Abstract
Objectives:
The aim of this study was to assess the Dutch nationwide incidence of myelin oligodendrocyte glycoprotein (MOG)-IgG-associated acquired demyelinating syndromes (ADS) and to describe the clinical and serological characteristics of these patients.
Methods:
All serum samples for routine diagnostics from February 2014 to December 2017 were sent to the single central reference laboratory for the full-length MOG-IgG cell-based assay (CBA) in the Netherlands. Clinical data from patients known in our National ADS centre were available.
Results:
A total of 1414 samples of 1277 patients were received; of these, 92 patients (7%) were MOG-IgG-seropositive. The mean incidence was 0.16/100,000 people, with higher seropositivity in children (0.31/100,000) than in adults (0.13/100,000). In MOG-IgG-positive patients at the National ADS centre (61/92, 66%), the most common presenting phenotype is acute disseminated encephalomyelitis (ADEM, 56%) in children and optic neuritis (ON, 44%) in adults. Relapsing disease occurred in 9/34 (26%) children and 11/27 (41%) adults during median follow-up of 27.5 months. Patients were tested MOG-IgG-positive >200 months after the initial attack, suggesting an extended time to first relapse (TTFR). Longitudinal analysis of MOG-IgG (25/61, 41%) showed that 67% of the monophasic patients remain seropositive and 60% in relapsing patients. Majority of seronegative patients had no relapses (89%).
Conclusion:
This nationwide study shows that the overall incidence of MOG-IgG-seropositive disorders is 0.16 per 100,000 people. The distribution over the clinical phenotypes differs between adults and children. Seropositivity can be maintained over years even without clinical activity, while seronegative patients generally had no relapses.
Keywords
Introduction
Myelin oligodendrocyte glycoprotein (MOG) is a protein expressed on the surface of myelin sheaths and oligodendrocytes.1,2 Anti-MOG antibodies (MOG-IgG) can cause demyelination in vitro and induce experimental autoimmune encephalomyelitis.3,4 MOG-IgG are found in subtypes of central nervous system (CNS) acquired demyelinating syndromes (ADS) in both adult and paediatric patients, for example, in neuromyelitis optica spectrum disorders (NMOSD), 5 acute disseminated encephalomyelitis (ADEM) and in a small subgroup of adult multiple sclerosis (MS) patients.6,7 Since the optimisation of the MOG-IgG cell-based assay (CBA), this CBA has become available for routine clinical practice. 8 Although research on the clinical aspect of MOG-IgG-associated demyelinating syndromes has taken a great leap in the recent years, the incidence figures of MOG-IgG seropositivity in the general population have not yet been investigated. In the Netherlands, one single centralised laboratory performs the diagnostic testing of MOG-IgG. 6 This provides an unique opportunity to gain insight into the nationwide incidence of MOG-IgG seropositivity in both children and adults presenting with CNS demyelinating diseases. In addition, a recent study showed that the distribution of clinical phenotypes differs between MOG-IgG-seropositive children and adults. 9 We here aim to provide an incidence estimate of MOG-IgG seropositivity in a typical western European country and to describe the clinical and serological characteristics of patients within the MOG-IgG spectrum.
Methods
Patients
The Dutch National ADS centre includes the NMOSD centre, Paediatric MS centre (Rotterdam) and Sanquin diagnostic services (Amsterdam). Basic demographic data (age and treating centre) were available from all serum samples sent in for routine MOG-IgG diagnostics. All serum samples were tested blindly, centrally and in duplicate at Sanquin Diagnostics with a CBA.
6
All patients were tested negative for anti-aquaporin 4 antibodies (AQP4-IgG). Details on the CBAs will follow in the next section. The CBA for MOG-IgG has become available nationwide since February 2014. Data were collected from four consecutive years (1 February 2014 to 31 December 2017). Samples that were sent in from abroad, mainly from the Dutch Caribbean and Belgium, were excluded from this study (
From the patients known in the National ADS centre and whose serum was tested between February 2014 and December 2017, clinical data were available. Patients were diagnosed with NMOSD or ADEM by the international consensus criteria.11,12 Magnetic resonance imaging (MRI) lesions were scored on T2 and FLAIR sequences: poorly demarcated (deep) grey and/or white matter lesions, gyral filling, extensive confluent white matter lesions, well-demarcated ovoid lesions (MS-like) and non-specific lesions.13,14 Spinal MRIs were evaluated if available and scored for lesion location (cervical, thoracic, and lumbar) and presence of longitudinally extensive transverse myelitis (LETM; ⩾3 segments). 12 For the serial sample analyses, only samples ⩾3 months after the previous sample were taken into account.
CBAs
CBAs were used for MOG-IgG and AQP4-IgG detection as described elsewhere.6,15 Briefly, patient serum was incubated with HEK 293 cells transiently transfected with AQP4-M23 (enhanced green fluorescent protein (eGFP) tagged; final serum dilution 1:20) or LN18 cells stably transfected with full-length MOG (eGFP tagged; final serum dilution 1:200). After washing, cells were subsequently incubated with goat anti-human IgG allophycocyanin-conjugated secondary antibody (specific for human IgG; Jackson ImmunoResearch Laboratories and Brunschwig Chemie B.V., Amsterdam, The Netherlands) and analysed after washing using fluorescence-activated cell sorting. We expressed the quantitative levels of antibody titers as the difference in median fluorescence intensity (ΔMFI) between the AQP4- and MOG-transfected with, respectively, the untransfected HEK 293 cells and untransfected LN18 cells. The cut-off was determined in every assay as the average mean fluorescence intensity + 10 standard deviations of 8 individual negative control sera. Both assays have an anti-IgG-specific detection antibody and thus no IgM anti-MOG or IgM anti-AQP4 was detected.
Ethical approval
This study was approved by the Medical Ethical Committee of the Erasmus MC in Rotterdam. All patients from the Erasmus MC provided written informed consent.
Statistical analysis
Demographic data of the general Dutch population were provided by Statistics Netherlands and were used to calculate the incidence rates. For statistical analyses, we used IBM SPSS software, version 24.0 (IBM Corp., Armonk, NY) and GraphPad Prism 5 (GraphPad Software, San Diego, CA). The chi-square test and Mann–Whitney’s
Data availability
The raw data used in this paper could be shared in an anonymised format by request from a qualified investigator through contacting the corresponding author.
Results
Incidence figures
From 1 February 2014 to 31 December 2017, 1414 serum samples of 1277 patients were tested for MOG-IgG. Of all included patients, 92 (7%) were seropositive. In total, 196 children and adolescents younger than 18 years were tested. Of these paediatric patients, 34 (17%) were MOG-IgG-seropositive. To calculate the representative mean incidence of MOG-IgG seropositivity in the Netherlands, data from 2015 to 2017 were used resulting in an incidence of 0.16 per year per 100,000 people (95% confidence interval (CI): 0.11–0.23). After dividing the MOG-IgG patients into children and adults, we observed a mean incidence of 0.31 per 100,000 children (95% CI: 0.17–0.51) and 0.13 per 100,000 adults per year (95% CI: 0.08–0.19). The incidence figures for children, adults and all patients of the included years are shown in Table 1.
Incidence figures of anti-MOG seropositivity in the Netherlands.
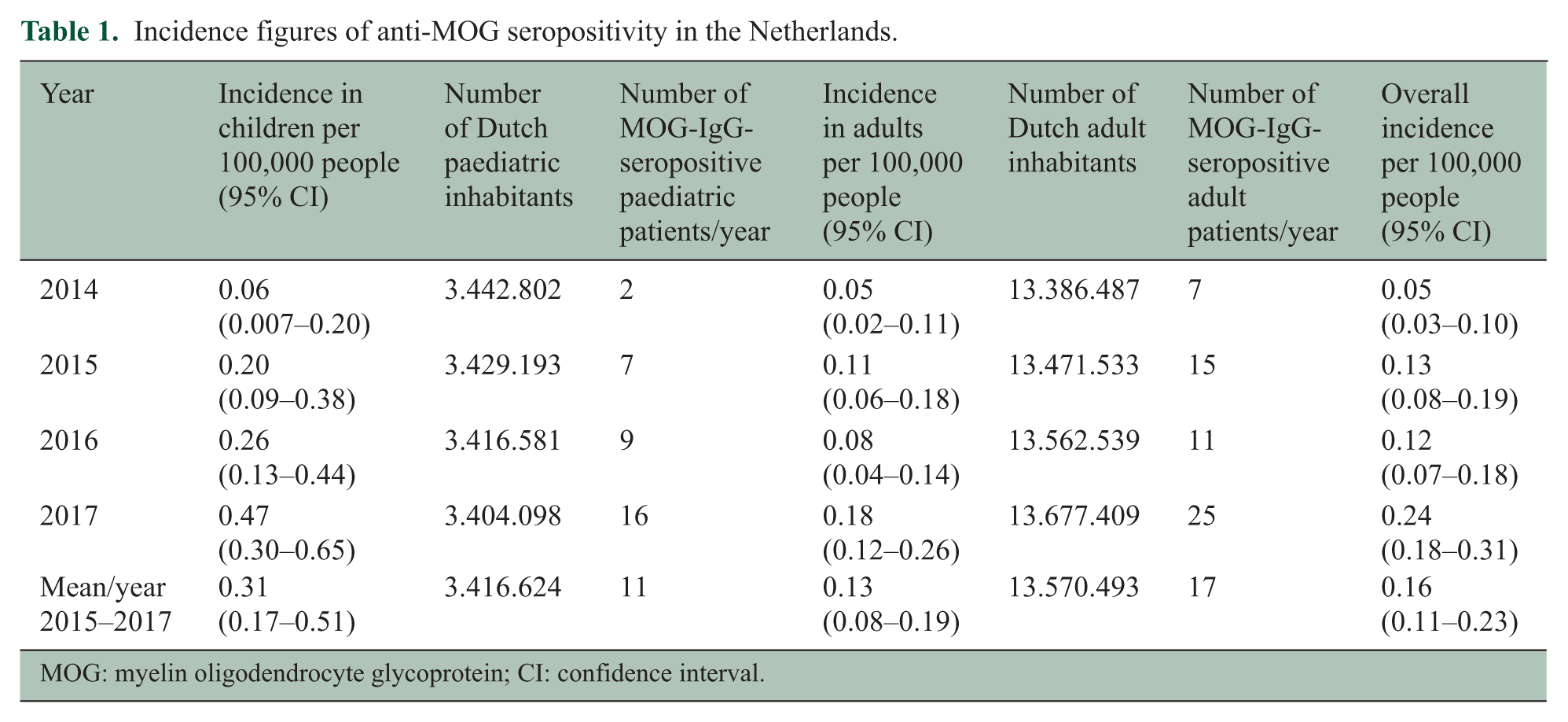
MOG: myelin oligodendrocyte glycoprotein; CI: confidence interval.
Baseline characteristics
In total, 61 of 92 (66%) MOG-IgG-seropositive patients were treated in the Dutch ADS centre (paediatric disease onset,
Clinical characteristics of MOG-IgG-seropositive patients.
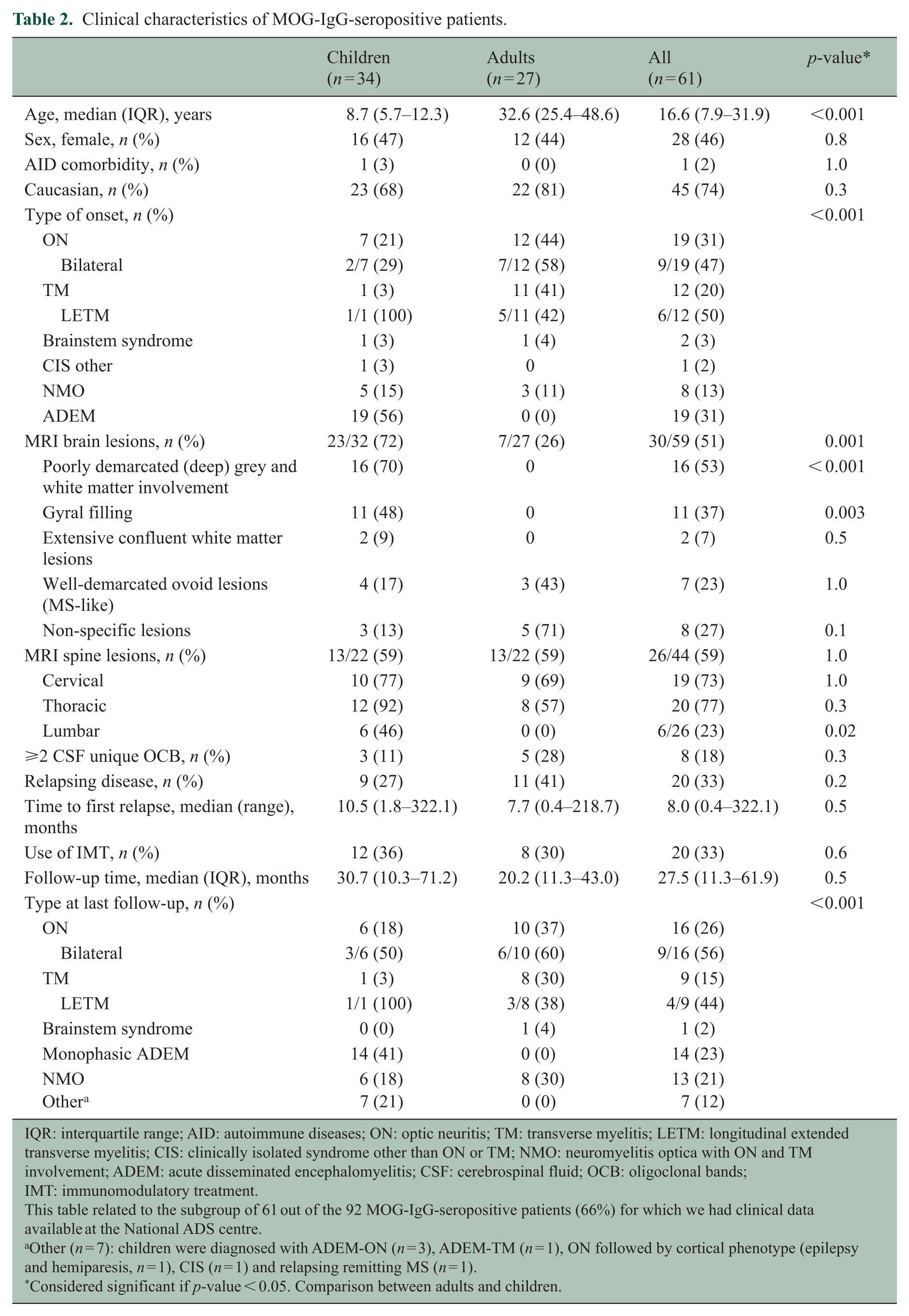
IQR: interquartile range; AID: autoimmune diseases; ON: optic neuritis; TM: transverse myelitis; LETM: longitudinal extended transverse myelitis; CIS: clinically isolated syndrome other than ON or TM; NMO: neuromyelitis optica with ON and TM involvement; ADEM: acute disseminated encephalomyelitis; CSF: cerebrospinal fluid; OCB: oligoclonal bands; IMT: immunomodulatory treatment.
This table related to the subgroup of 61 out of the 92 MOG-IgG-seropositive patients (66%) for which we had clinical data available at the National ADS centre.
Other (
Considered significant if
Follow-up characteristics
The most common phenotypes at end of FU in children were monophasic ADEM (14/34, 41%), followed by ON (6/34, 18%) and NMO (6/34, 18%). One MOG-IgG-seropositive child was diagnosed with relapsing remitting MS (RRMS; low delta-MFI: 330, cut-off: 104). Another child had a first episode of ON and had multiple relapses with a cortical phenotype (epilepsy, hemiparesis) with new corresponding lesions on brain MRI. In adults, the ON phenotype was most common (10/27, 37%), followed by TM (8/27, 30%) and NMO (13/27, 30%). During FU, optic nerve involvement was observed at least once in 16/34 children (47%) and in 17/27 adults (63%). Two out of 34 children showed relapsing ON (6%) and 3 out of 27 adults (11%). One of these adult patients (4%) was diagnosed with chronic relapsing inflammatory optic neuritis (CRION), suffering from multiple relapses when tapering off oral prednisone and showing quick and complete recovery after steroid administration. The distribution of clinical phenotypes at presentation and at last follow-up is presented in Figure 1 for adults and children.
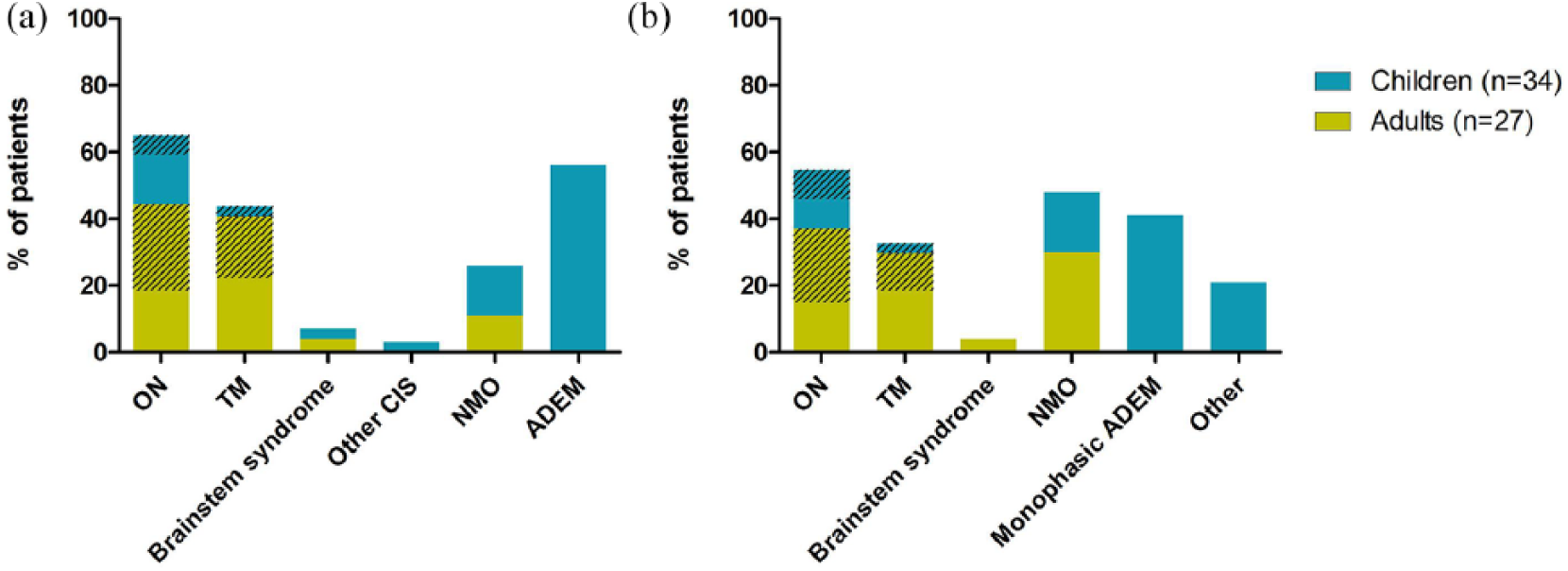
Distribution of clinical phenotypes in adults and children: (a) phenotypes at first presentation; (b) phenotypes at last follow-up. Other (
At the end of FU (median: 27.5 months, IQR: 11.3–61.9, range: 1.0–329.1), 20/61 (33%) patients showed a relapsing disease course (paediatric onset 9/34, 26%; adult onset 11/27, 41%). The proportion of patients with a first relapse within the first year after onset was 75% (median time to first relapse (TTFR): 5.8 months, IQR: 2.5–10.1 months). After 2 and 3 years, this proportion was 80% (16/20) for both time intervals. Within 5 years, 17/20 (85%) of the relapsing patients had a relapse. The remaining 3/20 relapsing patients were tested MOG-IgG-positive >200 months after the initial attack, suggesting an extended TTFR. Of these 20 relapsing patients, 15 (75%) were given immunomodulatory or immunosuppressive treatment (IMT), including azathioprine, rituximab (RTX), mycophenolate, cyclophosphamide, monthly intravenous immunoglobulins (IvIG) and teriflunomide.
Longitudinal analysis of MOG-IgG
Serial sampling was performed in 25/61 (41%) National ADS centre patients for routine clinical care (40 serum samples) with a median interval between first and last sample of 13.7 months (IQR: 7.2–25.4) with a median follow-up of 31.9 months (IQR: 12.3–57.7).
In Figure 2, we divided the 25 patients with serial samples into 4 groups based on their disease course and the last available test result: (1) relapsing patients with persisting MOG-IgG (
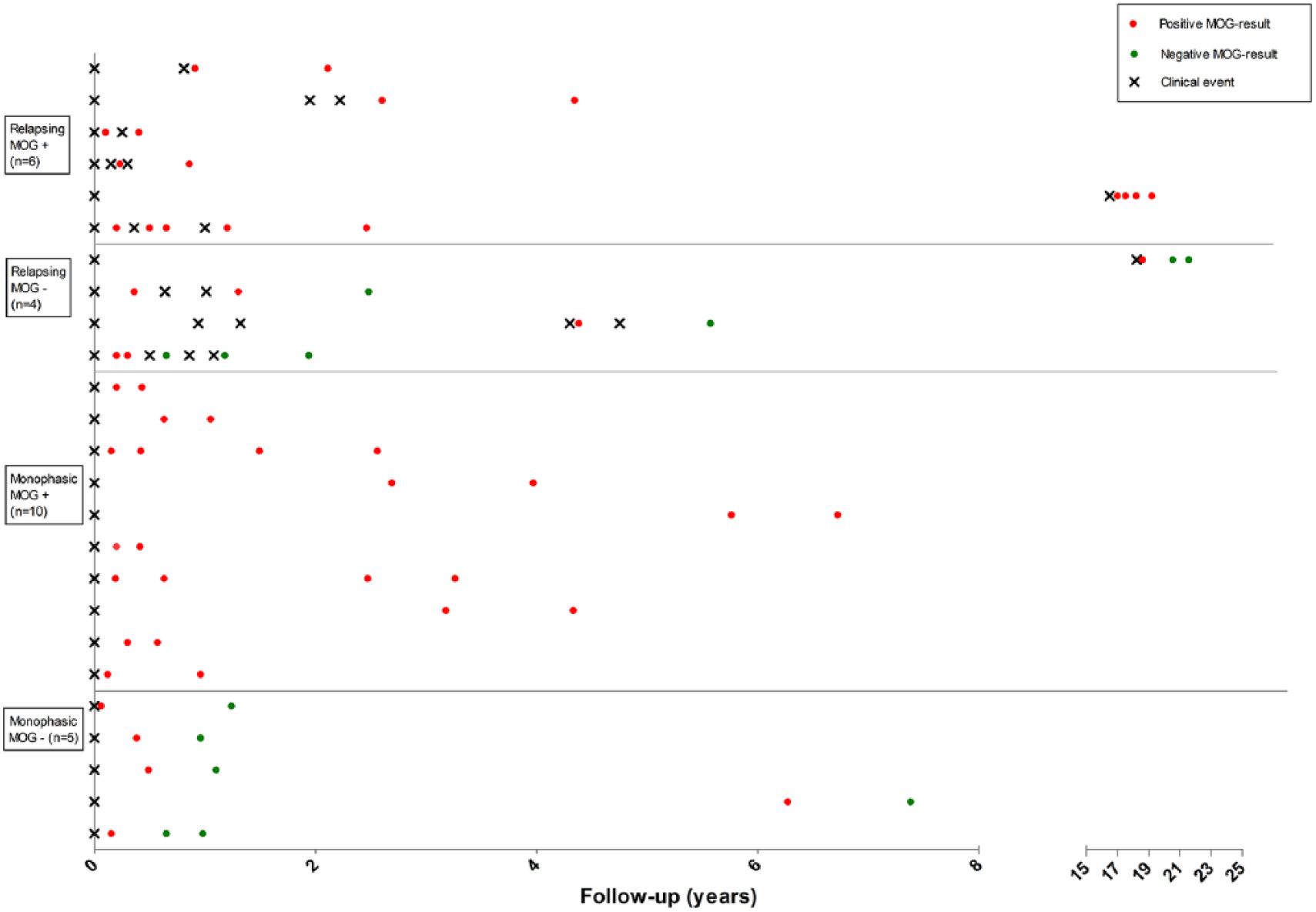
Longitudinal analysis of clinical and serological data; 25 of the 61 MOG-IgG-positive patients who were known at the National ADS centre were tested serially for MOG-IgG. Patients are classified into four groups according to their disease course (relapsing or monophasic) and the results of the last tested serum sample of that patient.
We observed that 10/15 (67%) monophasic patients remained seropositive (with a median FU time of 23.6 months; IQR: 9.7–50.1; median onset-to-last-sampling time of 21.7 months). The 5/15 (33%) monophasic who turned seronegative had a median FU of 30.4 months (IQR: 19.5–75.4). Relapsing patients (
IMT was initiated in 6/16 MOG-IgG-positive patients who remained positive during FU. Nine patients converted into seronegativity during FU. In four of these patients, IMT was initiated. For these individuals, we cannot rule out that this has affected their serostatus and delta MFI. Overall, we found no relation between IMT use and the last serostatus (
Discussion
We report the incidence estimate of MOG-IgG seropositivity in both adults and children with CNS-ADS in the Netherlands, from data derived from the National ADS centre. All samples were tested in one central laboratory, resulting in a nationwide MOG-IgG seropositivity incidence of 0.16/100,000 people per year (95% CI: 0.11–0.23), with a higher incidence in children (0.31/100.000; 95% CI: 0.17–0.51) than in adults (0.13/100,000; 95% CI: 0.08–0.19), reflecting the rarity of MOG-IgG seropositivity. These incidences result in 11 MOG-IgG-positive children and 17 MOG-IgG-positive adults per year. Previous studies showed that 18%–32% of the paediatric ADS patients are MOG-IgG-positive at baseline.6,16,17 Recently, we reported an update of the paediatric ADS incidence in the Netherlands of 0.80/100,000 children. 18 When extrapolating these results, the incidence of MOG-IgG-positive patients in the paediatric population could vary between 0.14 and 0.26 per 100,000 children. Our incidence figure of 0.31/100,000 (95% CI: 0.17–0.51) approximates this range, though somewhat higher. A slight difference is that in the current study, only patients with a positive test result were included. Incidence figures of MOG-IgG testing bear relevance for clinical care and counselling and also for survey of these ailments, implementation of assays in regional/national laboratories, planning with health care authorities and organisation of potential future clinical trials.
We had access to the clinical data of 61 MOG-IgG-seropositive patients who were all known in the National ADS centre (66%). In keeping with previous studies, the optic nerve was frequently involved in children (47%) and adults (63%) with MOG-IgG.5,19–22 Isolated brainstem syndromes were rare in both children and adults, but 10% of our patients showed brainstem involvement at first presentation, in contrast to another study (30% in total at presentation and during follow-up). 23 This could be due to different inclusion criteria between the studies. The clinical phenotypes that our patients showed at the end of follow-up were similar to previously published literature. Yet, one girl in our study experienced one ON and relapsing disease with predominantly cortical presentations (unilateral cerebral cortical epilepsy and hemiparesis). This presentation with seizures was described earlier in a case report with four MOG-IgG-seropositive adult patients (of whom two also experienced ON prior to seizures) and in a recent study, two children are identified.9,24 Earlier comparison with AQP4-IgG-positive patients also indicated a higher occurrence of seizures in MOG-IgG-positive patients. 25 The clinical spectrum of MOG-IgG-positive patients may further expand since the CBAs are now being established worldwide and more patients with MOG-IgG-related diseases are being identified. Clinicians should be aware of less frequently reported clinical manifestations associated with MOG-IgG positivity.
A relapsing disease course was observed in 33% of our patients, in line with other studies.5,26 In children, the proportion of MOG-IgG-positive patients with relapses (27%) is somewhat lower than in previous literature (up to 50%).6,16,17 In adults, 41% of the patients had a relapsing disease course which was comparable to other studies.5,19,21,26 However, a few studies reported a much higher proportion of relapsing patients (62%–80%) after a longer period of follow-up.14,27,28 This high proportion could be explained by longer follow-ups and the retrospective character of the studies. Our current figures of relapsing patients support a wait-and-see policy in at least a subgroup after the first attack of MOG-IgG-associated CNS demyelination.
Of note is that the majority of our relapsing patients (75%) have their first attack within 1 year after onset with a median TTFR of 5.8 months, comparable with other studies.19,29 Compared to AQP4-positive patients, the TTFR in the MOG-IgG patients is similar. 19 In our previous work, we reported a significantly longer TTFR of 28 months which can be explained by the small sample size. 5 A minor subgroup of the MOG-IgG-positive patients showed a relapse after an exceptional long time interval of >20 years after onset, in line with other studies.5,29,30
The longitudinal analysis of MOG-IgG patients showed that a considerable proportion of relapsing patients remains seropositive (64%). Yet, in our study, one patient had new relapses after turning seronegative, while being treated with potent immune suppression with RTX and monthly IvIG. A possible explanation is the timing of the blood sampling, as the first blood sample with a negative result was taken 4 months after the previous attack while being treated with immunosuppression, and the second negative blood sample was taken 2 days after a two cycles of RTX therapy. This might have influenced the MOG-IgG results. Beside from this patient, no relapses were observed in relapsing patients who eventually converted to seronegativity. This is in line with another study that described 10 MOG-IgG-positive patients with serial sampling, and observed that no relapses occur after patients turned seronegative. 31
One child was diagnosed with relapsing remitting MS with a typical MS disease course, including MS-like MRI fulfilling both the McDonald 2010 and 2017 criteria and had unique CSF oligoclonal bands and was therefore treated with teriflunomide. The delta-MFI was low (330; cut-off: 104). Low levels of MOG-IgG are observed in paediatric RRMS patients in previous studies.6,32
Our study has several limitations. First, these current incidence estimates are minimum figures, since mild cases and forme fruste types of the disease could have been missed. Rather than bona fide incidence, this can also be interpreted as the number of cases detected at the nation-level per year; this is by itself of interest and reflects the change of clinical practice. The incidence of 2014 is excluded from the mean incidence calculation, since the number is exceptionally low compared to the following years. This is most likely due to the low awareness of CBA availability in the first year after launching the CBA as a routine diagnostic test. Increased awareness of treating physicians could potentially contribute to even higher incidence figures in the future by testing more patients more quickly, although an asymptote may have been reached in this relatively small country. Second, it has not been feasible to have access to full clinical data of all MOG-IgG-seropositive patients in this country apart from the patient’s gender, as we did not have ethical permission to approach the patients who have not visited the Erasmus MC. Still, the more detailed analysis of this study covered 66% of the study population as these patients were referred to our National ADS centre. It is possible that certain phenotypes are over-represented in this group, for example, because of a lower threshold for referral in case of recurrent disease. Third, for definite conclusions on the temporal dynamics of MOG-IgG and the effect of immunosuppressive drugs on MOG-IgG serostatus, we lacked sufficient sample size and systematic schedule for blood sampling. Well-designed prospective studies including blood sampling at pre-defined time points will be needed to address these important questions.
In conclusion, this nationwide study of a typical western European country shows that the overall incidence of MOG-IgG-seropositive disease in children and adults is low, with an average of 0.16 per 100,000 people (95% CI: 0.11–0.23). The distribution over the different clinical phenotypes differs between adults and children. Seropositivity can be maintained over years even without clinical activity, while patients who converted to seronegativity during follow-up generally had no new relapses. Further studies with detailed clinical and serological follow-up in an international collaborative setting will set the stage for further improved insight on the use of this diagnostic test in clinical practice.
Footnotes
Acknowledgements
The authors thank the children, adults and their families for their participation in this study. They thank all the analysts in the autoimmune laboratory of Sanquin Diagnostic Services for performing the AQP4-IgG and MOG-IgG CBAs.
Author Contribution
C.L.M. contributed to study design, statistical analysis, interpretation of data and draft of the manuscript; Y.Y.M.W. contributed to study design, acquisition of data, statistical analysis, interpretation of data and draft of the manuscript; E.D.P., B.H.A.W., T.A.M.S., R.F.N. and D.H. contributed to acquisition of data and revision of manuscript for content; and R.Q.H. contributed to study design, study supervision, interpretation of data, and revision of manuscript for content. All authors reviewed and agreed on the content of this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: R.F.N. participated in trials with Sanofi and Novartis. R.Q.H. received honoraria for serving on advisory boards for Biogen Idec, Roche and Sanofi; he participated in trials with Biogen Idec, Merck-Serono, Roche, Genzyme and Novartis
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was supported by the Dutch MS Research Foundation. This study was not industry-sponsored.
