Abstract
Background:
Little is known about the effects of changes in the presence or absence of psychiatric disorders on health care utilization in multiple sclerosis (MS).
Objective:
To evaluate the association between “active” mood and anxiety disorders (MAD) and health care utilization in MS.
Methods:
Using administrative data from Manitoba, Canada, we identified 4748 persons with MS and 24,154 persons without MS matched on sex, birth year, and region. Using multivariable general linear models, we evaluated the within-person and between-person effects of any “active” MAD on annual physician visits, hospital days, and number of drug classes dispensed in the following year.
Results:
Annually, the MS cohort had an additional two physician visits, two drug classes, and nearly two more hospital days versus the matched cohort. Individuals with any MAD had more physician visits, had hospital days, and used more drug classes than individuals without a MAD. Within individuals, having an “active” MAD was associated with more utilization for all outcomes than not having an “active” MAD, but the magnitude of this effect was much smaller for visits and drugs than the between-person effect.
Conclusion:
Within individuals with MS, changes in MAD activity are associated with changes in health services use.
Introduction
The prevalence of psychiatric comorbidity, particularly depression and anxiety disorders, is high in the multiple sclerosis (MS) population. 1 Psychiatric comorbidity adversely affects quality of life and disability progression in MS,2,3 and depression synergistically affects mortality. 4 However, little is known about the effect of psychiatric comorbidity on health care utilization (HCU) in MS, although this is a key knowledge gap. 5
The MS population has higher rates of HCU than non-MS populations. 6 One study suggested that depression and bipolar disorder were associated with increased hospitalization rates in persons with MS, 7 while another study found that depression was not associated with increased health care costs. 8 While both studies compared HCU between individuals having MS with and without psychiatric disorders, neither examined whether changes in the presence or absence of psychiatric disorders within individuals affected HCU. Such information is important for understanding the potential effects of interventions for psychiatric disorders on the health system. Specifically, psychiatric disorders are treatable, thus optimized management of psychiatric disorders may reduce other health care needs while improving quality of life.
We compared HCU (hospitalizations, physician visits, medication use) in persons with MS with comorbid mood and anxiety disorders (MAD); persons with MS without comorbid MAD; and a matched cohort from the general population. We specifically aimed to determine the within-person effects of MAD on utilization over time.
Methods
Manitoba is a Canadian province with a population of ~1.3 million. The province provides universal, publicly funded health services, which are captured in administrative databases. We obtained approval to conduct this population-based study from the University of Manitoba Health Research Ethics Board and Manitoba’s Health Information Privacy Committee.
Data sources
We accessed administrative databases housed at the Population Research Data Repository at the Manitoba Centre for Health Policy, covering the fiscal years April 1, 1984 to March 31, 2016, including the population registry, physician claims, the discharge abstract database (DAD), and Drug Program Information Network (DPIN). The registry includes sex; dates of birth, death, and health care coverage; and residential region (postal code) for each provincial resident. Physician claims record the service date and diagnosis according to the International Classification of Diseases (ICD), 9th revision, Clinical Modification (ICD-9-CM). The DAD captures hospitalizations, including admission and separation dates, and diagnoses recorded using (ICD)-9-CM codes up to 2004 and ICD 10th revision, Canadian version (ICD-10-CA) codes thereafter. Since 1995, DPIN has recorded all outpatient prescription dispensations, including date and drug identification number (DIN). The DIN is linked to the World Health Organization’s Anatomical Therapeutic Chemical (ATC) Classification System. 9 The databases were linked at the individual level using an encrypted unique identifier.
Study populations
We identified Manitobans with MS during the period 1984–2016 using a validated case definition which required an individual to have ⩾3 health care encounters for MS (see Appendix). 10 For each case, we defined the first central nervous system demyelinating disease claim as the index date. Then, we identified a general population cohort matched up to 5:1 on sex, year of birth within ±5 years, and forward sortation area (first three digits of postal code) to each individual with MS. This cohort excluded anyone with any ICD-9-CM and ICD-10-CA codes for demyelinating disease or use of any MS-specific disease-modifying therapies. We also excluded individuals with diagnosis codes for inflammatory bowel disease (IBD) and rheumatoid arthritis (RA) as we were conducting concurrent studies for these diseases. 11 Controls were assigned the index date of their matched cases. Due to observed temporal trends that would complicate interpretation of findings, we limited the analysis period to April 1, 2006 to March 31, 2016 (Figure e1 in Supplemental Material). This reduced the number of matched controls available for some cases, but our sample size was sufficiently large that this did not affect statistical power.
Psychiatric comorbidity
Using validated case definitions that relied on hospital, physician, and prescription claims (see Appendix), we identified persons in both cohorts with depression, anxiety, bipolar disorder, and any mood or anxiety disorder (MAD, which included ⩾1 of depression, anxiety disorders, bipolar disorders), 12 who were ever affected (lifetime prevalence). The date of the first claim for each condition was considered the diagnosis date. However, psychiatric disorders can remit and recur;13–15 and HCU may decrease following remission and increase following recurrence. 16 Annually, therefore, we considered the affected person to be an annual prevalent (hereinafter “active”) case if there were ⩾2 physician claims or one hospital claim for the psychiatric disorder in that year; for hospital claims, the psychiatric disorder had to be the most responsible diagnosis, 17 as judged by trained coders. Prescription claims alone were not deemed a marker of “active” disease due to their potentially frequent off-label use. Thus, an individual who ever met the case definition for a psychiatric disorder could vary with respect to their status “active” or “inactive” from 1 year to another.
HCU
Outcomes of interest included the number of physician visits; number of classes (types) of prescription medications dispensed, at the fourth level of the ATC system (e.g. by chemical subgroup such as biguanide); and number of hospital days. We examined these outcomes annually, first considering HCU for any cause. Second, to better assess the effects of psychiatric morbidity on non-mental health use, we examined physician visits after excluding those related to mental health. We also examined physician visits by provider type, categorized as primary care, neurologist, psychiatrist, and other specialist.
Covariates
Covariates included sex (male as reference group), current age as updated annually, year of diagnosis (continuous), socioeconomic status (SES), region, physical comorbidity, and disease duration from the index date (continuous). Age was categorized as 18–24 (reference), 25–44, 45–64, and ⩾65 years due to a non-linear association with the outcomes of interest, and to ensure sufficiently large cell sizes. We determined SES by linking postal code to dissemination-area-level census data then calculating the Socioeconomic Factor Index version 2 (SEFI-2), which incorporates information regarding average household income, percent of single-parent households, unemployment and high-school education rates; scores <0 indicate higher SES. 18 SES was categorized into quintiles (lowest quintile as reference) at the index date. Regions were classified as urban or rural. Physical comorbidity was identified using the John Hopkins Adjusted Clinical Group System Aggregated Diagnosis Groups (ADGs™), a case-mix system developed in the United States which predicts HCU, and mortality in Canadian populations also.19–22 Specifically, we used major physical ADGs which were not time-limited to account for chronic physical conditions (see Appendix). To ensure adequate cell sizes and consistency with prior work, they were categorized as 0, 1, or ⩾2 and updated annually.
Analysis
We characterized the cohorts using mean (standard deviation (SD)), median (interquartile range (IQR)), and frequency (percent). We report standardized differences for these comparisons; 0.20 represents a small effect size.
We used multivariable negative binomial regression models with generalized estimating equations (GEE) to test for statistically significant differences between the association of cohort (MS vs. matched controls), and MAD status (yes vs. no), on the utilization outcomes of interest. The definition of active MAD status requires ongoing physician visits. Therefore, we examined utilization in the year following determination of MAD status to prevent confounding due to the definition. We accounted for differences in follow-up time by including the natural logarithm of person-years as the model offset. We modeled the risk difference for each outcome, as this allowed estimation of the absolute (incremental) effects of the independent variables of interest. Absolute effects allow the determination of number needed to treat and are more useful for policymakers than are relative risks. These models provide population averages of within-person and between-person effects, but can be parameterized to separate these effects using a person-mean variable and a within-person centered variable. 23 We also tested for the presence of a two-way interaction between cohort and MAD status for each outcome. If a two-way interaction was not statistically significant, we would expect the effects of each exposure (cohort, MAD) on HCU to be additive on the risk difference scale. A positive (synergistic) interaction would indicate the effect of cohort and MAD together exceeded the sum of the individual effects. A negative interaction would indicate the joint effect of these exposures was less than effect of the separate exposures. We report risk differences and 95% confidence intervals (95% CI).
In complementary analyses, we repeated our analyses using depression and anxiety disorders instead of the aggregate MAD. The prevalence of bipolar disorder was too low for such analyses. We also repeated the MAD analyses in an incident sub-cohort (index dates 2006–2016) to assess robustness of study findings.
Statistical analyses used SAS V9.4 (SAS Institute Inc., Cary NC).
Results
After restriction of the cohorts to the period 2006–2016, we identified 4748 individuals with MS and 24,154 matched controls. We identified five controls for each of 2734 (57.6%) cases; the mean (SD) matching ratio was 4.2 (0.05). The cohorts were well-matched at the index date for age, sex, and SES (Table 1). Most participants were female. At the index date, the MS cohort had a higher lifetime prevalence and active prevalence of any MAD, depression, and anxiety disorders than the matched cohort. The burden of physical comorbidity (number of ADGs) was higher in the MS population.
Characteristics of multiple sclerosis (MS) and matched cohorts.
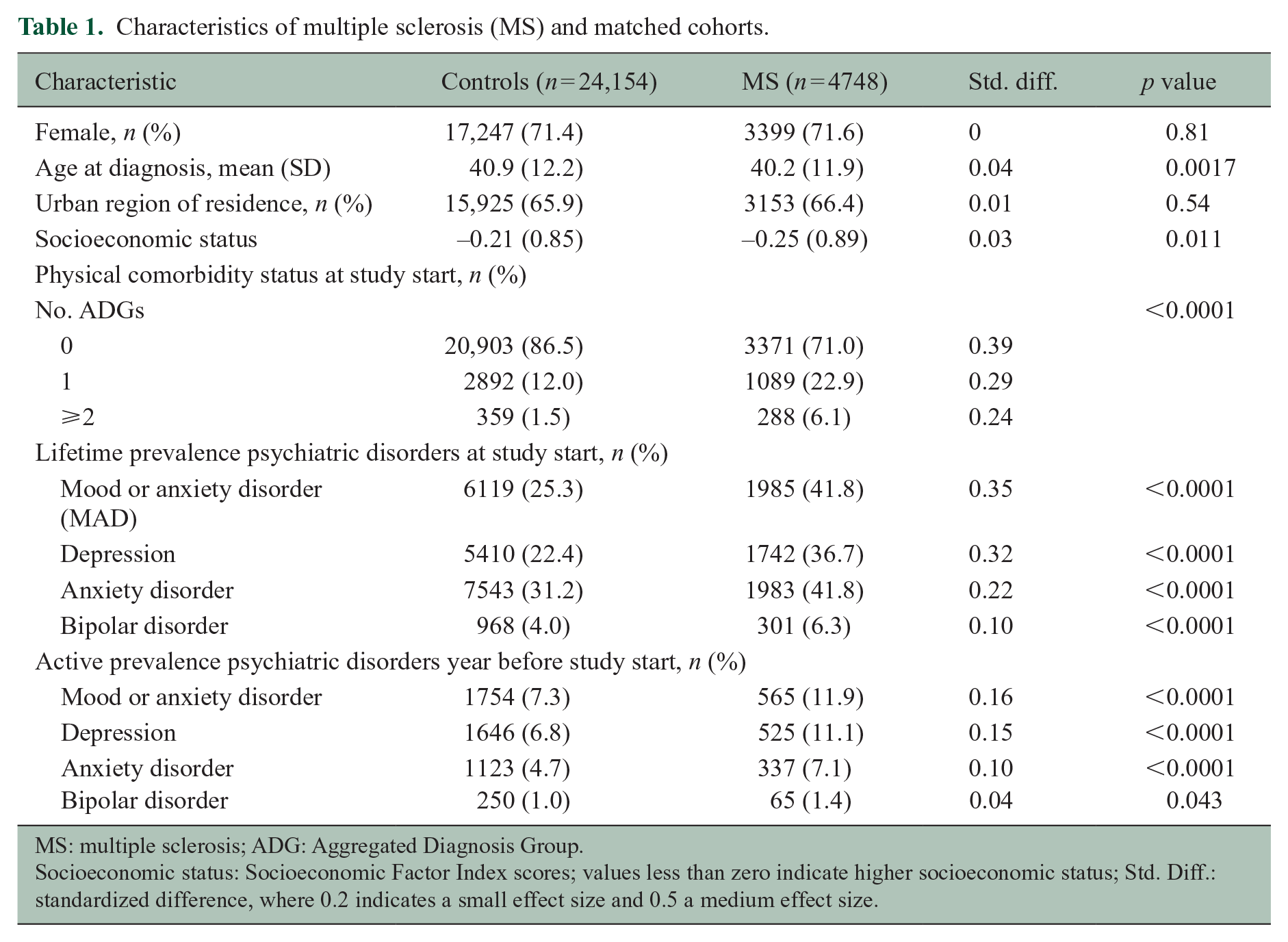
MS: multiple sclerosis; ADG: Aggregated Diagnosis Group.
Socioeconomic status: Socioeconomic Factor Index scores; values less than zero indicate higher socioeconomic status; Std. Diff.: standardized difference, where 0.2 indicates a small effect size and 0.5 a medium effect size.
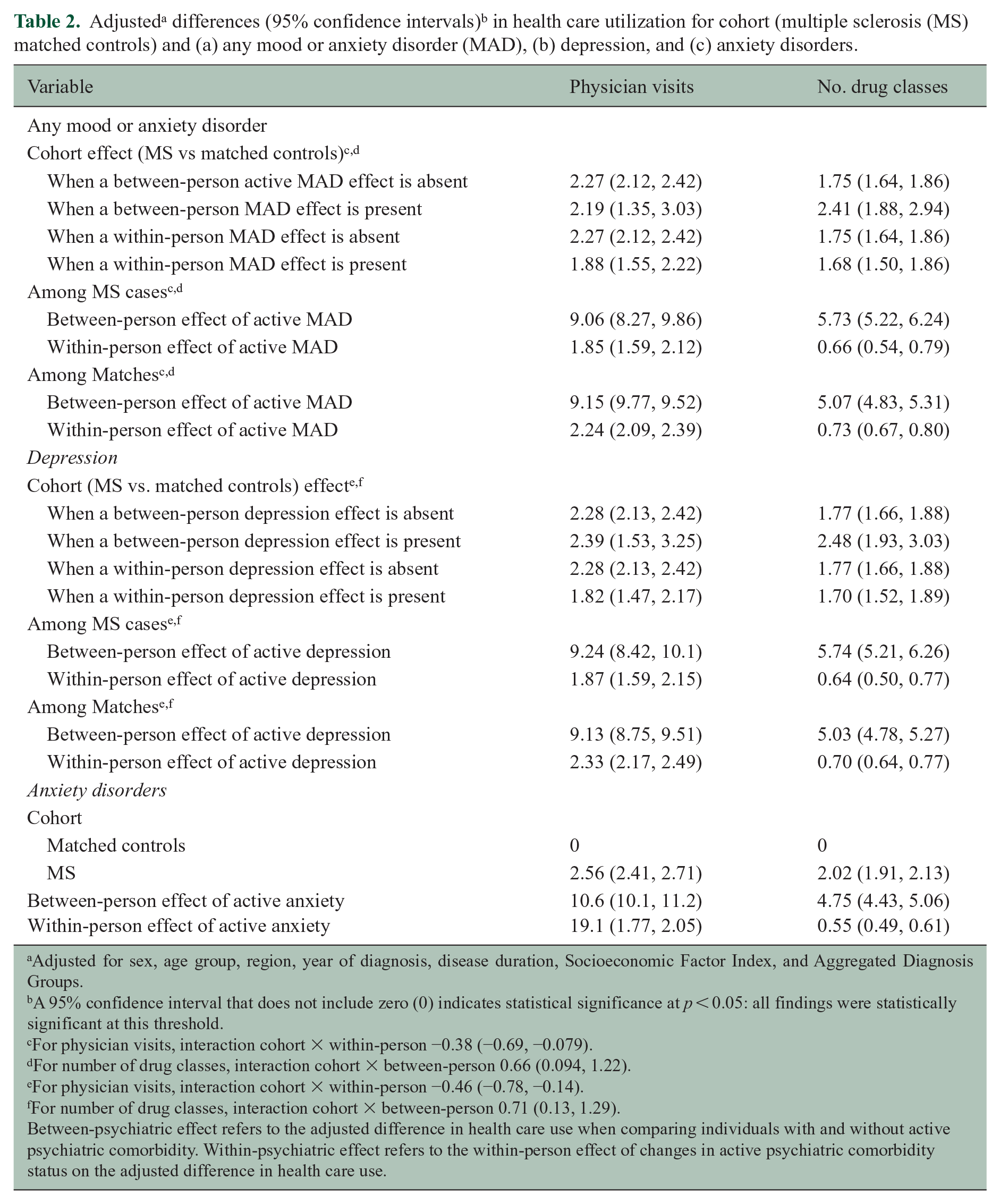
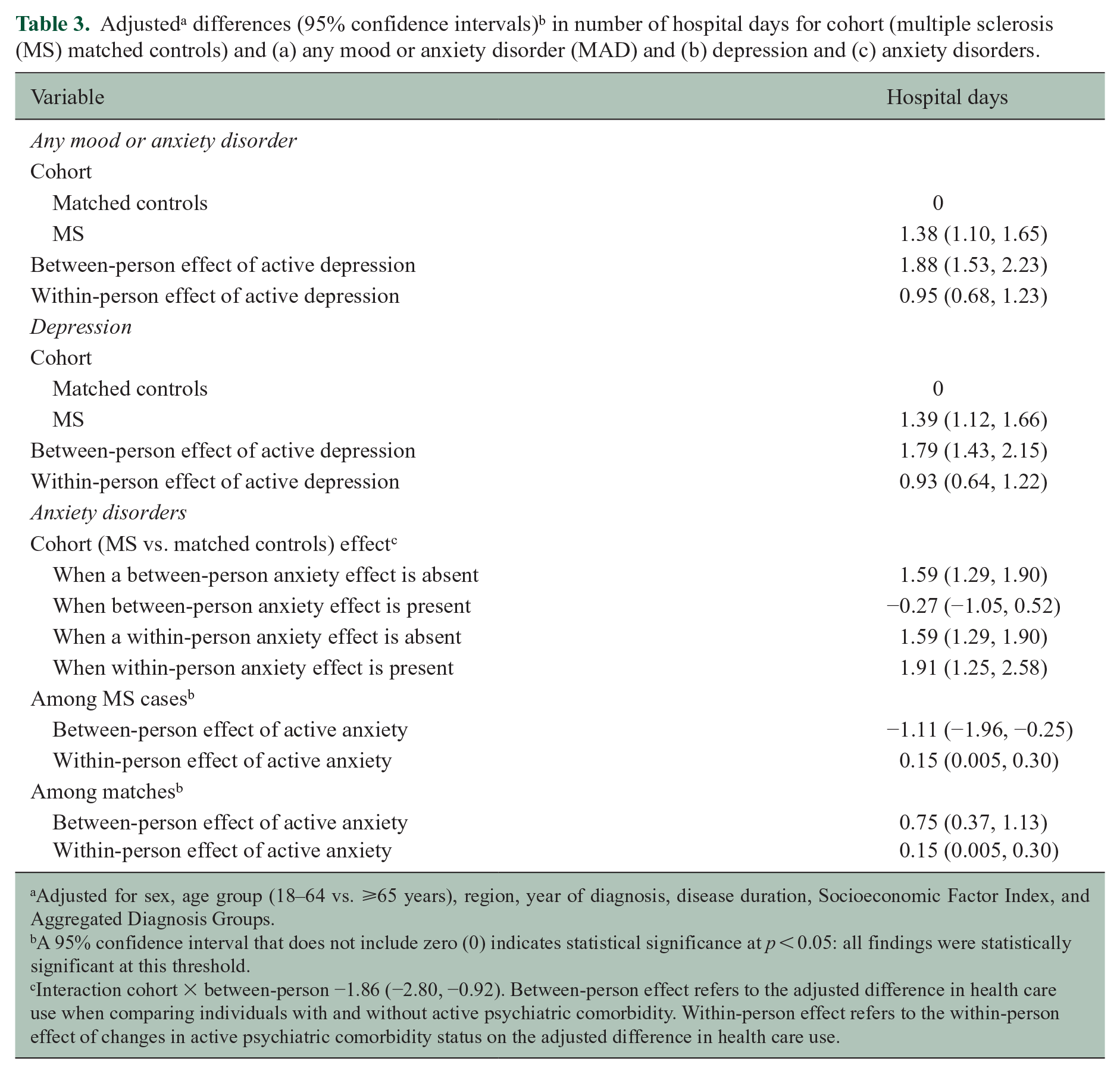
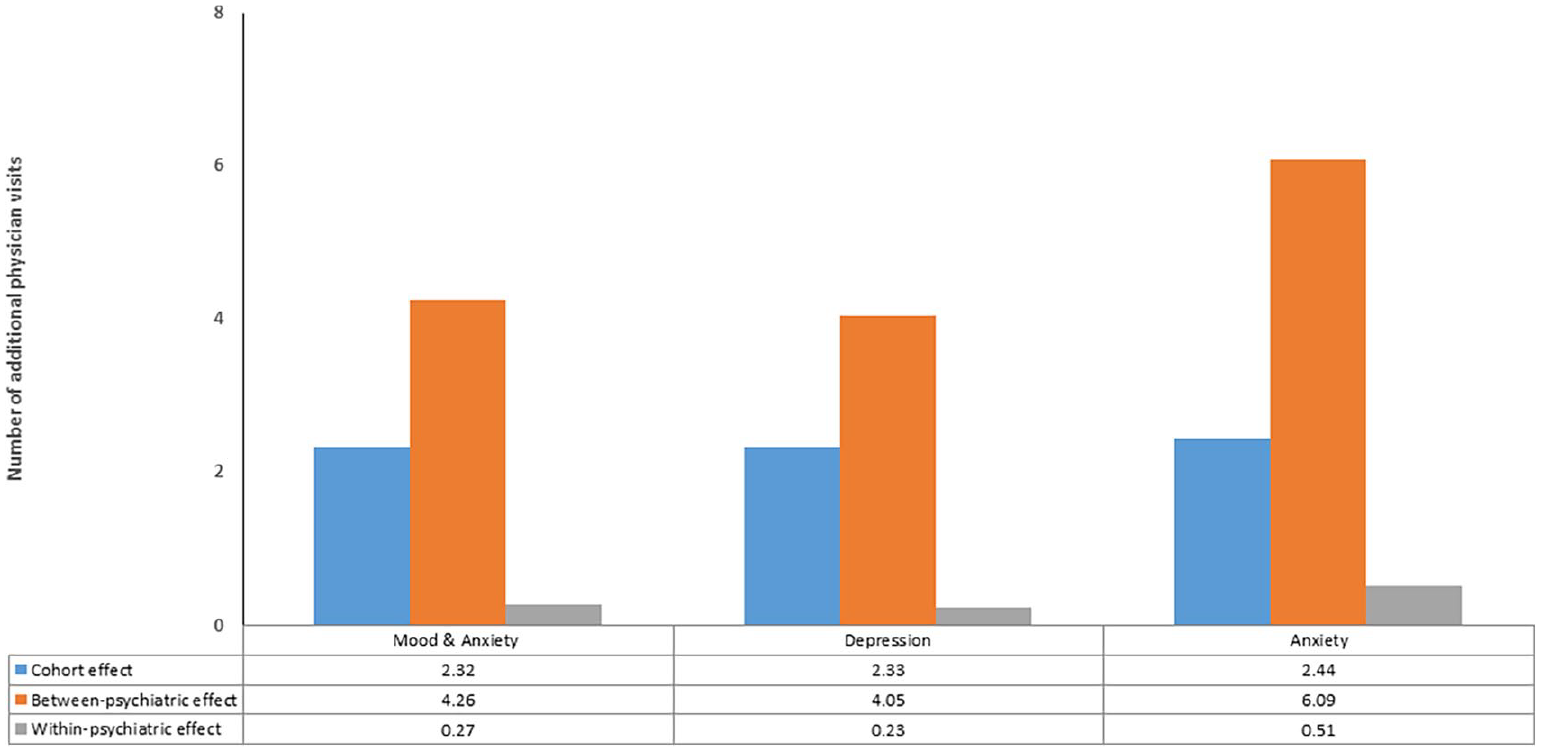
After adjusting only for any MAD, the MS cohort had over two additional physician visits, just under two more hospital days and two more classes of drugs annually versus the matched cohort (Figure 1). As compared to persons without any MAD, those with any MAD had more physician visits, had more hospital days, and used more classes of drugs annually. Within individuals, having an “active” MAD was also associated with more physician visits, hospital days, and classes of drugs dispensed than not having an “active” MAD.
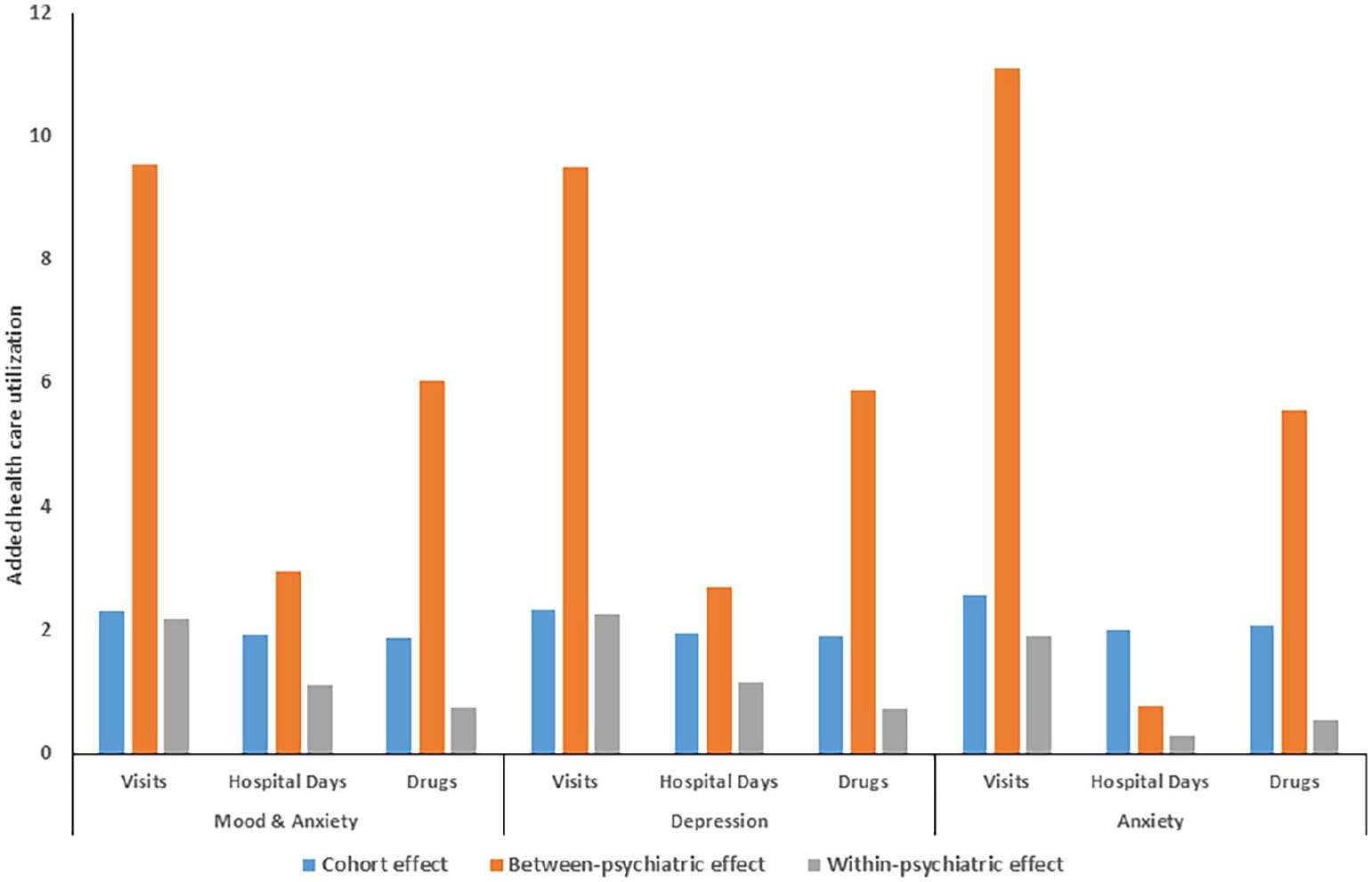
Additional health care utilization related to multiple sclerosis (cohort effect) and related to psychiatric comorbiditya.
After adjusting for cohort, individuals with any MAD had nearly 10 more physician visits than individuals without a MAD (Table 2). Similarly, individuals with any MAD had more hospital days (Table 3) and used more prescription classes (Table 2). Within individuals, having an “active” MAD was associated with more utilization for all outcomes than not having an “active” MAD. The magnitude of this effect was smaller for physician visits and drug classes dispensed than the between-person effect. When we examined the within-person and between-person effects of any MAD on physician visits after excluding visits for mental health diagnoses, findings were similar, although the effect was smaller (Figure 2).
Adjusted for sex, age group, region, year of diagnosis, disease duration, Socioeconomic Factor Index, and Aggregated Diagnosis Groups.
A 95% confidence interval that does not include zero (0) indicates statistical significance at p < 0.05: all findings were statistically significant at this threshold.
For physician visits, interaction cohort × within-person −0.38 (−0.69, −0.079).
For number of drug classes, interaction cohort × between-person 0.66 (0.094, 1.22).
For physician visits, interaction cohort × within-person −0.46 (−0.78, −0.14).
For number of drug classes, interaction cohort × between-person 0.71 (0.13, 1.29).
Between-psychiatric effect refers to the adjusted difference in health care use when comparing individuals with and without active psychiatric comorbidity. Within-psychiatric effect refers to the within-person effect of changes in active psychiatric comorbidity status on the adjusted difference in health care use.
Adjusted for sex, age group (18–64 vs. ⩾65 years), region, year of diagnosis, disease duration, Socioeconomic Factor Index, and Aggregated Diagnosis Groups.
A 95% confidence interval that does not include zero (0) indicates statistical significance at p < 0.05: all findings were statistically significant at this threshold.
Interaction cohort × between-person −1.86 (−2.80, −0.92). Between-person effect refers to the adjusted difference in health care use when comparing individuals with and without active psychiatric comorbidity. Within-person effect refers to the within-person effect of changes in active psychiatric comorbidity status on the adjusted difference in health care use.
Additional physician visits related to psychiatric comorbidity after excluding visits for psychiatric disorder diagnoses.
We observed a less than additive (i.e. negative) interaction between cohort and within-person effects of any MAD on physician visits, whereas the interaction of cohort and between-person effects on the number of prescription classes dispensed was greater than additive (i.e. synergistic; Table 2). We did not observe interactions between cohort and MAD on number of hospital days.
Findings for depression were similar to those for any MAD, including with respect to the interactions for physician visits and drug classes (Table 2). Findings for anxiety disorders were similar to those for any MAD and depression, but no interactions were observed (Table 2). When we examined the between-person and within-person effects of depression and anxiety on number of physician visits by physician type, the pattern of findings was similar (Figure e2 in Supplemental Material).
In the incident cohort (Table e4 in Supplemental Material), the within-person effects of any MAD on physician visits and drug classes were the same as those in prevalent cohort, while the between-person effects were slightly smaller. However, we did not observe significant interactions of cohort and the between-person effect on physician visits or drugs classes (Table e5 in Supplemental Material). The between-person effect of any MAD on hospital days was the same as in the prevalent cohort, while the within-person effect was smaller but fell within the bounds of the estimate in the prevalent cohort.
Discussion
In this population-based study, the prevalence of MAD, depression, and anxiety disorders was higher in persons with MS than in persons without MS. These estimates are similar to those reported previously.24,25 We also found that persons with MS had more hospital days and physician visits and used more prescription classes than persons without MS. Separating between-person and within-person effects of MAD allowed us to identify differences in the magnitude of their effects on HCU, although both were associated with additional hospital days, physician visits, and drug classes. The effects of MAD on physician visits were present even when we excluded visits for mental health diagnoses.
Our findings of increased HCU in the MS population than in the non-MS population are similar to prior reports. In the 2000 Canadian Community Health Survey, persons with MS had 12.2% more hospitalizations and 3.9 more physician visits than the general population. 26 An American study found that persons with newly diagnosed MS had 10.9% more hospitalizations and had 12.3% more emergency room visits than controls without MS in the 12 months post-diagnosis. 27 In Denmark, persons with MS also used more healthcare services than persons without MS. 6
Few studies have examined the effects of psychiatric disorders on HCU in MS. In 340 persons with MS, anxiety was associated with more physician visits and prescription dispensations. 28 Another study of 136 persons with MS did not find an association between depression and health care costs. 8 Our prior study in Manitoba conducted over the period 2007–2011 found that lifetime diagnoses of depression and bipolar disorder were associated with an increase in all-cause, but not MS-specific hospitalizations. 7 A population-based Swedish study of 4519 adults with MS found that those with psychiatric disorders had a greater risk for disability pension. 29 The between-person effects of psychiatric disorders on HCU were large, even after accounting for potential confounders. This suggests that individuals with and without psychiatric disorders differ in important ways that affect their utilization behaviors, such as other morbidities and contextual factors.
After controlling for between-person effects of psychiatric disorders, within-person variability in MAD was associated with differences in HCU across all outcomes. Specifically, being “active” with respect to any MAD, depression, or anxiety disorders rather than “inactive” was associated with average annual increase of nearly two physician visits in the following year. No prior studies have examined within-person effects of psychiatric disorders in MS. However, a study of 22,236 Americans with diagnoses of major depressive disorder identified via health claims data found that inpatient hospital days and emergency room visits were more than two-fold higher after individuals suffered a relapse of their depression. 16 The within-person effects were substantially smaller than the between-person effects, possibly reflecting that even individuals considered inactive in terms of their depression had not achieved full remission, thereby reducing apparent differences in HCU between the active and inactive states. These smaller differences could suggest that effective treatment of MAD may not reduce health care use much. However, a study of children with IBD and depression found that all HCU decreased post-psychotherapy as compared to pre-psychotherapy; effect sizes were moderate. 30
In the prevalent MS cohort, having an active MAD was associated with only a slight, less than additive effect on the number of physician visits. Partly, this may reflect a ceiling effect due to the already high number of physician visits. In the incident cohort, which had a shorter disease duration and fewer visits, the effect of an active MAD on physician visits was additive, supporting this concept. In the 2015 Medical Expenditure Panel Survey, mental health functioning did not interact with most of the chronic conditions examined to increase costs related to medical provider use. 31 However, small synergistic effects were observed for diabetes, chronic obstructive pulmonary disease, and cancer. While we found less than additive effects of MAD in MS on physician visits, having an active MAD was associated with a synergistic effect on the number of drug classes. This would amount to more than 370 additional drug classes annually than would be expected for the joint effects of MS and a MAD based on our study population of 4748 persons with MS. We can only speculate as to the reasons for these findings. One possibility lies with the symptoms of pain, fatigue, and sleep disturbance, all of which occur frequently in association with MS and are also commonly seen in MAD. Conceivably, for persons with MS and the presence of active depression or anxiety, these symptoms may either worsen these symptoms directly or result in less effective management via non-pharmacologic strategies, leading to the prescription of additional classes of medication.
Study strengths included the use of a large population-based cohort and validated case definitions for MS and psychiatric disorders, the assessment of the within-person effects of psychiatric disorders on HCU, and adjustment for medical comorbidities. Limitations should also be noted. We included only one Canadian province, and although data regarding increased HCU in MS are generally similar to those reported elsewhere, local practice patterns may have influenced our findings. Use of administrative data will underestimate the number of people affected by psychiatric disorders because some people will not seek treatment, and others will seek treatment from providers such as counselors who work outside the publicly funded health system. However, the rates of help-seeking from these other sources should not differ between the MS and matched cohorts. As administrative data lack clinical information, we could not account for the severity of the psychiatric disorders, or of MS; however, in the within-person analyses, each person acted as their own control. To limit confounding related to our definition of MAD status, we examined utilization in the year following the “active” determination, which may have underestimated the effect of active MAD status on utilization and limits the scope of our understanding of the active period. Although we defined the first claim for the psychiatric disorder as the diagnosis date, this has not been validated. We did not examine the reasons for the increased HCU associated with “active” psychiatric disorders, and this should be explored further.
Consistent with earlier studies, we found that HCU is higher in persons with MS than without MS. Importantly, as compared to persons without psychiatric comorbidity, persons with MS who have comorbid MAD attend more physician visits, use more hospital days, and use more prescription drug classes. Within persons with MS, those with active MAD also have more HCU, though this is less than the between-person effect of having a MAD or not. Together, these findings highlight the substantial impact of psychiatric comorbidity on HCU among persons with MS and potential benefits of optimized treatment of psychiatric comorbidity in this population.
Supplemental Material
MSJ963880_supplemental_material – Supplemental material for Effect of mood and anxiety disorders on health care utilization in multiple sclerosis
Supplemental material, MSJ963880_supplemental_material for Effect of mood and anxiety disorders on health care utilization in multiple sclerosis by Ruth Ann Marrie, Randy Walld, James M Bolton, Jitender Sareen, Scott B Patten, Alexander Singer, Lisa M Lix, Carol A Hitchon, Renée El-Gabalawy, Alan Katz, John D Fisk, James J Marriott and Charles N Bernstein in Multiple Sclerosis Journal
Footnotes
Acknowledgements
Members of the CIHR Team in Defining the Burden and Managing the Effects of Psychiatric Comorbidity in Chronic Immunoinflammatory Disease are Ruth Ann Marrie, James M Bolton, Jitender Sareen, John R Walker*, Scott B Patten, Alexander Singer, Lisa M. Lix, Carol A Hitchon, Renée El-Gabalawy, PhD, Alan Katz, John D Fisk, Charles N Bernstein, Lesley Graff, Lindsay Berrigan, Ryan Zarychanski, Christine Peschken, James Marriott, Kaarina Kowalec.
* Deceased
The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript. The use of Aggregated Diagnosis Groups™ (ADGs™) codes for risk adjustment in regression models were created using The Johns Hopkins Adjusted Clinical Group® (ACG®) Case-Mix System version 9. The authors acknowledge the Manitoba Centre for Health Policy for use of the Manitoba Population Research Data Repository under project #2014-030 (HIPC #2014/2015-19A). The results and conclusions presented are those of the authors and no official endorsement by the Manitoba Centre for Health Policy, Manitoba Health, or other data providers is intended or should be inferred
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ruth Ann Marrie receives research funding from CIHR, Research Manitoba, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, CMSC. She is supported by the Waugh Family Chair in Multiple Sclerosis. Randy Walld reports no disclosures. James Bolton receives research funding from CIHR, Brain and Behavior Research Foundation, and the MS Society of Canada. Jitender Sareen receives research funding from CIHR and holds stocks in Johnson and Johnson. Scott Patten receives research funding from CIHR, the MS Society of Canada, Roche, Biogen, and the Government of Alberta. Alexander Singer has received financial and in-kind support from an IBM/CIMVHR Advanced Analytics Grant and Calian Inc. Lisa Lix receives research funds from CIHR and the Arthritis Society. Carol Hitchon has research funds for unrelated studies from UCB Canada and Pfizer. Renee El-Gabalawy receives research funds from CIHR, University of Manitoba Start-Up Funds. Alan Katz receives research funds from CIHR, the Heart and Stroke Foundation, and Research Manitoba. John Fisk receives research funds from CIHR, the MS Society of Canada, Crohn’s and Colitis Canada, and the Nova Scotia Health Authority Research Fund and licensing and distribution fees from MAPI Research Trust. James Marriott has conducted clinical trials for Biogen Idec and Roche and receives research funding from the MS Society of Canada, the MS Scientific Foundation, and Research Manitoba. Charles Bernstein has consulted to Abbvie Canada, Janssen Canada, Pfizer Canada, Shire Canada, and Takeda Canada and has received unrestricted educational grants from Abbvie Canada, Janssen Canada, Shire Canada, and Takeda Canada. He has been on speaker’s bureaus of Abbvie Canada and Shire Canada.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Canadian Institutes of Health Research (THC-135234), Crohn’s and Colitis Canada, and the Waugh Family Chair in Multiple Sclerosis (to RAM). Dr Bernstein is supported in part by the Bingham Chair in Gastroenterology. Dr Sareen is supported by CIHR #333252. Dr Katz is supported by a Research Manitoba Chair. Dr Lix is supported by a Tier 1 Canada Research Chair. Dr El-Gabalawy is supported by University of Manitoba Start-Up Funding. Dr Patten holds the Cuthbertson & Fischer Chair in Pediatric Mental Health at the University of Calgary.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.