction
Coronavirus disease 2019 (COVID-19) has resulted in millions of deaths worldwide. 1 Vaccination against SARS-CoV-2, the virus that causes COVID-19, is the most effective means of protecting populations, including people with multiple sclerosis (PwMS), 2 and ending the COVID-19 pandemic. 3 However, as a result of immunomodulatory effects or depletion of circulating lymphocytes, MS disease-modifying therapies (DMTs) may reduce humoral or cellular immune responses to vaccines, including against SARS-CoV-2.1,4,5 Although the clinical significance of these varying responses, including the impact on risk of infection, is not fully understood, the COVID-19 pandemic makes better characterization of how MS DMTs influence vaccine effectiveness critically important.1,4,5
Limited data relating to the efficacy of COVID-19 vaccines in DMT-treated PwMS have led to varying vaccination guidelines from MS organizations. 4 An initial study suggested that immunoglobulin G (IgG) antibody responses to SARS-CoV-2 vaccination are attenuated in PwMS treated with the anti-CD20 therapy ocrelizumab or the sphingosine 1-phosphate receptor modulator (S1PRM) fingolimod. 5 This observation is consistent with previous studies of non-COVID vaccine responses in PwMS treated with these DMTs.6,7 Thus, robust data on immune responses to COVID-19 vaccines across multiple DMTs are needed to support evidence-based COVID-19 vaccination recommendations. We report initial results from a large real-world study of the impact of different DMT classes on IgG responses to SARS-CoV-2 vaccines.
Methods
Study design
Participants with MS enrolled in the Multiple Sclerosis Partners Advancing Technology and Health Solutions (MS PATHS) network in the United States, Germany, and Spain were recruited to voluntarily provide pre- and post-vaccination blood serum samples. MS PATHS comprises 10 MS centers that collect standardized data during routine office visits with the aim of improving outcomes for PwMS. Sample collection is ongoing; initial post-vaccination data from DMT classes (with minimum ⩾10 patients) are reported here. DMTs included in the preliminary analysis were grouped by mechanisms of action: alemtuzumab, anti-CD20, fumarates, glatiramer acetate (GA), interferons (IFNs), natalizumab, S1PRMs, teriflunomide, and no DMT.
For this study, serum samples could be collected up to 30 days pre-vaccination relative to the first injection of the vaccine and 28–90 days after the final vaccine dose. Post-vaccination blood samples, collected with or without a corresponding pre-vaccination sample, were taken after two injections for the AstraZeneca, Pfizer, and Moderna vaccines, and one injection for the Johnson & Johnson vaccine. The Siemens Healthineers Atellica® IM* SARS-CoV-2 IgG assay was used to measure semi-quantitative (index value) IgG response to the SARS-CoV-2 spike protein. 8 Univariate logistic regression analyses assessed the relationship between post-vaccination IgG response (reactive/non-reactive) and demographic, disease, and vaccine characteristics. These analyses were restricted to participants on anti-CD20 or S1PRM therapy since there was no heterogeneity in IgG response in other DMT categories. Univariate linear regression was used to assess the relationship between time of last anti-CD20 dose and IgG index value.
The study was approved by central and local ethics committees and conducted in accordance with the International Council on Harmonization guidelines for Good Clinical Practice and the Declaration of Helsinki. All patients provided written, informed consent.
Results
As of 24 August, 2021, post-vaccination serum samples were obtained from 322 participants: alemtuzumab (n = 23), anti-CD20 (n = 80; ocrelizumab n = 57, rituximab n = 22, ofatumumab n = 1), fumarates (n = 37; dimethyl fumarate n = 32, diroximel fumarate n = 5), GA (n = 33), IFNs (n = 23; IM IFN beta-1a n = 16, pegylated IFN beta-1a n = 6, IFN beta-1b, n = 1), natalizumab (n = 44), S1PRMs (n = 39; fingolimod n = 32, ozanimod n = 4, siponimod n = 3), teriflunomide (n = 15), and no DMT (n = 28). Post-vaccination samples were collected 47.4 (16.2) days after the last vaccine dose; 296/322 (91.9%) were following an mRNA vaccine. No pre-vaccination samples were available for these participants.
Baseline demographics and disease characteristics by DMT class are shown in Table 1. Reactive IgG rates (IgG index >1) from initial post-vaccination testing were 32/80 participants (40%) for anti-CD20, 16/39 (41%) for S1PRMs, and 100% for all other classes, including alemtuzumab (n = 23), fumarates (n = 37), GA (n = 33), IFNs (n = 23), natalizumab (n = 44), teriflunomide (n = 15), and no DMT (n = 28). SARS-CoV-2 IgG index value by DMT is shown in Figure 1. IgG response was not correlated with time of last dose of anti-CD20 therapies (Figure 2). In the anti-CD20 and S1PRM groups, for 1 year increase in the duration of therapy, odds of a reactive IgG results are: ORanti-CD20 = 0.53, 95% confidence interval (CI) = (0.34, 0.83) and ORS1PRM = 0.70, 95% CI = (0.53, 0.93). There was no association between probability of a reactive IgG result and patient characteristics, and no association between reactive IgG status and MS subtype (Figure 1).
Clinical and demographic characteristics of DMT-treated and DMT-untreated participants with MS who received SARS-CoV-2 vaccination.
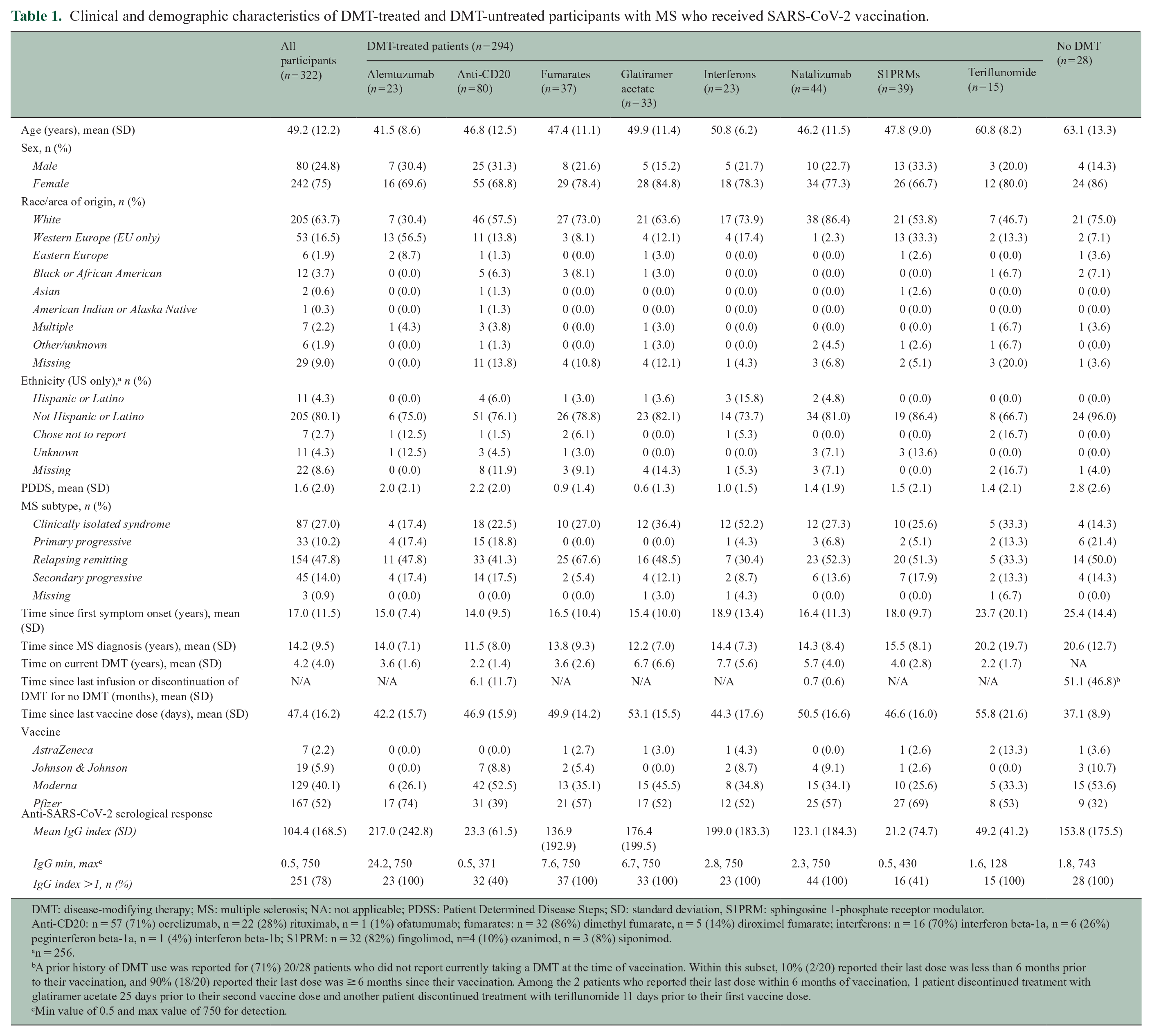
DMT: disease-modifying therapy; MS: multiple sclerosis; NA: not applicable; PDSS: Patient Determined Disease Steps; SD: standard deviation, S1PRM: sphingosine 1-phosphate receptor modulator.
Anti-CD20: n = 57 (71%) ocrelizumab, n = 22 (28%) rituximab, n = 1 (1%) ofatumumab; fumarates: n = 32 (86%) dimethyl fumarate, n = 5 (14%) diroximel fumarate; interferons: n = 16 (70%) interferon beta-1a, n = 6 (26%) peginterferon beta-1a, n = 1 (4%) interferon beta-1b; S1PRM: n = 32 (82%) fingolimod, n=4 (10%) ozanimod, n = 3 (8%) siponimod.
n = 256.
A prior history of DMT use was reported for (71%) 20/28 patients who did not report currently taking a DMT at the time of vaccination. Within this subset, 10% (2/20) reported their last dose was less than 6 months prior to their vaccination, and 90% (18/20) reported their last dose was ≥ 6 months since their vaccination. Among the 2 patients who reported their last dose within 6 months of vaccination, 1 patient discontinued treatment with glatiramer acetate 25 days prior to their second vaccine dose and another patient discontinued treatment with teriflunomide 11 days prior to their first vaccine dose.
Min value of 0.5 and max value of 750 for detection.
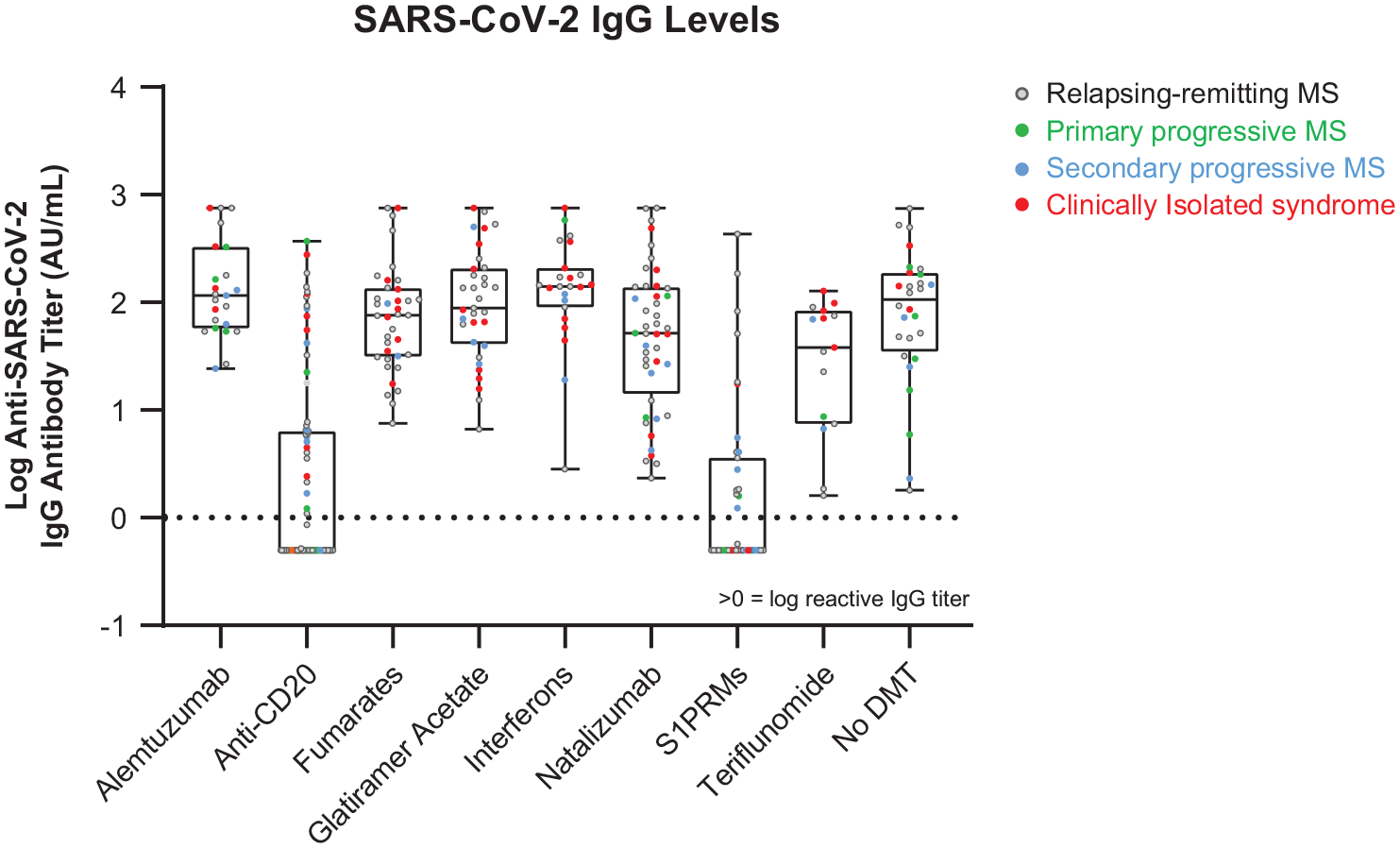
Unadjusted post-vaccination SARS-CoV-2 IgG levels by DMT class. Post-vaccination blood serum samples, collected 28–90 days after the final vaccine dose, were assessed for SARS-CoV-2 immunoglobulin G (IgG) response to the spike protein using Siemens Healthineers Atellica® IM* SARS-CoV-2 IgG assay. Unadjusted log-transformed anti-SARS-CoV-2 IgG index values are presented. Log-transformed IgG index values >0 are interpreted as reactive.
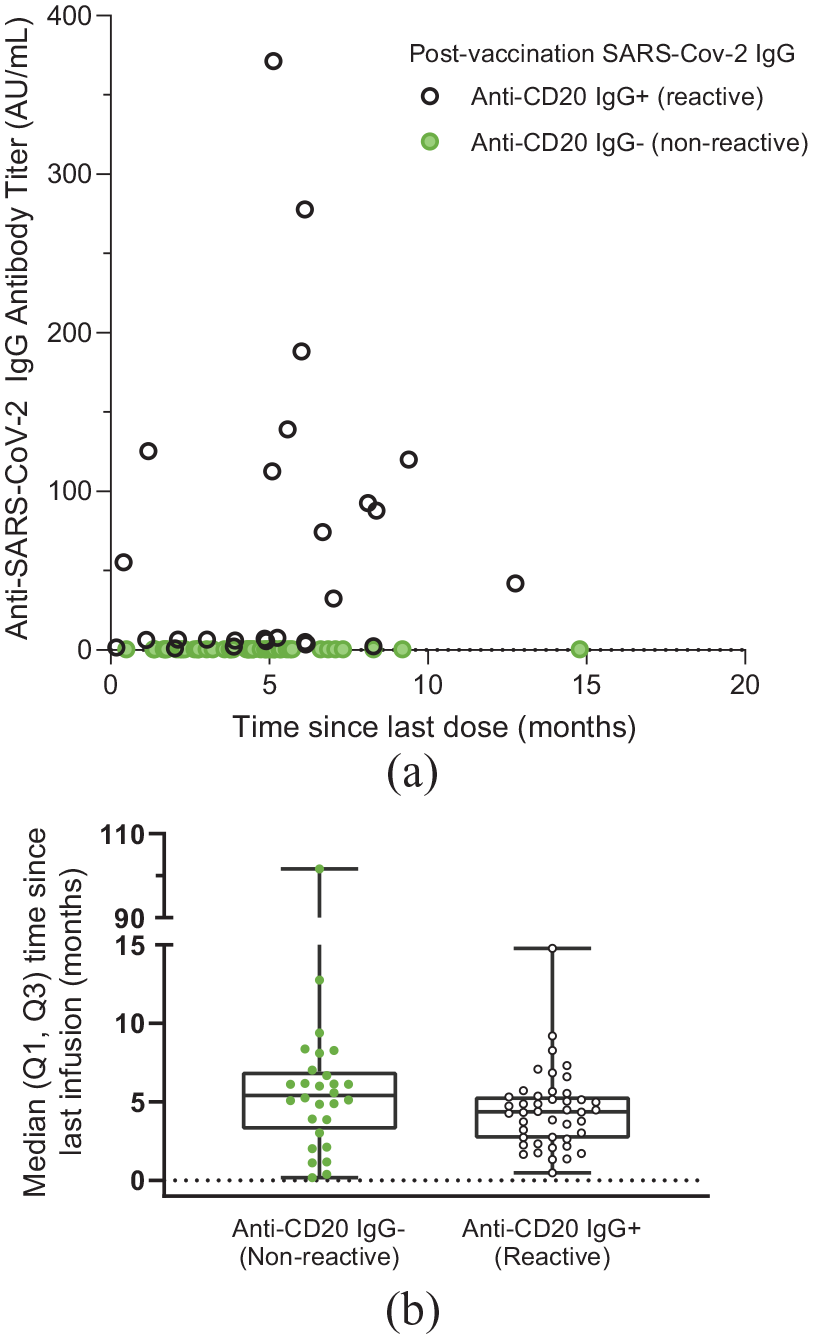
SARS-CoV-2 IgG serologic response stratified by time since last dose in anti-CD20-treated patients. (a) SARS-CoV-2 IgG serologic response is shown for patients with time since last dose data available treated with anti-CD20 (n = 72 of 80 total). Black symbols indicate the IgG positivity (28 of 72 (39%)) and green symbols indicate the IgG response below the threshold of 1 AU/mL (44 of 72 (61%)). (b) Median (Q1, Q3) time since last anti-CD20 infusion in months.
For the anti-CD20 group, the percent of patients with a reactive anti-SARS-CoV-2 response of >1.0 was: Pfizer (45% (14/31)), Moderna (38% (16/42)), and Johnson & Johnson (29% (2/7)). For the S1PRM group, the percent of patients with a reactive anti-SARS-CoV-2 response of >1.0 was: Pfizer (33% (9/27)), Moderna (60% (6/10)), Johnson & Johnson (100% (1/1)), and AstraZeneca (0% (0/1)).
Discussion
Our preliminary results suggest that anti-CD20 and S1PRM DMTs reduce the IgG response to SARS-CoV-2 vaccination in PwMS, consistent with other studies.5,9,10 We also report an association between duration anti-CD20 and S1PRM treatment and reduced odds of an SARS-CoV-2 vaccination IgG response. Conversely, the IgG response to SARS-CoV-2 vaccination appears to be preserved in PwMS treated with other DMTs, including alemtuzumab (n = 23), fumarates (n = 37), GA (n = 33), IFNs (n = 23), natalizumab (n = 44), teriflunomide (n = 15), and no DMT (n = 28). SARS-CoV-2 IgG index value by DMT is shown in Figure 1.
Based on the mechanism of action, the failure to mount a humoral response to SARS-CoV-2 vaccination with anti-CD20 therapy is likely due to depletion of naive and memory B lymphocytes. 5 The mechanism underlying reduced vaccine response in the S1PRM class is not clear even though reduced influenza vaccine response has also been observed in fingolimod treatment. 11 Although the number of circulating lymphocytes is reduced with S1PRM treatment, cells are sequestered in lymphoid organs but not killed. However, besides inhibiting lymphocyte egress from lymph nodes, fingolimod has a variety of effects on T-cell and B-cell mediated immune responses that might be expected to quantitatively or qualitatively affect antibody responses to vaccines. 12
Limitations include the relatively small number of participants in some DMT groups. A larger cohort, including additional DMT classes, is being accrued in an ongoing study. A further limitation is the lack of pre-vaccination samples or cross-testing post-vaccination samples using nucleocapsid-based assay to screen out patients previously infected with SARS-CoV-2; both are being tested in the ongoing study. This study used a commercially available assay to semi-quantitatively assess the SARS-CoV-2 IgG response to spike protein as a measure for SARS-CoV-2 immunity. 13 We did not directly measure neutralizing activity; however, the test manufacturer has reported a 0.81 correlation between IgG index and neutralization titers. 14 We also did not assess T-cell response to SARS-CoV-2. The relative importance of both humoral and cell-mediated mechanisms in protective immunity to SARS-CoV-2 in the context of DMTs has not been fully elucidated and will be crucial to understand long term.
This study highlights the complexity of post-vaccination IgG responses across DMTs, knowledge of which is necessary to optimize MS management and to provide guidance to PwMS around recommended behaviors and precautions given their likelihood of protection after vaccination. Additional studies are needed to further evaluate humoral and especially cellular responses to SARS-CoV-2 vaccination in DMT-treated PwMS. Vaccinations should be discussed as an integral part of MS care, particularly in relation to how some DMTs may impact effective immune responses to routine vaccines, as well as the SARS-CoV-2 vaccine.
Footnotes
Acknowledgements
This study was sponsored by Biogen (Cambridge, MA, USA). Karen Spach, PhD, of Excel Scientific Solutions (Fairfield, CT, USA) provided initial editing support based on a draft from authors and incorporated author comments during revisions, and Nathaniel Hoover from Excel Scientific Solutions copyedited and styled the manuscript per journal requirements.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.A.C. reports receiving personal compensation for consulting from Biogen, Bristol Myers Squibb, Convelo, Genentech, Janssen, NervGen, Novartis, and PSI; speaking for H3 Communications; and serving as an Editor of Multiple Sclerosis Journal. R.A.B. reports being on the scientific advisory board and/or doing consulting for Biogen, EMD Serono, Genentech/Roche, Novartis, Sanofi/Genzyme, and Viela Bio and receiving research support from Biogen, Novartis, and Roche. C.M.H. reports receiving speaking and consulting fees from Biogen, Bristol Myers Squibb, EMD Serono, Genentech, Genzyme, and Novartis and grants/research support from Biogen, Genentech, NIH–NINDS 1U01NS111678-01A1 sub-award, Novartis, and Patient-Centered Outcomes Research Institute (PCORI). M.H. reports receiving research support from Biogen and PCORI. E.M.M. reports receiving research support from Biogen, Genentech, and Teva and royalties from UpTo Date (editorial duties). R.N. reports being on the scientific advisory board and/or doing consulting for Avidea, Biogen, Bristol Myers Squibb, Genentech, Genzyme, GW Therapeutics, Horizon Therapeutics, Janssen, Lundbeck, Nervgen, TG Therapeutics, Third Rock Ventures, and Viela Bio. J.N. reports receiving research grants from ADAMAS, Biogen, Genzyme, Novartis, and PCORI and consulting and/or speaking fees from Alexion, Bristol Myers Squib, EMD Serono, Genetech, Greenwich Biosciences, Novartis, and Viela Bio. M.T. has received compensation for consulting services and speaking honoraria from Almirall, Bayer Schering Pharma, Biogen, Genzyme, Janssen, Merck-Serono, Novartis, Roche, Sanofi-Aventis, Viela Bio, and Teva Pharmaceuticals and is the co-editor of Multiple Sclerosis Journal-ETC. A.Z. has received travel expenses for scientific meetings from Biogen and Novartis, speaking honoraria from Eisai, and a study grant from Novartis. T.Z. reports being on the scientific advisory board and/or doing consulting for Biogen, Celgene, Merck, Novartis, and Roche; receiving compensation for serving on speakers bureaus for Biogen, Celgene, Merck, Novartis, Roche, and Sanofi; and receiving research support from Biogen, Celgene, Merck, Novartis, and Sanofi. X.M. reports receiving speaking honoraria and/or travel expenses for participation in scientific meetings, and/or has been a steering committee member of clinical trials and/or participated in advisory boards of clinical trials in the past years with Actelion, Alexion, Bayer, Biogen, Bristol Myers Squibb/Celgene, EMD Serono, Genzyme, Hoffmann-La Roche, Immunic, Janssen Pharmaceuticals, MedDay, Merck, Mylan, Nervgen, Novartis, Sanofi-Genzyme, Teva Pharmaceutical, TG Therapeutics, Excemed, MSIF and NMSS. C.I.G., C.M.S., and J.R.W. are employees of and shareholders/stockholders of Biogen. R.R. and M.L.N. are shareholder/stockholders and former employees of Biogen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Biogen (Cambridge, MA, USA). Funding for writing and editorial support was provided by Biogen.
