Abstract
Background:
Meningeal inflammation may contribute to gray matter (GM) involvement in multiple sclerosis (MS) and is proposed to manifest as magnetic resonance imaging (MRI) leptomeningeal enhancement (LME).
Objective:
To investigate how LME relates to GM lesions in relapsing-remitting multiple sclerosis (RRMS) at 7T.
Methods:
A total of 30 RRMS subjects (age (mean ± standard deviation (SD)): 44.0 ± 11.3 years, 93% on disease-modifying treatment) and 15 controls underwent gadolinium-enhanced three-dimensional (3D) MP2RAGE (magnetization-prepared 2 rapid gradient-echo) and fluid-attenuated inversion recovery (FLAIR) MRI. LME, cortical lesions (CLs), thalamic lesions (TLs), and white matter (WM) lesions were expert-quantified. Wilcoxon rank-sum, two-sample t-tests, Spearman correlations, and regression models were employed.
Results:
Two-thirds (20/30) of MS subjects and 1/15 controls (6.7%) had LME. LME+ MS subjects had 2.7 ± 1.5 foci, longer disease duration (14.9 ± 10.4 vs. 8.1 ± 5.7 years, p = 0.028), increased CL number (21.5 ± 12.6 vs. 5.5 ± 5.0, p < 0.001) and volume (0.80 ± 1.13 vs. 0.13 ± 0.13 mL, p = 0.002), and increased TL number (3.95 ± 2.11 vs. 0.70 ± 1.34, p < 0.001) and volume (0.106 ± 0.09 vs. 0.007 ± 0.01 mL, p < 0.001) versus LME– subjects. LME focus number correlated more highly with CL (rs = 0.50, p = 0.01) and TL (rs = 0.81, p < 0.001) than WM lesion (rs = 0.34, p > 0.05) volume. Similar LME–CL number associations were observed in unadjusted and WM lesion–adjusted comparisons (both p < 0.001).
Conclusion:
Cerebral LME is common in RRMS at 7T and is independently associated with GM injury. We hypothesize that cerebrospinal fluid (CSF)-related inflammation links cortical and thalamic injury.
Introduction
Multiple sclerosis (MS) is traditionally characterized by white matter (WM) inflammatory demyelinating lesions, presenting most commonly with a relapsing-remitting (RR) course, with risk for transition to a secondary progressive (SP) phase where disability accrues in the absence of relapses. 1 Recent histopathologic and imaging studies show a more complex pathobiology, with cortical demyelination and atrophy of the cortical and deep gray matter (GM), and meningeal inflammation appearing early in the disease course and accelerating in progressive stages.2–17 Histopathologic studies have linked cortical pathology to meningeal inflammation and the presence of lymphoid follicles at the cortical leptomeningeal surface.15–19 A proposed magnetic resonance imaging (MRI) marker of meningeal inflammation is leptomeningeal enhancement (LME), which can be detected by gadolinium-enhanced high-resolution fluid-attenuated inversion recovery (FLAIR) MRI.8,20–29 LME is found in a variety of central nervous system (CNS) infectious and inflammatory illnesses, though it is more common in inflammatory than non-inflammatory CNS conditions. 23
Prior 3T MRI MS studies have shown that LME has a prevalence of 20%–50%, is found more commonly in progressive MS,22,25–27 and is linked to physical disability22,25 and whole-brain and cortical atrophy.22,25,26 However, LME prevalence at 7T in MS subjects is higher, that is, ~90%. 8 Unlike conventional MRI-defined parenchymal WM-enhancing lesions, LME foci commonly persist for months to years,22,29 particularly in patients with progressive disability. 29 The main goal of our study was to use 7T MRI to investigate the relationship between LME and GM (cortical and thalamic) lesions in relapsing-remitting multiple sclerosis (RRMS).
Methods
Subjects
A total of 30 patients with RRMS 30 (see Table 1 for characteristics) and 15 healthy controls underwent MRI, with the following inclusion criteria: (1) age 18–60, (2) no contraindications to MRI, (3) no severe acute or chronic medical comorbidities, and (4) no clinical relapses or corticosteroids in the month preceding MRI. An MS-specialist neurologist determined Expanded Disability Status Scale (EDSS) scoring and timed 25-foot walk (T25FW). Subjects with progressive MS were excluded to minimize cohort heterogeneity given the small sample size. Controls were selected to comprise similar age and sex characteristics versus the MS subject cohort. The Partners Human Research Committee approved the study; all subjects gave informed consent.
Cohort demographics, MRI, and clinical characteristics.
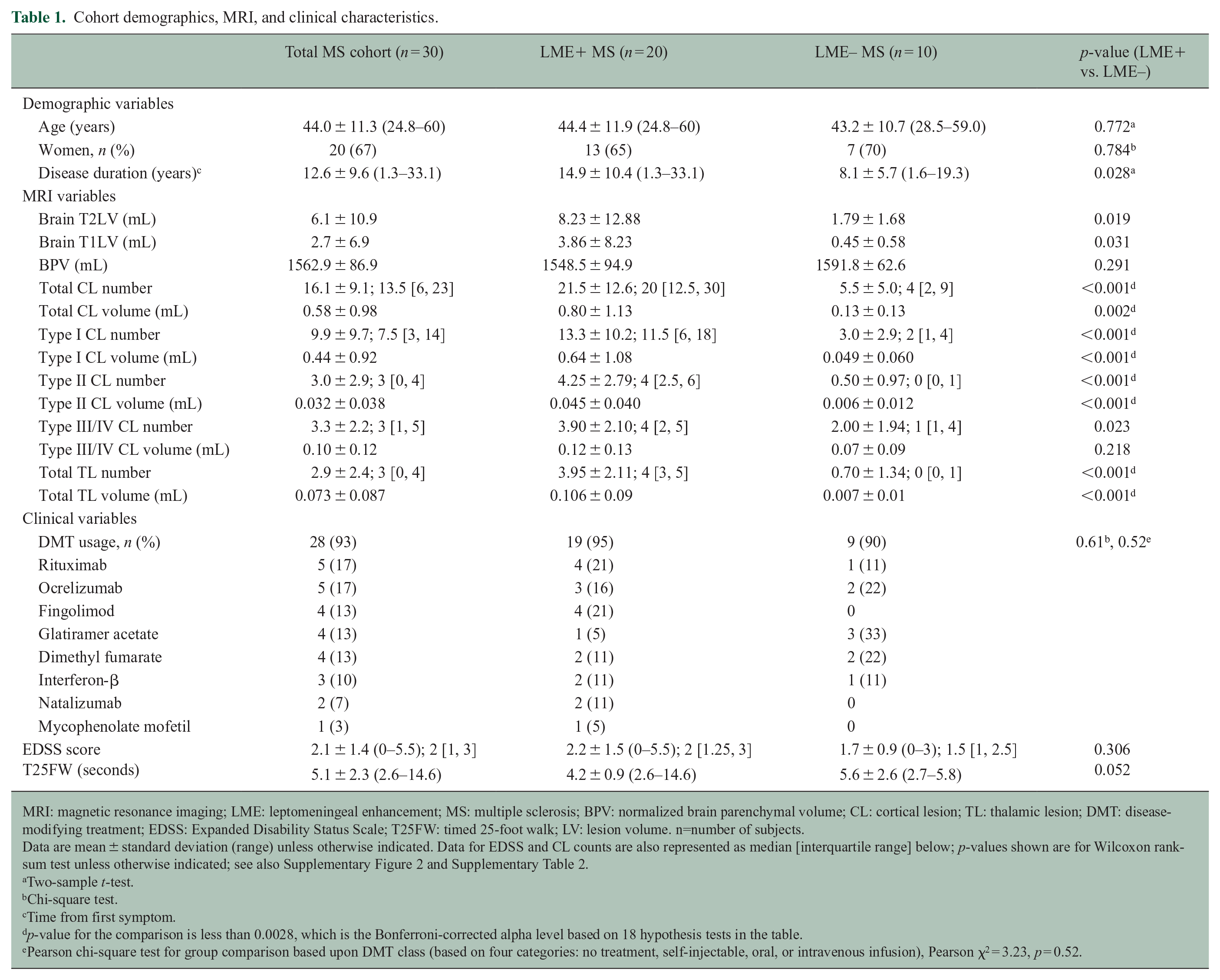
MRI: magnetic resonance imaging; LME: leptomeningeal enhancement; MS: multiple sclerosis; BPV: normalized brain parenchymal volume; CL: cortical lesion; TL: thalamic lesion; DMT: disease-modifying treatment; EDSS: Expanded Disability Status Scale; T25FW: timed 25-foot walk; LV: lesion volume. n=number of subjects.
Data are mean ± standard deviation (range) unless otherwise indicated. Data for EDSS and CL counts are also represented as median [interquartile range] below; p-values shown are for Wilcoxon rank-sum test unless otherwise indicated; see also Supplementary Figure 2 and Supplementary Table 2.
Two-sample t-test.
Chi-square test.
Time from first symptom.
p-value for the comparison is less than 0.0028, which is the Bonferroni-corrected alpha level based on 18 hypothesis tests in the table.
Pearson chi-square test for group comparison based upon DMT class (based on four categories: no treatment, self-injectable, oral, or intravenous infusion), Pearson χ2 = 3.23, p = 0.52.
MRI acquisition
Brain 7T MRI employed a MAGNETOM Terra scanner (Siemens, Erlangen, Germany) with a single-transmit/32-channel receive head coil and uniform scan protocol, covering the whole brain with voxel sizes: 0.7 × 0.7 × 0.7 mm3 including T1-weighted (T1w) magnetization-prepared 2 rapid gradient-echo (MP2RAGE) and T2-weighted FLAIR. Acquisition parameters are shown in Supplementary Table 1. Post-contrast imaging was performed after intravenous (IV) 0.1 mmol/kg of gadoterate meglumine (Dotarem®) which commenced with repeating both sequences (equating to a ~10-minute delay between contrast administration and FLAIR initiation).
MRI analysis
LME detection was performed by a neurologist with experience in neuroimaging (J.Z.), supervised by a senior neurologist-neuroimager (R.B.) by manual comparison of pre- and post-contrast sagittal three-dimensional (3D) FLAIR. Scans were evaluated for hyperintense signal on post-contrast FLAIR with no signal on pre-contrast FLAIR. All suspected LME foci were confirmed on resampled axial or coronal planes. Enhancement of vessel wall structures with pre-contrast FLAIR hyperintensity and dura non-contiguous with the cortical surface was excluded. Two variants of LME 8 were quantified: (1) “nodular”—discrete, pial, or subarachnoid spherical foci and (2) “spread/fill”—larger enhancing areas extending through the subarachnoid space.
Cortical lesions (CLs) were identified based on previous 7T methods,13,31 required to be hypointense on T1w-MP2RAGE versus adjacent normal-appearing cortex, at least 1 mm wide, and distinct from blood vessels. The CL rater was blinded to LME status. The corresponding FLAIR hyperintensity frequently provided confirmation, though it was not a strict requirement (as FLAIR cortical ribbon artifact can obscure lesions). Coronal and axial T1w-MP2RAGE and FLAIR resampling helped verify lesions. CL subtypes were classified as type I (leukocortical, involving both GM and WM), type II (purely intracortical), and type III/IV (subpial: diffuse demyelination extending inward from the pial surface).1,2,32–34 After the blinded LME and CL analysis was complete, post-contrast FLAIR and MP2RAGE images were assessed for LME–CL co-localization (defined by the presence of LME anywhere along the pial surface adjacent to the CL).
Axial T1w-MP2RAGE and FLAIR resampled sequences were used for the detection of thalamic lesions (TLs) and lesions were required to be both FLAIR hyperintense and T1w-MP2RAGE hypointense. TLs were traced on the axial resampled FLAIR pre-contrast sequence.
Whole-brain WM T2 hyperintense (T2LV) and T1 hypointense lesion (T1LV) volumes were quantified using expert identification on the pre-contrast sagittal FLAIR (T2LV) and T1w-MP2RAGE (T1LV) sequences. Normalized brain parenchymal volume (BPV) was obtained using a fully automated segmentation pipeline (SIENAX v5.0, https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/SIENA) and segmentation maps were expert-reviewed (R.C.) to ensure quality. The analysis of CLs, TLs, and WM lesions was performed with a semi-automated edge-finding tool in the Jim 7.0 software (Xinapse Systems Ltd, West Bergholt, UK; http://www.xinapse.com) and was blinded to clinical variables and LME status (operators had no access to the post-contrast images). Intra-rater reliability analyses for LME status, LME focus number, and CL number were performed, with intraclass coefficients of 1.0, 0.970, and 0.995, respectively, from five cases chosen to have a wide range of involvement.
Statistical analysis
Clinical characteristics were compared between groups using a two-sample t-test or one-way analysis of variance for continuous measures and a chi-square test for categorical measures. MRI was compared using a Wilcoxon rank-sum test for two-group comparisons. Spearman correlation coefficients assessed the relationship between lesion numbers and other variables. In addition, we compared the correlation coefficient between LME lesion number and T1 lesion volume/T2 lesion volume with the correlation coefficients between LME lesion number and CL/TL number and volume using Choi’s test. A negative binomial regression model was used to assess the association between LME positivity and the CL number adjusting for T2 lesion volume. A p < 0.05 was considered statistically significant. This was an exploratory study due to the limited data available from 7T MRI on the detection of LME in RRMS. As such, for the primary interpretation of the data, we did not correct for multiple comparisons. To guide hypothesis testing for future studies, we also provide a Bonferroni-corrected alpha level for each set of analysis.
Results
Demographics
MS subjects were similar in age and sex distribution to controls (mean age: 44.0 ± 11.3 vs. 44.1 ± 10.0 years; 67% women for both groups). The majority of MS subjects were receiving disease-modifying treatment (n = 28/30; 93%) at the time of MRI, with anti-B-cell therapy (rituximab and ocrelizumab) mostly commonly used (n = 10/30; 33%) (Table 1). We performed a statistical analysis to assess the relationship between anti-CD20 therapy use and LME positivity and found no association (70% LME positivity among anti-CD20-therapy-treated subjects and 65% LME+ among patients not receiving such therapy; p = 0.78, chi-square test). Furthermore, there were no differences in the CL number (p = 0.83) or volume (p = 0.79) between these treatment groups.
LME: prevalence, morphology, and anatomic location
LME examples are shown in Figure 1 and Supple-mentary Figure 1. LME was found in 20/30 (66.7%) RRMS subjects and in 1/15 (6.7%) healthy controls. A total of 54 LME foci were identified in 20 RRMS subjects—a mean of 2.7 ± 1.5 (range 1–6) foci per subject, most commonly 2 foci per subject (n = 7). All 20 LME+ MS subjects had at least 1 nodular LME focus (Figure 1). Spread LME morphology (Figure 1 and Supplementary Table 2) was found in 8/20 (40%) MS subjects. LME focus distribution was as follows: n = 23 (43%) left hemisphere, n = 31 (57%) right hemisphere, n = 28 (52%) frontal, n = 17 (31%) parietal, n = 7 (13%) temporal, and n = 2 (4%) occipital. A single left frontal LME focus was identified in a 51-year-old control (Supplementary Figure 1).
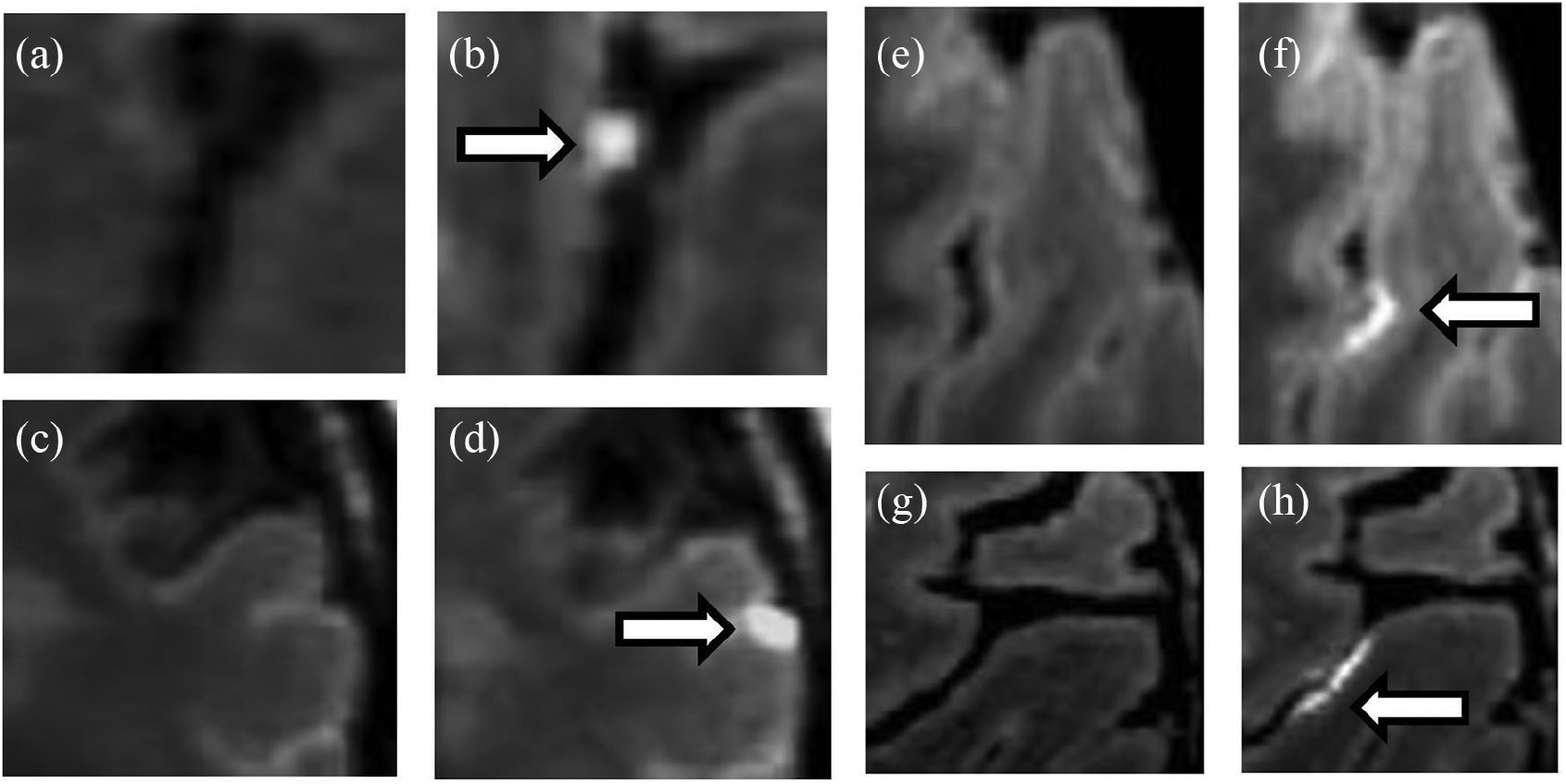
Detection of LME using 7T MRI in relapsing-remitting multiple sclerosis. Examples of LME in RRMS subjects as detected by 3D FLAIR sagittal sequences at 7T MRI. Pre-contrast (a, c, e, g) and post-contrast (b, d, f, h) images showing representative foci of LME arrows. Characteristic nodular LME foci (b, d; arrows) found within a sulcus (b) and at the pial surface (d). Spread/fill LME focus examples (f, h), with contrast layering within the sulcal depth (arrows) (see also Supplementary Figure 1).
LME: relationship to demographic and treatment variables in the MS group
Table 1 shows the comparison of MS subjects who had at least 1 focus of LME (LME+) versus those who were free of any foci (LME–). The mean age and sex were similar; however, the LME+ group had a longer disease duration. Distribution of disease-modifying treatment (self-injected, oral, monoclonal antibody, or other) was not different between groups (Pearson χ2 = 3.23, p = 0.52).
LME is associated with CL burden in the MS group
Table 1 shows the comparison of CLs between LME+ versus LME– MS subjects. Figure 2 and Supplementary Figure 2 also show the link between LME and CLs. In total, 484 CLs were detected in the MS subjects (mean: 16.1 lesions per subject), distributed as follows: type I (leukocortical), n = 296 (61% of CLs, mean: 9.8 lesions per subject); type II (intracortical), n = 90 (19%, mean: 3.0 lesions); and type III/IV (subpial), n = 98 (20%, mean: 3.3 lesions).
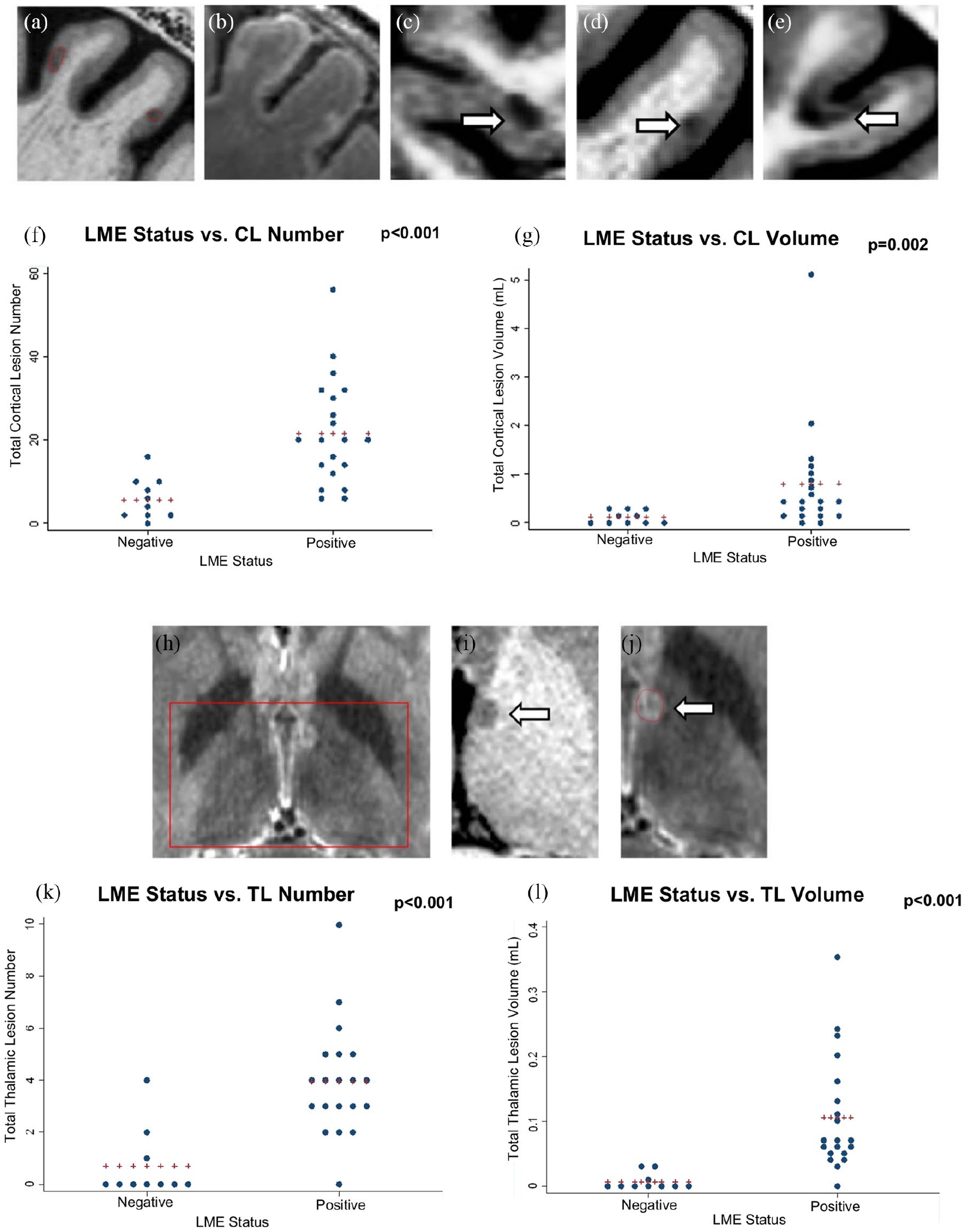
LME is associated with cortical and TL burden in multiple sclerosis. Upper panel (a–g): (1) CLs: (a–e) example CLs in RRMS subjects detected by 7T MRI (pre-contrast sagittal 3D images shown); (a, b) two small intracortical type II CLs evident on pre-contrast sagittal 3D MP2RAGE (a; red tracing) with the corresponding hyperintensity on 3D FLAIR confirming the presence of the CL (b); (c–e) examples of CL subtypes (arrows) detected on pre-contrast sagittal MP2RAGE sequence are shown: (c) type I—leukocortical, (d) type II—intracortical, and (e) type III/IV—subpial. (2) LME status versus CL burden: (f, g) cluster plots display CL burden versus LME status for (f) total CL number and (g) total CL volume. LME+ status is associated with increased CL burden for each analysis (red “+” bar indicates the mean value for the group; dots indicate individual patients; p < 0.05 for both analyses; see also Table 1). Lower panel (h–l): (1) TLs: (h–j) example TL in a 48-year-old woman with RRMS detected by 7T MRI (resampled 3D images shown); (h) axial FLAIR showing the bilateral hemisphere deep GM, with box (red) indicating the region of the bilateral thalami; note the hyperintense focus within the left TL; (i, j) the same region, magnified, shows (i) hypointensity on the axial MP2RAGE image (arrow) and (j) hyperintensity on the axial FLAIR image indicating a small ovoid TL (arrow; red tracing). (2) LME status versus TL burden: (k, l) cluster plots display TL burden versus LME status for (k) total TL number and (l) total TL volume. LME+ status is associated with an increased TL burden for both analyses (red “+” bar indicates the mean value for the group; dots indicate individual patients; p < 0.05 for both analyses; see also Table 1).
LME+ subjects had, on average, a fourfold increased CL number (21.5 ± 12.6 vs. 5.5 ± 5.0, p < 0.001) and a sixfold increased volume (0.80 ± 1.13 vs. 0.13 ± 0.13 mL, p = 0.002) versus LME– subjects (Figure 2 and Table 1), evident across all CL subtypes: type I (13.3 ± 10.2 vs. 3.0 ± 2.9 lesions, p < 0.001), type II (4.25 ± 2.79 vs. 0.50 ± 0.97, p < 0.001), and type III/IV (3.90 ± 2.10 vs. 2.00 ± 1.94, p = 0.023) (Supplementary Figure 2).
An LME–CL linkage was also suggested by correlation analyses (Table 2), showing a moderate to strong association between the LME number and both total CL number (rs = 0.59, p = 0.001) and volume (rs = 0.50, p = 0.01), the former surviving a Bonferroni correction. Topographical LME–CL co-localization (LME adjacent to a CL) was only found in 4 of 54 LME foci (7.4%) with no relationship to the CL subtype. LME correlations with brain T2LV (rs = 0.34) and T1LV (rs = 0.31) were non-significant (both p > 0.05; Table 2).
Clinical and MRI correlation with LME focus number and TL number.
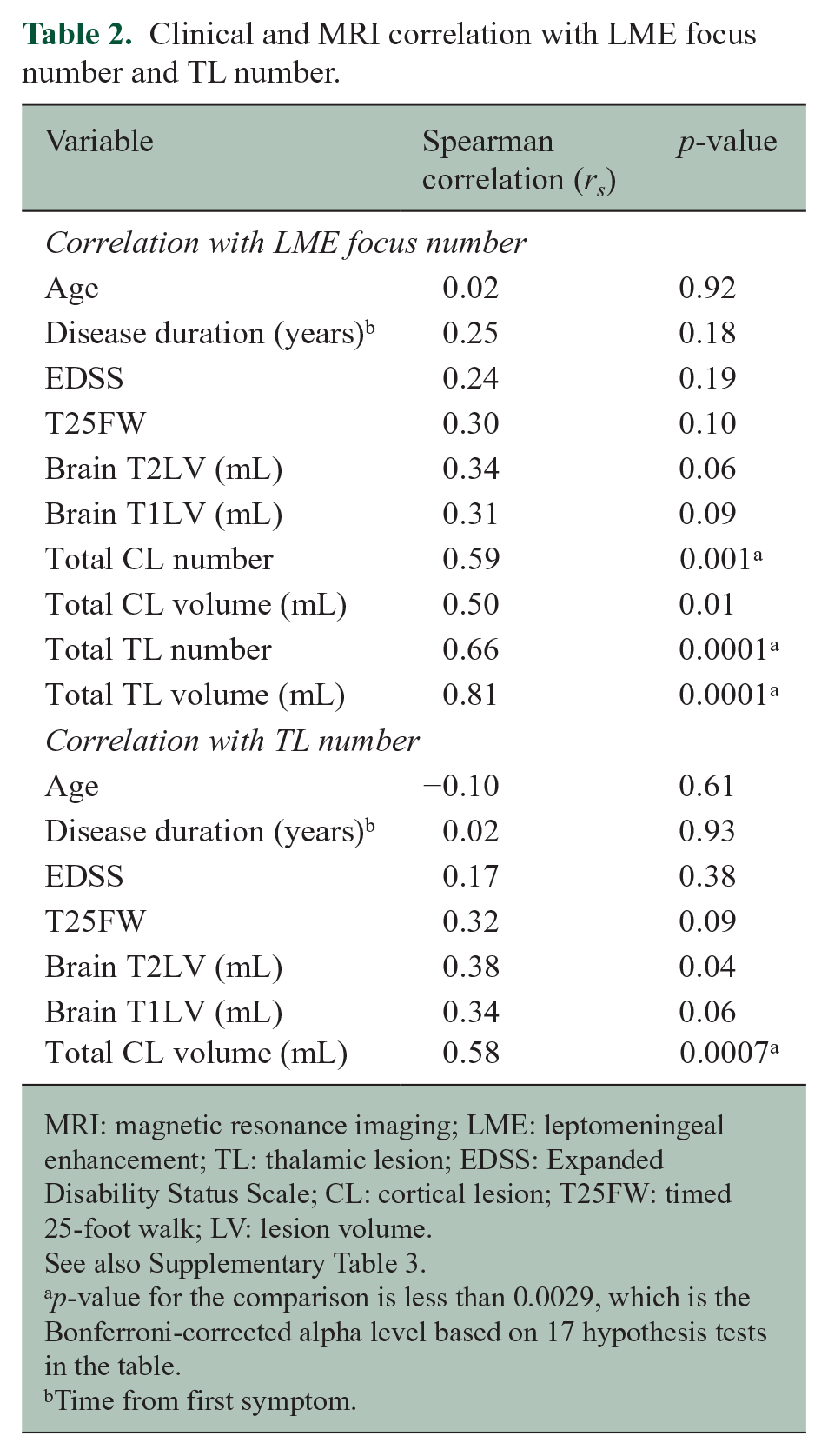
MRI: magnetic resonance imaging; LME: leptomeningeal enhancement; TL: thalamic lesion; EDSS: Expanded Disability Status Scale; CL: cortical lesion; T25FW: timed 25-foot walk; LV: lesion volume.
See also Supplementary Table 3.
p-value for the comparison is less than 0.0029, which is the Bonferroni-corrected alpha level based on 17 hypothesis tests in the table.
Time from first symptom.
LME is associated with TL burden in the MS group
Examples of TLs are shown in Figure 2. Table 1, Figure 2, and Supplementary Table 3 show a relationship between TLs and both LME and CLs. TLs were detected in 73% (n = 22) of MS subjects. Those who were LME+ had, on average, a 5-fold increased TL number (3.95 ± 2.11 vs. 0.70 ± 1.34, p < 0.001) and a 15-fold increased total TL volume (0.106 ± 0.09 vs. 0.007 ± 0.01 mL, p < 0.001) compared to LME– MS subjects.
The LME focus number showed a moderate to strong correlation with the TL number (rs = 0.66, p = 0.0001) and volume (rs = 0.81, p = 0.0001) (Table 2). The strength of LME–TL correlations was similar to that observed between LME and CLs (Table 2) and higher than that between the LME number and WM lesion volume. Additional analyses showed a link between CLs and TLs (Supplementary Table 3): The TL number showed higher correlations with the total CL volume (rs = 0.68, p < 0.0001) than with either T2 (rs = 0.49, p = 0.01) or T1 WM lesion volume (rs = 0.48, p = 0.01). Furthermore, using Choi’s test, we found that the LME focus number correlation with the TL volume was stronger than the correlation with either T1LV (p = 0.008) or T2LV (p = 0.01).
Comparison of LME groups based on morphology
Figure 1 shows examples of the two morphological variants of LME—“nodular” and “spread/fill.” Supplementary Table 2 shows the comparison of patients with any LME “spread” morphology (n = 8) to those having only “nodular” morphology (n = 12), showing no age, sex, or disease duration differences. Two-group comparisons between “spread” LME+ and “nodular only” showed that the total CL volume was significantly higher (p = 0.045) in the “spread” LME group, while differences in other MRI metrics (TL, T1, and T2 lesion volumes) were not significant (Supplementary Table 2).
LME: relationship to conventional MRI and clinical measures of disease severity in the MS group
Table 1 shows the comparison of conventional MRI brain WM lesion characteristics of LME+ versus LME– MS subjects. LME+ subjects had increased T2LV (p = 0.019) and T1LV (p = 0.031) versus LME– subjects. However, these did not survive Bonferroni correction and the relationship between LME and WM lesions was inconsistent across analyses (the LME focus number did not show any significant correlation with T1LV or T2LV; Table 2). Negative binomial regression assessed the effect of T2LV on the LME–CL association. There was only a minimal difference between unadjusted and WM-lesion-adjusted estimated rate ratios regarding the association between LME status and the CL number (3.9 (95% confidence interval (CI): 2.3, 6.6; p < 0.001) versus 3.1 (95% CI: 1.9, 4.9; p < 0.001)). Differences in BPV between LME+ MS subjects (1548.5 ± 94.9 mL), LME– MS subjects (1591.8 ± 62.6 mL), and healthy controls (1587.9 ± 34.4 mL) did not reach significance (three-group comparison, p = 0.20). Comparing LME+ to LME– MS subjects, physical disability (EDSS score, T25FW) was not significantly different (p > 0.05 for both comparisons) (Table 1). Similarly, no association was found between LME morphology and disability or BPV (Supplementary Table 2).
Intraparenchymal contrast enhancement
None of the subjects showed gadolinium enhancement of CLs, TLs, or WM lesions.
Discussion
We investigated the relationship between 7T cerebral LME and GM (cortical and thalamic) lesions in RRMS. We showed that LME was common in RRMS versus healthy controls and was linked to increased CL and TL burden. The association between LME and CLs was broadly distributed across CL subtypes and was independent of WM lesions. The link between LME and GM lesions was consistent throughout our analysis, whether assessing LME+ versus LME– patients or in correlation analysis using the number of LME foci. Moreover, the data suggest that cerebral LME was more strongly associated with cortical/thalamic GM than with WM lesion burden, both in group comparisons of LME+ versus LME– subjects and in correlation analyses comparing the LME focus number to lesion burden; however, this was not an a priori hypothesis.
We found LME in a majority of RRMS subjects (67%, n = 20/30), at a rate significantly higher than reported in RRMS subjects at 3T (ranging from 19% (n = 35/181) 22 to 45% (n = 14/31)). 26 One prior study of LME at 7T 8 showed an even higher prevalence in RRMS (~85%) (Harrison, personal communication, 2019). The fact that our study did not observe such high LME prevalence may be reflective of differences in demographics (our cohort had fewer male subjects, no whole-brain atrophy, longer disease duration, and a higher percentage of patients on immunotherapy) and/or MRI technique/analysis (manual vs. subtraction comparison of post- and pre-contrast images; FLAIR vs. magnetization-prepared, fluid-attenuated inversion recovery (MPFLAIR) acquisition, and scanner hardware).
We demonstrated an association between LME and CL burden in RRMS. Histological data indicate that cortical demyelination may be a feature of early-stage MS (prevalence ~37% in early RRMS) 11 and it increases in progressive disease, where the extent of meningeal inflammation correlates with the extent of cortical demyelination.9,12,17,19 An association between LME and GM/cortical atrophy has been noted by several groups8,22,25,26 with topographical associations between 3T LME, meningeal inflammation, and subpial CLs noted in one postmortem case. 22 We did not find a 1:1 LME–CL relationship and instead found that LME+ status is a marker of increased CL burden broadly. Although there is an improved detection of CLs at 7T versus 3T, 33 type I (leukocortical) lesions are the most common subtype visualized,8,35 while type II (intracortical) and type III/IV (subpial lesions), which are the most common CLs found at autopsy,2,32,33 remain elusive at 7T. Thus, even at 7T, there are technical limitations to the CL assessment, which may have influenced our ability to detect a topographic CL-LME co-occurrence.
Our results indicated that “spread” LME morphology was rarer, seen in 40% of subjects (n = 8/20), versus “nodular” LME, which was evident in all LME+ subjects. By contrast, a prior 7T study that included patients with progressive forms of MS found spread/fill LME in 76% (n = 22/29) and nodular in 51% (n = 12/29) of cases. 8 This may be due to differences in cohort and/or LME detection. In our study, three-group comparisons between “spread” LME, “nodular”-only LME, and LME– subjects showed significant differences in the CL number and volume (with a trend toward higher CL burden in “spread” LME patients). However, the study was not powered to assess the relationship between LME morphology subgroups and MRI disease severity. In prior work, “spread” LME has been linked to reduced cortical GM volume 8 and risk for EDSS progression in the next year, 29 though relevance in RRMS is unclear, so longitudinal study of LME morphology variants is needed.
Our data indicate a potential novel link between LME and TLs in RRMS patients. Prior histopathological studies have shown that demyelination and atrophy of the cerebral deep GM are clinically relevant elements of MS pathology, may occur early in the disease course, and accelerate in progressive stages.5,36–39 7T MRI affords new capability to characterize TLs, which are poorly visualized at 3T (data not shown). TLs are evident in ~70% of MS patients at 7T and are associated with CL burden. 39 We showed similar TL prevalence and found associations between LME+ status and increased CL and TL burden. In addition, the LME focus number had moderate to strong correlations with the TL number and volume. In line with prior work, 39 the TL number showed a moderate to strong correlation with the CL volume as compared to the WM lesion volume, which showed borderline statistical significance.
There are several concepts that may illuminate the link between meningeal inflammation and both cortical and deep GM damage. First, the proximity of cortical and deep GM to the cerebrospinal fluid (CSF; whether via the subarachnoid space or the ventricles) may implicate CSF-related inflammation/toxins. The blood–choroid plexus–CSF barrier, with its lack of endothelial tight junctions or glia limitans, but the presence of adhesion molecules and chemokines, is more permissive than the blood–brain barrier to immune cell entry from the circulation. 40 Choroid plexus–recruited effector T cells and macrophages have been implicated in both experimental autoimmune encephalitis 41 and in human MS autopsied brain tissue. 42 Increased HLA-DR (human leukocyte antigen—DR isotype)-positive cells in the choroid plexus may promote inflammatory cytokines, chemokines, and nitrous oxide in the CSF. 42 Moreover, increased vascular cell adhesion molecule 1 (VCAM-1) expression on choroid plexus endothelial cells in MS versus control brains may lead to CSF entry of lymphocytes. 42 Similar inflammatory intrathecal profiles (indicative of increased B-cell activity and lymphoid neogenesis) have been identified in both secondary progressive multiple sclerosis (SPMS) patients with extensive GM injury and meningeal inflammation and in early-stage MS patients with increased CL burden at diagnosis. 14 Several MRI studies have suggested that there is a gradient of MRI abnormality extending from the CSF surface to affect GM and normal-appearing WM in a pattern distinct from WM lesions,43,44 and a surface-in thalamic gradient of damage is evident even in pediatric RRMS. 45 This “toxic CSF” hypothesis may provide a key to understanding GM injury throughout the disease course, providing a bridge to progressive disease. 14
Alternatively, the cerebral cortex and thalamus may have common GM-specific antigen expression, which differs from WM, providing a unique target for the immune response. 46 In favor of this concept, widespread cortical GM demyelination can occur in the absence of cerebral WM lesion accumulation in a subset (12%) of MS patients, usually accompanied by a diagnosis of progressive MS. 47 Furthermore, it is well known that WM lesions may dissociate from the level of brain atrophy in MS, both in patients with a high lesion load with minimal atrophy or vice versa, linked to specific serum microRNA profiles. 48 Thus, topographic and pathobiologic involvement in MS may proceed along distinct immune-driven pathways.
Our work has a number of limitations that warrant further consideration. This is a single-center study, with a small sample size composed of RRMS subjects with generally mild clinical involvement, and no whole-brain atrophy. There is no longitudinal follow-up to assess how LME–MRI relationships change with disease course. LME and CLs were detected manually, and, although we show acceptable intra-rater reliability for our detection methods, comparison to standardized subtraction imaging techniques and evaluation of inter-rater reliability will be important for broader applications. Detection of nodular LME foci may particularly be prone to false positives (as they may resemble normally enhancing vasculature) and/or false negatives (due to their small size). Even with ultra-high-field MRI, only a fraction of CLs are visible, and thus relationships between LME and CL may not accurately reflect the pathological ground truth. Furthermore, regional atrophy investigations (e.g. cortical and deep GM) are necessary to validate these findings. LME showed no relationship to clinical outcomes, but this was likely limited by sample size, narrow clinical scales, and lack of longitudinal observation. Future studies to assess the relationship of LME to broader clinical outcomes, including cognitive status, fatigue, and patient-reported metrics are needed. This study was not designed to assess treatment impact on LME. The recent incorporation of routine clinical use of 7T brain MRI at our center, however, will facilitate well-powered, long-term investigations to assess the relationship of LME to treatment and clinical progression.
Conclusion
Cerebral LME is commonly detected in RRMS by 7T MRI and is associated with cortical and thalamic GM lesions. These data suggest common links in the pathogenesis of MS-mediated GM disease, such as CSF-related inflammation, given the proximity of these regions to the CSF space, or common antigen-mediated autoimmunity. LME may be a sign of such inflammation.
Supplemental Material
MSJ885106_supplementary_figures – Supplemental material for 7T MRI cerebral leptomeningeal enhancement is common in relapsing-remitting multiple sclerosis and is associated with cortical and thalamic lesions
Supplemental material, MSJ885106_supplementary_figures for 7T MRI cerebral leptomeningeal enhancement is common in relapsing-remitting multiple sclerosis and is associated with cortical and thalamic lesions by Jonathan Zurawski, Shahamat Tauhid, Renxin Chu, Fariha Khalid, Brian C Healy, Howard L Weiner and Rohit Bakshi in Multiple Sclerosis Journal
Supplemental Material
MSJ885106_supplementary_tables – Supplemental material for 7T MRI cerebral leptomeningeal enhancement is common in relapsing-remitting multiple sclerosis and is associated with cortical and thalamic lesions
Supplemental material, MSJ885106_supplementary_tables for 7T MRI cerebral leptomeningeal enhancement is common in relapsing-remitting multiple sclerosis and is associated with cortical and thalamic lesions by Jonathan Zurawski, Shahamat Tauhid, Renxin Chu, Fariha Khalid, Brian C Healy, Howard L Weiner and Rohit Bakshi in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors would like to thank Ravi Seethamraju, Aida Faria, and Vera Kimbrell for technical expertise and assistance with 7T MRI sequence development and research scanning at the Brigham and Women’s Hospital.
Authorship
All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval to the version to be published.
Declaration of Conflicting Interests
J.Z. has received consulting fees from ERT Consulting. B.C.H. has received research support from Merck Serono, Genzyme, Novartis, and Verily Life Sciences. H.L.W. has received research support from NIH, NMSS, EMD Serono, Novartis, Sanofi Genzyme, Teva Neurosciences, and Verily Life Sciences and personal and consulting fees from Genentech, Tiziana Life Sciences, Tilos Therapeutics, IM Therapeutics, vTv Therapeutics, and MedDay Pharmaceuticals. R.B. has received consulting fees from Bayer, Biogen, Celgene, EMD Serono, Genentech, Guerbet, Sanofi Genzyme, and Shire and research support from EMD Serono and Sanofi Genzyme. The other authors have nothing to disclose.
Ethical Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964, as revised in 2013.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by The Ann Romney Center for Neurologic Diseases and The Watercove Foundation.
Informed Consent
Informed consent was obtained from all patients for being included in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
