Abstract
Background:
The timed 25-foot walk (T25FW) is a key clinical outcome measure in multiple sclerosis patient management and clinical research.
Objectives:
To evaluate T25FW performance and factors associated with its change in the Multiple Sclerosis Outcome Assessments Consortium (MSOAC) Placebo Database (n = 2465).
Methods:
We created confirmed disability progression (CDP) variables for T25FW and Expanded Disability Status Scale (EDSS) outcomes. We used intraclass correlation coefficients (ICCs) and Bland Altman plots to evaluate reliability. We evaluated T25FW changes and predictive validity using a mixed-effects model, survival analysis, and nested case–control analysis.
Results:
The mean baseline score for the T25FW in this study population was 9.2 seconds, median = 6.1 (standard deviation = 11.0, interquartile range (IQR) = 4.8, 9.0). The T25FW measure demonstrated excellent test–retest reliability (ICC = 0.98). Walk times increased with age, disability, disease type, and disease duration; relapses were not associated with an increase. Patients with T25FW progression had a faster time to EDSS-CDP compared to those without (hazards ratio (HR): 2.6; confidence interval (CI): 2.2, 3.1). Changes in the T25FW were more likely to precede changes in EDSS.
Conclusion:
This research confirms the association of the T25FW with disability and provides some evidence of predictive validity. Our findings support the continued use of the T25FW in clinical practice and clinical trials.
Keywords
Background
Multiple sclerosis
Multiple sclerosis (MS) is a demyelinating and chronic neurologic condition affecting over 900,000 patients in the United States. 1 Among all of the symptoms in MS, walking dysfunction has long been recognized as a predominant impairment from early accounts of the disease. 2 Of persons with MS who have difficulty walking, 70% report it as being the most challenging aspect of having MS. 3
The timed 25-foot walk (T25FW) is the most commonly used standalone measure of ambulation. 4 The T25FW is typically conducted as a component of the MS functional composite (MSFC), though it has been used as a standalone measure in clinical studies. Based on existing research, the T25FW demonstrates strong content validity, criterion validity, construct validity, responsiveness, and clinical meaningfulness. 5 Prior research has not established predictive validity of the test and has not explored the test in a treatment-naïve population. In-depth patient-level analysis for T25FW has not been reported for a large heterogeneous cohort of MS patients.
The two largest known studies to date on the T25FW include research on the full Multiple Sclerosis Outcomes Assessment Consortium (MSOAC) database of 14 clinical trials (12,776) patients6,7 and a meta-analysis of T25FW outcomes across 50 individual studies (REF). 8 These studies included both treated and untreated patients. We focused our research on outcomes in the placebo arm subset of the MSOAC database, which approximates the course of ambulatory changes in an untreated population.
Objectives
Based on our review of the literature, we established four aims to support further use of the T25FW as a clinical outcome. First, we investigated the association between demographic and clinical characteristics with baseline T25FW. Second, we examined the consistency of the measure through assessments of test–retest reliability at individual patient visits. Third, we characterized the association of demographics and clinical factors with changes over time in the T25FW. Fourth, we investigated associations of a 20% change in the T25FW with sustained disability progression on the Expanded Disability Status Scale (EDSS).
Methods
Study population
Our study population is drawn from the MSOAC database, established in 2012 to test and validate outcome measures in MS across clinical trials.9,10 The full database includes outcomes from 16 trials conducted primarily over the years 2000–2013 that were harmonized based on a clinical data standard created by the consortium. Key results from the full dataset have been published.6,7 The placebo arms for nine of these trials, totaling 2465 patients, was made available for research by the MSOAC. Datasets were anonymized with respect to both patient and clinical trial identifiers prior to our analysis.
Outcomes
The T25FW score is calculated by averaging the two trials conducted at a single visit. In alignment with National MS Society Guidelines, we set T25FW maximum values to 180 seconds. 11 We defined T25FW confirmed disability progression (CDP) as a change that was ⩾ 20% from the baseline score with a ⩾ 20% increase sustained at a time point ⩾ 90 days from this first increase. We constructed an EDSS confirmed progression variable using the same time intervals and defined worsening as: baseline 0 required a 1.5-point increase, baseline 0–5.5 required a 1-point increase, baseline ⩾6.0 required a 0.5 increase. 12 We created a binary variable to represent low disability (EDSS ⩽ 4) and high disability (EDSS > 4). Relapses were neurologist-confirmed according to study protocols. There is general consistency in the definitions of relapses among studies. Variables for age, sex, race, disease type, and disease duration were drawn directly from MSOAC datasets.
Statistical analysis
We calculated basic descriptive statistics for demographic and clinical characteristics, including measures of central tendency (means and medians) and variability (interquartile ranges (IQRs), standard deviation). We measured intraclass correlation coefficients (ICCs) for the two T25FW trials at the baseline and final visits for each patient.13,14 We examined concordance through a Bland Altman plot of the two trials at the baseline visit. We calculated annualized changes in T25FW over time based on first and last T25FW scores. We categorized T25FW groups using previously validated cutoff scores of <6 seconds, 6–7.99 seconds, and >8 seconds. 15 We used mixed-effects linear regression models to evaluate changes in the T25FW (n = 16,552). As predictors, we included age as a continuous centered variable, gender, baseline EDSS, relapses during the study period, EDSS-CDP, and disease type. We utilized an unstructured covariance model and included both a random intercept and random slope. We used Akaike information criterion (AIC) scores to evaluate relative model fit.
We used the Kaplan–Meier estimator and Cox proportional hazards ratio to estimate the survival function of EDSS-CDP in patients with and without T25FW CDP. In patients with both T25FW and EDSS-CDP, we examined when these events occurred. We used an incidence density sampling method in a nested case–control study and matched cases (patients with T25FW progression) to controls (patients without) based on age, gender, and disease type to assess the relationship to EDSS-CDP. All p-values were two-tailed and p < 0.05 was considered significant.
We did not impute missing data. We used pairwise analysis rather than listwise analysis to maximize use of all available data and the mixed model assumes missingness at random. We investigated cases where > 10% of variables were missing to determine potential impact on our results. Statistical analysis was performed using SAS Studio 3.8 statistical software (SAS Institute, Cary, NC). ICCs were estimated using the Chinese University of Hong Kong ICC online program based on the Portney and Watkins model. 16
Results
The MSOAC placebo database reflects a typical heterogeneous MS trial population. The mean age was 41.8 (SD = 10.4) years, a majority of patients were females (67.3%), and, where race was collected, 90.7% were identified as White (Table 1). Most patients had relapsing-remitting MS (RRMS; 64.1%); 22.4% had secondary progressive MS (SPMS), and 13.5% had primary progressive MS (PPMS). The average EDSS score was 3.4 (SD = 1.8) and IQR = (2.0, 4.5). Eighty percent of patients had been diagnosed less than 10 years prior to the study start date.
Baseline characteristics for the MSOAC placebo database.
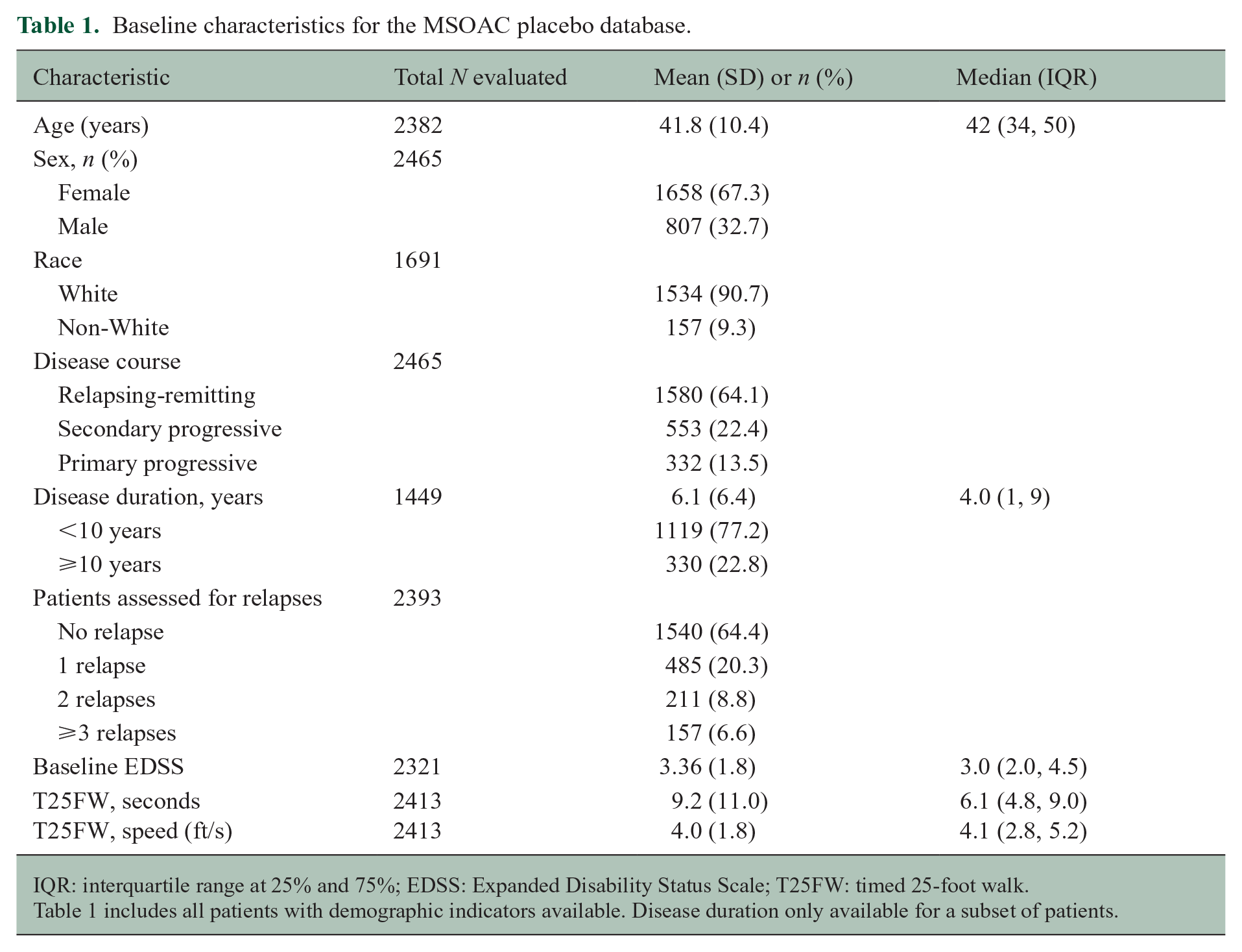
IQR: interquartile range at 25% and 75%; EDSS: Expanded Disability Status Scale; T25FW: timed 25-foot walk.
Table 1 includes all patients with demographic indicators available. Disease duration only available for a subset of patients.
The mean baseline score for the T25FW was 9.2 seconds, median = 6.1 (standard deviation = 11.0, interquartile range (IQR) = 4.8, 9.0) (Figure 1). Baseline T25FW scores varied considerably with respect to age, disability status baseline score, and disease subtype (Table 2). There were no sex differences, but older patients (age 50–60 years) took twice as long to complete the test as younger patients (age 18–30 (12.5 vs 6.0 seconds)). T25FW scores were substantially lower among RRMS patients (mean 6.6 seconds) than among SPMS and PPMS patients (11.8 and 15.3 seconds, respectively).
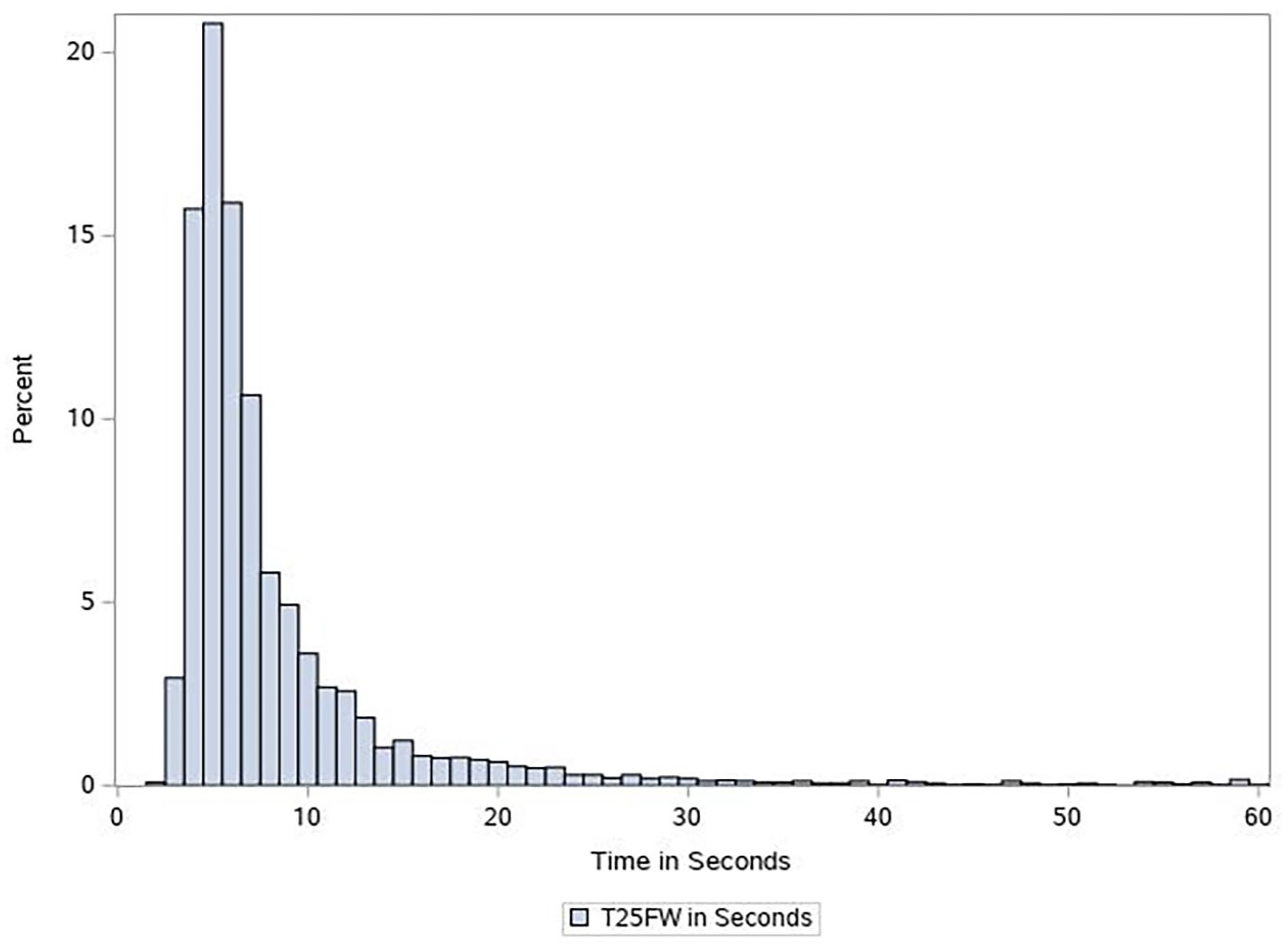
Distribution of T25FW outcome measured at baseline in seconds, n = 2413.
Baseline timed 25-foot walk scores by patient characteristics.
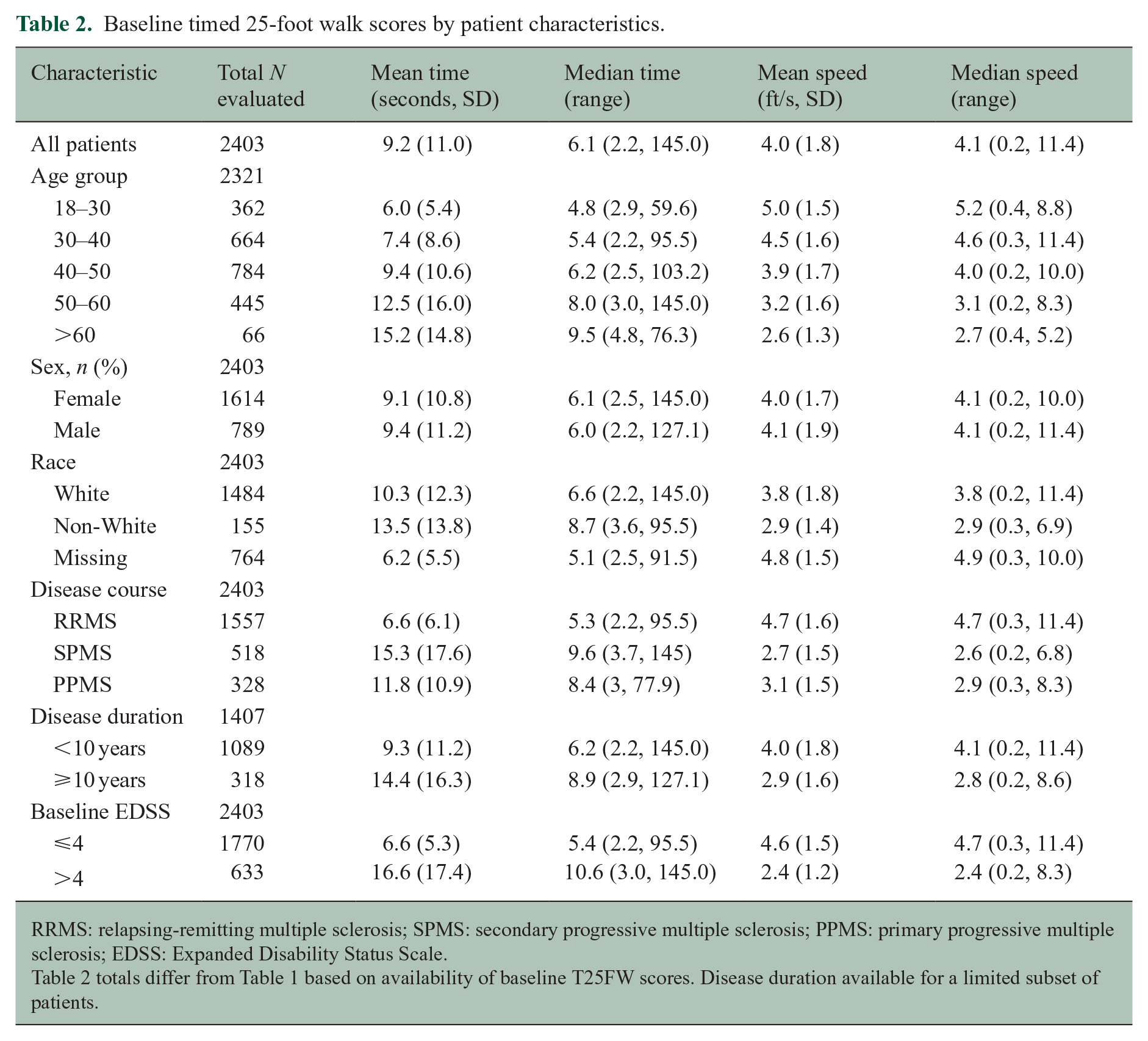
RRMS: relapsing-remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis; PPMS: primary progressive multiple sclerosis; EDSS: Expanded Disability Status Scale.
The T25FW demonstrated strong reliability with high concordance of the two replicate test measurements taken during each of the first and last visits. Among all patients, the mean value ICC for the two measurements was 0.987 at the first visit and 0.979 at the last visit. The Bland Altman plot further supports consistency of the T25FW (Figure 2).
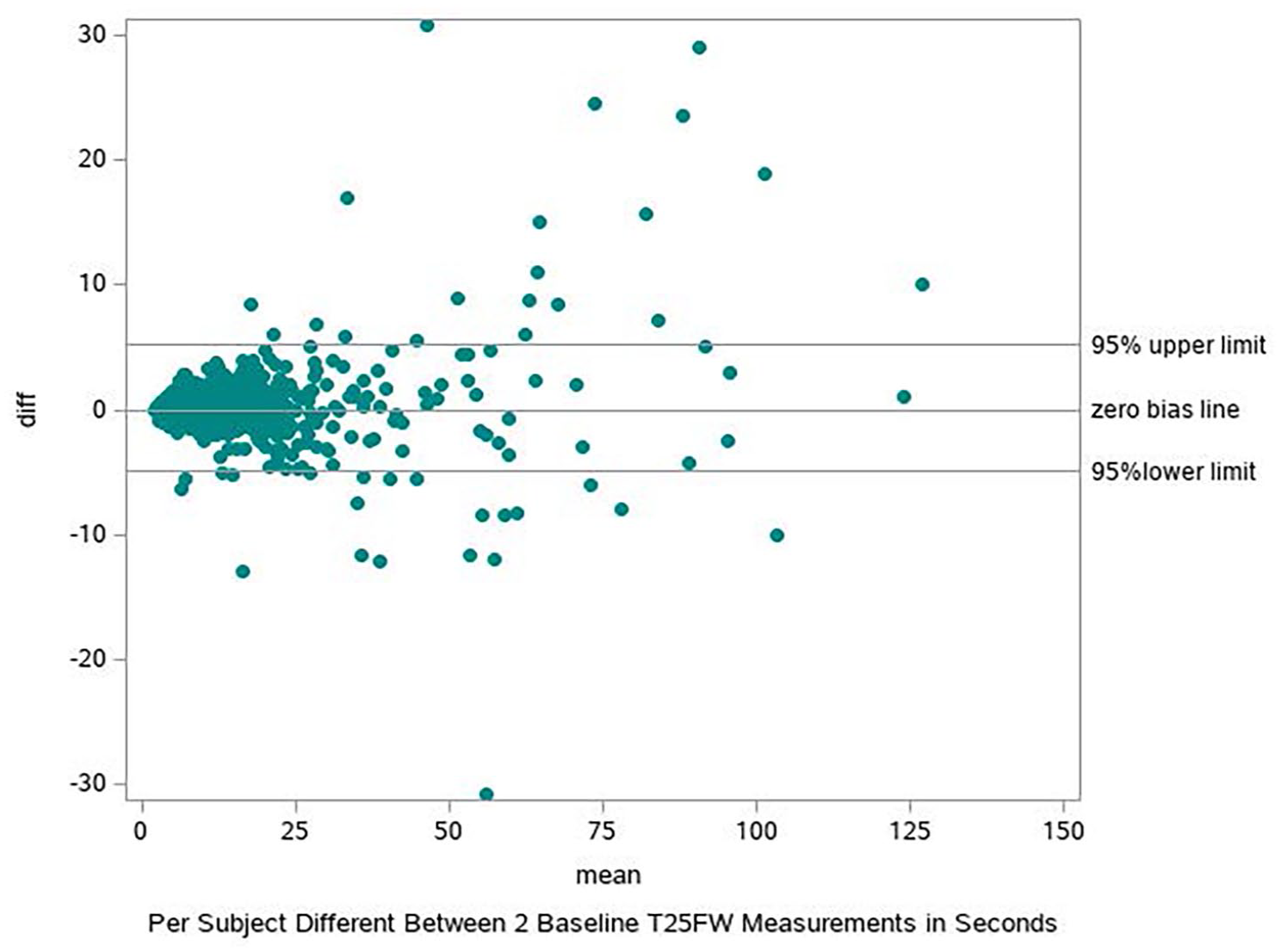
Estimate of concordance for the two replicate T25FW tests conducted at baseline visit, n = 2403.
Higher annualized increases were significantly associated with baseline performance, male gender, older age, and baseline MS type (Table 3). Groups that have significant increases in change on the T25FW (e.g. EDSS > 4), are further characterized by high variability in scores. Men had a higher annualized rate of increase compared to women (3.4 vs 2.2 seconds), but this difference was attributable to a higher proportion of men having PPMS, which accelerated worsening compared to other MS subtypes. Baseline walk times affected rates of change. Patients with a baseline T25FW of < 6 seconds had a 0.6 second annualized increase, baseline of 6–8 seconds experienced a 1.8 second increase, and baseline > 8 seconds averaged a 6.2 second increase.
Annualized changes in the timed 25-foot walk (T25FW) in the MSOAC placebo database.
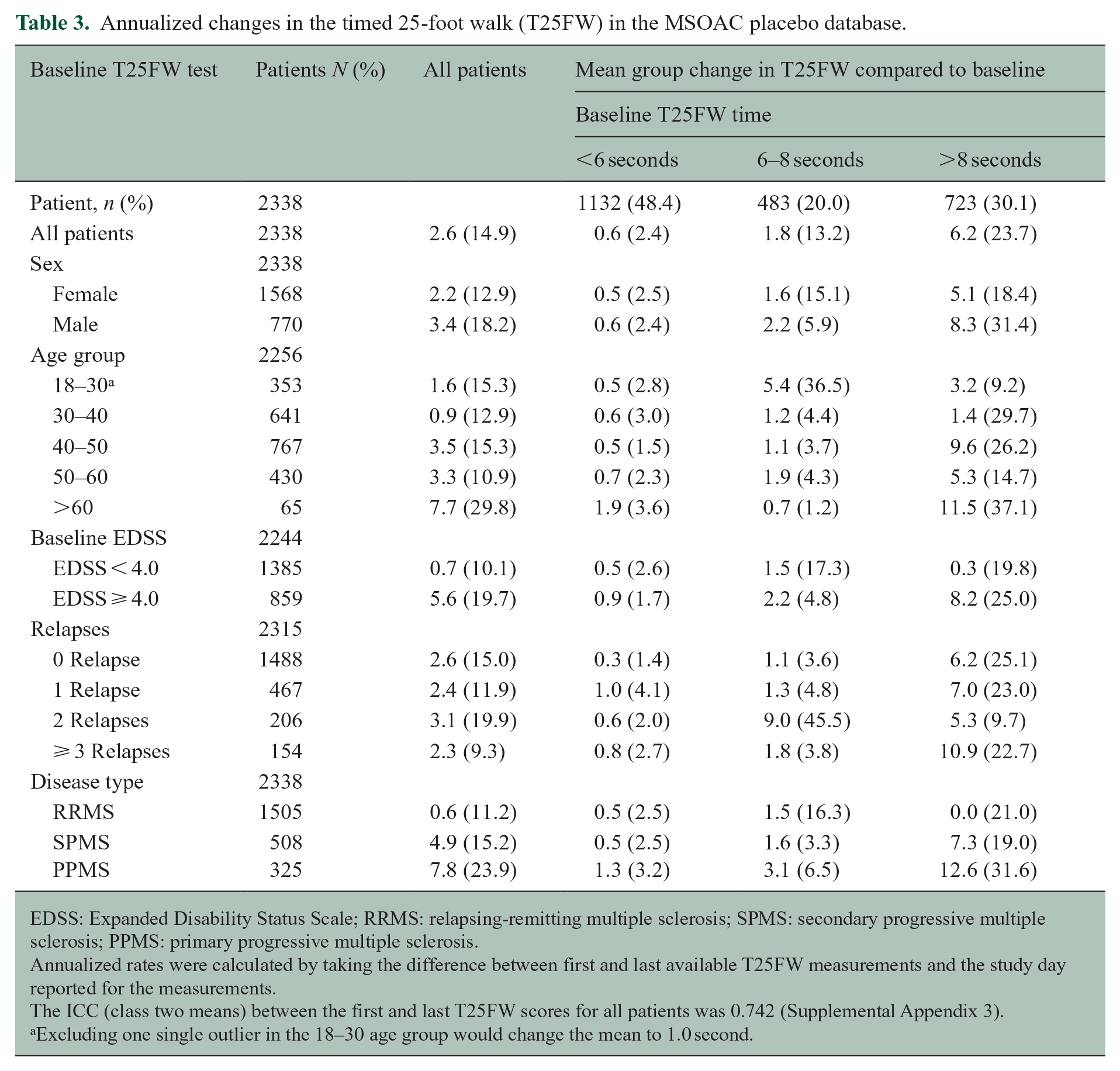
EDSS: Expanded Disability Status Scale; RRMS: relapsing-remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis; PPMS: primary progressive multiple sclerosis.
Annualized rates were calculated by taking the difference between first and last available T25FW measurements and the study day reported for the measurements.
The ICC (class two means) between the first and last T25FW scores for all patients was 0.742 (Supplemental Appendix 3).
Excluding one single outlier in the 18–30 age group would change the mean to 1.0 second.
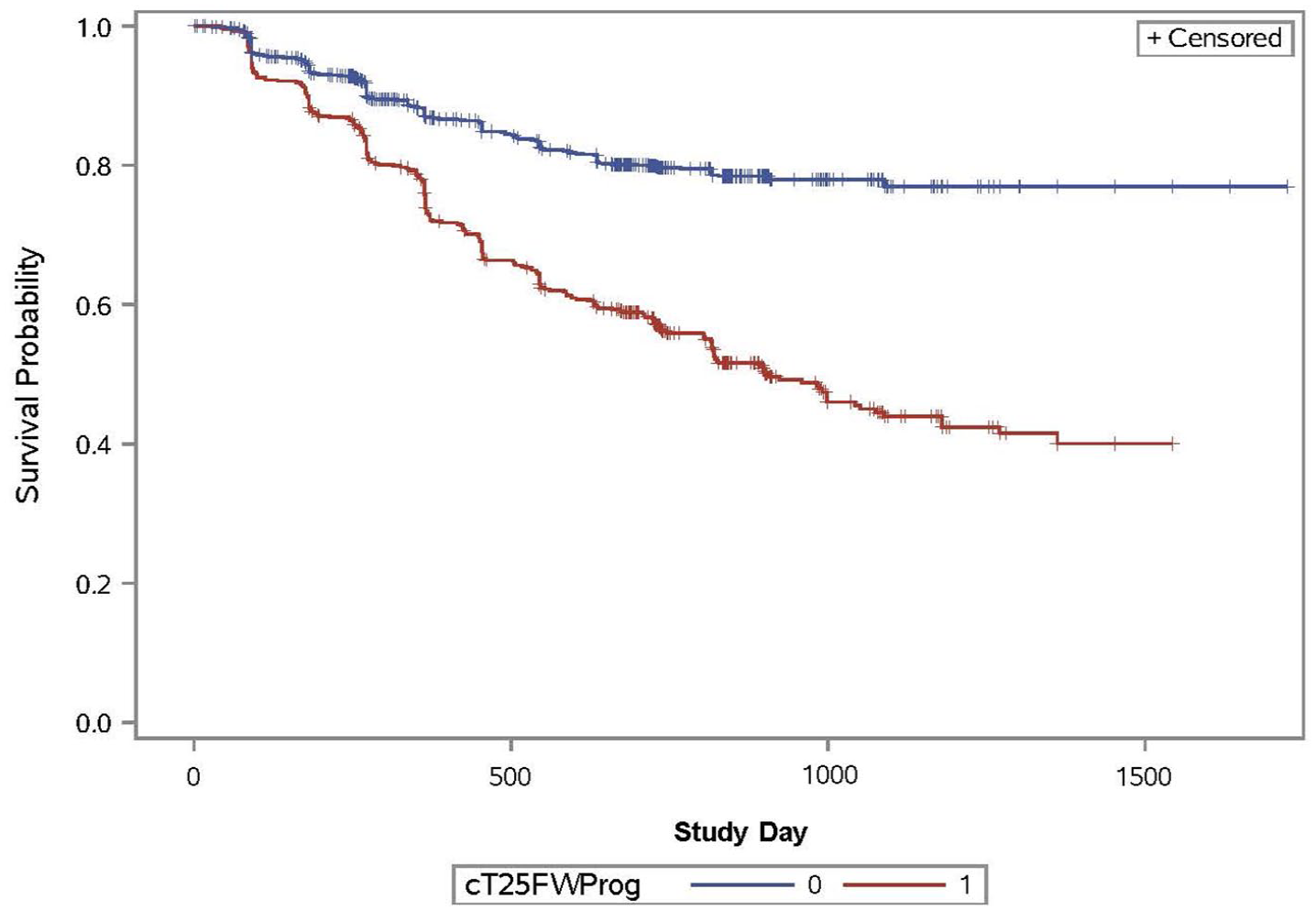
Survival analysis for confirmed disability progression among patients with and without T25FW progression, defined as ⩾ 20% increase sustained at ⩾ 3 months; N = 2395.
Higher annualized increases were associated with older age groups. The 18–30 years age group had a mean increase of 1.0 second, and patients in the oldest group of > 60 (representing SPMS and PPMS patients exclusively) had an increase of 7.7 seconds with considerable variability (SD 29.8). MS subtype was related to changes in walk times, with average annualized increases of 0.6 for relapsing-remitting, 4.9 for secondary progressive, and 7.8 for primary progressive.
The number of relapses did not appear to have a significant correlation with T25FW increases. The average annualized increase was 2.4 in those with one relapse, 3.1 in those with two relapses, 2.3 in those with three or more relapses, and 2.6 seconds among those with no relapses.
Results from the mixed-effects models (Table 4) corroborated the findings from the raw data and offered new insights. After adjusting for other variables, changes in the T25FW between genders were not statistically significant (LS mean difference −0.4 seconds, p = 0.33). Baseline EDSS levels were associated with the biggest changes over time: 4.8 seconds per year for high versus low baseline EDSS (p < 0.0001). Disease course and EDSS progression were associated with changes over time. At 3 years, patients with no progression were estimated to have a T25FW of ~12 seconds. Patients with EDSS-CDP during this period were estimated to have a T25FW of ~23 seconds. We note that the interaction of time and age appears to suggest small but paradoxical improvement. This is likely due to confounding and multicollinearity in the model, with the effect for age being distributed across EDSS and disease course variables, which are correlated (Pearson correlation .61).
Annualized changes in T25FW from a mixed-effects linear regression model.
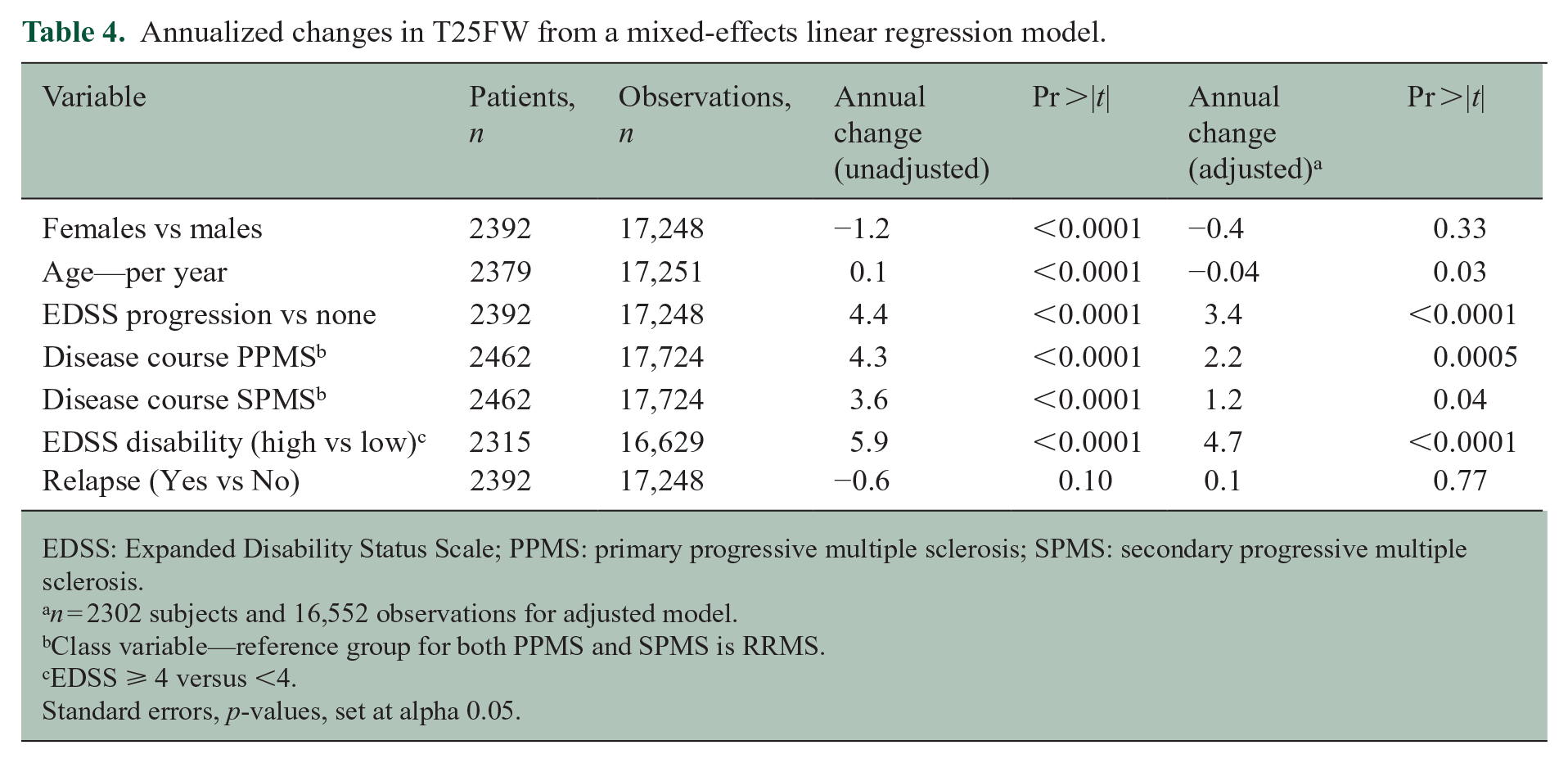
EDSS: Expanded Disability Status Scale; PPMS: primary progressive multiple sclerosis; SPMS: secondary progressive multiple sclerosis.
n = 2302 subjects and 16,552 observations for adjusted model.
Class variable—reference group for both PPMS and SPMS is RRMS.
EDSS ⩾ 4 versus <4.
Standard errors, p-values, set at alpha 0.05.
We further characterized the association of EDSS and T25FW disability measures through a Kaplan–Meier analysis. We observed separation in the curves for patients with and without T25FW-CDP based on time to EDSS progression (Figure 3). The log-rank chi-square was 144.7 and the Wilcoxon chi-square was 98.7 (p for each < 0.0001). The hazard ratio was 2.6 with a log-rank chi-square of 135.1 (p < 0.0001).
Patients with higher levels of disability who experience progression tend to have T25FW disability preceding EDSS disability (Table 5). T25FW disability preceded EDSS by 273.1 days (SD 241). A survival curve for this population is included in the Supplemental Appendix. In addition, among patients with EDSS > 4 who progressed on both EDSS and T25FW at different visits, 72.1% of patients had T25FW progression first, compared to 28.0% who had EDSS progression first. Across all patients, 23% (n = 573) of patients experienced EDSS-CDP, 24% (n = 593) experienced T25FW-CDP, and 11% (n = 282) experienced both.
MSOAC disability progression on T25FW and EDSS measures.
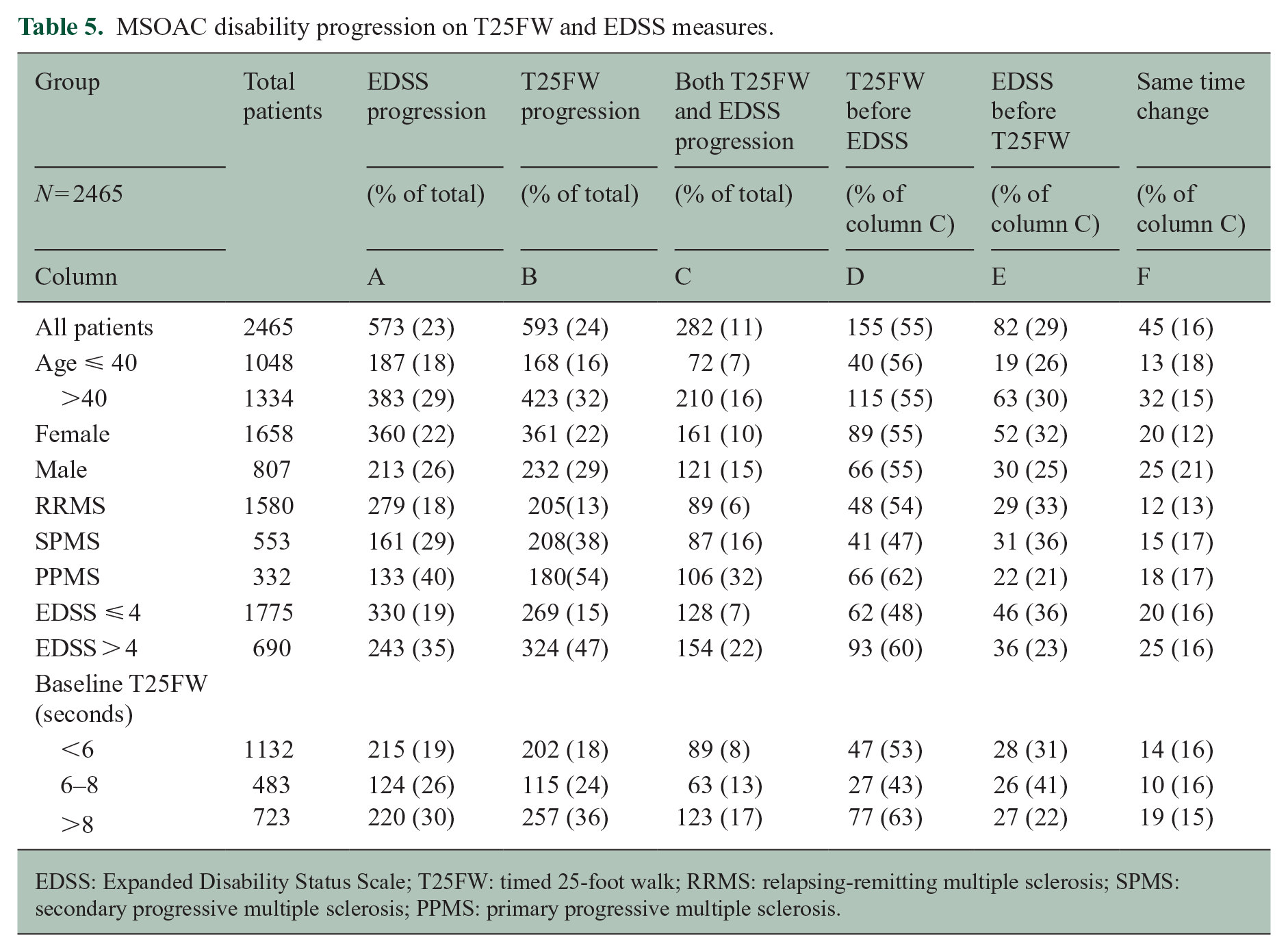
EDSS: Expanded Disability Status Scale; T25FW: timed 25-foot walk; RRMS: relapsing-remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis; PPMS: primary progressive multiple sclerosis.
In an effort to provide additional information on whether change in the T25FW-CDP is an earlier marker of subsequent EDSS-CDP, our nested case–control identified 463 pairs matched on age, gender, and MS subtype. Cases with confirmed T25FW-CDP were matched with controls that did not have progression at the same time point in the study. There was a clear separation in curves (figure in Supplemental Appendix); the hazard ratio was 2.5 (chi-square 42.6, p < 0.0001) and the incidence density ratio was 3.1 (confidence interval (CI): 2.3, 4.3). This suggests three times higher odds for EDSS-CDP among patients who have T25FW-CDP compared to those who do not.
Discussion
The mean value for T25FW at baseline for our population is identical to that from a recently completed meta-analysis of 50 studies of the T25FW in MS—9.2 seconds. 8 On the contrary, this baseline score is somewhat higher than that reported in the full MSOAC database (7.6 seconds). 6 This difference may be due to higher patient disease severity of the clinical trials in our dataset, evidenced by a baseline EDSS of 3.4 in our sample versus 2.9 in the full dataset.
Literature suggests that PPMS and SPMS patients have similar courses of disease activity over time. 17 We found that, even though PPMS patients started with a faster baseline T25FW than SPMS patients (11.8 vs 15.3), the changes among PPMS patients were more rapid. PPMS patients had an annualized increase of 7.8 seconds compared to 4.9 seconds for SPMS patients, despite comparable EDSS. The difference in genders in our study was influenced by a higher proportion of male patients in the PPMS subpopulation and did not persist after adjusting for other covariates.
We found evidence to support that the previously identified T25FW baseline benchmarks of <6 seconds, 6–8 seconds, and >8 seconds are meaningful markers of expected change. These performance benchmarks had been found in earlier research to be associated with real-life anchor measures of functional independence, and physiologic measurements of gait. 15 Across subgroups in our study, patients who took ⩾6 seconds at study baseline were likely to have higher rates of increase in T25FW scores than patients who walked in <6 seconds. Differences in EDSS were observed across these baseline groups (EDSS 2.3, 3.5, and 5.0, respectively for the <6, 6–8, and >8-second groups), suggesting that baseline T25FW may serve as a surrogate or marker for disability.
While it is not surprising that T25FW scores were higher in the older age groups, we found it interesting that the rate of change also increased by age groups. While we are aware of research associating slower ambulation with age, we did not find literature associating changes in the T25FW with aging. 18 In particular, older age groups with slower walk times had faster rates of change over time (Table 3). This finding was confirmed in our mixed-effects model and Kaplan–Meier analysis. The model suggests that, adjusting for other factors (gender, disease type, baseline EDSS, EDSS-CDP), a 55-year-old with MS would have an annualized rate of increase 2.1 seconds higher than a 25-year-old with MS.
Our results may suggest that the number of MS relapses do not have as strong an impact on long-term disability accumulation as previously thought. There was not a strong association between the number of relapses and T25FW scores. Even patients with three or more relapses (n = 157) had on average increases in walk times that were similar to the overall population mean change (2.3 vs 2.6 seconds). Other research has reported moderate or strong associations between relapses and T25FW worsening.19,20 This difference may be due to the fact that other research examines the more immediate effect of relapses, whereas our rates of change track to changes over an average of 1–2 years. A caveat to this observation is that patient drop-out and switching arms due to relapses may have occurred and introduced selection bias.
In our study, we confirmed test–retest reliability in a large study across a heterogeneous population, incorporating more than 47,000 individual trials of the T25FW. This builds on multiple smaller studies also showing reliability of the T25FW test in a clinical setting. 21 Newer measures of ambulation, while they aim to more precisely capture other aspects of patient disability, will have a high benchmark to overcome in order to deliver the same level of consistency and reliability as the T25FW.
Our examination of patients with both EDSS and T25FW progression suggests a temporal element extremely valuable for use of the T25FW as a primary outcome measure—predictive validity for EDSS progression. In the development of the MSFC, one of the key criteria for an MS outcome was predictive validity. 22 In our study, T25FW progression occurred before EDSS progression 65% of the time, excluding patients who changed at the same time. Since many patients for almost any marker in MS would ultimately experience progression, it was important to control for an exposure time bias, which we did by using a case–control design. The results of our nested case–control suggest that, when starting at the same time point, patients who have TF25FW progression will have EDSS progression sooner than those who do not have T25FW progression. In addition, we find that, among patients with EDSS-CDP, 37% have T25FW progression, whereas only 9% of patients without EDSS-CDP have progression on T25FW. Taken together, these analyses suggest that the T25FW may have predictive validity and serve as a sensitive measure of future EDSS changes. The utility of this measure is also supported by the fact that a greater proportion of patients progress on T25FW than on other measures of disability in our study and in other studies in SPMS.23,24
Our study benefits from the large amount of participant data in the MSOAC database that enabled well-powered analyses. We were able to rely on more than 47,000 individual T25FW measurements. Another benefit of the MSOAC database is the detailed taxonomy, which enables analysis of endpoints on a participant level, yielding more flexibility than meta-analyses. An additional strength is that the outcomes were measured in the context of regulated clinical trials, with monitoring and control measures in place, yielding high quality and generally consistent reporting of results.
Our study has limitations. A key factor potentially affecting internal validity is confounding. For methods like the mixed-effects linear regression model, we have unmeasured known possible confounders (e.g. magnetic resonance imaging (MRI) activity, socio-economic status) that are not included. As we cannot identify the studies individually, we have some confounding by entrance criteria. However, as most of our analyses are within person analyses, unless there are interactions between study entrance criteria and these results, we are somewhat insulated from this limitation. We also have unknown confounders arising from differences in standards of care over time and clinical trial protocols. We may have measurement error in our analysis that arises from taxonomy differences or missing data due to variability in data collection across protocols. There are also external validity limitations. Patients who participate in clinical studies are not entirely representative of typical MS patients in measured factors including disease severity and unmeasured factors such as psychological characteristics. Another limitation related to the measure itself is that the T25FW does not capture important facets of ambulation in MS—for example gait, endurance, or fatigue.
In conclusion, the T25FW has excellent features as an outcome measure with high test–retest reliability and is sensitive to clinical change, indicating that it has merit as a standalone measure in MS trials. Clinicians may benefit from using the T25FW in clinical practice as it might serve as a relevant indicator of both current and future disability and is easier to implement than the EDSS. Clinicians may find it valuable to assess individual patient performance against the T25FW benchmarks generated for characteristics such as age, sex, baseline values, and MS subtype.
Future research can strengthen the relevance of our findings. First, there is an opportunity to engage clinical trial researchers to further harmonize the taxonomy used in MS clinical research so that data across trials can be more effectively aggregated and analyzed. Second, there is an opportunity to further explore aspects of the T25FW in patients with truly minimal disease. In addition, the clinical sensitivity of the 20% cut-off could be re-examined to optimize use of the measure in clinical trials.
Supplemental Material
sj-docx-1-msj-10.1177_13524585211017013 – Supplemental material for The timed 25-foot walk in a large cohort of multiple sclerosis patients
Supplemental material, sj-docx-1-msj-10.1177_13524585211017013 for The timed 25-foot walk in a large cohort of multiple sclerosis patients by Anissa Kalinowski, Gary Cutter, Nina Bozinov, Jessica A Hinman, Michael Hittle, Robert Motl, Michelle Odden and Lorene M Nelson in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors would like to thank members of the MS Outcomes Assessment Consortium and Critical Path Institute for generating and making the data available for analysis; and Kristin Sainani, Julia Simard, and Rita Popat from the Stanford Epidemiology and Population Health for contributions to study design and statistical analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.B., R.M., J.A.H., and M.H. declare no conflicts of interest in preparing this article. A.K. was previously employed by Roche and Biogen. M.O. serves as consultant for Cricket Health. G.C. served on Data and Safety Monitoring Boards for Astra-Zeneca, Avexis Pharmaceuticals, Biolinerx, Brainstorm Cell Therapeutics, Bristol Meyers Squibb/Celgene, CSL Behring, Galmed Pharmaceuticals, GreenValley Pharma Ltd, Mapi Pharmaceuticals Ltd, Merck, Merck/Pfizer, Opko Biologics, OncoImmune, Neurim, Novartis, Ophazyme, Sanofi-Aventis, Reata Pharmaceuticals, Teva pharmaceuticals, VielaBio Inc, Vivus, NHLBI (Protocol Review Committee), and NICHD (OPRU oversight committee). G.C. served on consulting or Advisory Boards for Biodelivery Sciences International, Biogen, Click Therapeutics, Genzyme, Genentech, GW Pharmaceuticals, Immunic, Klein-Buendel Incorporated, Medimmune/Viela Bio, Medday, Merck/Serono, Neurogenesis LTD, Novartis, Osmotica Pharmaceuticals, Perception Neurosciences, Recursion/Cerexis Pharmaceuticals, Regeneron, Reckover Pharmaceuticals, Roche, and TG Therapeutics. G.C. is President of Pythagoras, Inc., a private consulting company located in Birmingham AL. L.M.N. has received grants from the Centers for Disease Control and Prevention, National Institutes of Health, and National MS Society and contracts from the Agency for Toxic Substances and Diseases Registry. L.M.N. receives compensation for serving as a consultant to Acumen, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
