Abstract

In childhood, multiple sclerosis (MS) with a primary progressive disease course is very rare.1,2 Therefore, we read the recent article of Abdel-Mannan et al. 3 presenting six pediatric patients fulfilling McDonald 2017 criteria for primary progressive MS (PPMS) with great interest. All patients presented with a progressive myelopathy, including progressive balance, and/or lower limb function impairment for at least 1 year. During follow-up, they had an ongoing progressive disease course, without sustained improvement and with a faster decline of Expanded Disability Status Scale (EDSS) compared to pediatric relapsing-remitting MS patients. In the four patients treated with intravenous methylprednisolone (ivMP), this treatment was unsuccessful. Patients continued to progress despite immunomodulatory treatment (azathioprine or hematopoietic stem cell transplantation). Only one patient was started on first-line disease-modifying therapy (DMT) for MS (interferon-beta-1a) but did not tolerate this. Although rare, Abdel-Mannan et al. 3 plead for recognition of PPMS in childhood in order to start registered PPMS treatment in these patients in hope to prevent further deterioration.
In our national cohort of pediatric patients with demyelinating diseases in the Netherlands (PROUD-kids study; PRedicting OUtcome in acquired Demyelinating syndromes in childhood), we identified two pediatric MS patients who presented with a progressive decline of balance, coordination, and motor function. In addition, one patient had progressive vision loss. At symptom onset, these patients were 8 and 13 years old (a boy and a girl, respectively). At the time of presentation in our National Pediatric MS center in Rotterdam, they already deteriorated for 9.2 and 1.3 years, respectively. Comparable with the cases described by Abdel-Mannan et al., 3 magnetic resonance (MR) imaging of our patients showed typical MS lesions with presence of periventricular, juxtacortical, infratentorial, and spinal cord lesions (Figure 1); presence of unique oligoclonal bands in cerebrospinal fluid; and no indication for other diagnoses in additional investigations in both. They also responded poorly to ivMP, with no to minimal clinical improvement.
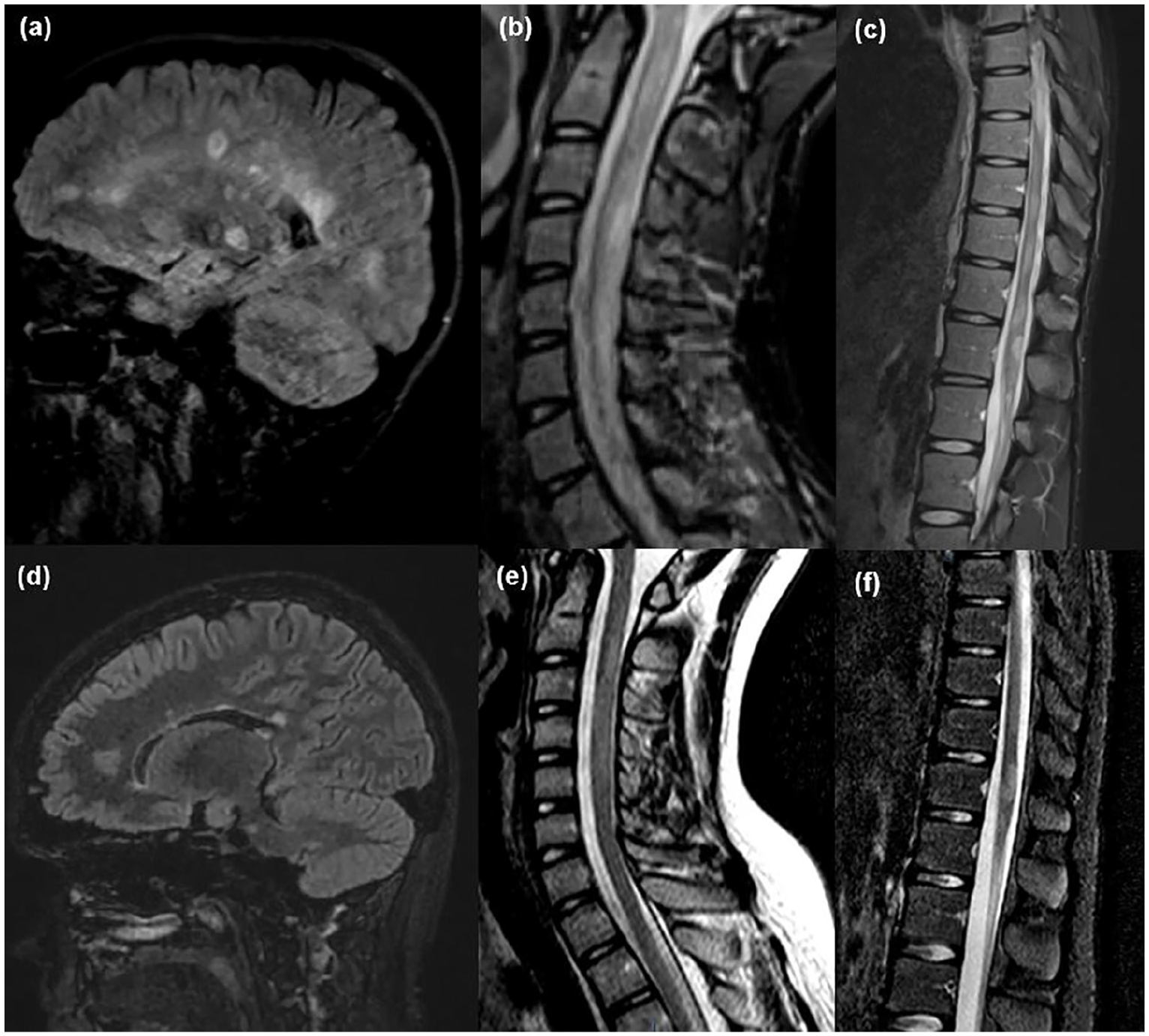
MR images of the (a–c) boy and (d–f) girl with a primary progressive-like disease course from first presentation. Sagittal FLAIR-weighted brain images show (a, d) multiple supratentorial and (d) infratentorial hyperintens white matter lesions. T2-weighted spinal cord MRI scan shows (b, e) cervical as well as (c, f) thoracolumbar involvement in both patients.
Interestingly, in contrast to the continuing progression Abdel-Mannan et al. 3 described in all their patients despite treatment (no DMTs), our patients stabilized after starting second-line DMTs. Directly after MS diagnosis, both were started on Natalizumab, and recently one patient changed to Fingolimod due to positive John Cunningham virus. Within a follow-up of 2.1 years after starting treatment, a relevant improvement in EDSS was observed from 3.5 to 3.0 and 4.0 to 3.5, respectively. In addition, they had no relapses and MR imaging of brain and spinal cord showed no new lesions during follow-up. Even though they presented with a primary progressive-like disease course and did not respond well to steroids; in our opinion, the observed response to second-line DMTs may indicate relevant inflammatory activity in these pediatric patients. This is in line with the higher inflammatory activity observed in pediatric compared to adult MS patients with a relapsing disease course.4,5 In conclusion, in our view pediatric MS patients with a primary progressive-like disease course differ from adult PPMS patients. Therefore, we argue that all pediatric MS patients with a primary progressive-like disease course should receive regular first-line or second-line MS treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.L.B., K.M.B., and B.H.A.W. declare no conflicts of interest. J.S. received lecture and/or consultancy fees from Biogen, Merck, Novartis, and Sanofi-Genzyme. R.F.N. participates in trials by Sanofi and Novartis, received honoraria from Novartis and Zogenix.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Dutch MS research Foundation.
