Abstract
Approximately 200,000 multiple sclerosis (MS) patients worldwide receive B-cell-depleting immunotherapy with rituximab (anti-CD20), which eliminates the ability to generate an antibody response to new infections. As severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)–specific antibodies might help viral clearance, these patients could be at risk of severe complications if infected. Here, we report on an MS patient who had received rituximab for ~3 years. The patient was examined 5 days before the onset of coronavirus disease 2019 (COVID-19) symptoms and was admitted to the hospital 2 days after. She recovered 14 days after symptom onset despite having a 0% B lymphocyte count and not developing SARS-CoV-2 immunoglobulin G (IgG) antibodies.
Keywords
Introduction
The first cases of coronavirus disease 2019 (COVID-19) were observed in China in December 2019. 1 The causative virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is the result of a zoonotic transmission either directly from bats or via an intermediate host species. 2 The virus has spread globally since January 2020. Morbidity and mortality rates increase with age and specific comorbidities. 3 Disease severity in patients is linked to not only the viral infection but also the host immune response, creating an urgent need to understand the virus–host interplay. Convalescent serum samples have been applied with apparently good clinical results in COVID-19, 1 raising concerns that multiple sclerosis (MS) patients receiving B-cell-depleting treatment might suffer severe complications during primary infection; such concerns are reflected by decisions to delay the use of anti-CD20 drugs in new patients and suggestions to consider extended-interval dosing for those already receiving anti-CD20 therapy, especially in those who are B-cell depleted or have low levels of immunoglobulin G (IgG). 4 This case report suggests that MS patients receiving B-cell-depleting therapy are not necessarily at higher risk and demonstrates that infection can resolve through cellular immune responses alone.
Report of case
The 59-year-old female MS patient was diagnosed in 2016, and rituximab treatment initiated in September 2017; no CD19+ B-cell reconstitution was detected at any follow-up visits during the 3 years of treatment (Figure 1(a)). On 1 April 2020, the patient had a mild paraparesis and paraspasticity and a limited walking distance, unchanged since the last admission in September 2019 (Expanded Disability Status Scale (EDSS) of 6), and a 0% B-cell count. No COVID-19 symptoms were present. Routine laboratory investigations were normal; magnetic resonance imaging (MRI) showed a stable disease course since initiation of rituximab; natural killer (NK), CD4+, and CD8+ T-cell counts were normal; and the CD4/CD8 T-cell ratio inverted, a common consequence of rituximab treatment. 5
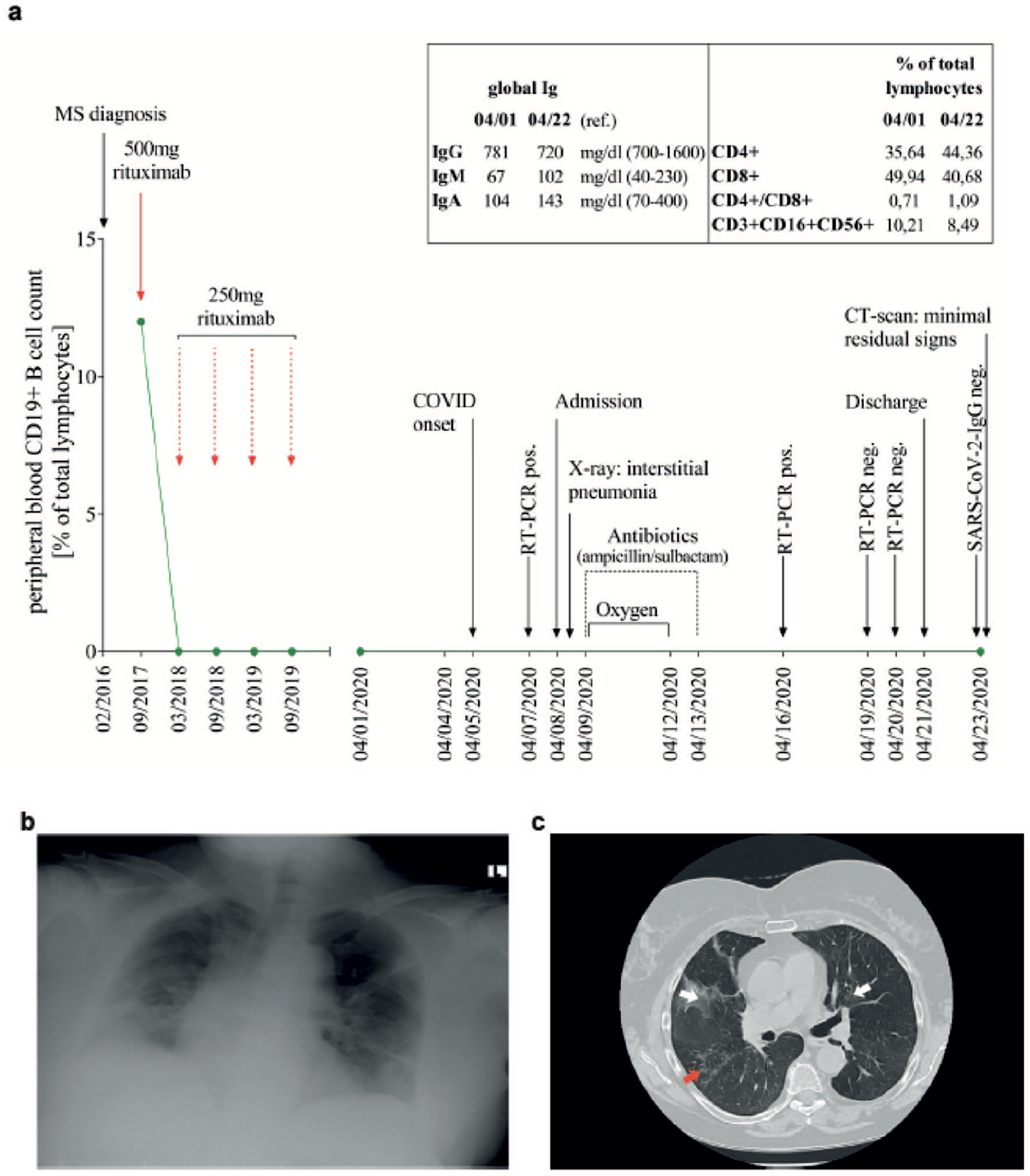
Clinical management of a 59-year-old female patient with progressive multiple sclerosis (MS) infected with SARS-CoV-2. (a) Disease course. The patient was diagnosed with MS in February 2016. Rituximab treatment was started in September 2017, and repeat doses were given every 6 months as outlined. Symptoms of COVID-19 developed on 5 April 2020. All symptoms resolved after 14 days. Box shows IgG, IgM, and IgA counts (left), and CD4+ and CD8+ T-cell counts, CD4+/CD8+ ratios, and NK (CD3+, CD16+, CD56+) cell counts (right). Antibody levels and T-cell and NK cell counts were measured on 1 April 2020 (5 days before the onset of COVID-19 symptoms) and on 22 April 2020 (1 day after discharge from hospital and 6 days after the first negative SARS-CoV-2 nasopharyngeal swab test). (b) Chest X-ray. The anteroposterior erect chest X-ray was taken on admission, 7 April, showing typical signs of interstitial pneumonia with severely decreased lung volumes and reticulation. (c) Thoracic high-resolution CT (HRCT) chest scan. The HRCT scan was performed 1 day after discharge from hospital (22 April) showing residual signs of interstitial pneumonia, that is, dorsobasal dystelectasis (white arrows) and minor honeycombing (red arrow).
On 5 April 2020, the patient developed a dry cough, which worsened 2 days later with dyspnea, fatigue, headache, and nausea. The nasopharyngeal swab test for SARS-CoV-2 was positive, and the patient was admitted to hospital with a fever of 39°C and low oxygen saturation. A chest X-ray revealed interstitial pneumonia (Figure 1(b)), and prophylactic intravenous antibiotic (ampicillin/sulbactam) treatment was instituted. The dyspnea rapidly improved, CD4+ T-cell counts increased, the CD4/CD8 ratio normalized, and the NK cell count slightly decreased but was still within the normal range.
On days 14 and 15, after symptom onset, two nasopharyngeal swab test and a cerebrospinal fluid (CSF) sample were negative for SARS-CoV-2 (Figure 1(a)). All symptoms resolved, and the patient was discharged 13 days after admission. A SARS-CoV-2 IgG antibody test was negative 18 days after symptom onset, and a thoracic chest computed tomography (CT) scan (Figure 1(c)) showed minimal residual signs of interstitial pneumonia (dorsobasal dystelectasis).
Discussion
As cross-neutralization of SARS-CoV-2 by antibodies to other coronaviruses is unlikely, 6 this case, suggests that SARS-CoV-2 infection can resolve solely through cellular immune responses. This finding is further supported by a study reporting the successful recovery from SARS-CoV-2 infection in a set of twins who lacked B-cells due to X-linked agammaglobulinemia. 7 These observations shed light on other COVID-19 studies,6,8 which demonstrated that patients with mild disease symptoms have low antibody levels 6 and those with weak IgG responses clear virus faster than those with strong IgG responses. 8 Collectively, these findings suggest that the best correlate of viral clearance might be obtained by measuring cellular immune responses rather than antibody levels. Furthermore, potentially harmful effects of SARS-CoV-2 antibodies toward the Spike protein are highlighted by experiments with a SARS-CoV-1-spike Modified vaccinia Ankara (MVA) vaccine in rhesus macaques. 9 In this trial, antibody-dependent enhancement (ADE) of infection of alveolar macrophages resulted in a switch from a wound healing to a proinflammatory phenotype. Subsequent interleukin-6 (IL-6), interleukin-8 (IL-8), and monocyte chemoattractant protein-1 (MCP-1/CLL2) secretion ignited a hyperinflammatory reaction, resulting in severe alveolar damage similar to that seen in some COVID-19 patients. 1 Thus, if ADE contributes to COVID-19 pathogenesis, 10 patients receiving rituximab might be less likely to develop alveolar hyperinflammation due to a lack of SARS-CoV-2 antibody production and the absence of effector-memory B-cell-mediated proinflammatory cytokine production. 5
Data from a cross-sectional questionnaire study (n = 712) from Iran found that MS patients on B-cell-depleting therapy had a 2.6-fold increase in the risk of being in the COVID-19-suspect group. This group comprised patients who reported fever and cough or fever and shortness of breath, and those who had a suggestive chest CT (n = 34); however, only two COVID-19-suspect patients were hospitalized and neither required intensive care. 11 This study suggests that MS patients as a group have a mild to moderate disease course regardless of the treatment regimen and also that those on B-cell-depleting therapy might be at higher risk of SARS-CoV-2 infection. As none of the patients were tested for SARS-CoV-2, these observations need to be verified in a setting where virus testing is possible.
Recovery from COVID-19 has previously been reported in one MS patient with hepatitis B infection who was treated for a year with the B-cell-depleting drug ocrelizumab and the antiviral drug lamivudine (3TC). 12 Whereas recovery from COVID-19 in this patient could potentially be facilitated by lamivudine, our report highlights that B-cell-depleted MS patients can clear the virus without the aid of antiviral therapy. These observations are in line with the lack of fatalities reported by an international pharmacovigilance case series of 30 ocrelizumab-treated MS patients infected by SARS-CoV-2. 13
It is unknown whether the patient described here, or any patient who has recovered from SARS-CoV-2 infection in the absence of B-cells, is at higher risk of reinfection. However, this case suggests that MS patients receiving B-cell-depleting therapy are not at higher risk of severe complications from primary SARS-CoV-2 infection, and demonstrates that viral clearance is possible without B-cell involvement and antiviral therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
Written consent has been obtained from the patient.
