Abstract
Background:
Alemtuzumab is given as two annual courses. Patients with continued disease activity may receive as-needed additional courses.
Objective:
To evaluate efficacy and safety of additional alemtuzumab courses in the CARE-MS (Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis) studies and their extensions.
Methods:
Subgroups were based on the number of additional alemtuzumab courses received. Exclusion criteria: other disease-modifying therapy (DMT); <12-month follow-up after last alemtuzumab course.
Results:
In the additional-courses groups, Courses 3 and 4 reduced annualized relapse rate (12 months before: 0.73 and 0.74, respectively; 12 months after: 0.07 and 0.08). For 36 months after Courses 3 and 4, 89% and 92% of patients were free of 6-month confirmed disability worsening, respectively, with 20% and 26% achieving 6-month confirmed disability improvement. Freedom from magnetic resonance imaging (MRI) disease activity increased after Courses 3 and 4 (12 months before: 43% and 53%, respectively; 12 months after: 73% and 74%). Safety was similar across groups; serious events occurred irrespective of the number of courses.
Conclusion:
Additional alemtuzumab courses significantly improved outcomes, without increased safety risks, in CARE-MS patients with continued disease activity after Course 2. How this compares to outcomes if treatment is switched to another DMT instead remains unknown.
Introduction
Alemtuzumab (LEMTRADA®; Sanofi Genzyme, Cambridge, MA, USA), a humanized monoclonal antibody, demonstrated efficacy in 2-year, phase 3 clinical trials (Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis (CARE-MS) I (NCT00530348) and II (NCT00548405))1,2 by significantly improving clinical and magnetic resonance imaging (MRI) outcomes in patients with relapsing-remitting multiple sclerosis (RRMS) versus subcutaneous interferon beta-1a (SC IFNB-1a; Rebif®; EMD Serono Inc., Rockland, MA, USA). Outcomes were maintained over a 4-year open-label CARE-MS extension study (CAMMS03409 (NCT00930553))3 –5 and continued into the ongoing 5-year long-Term follow-up study for multiple sclerOsis Patients who have completed the AlemtuZumab extension study (TOPAZ; NCT02255656).6,7
Alemtuzumab selectively targets the cell surface antigen CD52,8,9 causing depletion of B and T cells and a subsequent distinct pattern of repopulation, along with a shift toward a less inflammatory cytokine profile.9,10 The resulting immune system rebalancing is an important component of the mechanism of action leading to suppression of disease activity; however, the mechanism of action is not yet fully elucidated.8,11 –13
Alemtuzumab was initially approved as two 12 mg/day courses (5 consecutive days at treatment initiation; 3 consecutive days 12 months later).14,15 Product labeling was amended in 2018 to include additional as-needed 3-day courses in the United States (no limit on additional courses), and Europe and Australia (up to two additional courses), for disease activity defined by clinical or imaging features and given at least 12 months after the most recent course.14,15 To investigate the efficacy and safety of additional alemtuzumab in patients with disease activity following Course 2, post hoc analyses of 8-year data from the CARE-MS, extension, and TOPAZ studies were undertaken.
Patients and methods
Patients and procedures
Post hoc analyses of patient subgroups, defined by total number of alemtuzumab courses received (2-courses, 3-courses, 4-courses, and ⩾5-courses), were carried out with alemtuzumab-treated patients from CARE-MS I and II who subsequently entered the extension studies. Analysis included 8 years of follow-up after initiating alemtuzumab (analysis cut-off date: 4 October 2017). Patients who received another disease-modifying therapy (DMT) at any time, or received Courses 3, 4, or 5 after Month 85 (i.e. <12 months of follow-up after the last alemtuzumab course before data cut-off date), were excluded. Data from patients who received ⩾6 alemtuzumab courses were censored at the time of Course 6 administration to assess the efficacy of Course 5. Patients who received SC IFNB-1a in the core studies and switched to alemtuzumab in the CARE-MS extension were not included in this analysis.
Study designs for the two rater-blinded, active-controlled, head-to-head, phase 3 CARE-MS trials, and the open-label CARE-MS extension, have been published previously.1 –5 In the core studies, patients with active RRMS who were treatment-naive (CARE-MS I) 1 or had an inadequate response to prior therapy (CARE-MS II) 2 were randomized to either alemtuzumab 12 mg/day on 5 consecutive days at baseline and on 3 consecutive days 12 months later, or SC IFNB-1a 44 µg three times per week. Patients completing the core studies could enroll in the CARE-MS extension and receive additional courses of alemtuzumab 12 mg/day on 3 consecutive days ⩾12 months after the previous course as needed for disease activity (⩾1 protocol-defined relapses and/or ⩾2 unique lesions defined as either new/enlarging T2 hyperintense and/or gadolinium (Gd)-enhancing brain and/or spinal cord lesions on MRI) at investigator’s discretion, or receive another DMT. Disqualification criteria for additional courses included, but were not limited to, pregnancy, prior diagnosis of immune thrombocytopenia (ITP), anti-glomerular membrane disease, and/or history of malignancy (except basal cell carcinoma). Patients completing the 4-year CARE-MS extension study could enroll in the subsequent, ongoing, 5-year TOPAZ extension study and receive additional alemtuzumab 12 mg/day on 3 consecutive days ⩾ 12 months after the previous course at investigator’s discretion (no disease criteria), or receive another DMT at any time.
Efficacy and safety assessments
Details regarding relapse event assessment, Expanded Disability Status Scale (EDSS) assessment, and MRI scans were described previously.1 –5 Patients free of relapse and patients free of MRI disease activity were reported on an annual basis as percentages of patients per year. Patients were assessed for cumulative freedom from 6-month confirmed disability worsening (CDW; ⩾1-point EDSS score increase (⩾1.5 if baseline EDSS = 0)) and cumulative 6-month confirmed disability improvement (CDI; ⩾1.0-point decrease from core study baseline EDSS score, assessed in patients with baseline EDSS scores ⩾2.0). MRI lesion outcomes included percentages of patients with MRI disease activity, defined as new Gd-enhancing T1 lesions on current scan or new/enlarging T2 hyperintense lesions since last scan; additional MRI analyses included assessment of new T1 non-enhancing hypointense lesions.
Safety was evaluated over 8 years, and all patients were monitored for autoimmune adverse events (AEs) for ⩾48 months following the last course of alemtuzumab.1 –5 Infusion-associated reactions (IARs) were defined as any AE with onset during or ⩽24 hours after the end of infusion.
Statistical analysis
Analyses were based on all available data without imputation through Year 8. Annualized relapse rates (ARRs) were estimated through negative binomial regression. Comparison of ARR before and after receiving additional courses was evaluated using a repeated negative binomial regression model with robust variance estimation. Percentages of patients free of 6-month CDW or achieving 6-month CDI were determined using the Kaplan–Meier method. MRI disease activity by means of MRI lesions was summarized descriptively with percentages, and 95% confidence intervals (CIs) were obtained using the normal approximation to the binomial distribution. Percentages of patients free of MRI disease activity before and after receiving additional courses were compared using McNemar’s test. Safety data were reported as incidences (percentage of patients with ⩾1 event) through Year 8. All statistical analyses were conducted using SAS (version 9.4, The SAS Institute, Cary, NC, USA).
Protocol approval, registration, and patient consent
CARE-MS I, CARE-MS II, CAMMS03409, and TOPAZ are registered with ClinicalTrials.gov. Patients provided written informed consent, and all procedures were approved by local institutional ethics review boards of participating sites.
Results
Patients
Of 742 CARE-MS patients who entered the CARE-MS extension, 396 (53%) received no more than two courses over 8 years, 201 (27%) received exactly three courses, 94 (13%) received exactly four courses, and 51 (<7%) received five or more courses (Figure 1(a)). Courses 3 and 4 were received most frequently in Year 3 (143/742 (19%)) and Year 4 (45/742 (6%)), respectively (Figure 1(b)). For patients who received exactly three courses, the mean (median (range)) time between Courses 2 and 3 was 3.3 years (3.0 (1.0–6.8) years). The intervals between Courses 2 and 3, and subsequently Courses 3 and 4, in patients who received exactly four courses were 2.0 years (1.7 (1.0–5.0) years) and 2.3 years (2.1 (1.0–5.5) years), respectively. Reasons for additional treatment were reported as relapse (Course 3: 52%; Course 4: 48%), MRI activity (Course 3: 23%; Course 4: 20%), both relapse and MRI activity (Course 3: 21%; Course 4: 23%), other reasons (Course 3: 2%; Course 4: 6%), and no reason given (Course 3: 2%; Course 4: 3%). Supporting Tables 1–3 provide comprehensive frequencies of CARE-MS I and II patients who received additional alemtuzumab and/or other DMTs by year through Year 8.
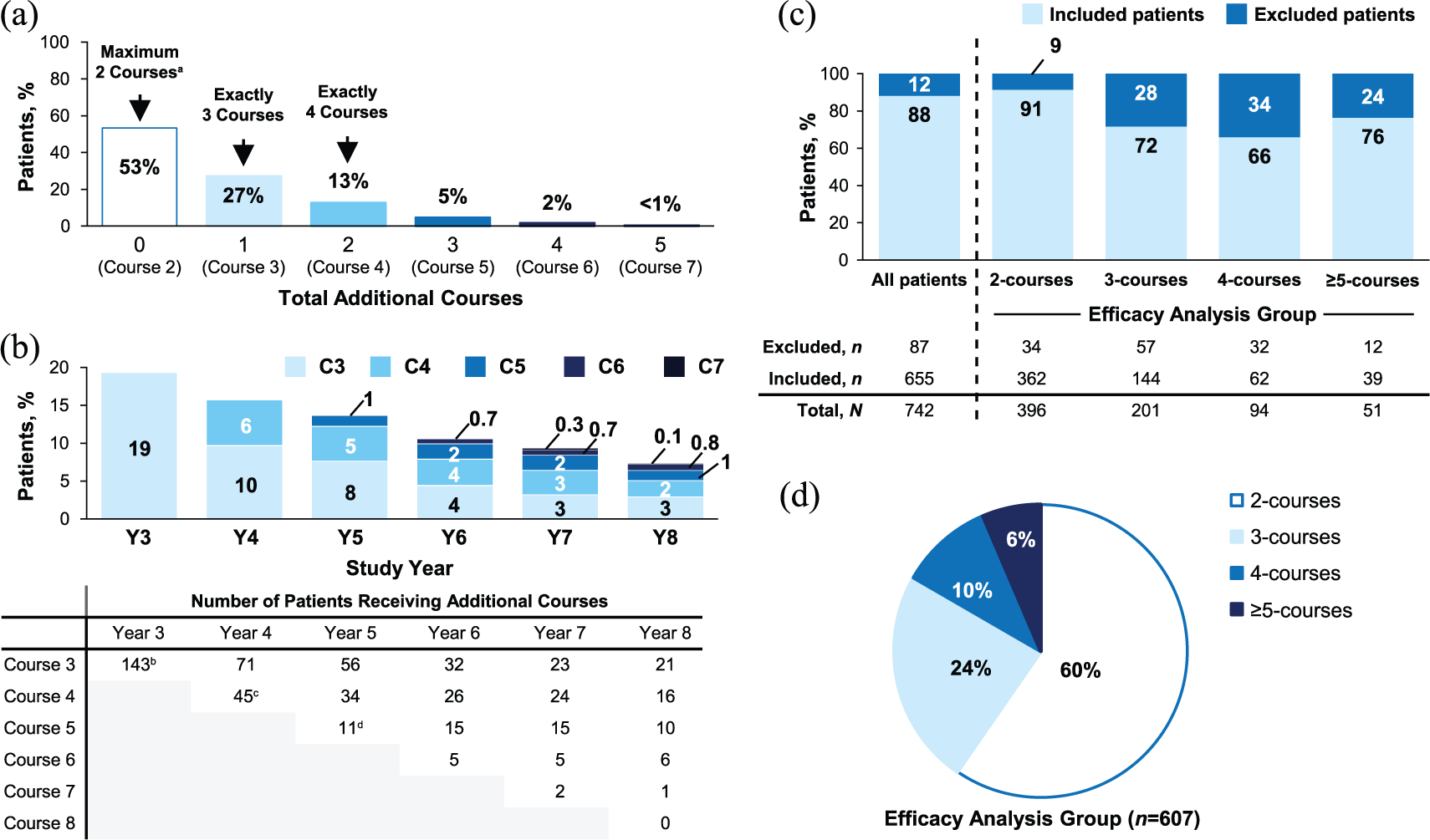
Percentages of patients receiving additional courses of alemtuzumab through Year 8. (a) Percentages of all patients who entered the CARE-MS extension (N = 742) and received additional courses of alemtuzumab 12 mg/day through Year 8. (b) Timing of administration of Courses 3, 4, 5, 6, 7, and 8 through Year 8. Percentages are based on all patients who entered the CARE-MS extension (N = 742). (c) Proportions of patients included and excluded from the analysis based on inclusion criteria for the 2-, 3-, 4-, and ⩾5-courses groups. (d) Proportional makeup of the analyzed patient population belonging to the 2-, 3-, 4-, or ⩾5-courses groups.
Pooled CARE-MS I and II core study baseline characteristics.
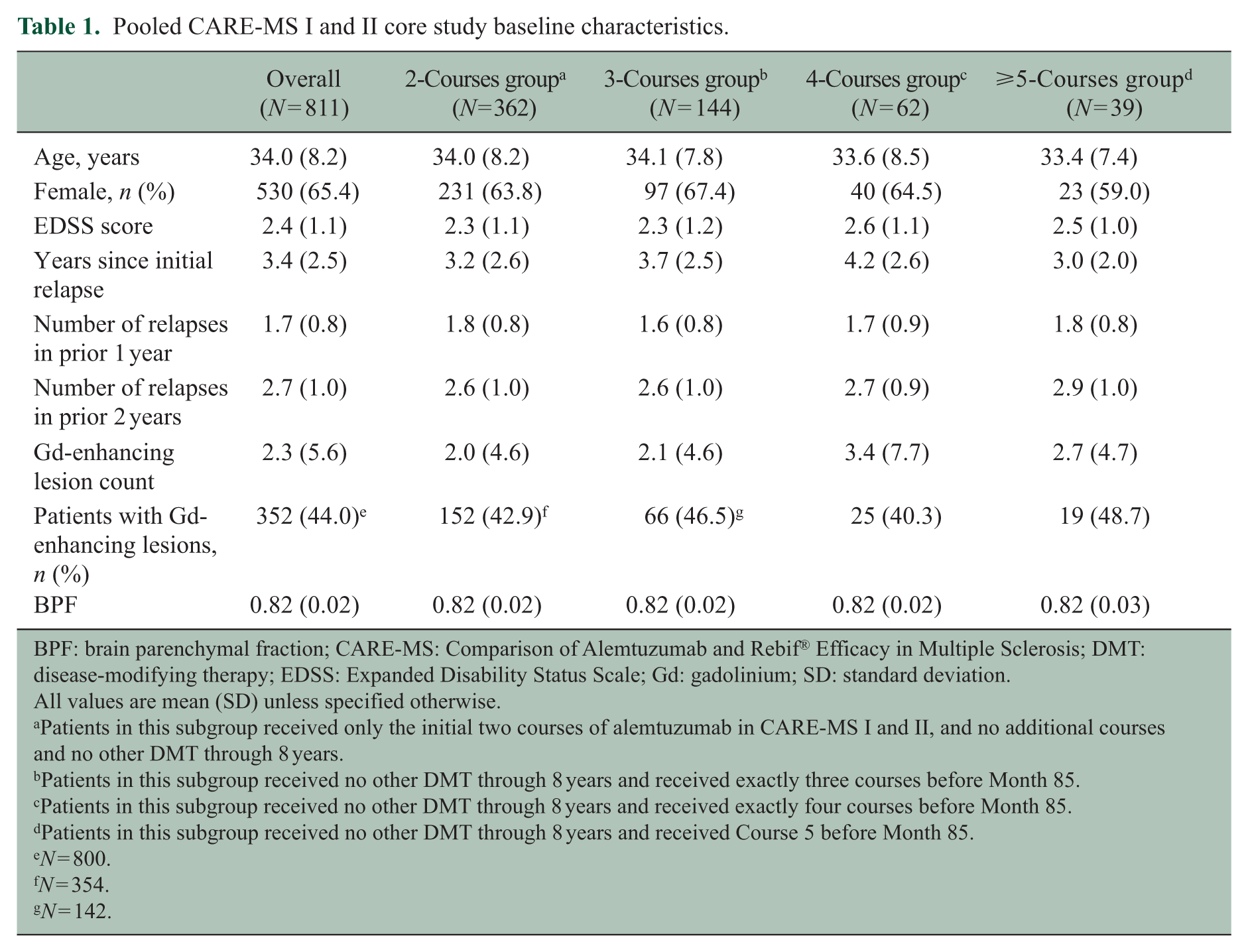
BPF: brain parenchymal fraction; CARE-MS: Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis; DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; Gd: gadolinium; SD: standard deviation.
All values are mean (SD) unless specified otherwise.
Patients in this subgroup received only the initial two courses of alemtuzumab in CARE-MS I and II, and no additional courses and no other DMT through 8 years.
Patients in this subgroup received no other DMT through 8 years and received exactly three courses before Month 85.
Patients in this subgroup received no other DMT through 8 years and received exactly four courses before Month 85.
Patients in this subgroup received no other DMT through 8 years and received Course 5 before Month 85.
N = 800.
N = 354.
N = 142.
A total of 655 patients had ⩾12 months of follow-up after their last course of alemtuzumab with no other DMT through Year 8 and were thus included in the analysis. Of these, 607 were included in the efficacy analysis subgroups, with 362 (60%), 144 (24%), 62 (10%), and 39 (6%) receiving exactly two, exactly three, exactly four, and ⩾5 courses, respectively (Figure 1(c) and (d)). Among these, 269/362 (74%) in the 2-courses group, 120/144 (83%) in the 3-courses group, 50/62 (81%) in the 4-courses group, and 34/39 (87%) in the ⩾5-courses group remained on study through Year 8 (Supporting Figure 1). Baseline characteristics were comparable between all groups; however, patients in the 4-courses and ⩾5-courses groups had higher core study baseline Gd-enhancing lesion counts (Table 1).
Efficacy of two courses
Relapse rate was highest in Year 1 (0.14) before receiving Course 2 and decreased in subsequent years. ARR was 0.04 in Year 8, with 88%–98% of patients per year relapse-free (Figure 2(a)). Mean EDSS score was stable through Year 8, with −0.30 (95% CI: −0.44 to −0.16) mean change from core study baseline (Figure 2(b)); 78% of patients were free of 6-month CDW and 53% achieved 6-month CDI cumulatively over 8 years (Figure 2(c)). In Year 8, 77% of patients were free of MRI disease activity (Figure 2(d)).
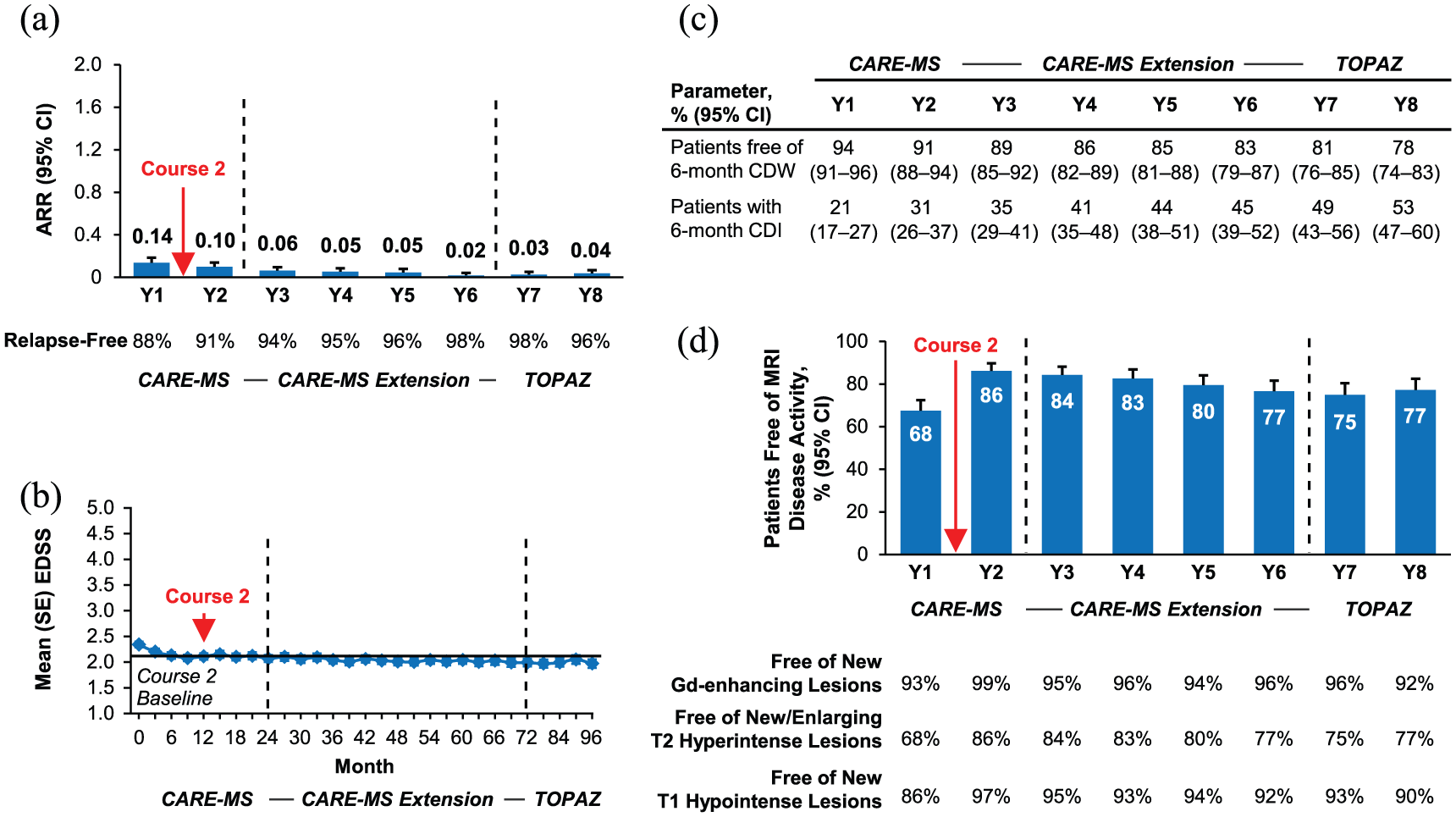
Efficacy outcomes in the 2-courses group. Results for patients who received exactly two courses and no other DMT through Year 8. (a) Yearly ARR from Year 1 of the core study through Year 8. (b) Mean (SE) EDSS scores from CARE-MS core study baseline through Year 8. EDSS scores were assessed every 3 months during the core studies and the CARE-MS extension, and every 6 months during TOPAZ. (c) Kaplan–Meier analyses of the percentages of patients free of 6-month CDW, and percentages of patients with 6-month CDI from Year 1 through Year 8. (d) Percentages of patients free of MRI disease activity, new Gd-enhancing T1 lesions, new/enlarging T2 hyperintense lesions, and new T1 hypointense lesions from Year 1 through Year 8.
Efficacy of Course 3
ARR was 0.73 during the 12 months before Course 3, due to disease activity that drove the administration of additional courses. ARR was significantly reduced in the 12 months after Course 3 (0.07; p < 0.0001), and remained low at Month 36 (0.09; Figure 3(a)). Percentages of patients relapse-free increased from 33% in the 12 months before Course 3 to 94%, 88%, and 85% per year in each year (Months 12, 24, and 36) post Course 3, respectively. Mean EDSS score increased prior to Course 3 administration and then was stable over 36 months after Course 3 (Figure 3(b)); change in mean EDSS score from Course 3 administration to Month 36 was −0.03 (95% CI: −0.30 to +0.25). Cumulatively over 36 months after Course 3, 89% of patients were free of 6-month CDW and 20% achieved 6-month CDI (Figure 3(c)). The percentage of patients free of MRI disease activity increased significantly from 43% in the 12 months before Course 3 to 73% (p < 0.0001) in the 12 months after Course 3, with similar proportions free of MRI disease activity in each subsequent year (Figure 3(d)).
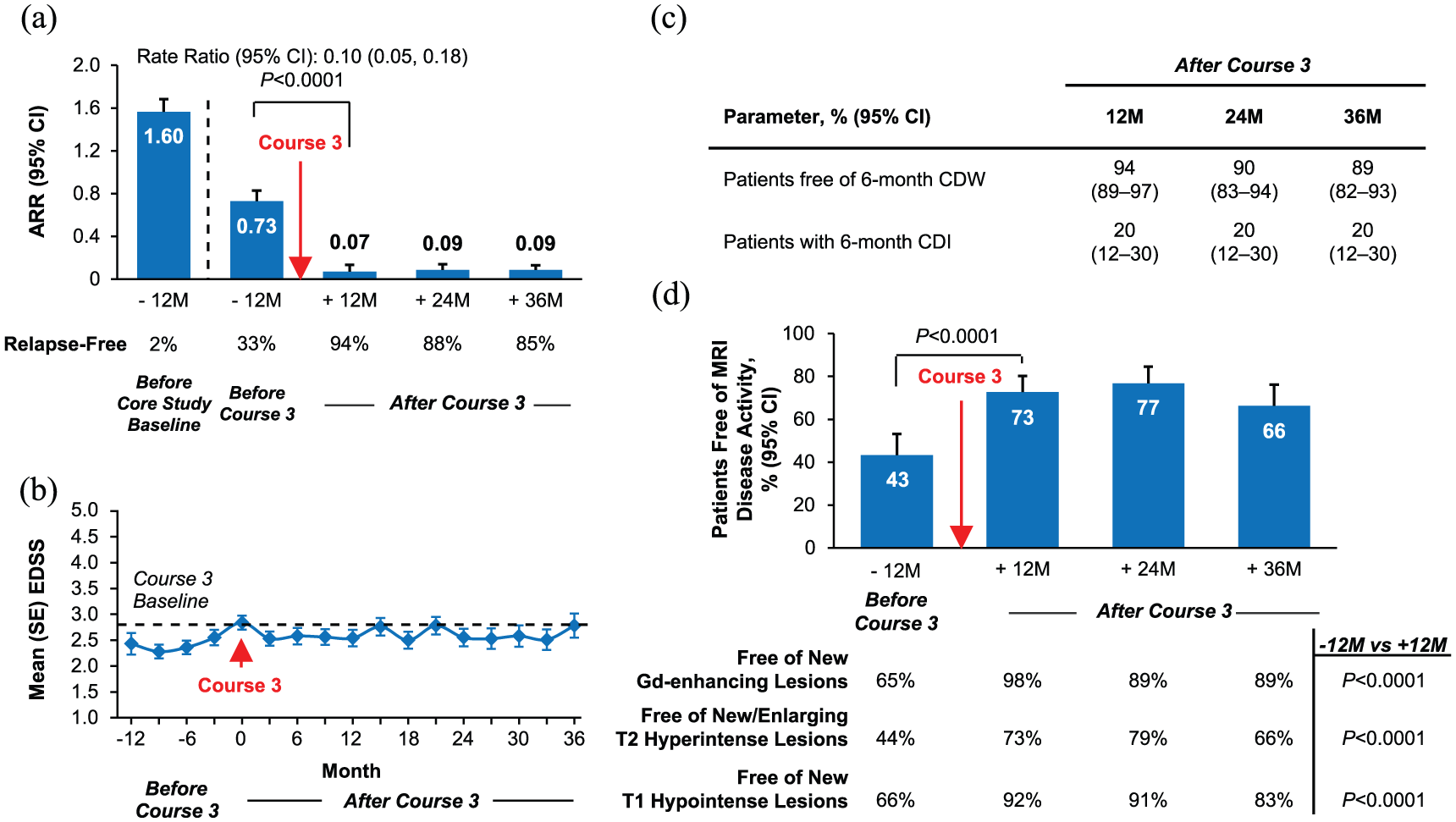
Efficacy outcomes in the 3-courses group. Results for patients who received exactly three courses within 85 months and no other DMT through Year 8. Through 24 and 36 months, respectively, 111 and 87 patients in the 3-courses group were available for follow-up after receiving Course 3. (a) ARR at 12 months before CARE-MS core study enrollment, at 12 months before Course 3, and at 12, 24, and 36 months after Course 3. (b) Mean (SE) EDSS scores from 12 months before Course 3 to 36 months after Course 3. (c) Kaplan–Meier analyses of the percentages of patients free of 6-month CDW, and percentages of patients with 6-month CDI from the time of Course 3 administration to 36 months after Course 3. (d) Percentages of patients free of MRI disease activity, new Gd-enhancing T1 lesions, new/enlarging T2 hyperintense lesions, and new T1 hypointense lesions from 12 months before Course 3 to 36 months after Course 3. p-values for ARR analyses are based on repeated negative binomial regression with robust variance estimation. p-values for MRI analyses are based on McNemar’s test. The MRI scan immediately before the start date for receiving Course 3 is considered to be “before Course 3,” and the MRI scan ⩾ 3 months after receiving Course 3 is considered to be “after Course 3.”
Efficacy of Course 4
In the 4-courses group, Course 4 significantly reduced ARR (12 months before: 0.74; 12 months after: 0.08 (p < 0.0001); 36 months after: 0.11; Figure 4(a)); patients per year who were relapse-free increased from 31% in the 12 months before Course 4 to 92%, 84%, and 79% per year in each year (Months 12, 24, and 36) post Course 4, respectively. Change in mean EDSS score from Course 4 administration to Month 36 was −0.23 (95% CI: −0.83 to +0.37; Figure 4(b)). Cumulatively through Month 36 after Course 4, 92% of patients were free of 6-month CDW and 26% achieved 6-month CDI (Figure 4(c)). Course 4 significantly increased the percentage of patients free of MRI disease activity per year from 53% in the 12 months before to 74% (p < 0.01) in the 12 months after treatment, with similar proportions free of MRI disease activity in each subsequent year (Figure 4(d)). Outcomes for patients who received ⩾5 courses are presented in Supporting Figure 2.
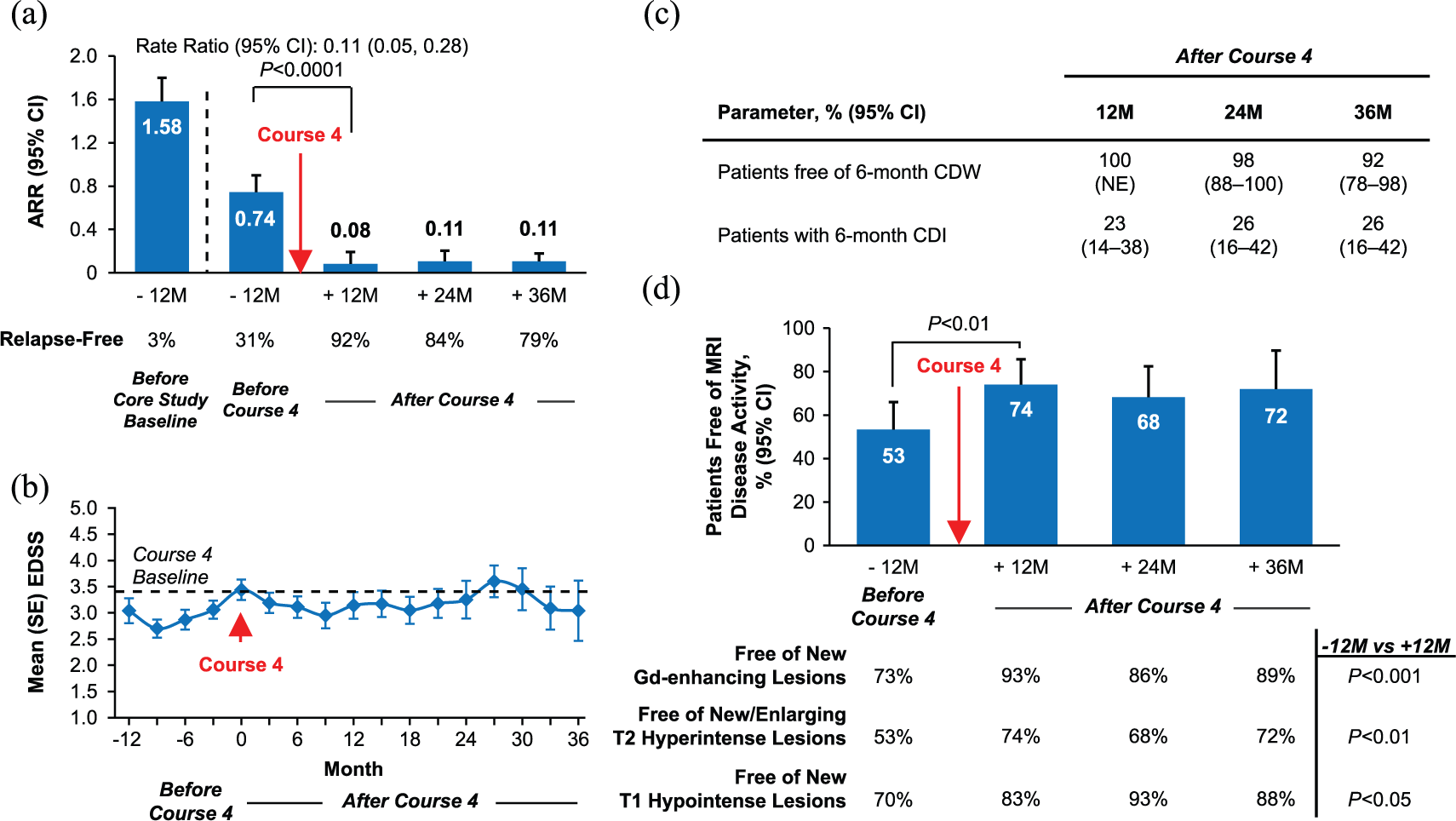
Efficacy outcomes in the 4-courses group. Results for patients who received exactly four courses within 85 months and no other DMT through Year 8. Through 24 and 36 months, respectively, 40 and 20 patients in the 4-courses group were available for follow-up after receiving Course 4. (a) ARR at 12 months before CARE-MS core study enrollment, at 12 months before Course 4, and at 12, 24, and 36 months after Course 4. (b) Mean (SE) EDSS scores from 12 months before Course 4 to 36 months after Course 4. (c) Kaplan–Meier analyses of the percentages of patients free of 6-month CDW, and percentages of patients with 6-month CDI from the time of Course 4 administration to 36 months after Course 4. (d) Percentages of patients free of MRI disease activity, new Gd-enhancing T1 lesions, new/enlarging T2 hyperintense lesions, and new T1 hypointense lesions from 12 months before Course 4 to 36 months after Course 4. p-values for ARR analyses are based on repeated negative binomial regression with robust variance estimation. p-values for MRI analyses are based on McNemar’s test. The MRI scan immediately before the start date for receiving Course 4 is considered to be “before Course 4,” and the MRI scan ⩾3 months after receiving Course 4 is considered to be “after Course 4.”
Safety
The safety profile of alemtuzumab through Year 8 in the additional-courses group was similar to those observed in the 2-courses group and the overall CARE-MS population (Table 2 and Supporting Table 4). Over 8 years, incidences were similar for the 2-courses group and the additional-courses group for infections (84.3% vs 89.1%), thyroid AEs (48.6% vs 45.4%), ITP (1.9% vs 1.0%), and nephropathy (0.3% vs 0.3%). Mean time (standard deviation) from first course of alemtuzumab to onset of first autoimmune AE (i.e. thyroid AE, ITP, or nephropathy) in the additional-courses group was 3.4 (1.9) years versus 2.5 (1.4) years in the 2-courses group (Supporting Table 5). ITP events in the additional-courses group (n = 3) had initial onset at approximately 10 months after Course 2 (Grade 3), 4 months after Course 4 (Grade 4), and 34 months after Course 3 (Grade 4); all patients recovered.1,16,17 A Grade 3 nephropathy event occurred approximately 4 months after Course 3 and is currently ongoing. 2 Five malignancy cases were reported in the additional-courses group, including Grade 2 squamous cell carcinoma approximately 19 months after Course 4, 17 Grade 2 malignant melanoma approximately 57 months after Course 3, 18 Grade 2 malignant melanoma in situ approximately 59 months after Course 3, 19 and Grade 3 papillary thyroid carcinoma approximately 10 months after Course 2 but prior to receiving additional treatment. 1 The fifth malignancy case began with Grade 2 basal cell carcinoma (first event) approximately 21 months after Course 4 followed by liver metastasis from an unknown primary site (second event) approximately 4 months after alemtuzumab Course 5; the patient died during Year 8.16,20 In addition to the malignancy fatality, six more deaths occurred in the additional-courses group; all were reported previously.6,7,18,19 In Year 7, one patient died from acute respiratory distress syndrome, sepsis, and acute renal failure approximately 46 months after Course 3 and had been treated for non-specific inflammation of the lungs within the previous year; 6 another patient died at home from unknown causes approximately 17 months after Course 3. 7 In Year 8, causes of death included septic shock (approximately 43 months after Course 5), 18 sudden death of unknown cause (approximately 14 months after Course 3), 18 atrioventricular block (approximately 26 months after Course 4), 19 and organizing pneumonia (approximately 17 months after Course 6). 19
Pooled CARE-MS I and II safety summary.
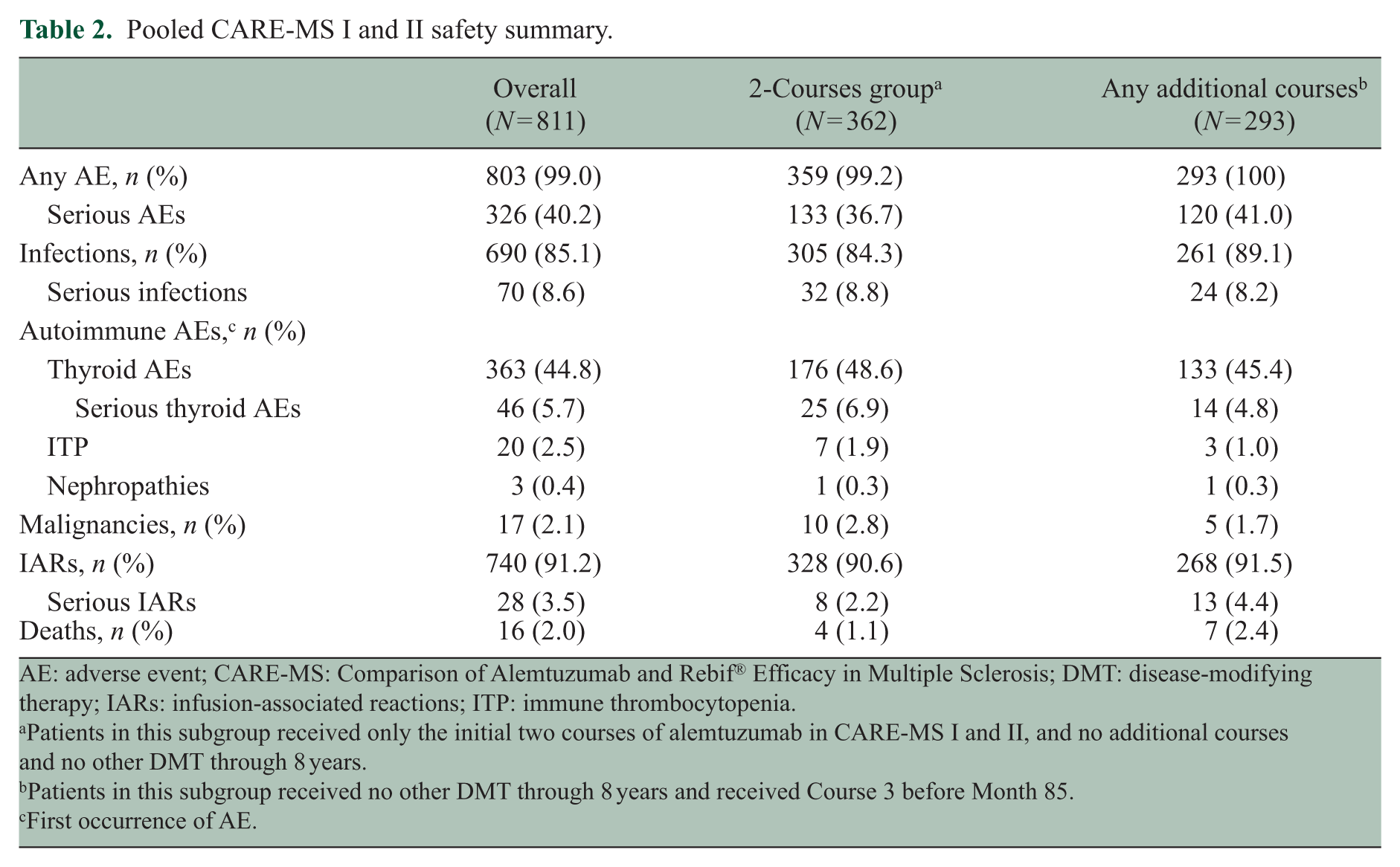
AE: adverse event; CARE-MS: Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis; DMT: disease-modifying therapy; IARs: infusion-associated reactions; ITP: immune thrombocytopenia.
Patients in this subgroup received only the initial two courses of alemtuzumab in CARE-MS I and II, and no additional courses and no other DMT through 8 years.
Patients in this subgroup received no other DMT through 8 years and received Course 3 before Month 85.
First occurrence of AE.
Discussion
RRMS presents with variable severity, including periods of both clinical latency and high activity regardless of treatment.21,22 Breakthrough disease activity has been described in RRMS patients receiving various DMTs and is not always considered a measure of treatment failure.23 –25 The present post hoc analyses of CARE-MS patients showed that additional courses of alemtuzumab effectively abated breakthrough disease activity for up to 3 years by significantly improving relapse and MRI outcomes, and stabilizing or improving disability in subsequent years. High retention rates (⩾74%) among the study population over 8 years, whether additional courses were received or not, add to the strength of the data set.
Approximately 60% of analysis patients received no more than two courses of alemtuzumab over 8 years. Notably, these patients, who had active disease at baseline, maintained low ARR, had low cumulative rates of disability worsening, and had low annual proportions of patients with MRI activity over 8 years despite receiving no additional treatment, with 53% achieving 6-month CDI. Additional courses were given to 40% of analysis patients due to breakthrough disease activity, with 6% receiving five or more courses through Year 8. The variable number of courses needed to control disease activity reflects the heterogeneity of the multiple sclerosis (MS) clinical course; two courses of alemtuzumab may be sufficient for controlling disease in many patients, whereas others may require additional courses to lower disease activity. Product labeling since 2018 provides the opportunity to tailor cumulative dosage based on an individual patient’s needs. Recent studies suggesting higher relapse activity before treatment may indicate increased likelihood of disease activity with treatment;18,26 –28 however, baseline disease activity in the 3-, 4-, and ⩾5-courses groups were similar to the 2-courses group, except for higher baseline Gd-enhancing lesion counts in the 4- and ⩾5-courses groups.
Immunomodulatory effects relating to repopulating lymphocytes following alemtuzumab treatment may underlie the need for additional courses in individual patients, but currently available data have not identified a cellular biomarker corresponding with breakthrough disease. Treatment with alemtuzumab increases suppressive function of regulatory T (Treg) cells and shifts the cytokine balance toward a less inflammatory environment lasting years.11,12 Analyses of total lymphocytes or subsets of CD4+ and CD8+ T cells (naïve, memory, Treg) and B cells (immature, mature, memory) detected no differences between CARE-MS patients with and without disease activity after alemtuzumab;29,30 however, one study found that increased interleukin-17-producing CD4+ cells may be associated with relapses in alemtuzumab-treated patients. 11 Further examination of more refined subsets of lymphocytes may allow prediction of disease activity and subsequent need for additional courses.
Alemtuzumab maintained a safety profile through Year 8 in patients who received additional courses that was consistent with both the 2-courses group and the overall CARE-MS population. There was no evidence of increased autoimmune AEs or malignancies associated with additional treatment, and rates of infections and serious infections were comparable in patients who did or did not receive additional courses. These findings suggest that in CARE-MS patients the risk of AEs does not increase with additional alemtuzumab courses. However, safety in the CARE-MS trials may not fully predict safety in real-world settings. In addition to AEs observed in clinical trials, including IARs, increased infection risk, and secondary autoimmunity (i.e. thyroid disorders, ITP, nephropathies, and autoimmune cytopenias),1,2 AEs have emerged in the postmarketing setting, including opportunistic infections, autoimmune hepatitis, other less common autoimmune events, acute acalculous cholecystitis, and cardiovascular and pulmonary events possibly related to infusion.14,15
A limitation of this analysis was the lack of a comparator group of patients who were eligible for additional alemtuzumab courses but did not receive them. Low rates of clinical and MRI disease activity were observed in the 2-courses group, suggesting that many patients did not meet disease activity criteria for additional courses. Reasons for not administering additional courses in those who did have disease activity were not captured in the clinical studies, but likely included meeting exclusion criteria for additional treatment or assessment of disease activity as mild or not warranting further treatment. Eligibility in patients not receiving additional courses was therefore not assessable, precluding analysis of patients who were eligible for additional treatment but did not receive it. In addition, some patients who received additional courses were not included in the current analysis due to insufficient follow-up time (<12 months) to assess efficacy and safety after the last course. Patients who were treated with Courses 3 or 4 after the inclusion criteria cut-off of Month 85 exhibited prolonged reduction in breakthrough disease activity following Course 2; however, their outcomes are not reflected in these results. Furthermore, these results do not reflect outcomes in patients who received another DMT (9%) for presumed disease activity either in lieu of additional alemtuzumab or between alemtuzumab courses.
Conclusion
This post hoc analysis indicates that up to two additional alemtuzumab courses help mitigate clinical and MRI disease activity in RRMS patients with breakthrough disease activity after the initial two-course regimen, with a safety profile consistent with the overall CARE-MS population and those who received two courses over 8 years.
Supplemental Material
MSJ888610_supplementary_material – Supplemental material for Efficacy of alemtuzumab in relapsing-remitting MS patients who received additional courses after the initial two courses: Pooled analysis of the CARE-MS, extension, and TOPAZ studies
Supplemental material, MSJ888610_supplementary_material for Efficacy of alemtuzumab in relapsing-remitting MS patients who received additional courses after the initial two courses: Pooled analysis of the CARE-MS, extension, and TOPAZ studies by Giancarlo Comi, Raed Alroughani, Aaron L Boster, Ann D Bass, Regina Berkovich, Óscar Fernández, Ho Jin Kim, Volker Limmroth, Jan Lycke, Richard AL Macdonell, Basil Sharrack, Barry A Singer, Patrick Vermersch, Heinz Wiendl, Tjalf Ziemssen, Alan Jacobs, Nadia Daizadeh, Claudio E Rodriguez and Anthony Traboulsee in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors and Sanofi would like to thank the patients for their participation in the CARE-MS I, CARE-MS II, CAMMS03409, and TOPAZ studies, as well as the Steering Committees and the investigators. Critical review of the manuscript was provided by Darren P. Baker, PhD, Ericka M. Bueno, PhD, and Colin Mitchell, PhD, of Sanofi. Editorial and writing assistance was provided by Rebecca L. Orndorff, PhD, and Valerie P. Zediak, PhD, of Eloquent Scientific Solutions and was funded by Sanofi. ClinicalTrials.gov Registration Numbers: CARE-MS I, II, extension, TOPAZ: NCT00530348, NCT00548405, NCT00930553, NCT02255656.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.C. reports receiving consulting fees from Actelion, Bayer, Merck Serono, Novartis, Sanofi, and Teva; lecture fees from Bayer, Biogen Dompé, Merck Serono, Novartis, Sanofi, Serono Symposia International Foundation, and Teva. R.A. reports receiving speaker honoraria and research grants from and serving on scientific advisory boards for Bayer, Biogen, GSK, Lundbeck, Merck Serono, Novartis, Roche, and Sanofi. A.L.B. reports receiving consulting fees and/or speaking fees for non-CME services from Biogen, Mallinckrodt, Medtronic, Novartis, Sanofi, and Teva. A.D.B. reports receiving research funding, compensation for medical advisory boards, and fees from speaker’s bureaus for Actelion, Biogen, EMD Serono, Genentech-Roche, Mallinckrodt, Novartis, Sanofi, and TG Therapeutics. R.B. reports participating in advisory boards and receiving consulting fees from Acorda, Avanir, Bayer, Biogen, Novartis, Questcor, Sanofi, and Teva. Ó.F. reports receiving speaking and/or consulting fees from Allergan, Almirall, Bayer-Schering, Biogen, Merck Serono, Novartis, Sanofi, and Teva; compensation for serving as a journal editor, associate editor, or member of an editorial advisory board for Revista Española de Esclerosis Múltiple; and research support from the Hospital Foundation FIMABIS. H.J.K. reports receiving consulting and/or speaking fees from Bayer, Biogen, Celltrion, Eisai, Genzyme, HanAll BioPharma, MedImmune, Merck Serono, Novartis, Teva-Handok, and UCB, and research support from Genzyme, Merck Serono, the Ministry of Science & ICT of the Republic of Korea, Teva-Handok, and UCB; he also reports being a member of a steering committee for MedImmune, being co-editor for Multiple Sclerosis Journal—Experimental, Translational, and Clinical, and being an associate editor for the Journal of Clinical Neurology. V.L. reports receiving honoraria for consulting and speaking at symposia for Bayer, Biogen, Merck Serono, Novartis, Roche, Sanofi, and Teva, with approval by the HR Department of Cologne General Hospital and the University of Cologne. J.L. reports receiving travel support and/or lecture honoraria from Biogen, Merck, Novartis, Sanofi, and Teva; he also reports serving on advisory boards for Almirall, Biogen, Merck, Novartis, Sanofi, and Teva, serving on the editorial board for Acta Neurologica Scandinavica, and receiving unconditional research support from Biogen, Novartis, and Teva. R.A.L.M. reports receiving compensation for advisory boards and/or speaking fees from Bayer, Biogen, Merck, Novartis, Roche, Sanofi, and Teva, and receiving research support from Biogen, Merck, Novartis, Sanofi, and Teva. B.S. reports research and travel grants, honoraria for expert advice on MS, and speaking fees from Biogen, Merck, Novartis, Roche, Sanofi, and Teva. B.A.S. reports receiving speaking and/or consulting fees from AbbVie, Acorda, Alexion, Biogen, EMD Serono, Genentech, Novartis, Roche, Sanofi, Teva, and TG Therapeutics, and receiving research support from AbbVie, Alkermes, Biogen, MedImmune, Novartis, Roche, and Sanofi. P.V. reports receiving consulting and/or speaking fees, and research support, from Almirall, Biogen, Celgene, Merck, Novartis, Roche, Sanofi, Servier, and Teva. H.W. reports receiving consulting and/or speaking fees from Bayer, Behring, Biogen, EMD Serono, Fresenius Medical Care, Merck Serono, Novartis, Roche, Sanofi, and Teva; he also reports receiving license fee payments from Huber-Verlag, and grant/research support from Neotope Bioscience, Novartis, and PML Consortium. T.Z. reports receiving consulting and/or speaking fees from Almirall, Bayer, Biogen, Merck, Novartis, Roche, Sanofi, and Teva, along with grant and research support from Biogen, Novartis, Sanofi, and Teva. A.J. and N.D. report receiving personal compensation as employees of Sanofi. C.E.R. reports personal compensation as an employee of Sanofi at the time of the analysis and is currently affiliated with Sunovion Pharmaceuticals, Marlborough, MA, USA. A.T. reports receiving consulting and/or speaking fees, and grant/research support from Biogen, Chugai, Roche, Sanofi, and Teva.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial support for the development of this paper was funded by Sanofi. The CARE-MS, CAMMS03409, and TOPAZ studies were funded by Sanofi and Bayer HealthCare Pharmaceuticals.
Supplemental material
Supplemental material for this article is available online.
Data Sharing
Qualified researchers may request access to patient level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and dataset specifications. Patient level data will be anonymized and study documents will be redacted to protect the privacy of trial participants. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at: ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
