Abstract
Background:
In the 2-year CLARITY study, cladribine tablets significantly improved clinical and magnetic resonance imaging (MRI) outcomes (vs placebo) in patients with relapsing–remitting multiple sclerosis (MS).
Objective:
To assess the safety and efficacy of cladribine treatment in a 2-year Extension study.
Methods:
In this 2-year Extension study, placebo recipients from CLARITY received cladribine 3.5 mg/kg; cladribine recipients were re-randomized 2:1 to cladribine 3.5 mg/kg or placebo, with blind maintained.
Results:
A total of 806 patients were assigned to treatment. Adverse event rates were generally similar between groups, but lymphopenia Grade ⩾ 3 rates were higher with cladribine than placebo (Grade 4 lymphopenia occurred infrequently). In patients receiving cladribine 3.5 mg/kg in CLARITY and experiencing lymphopenia Grade ⩾ 3 in the Extension, >90% of those treated with cladribine 3.5 mg/kg and all treated with placebo in the Extension, recovered to Grade 0–1 by study end. Cladribine treatment in CLARITY produced efficacy improvements that were maintained in patients treated with placebo in the Extension; in patients treated with cladribine 3.5 mg/kg in CLARITY, approximately 75% remained relapse-free when given placebo during the Extension.
Conclusion:
Cladribine tablets treatment for 2 years followed by 2 years’ placebo treatment produced durable clinical benefits similar to 4 years of cladribine treatment with a low risk of severe lymphopenia or clinical worsening. No clinical improvement in efficacy was apparent following further treatment with cladribine tablets after the initial 2-year treatment period in this trial setting.
Keywords
Introduction
Cladribine is an oral, synthetic deoxyadenosine analog prodrug, which preferentially depletes lymphocytes by exploiting the kinase-to-phosphatase enzyme profile in these cells. This produces moderate and discontinuous reductions in T and B cells with relatively minor and transient effects on innate immune cells, such as neutrophils and monocytes. 1 The dosing regimen of cladribine tablets involves very short treatment periods relative to the length of clinical effect (8–10 days annually). 2 In the Cladribine Tablets Treating Multiple Sclerosis Orally (CLARITY) study (ClinicalTrials.gov, NCT00213135), patients with active relapsing–remitting multiple sclerosis (RRMS) were randomized to placebo or one of two cumulative doses of cladribine tablets (3.5 or 5.25 mg/kg body weight) for 2 years; each dose showed significant benefits in rate of relapse, disability progression, and magnetic resonance imaging (MRI) measures. 2 The most commonly reported adverse event (AE) was mild to moderate lymphopenia, reflecting the mode of action of cladribine. 2
Here, we report findings from the CLARITY Extension study (ClinicalTrials.gov, NCT00641537), which investigated long-term safety, tolerability, and clinical benefits following administration of placebo or a cumulative dose of cladribine tablets 3.5 mg/kg body weight in patients who had completed CLARITY. The design allowed assessment of the effects of 2 years’ additional treatment with cladribine tablets beyond the 2-year CLARITY regimen and included an assessment of the duration of the therapeutic benefit by a comparison of 2 years’ treatment with cladribine tablets followed by 2 years of placebo, and 2 years’ treatment with cladribine tablets followed by two additional years of cladribine tablets treatment. In addition, early vs late treatment was compared by including a group receiving 2 years’ placebo treatment followed by 2 years’ cladribine tablets treatment.
Methods
Patients
Patients who completed the 2-year study period in CLARITY were eligible for the Extension if they had normal lymphocyte count and other normal hematological results within 28 days of the first planned dose (see Supplementary Materials). CLARITY Extension was not a pre-planned study, and as a consequence after completing CLARITY, there was a variable gap period before patients entered the Extension (the median gap duration for the overall population was 40.3 weeks). Patients who had received interferon beta or glatiramer acetate during the gap period had to discontinue their disease-modifying drug (DMD) therapy ⩾3 months before the first study day of the Extension.
Randomization and masking
Patients who entered the Extension were re-randomized using an interactive voice response system. Randomization and double-blinding followed CLARITY procedures 2 (see Supplementary Materials). The on-study treatment groups were as follows:
CP 3.5 mg/kg (cladribine tablets 3.5 mg/kg in CLARITY/placebo in CLARITY Extension);
CP 5.25 mg/kg (cladribine tablets 5.25 mg/kg in CLARITY/placebo in CLARITY Extension);
CC 7 mg/kg (cladribine tablets 3.5 mg/kg in CLARITY/cladribine tablets 3.5 mg/kg in CLARITY Extension);
CC 8.75 mg/kg (cladribine tablets 5.25 mg/kg in CLARITY/cladribine tablets 3.5 mg/kg CLARITY Extension);
PC 3.5 mg/kg (placebo in CLARITY/cladribine tablets 3.5 mg/kg in CLARITY Extension).
Procedures
The study design is represented in Figure 1. Patients in CLARITY were randomized (1:1:1 ratio) to one of two cumulative doses of cladribine tablets or placebo, taking one or two cladribine 10-mg tablets (or matching placebo) daily over 4–5 days in either Weeks 1 and 5 of Years 1 and 2 (to give a cumulative dose of 3.5 mg/kg) or Weeks 1, 5, 9, and 13 of Year 1 and Weeks 1 and 5 of Year 2 (giving a 5.25 mg/kg cumulative dose). 2 In the 96-week CLARITY Extension study, eligible patients who received placebo in CLARITY were assigned to cladribine tablets 3.5 mg/kg, and patients treated with cladribine tablets in CLARITY were re-randomized (2:1) to cladribine tablets 3.5 mg/kg or placebo. Treating physicians supervised study drug administration and monitored safety assessments including laboratory parameters and were responsible for the recording and treatment of AEs and MS relapses, whereas separate evaluating physicians assessed neurological findings and relapses (each was blinded to treatment group assignment). The blind was maintained from CLARITY in all treatment arms during the Extension. All patients treated with cladribine tablets during the Extension received a cumulative dose of 3.5 mg/kg, administered in Weeks 1, 5, 48, and 52 (only patients with Grade 0 or 1 lymphocyte counts were retreated at Weeks 48 and 52). The Extension was followed by a 24-week “supplemental follow-up” (SUPF) during which patients did not receive treatment with cladribine tablets but could receive DMDs.
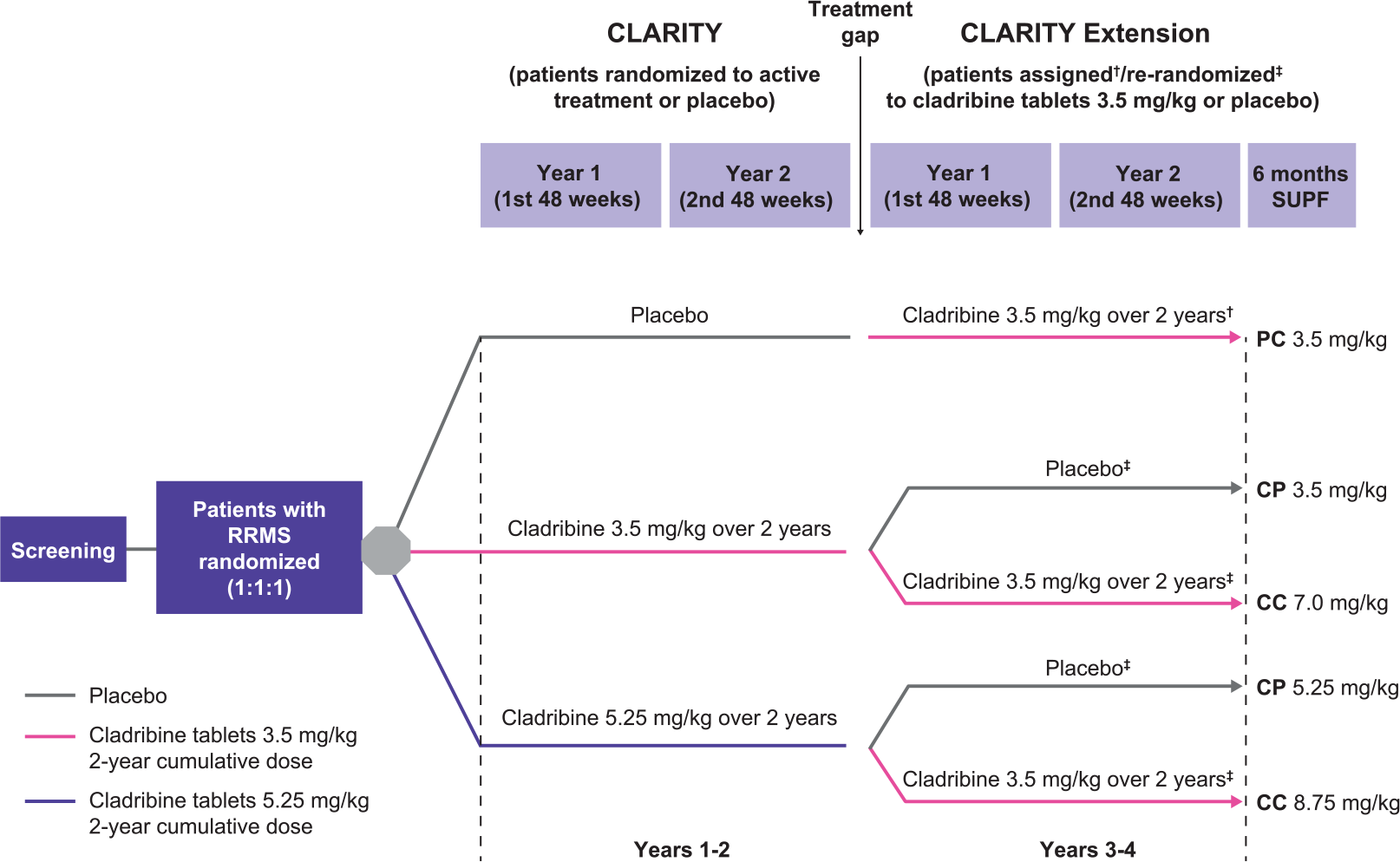
Study design. Each short course of treatment comprised one or two 10-mg cladribine tablets taken once daily for four or five consecutive days or an equivalent number of matching placebo tablets. There was a variable interval between the treatment periods in the CLARITY and Extension studies.
Endpoints
Safety and clinical endpoints, including amendments, are described in the Supplementary Materials. Clinical endpoints included the annualized relapse rate (ARR), the proportion of patients free of qualifying relapses, time to first qualifying relapse, and time to confirmed Expanded Disability Status Scale (EDSS) progression.
Statistical analysis
No sample size calculation was made as enrollment was limited to eligible patients from CLARITY. No hypotheses were pre-specified in the original protocol, but an amendment specified a benefit/risk analysis and a 2.5% significance level for the interim and final analyses. Efficacy endpoints were exploratory and all determinations of significance were nominal; a p value ⩽0.025 was considered nominally significant.
To investigate potential incremental effects of extended treatment during the Extension, comparisons between groups included CP 3.5 mg/kg vs PC 3.5 mg/kg (early vs late treatment with the 3.5 mg/kg dose), CC 7 mg/kg vs CP 3.5 mg/kg, CC 8.75 mg/kg vs CP 5.25 mg/kg (192 weeks of treatment vs 96 weeks of treatment), and CC 7 mg/kg + CC 8.75 mg/kg (combined data for patients who received treatment with cladribine tablets in CLARITY and CLARITY Extension) vs CP 3.5 mg/kg + CP 5.25 mg/kg (combined data for placebo in Years 3 and 4).
The intention-to-treat (ITT) population, which comprised patients randomized or assigned to treatment in the Extension, was analyzed for efficacy. The safety population, which included patients who received ⩾one dose of study medication and had follow-up safety data, was analyzed for safety. Patients who had completed CLARITY but were not eligible for treatment in the Extension were followed for safety only (SAFUP population).
Results
Patients
There were 1326 patients randomized into the CLARITY trial, of which, 1184 completed (89.3%). Of the 1184 that completed the CLARITY trial, 867 were enrolled in the Extension (73.2%, from 16 January 2008 to 20 July 2009). Of the 867 enrolled in the Extension, 61 comprised the SAFUP population, and 806 were randomized or assigned to treatment. The CLARITY study consisted of a total of 155 study sites; however, 22 fewer study sites were involved in CLARITY Extension. In total, there were five treatment groups in CLARITY Extension (CP 3.5 mg/kg, n = 98; CP 5.25 mg/kg, n = 92; CC 7 mg/kg, n = 186; CC 8.75 mg/kg, n = 186; and PC 3.5 mg/kg, n = 244). These patients comprised the ITT population and qualified for the safety population. In CLARITY Extension, 806 patients were treated, of which 738 (91.6%) completed the 2-year study (Figure 2), and 636 (78.9%) entered the SUPF. The characteristics of patients who entered the Extension were similar across groups at baseline, except that patients who had received placebo in CLARITY showed evidence of greater disease activity (Table 1). The characteristics of patients who did or did not enter CLARITY Extension at baseline of the CLARITY study were also similar (Supplementary Table 1). The gap between CLARITY and the Extension varied within each group but was distributed similarly across the treatment groups. Relapses during the gap period were not qualified by an evaluating physician. There were no major differences between groups in disease management during the gap period with few patients receiving other DMDs (Table 1). Details for concomitant DMD use during the gap between CLARITY and Extension and during the SUPF are shown in the Supplementary Materials.
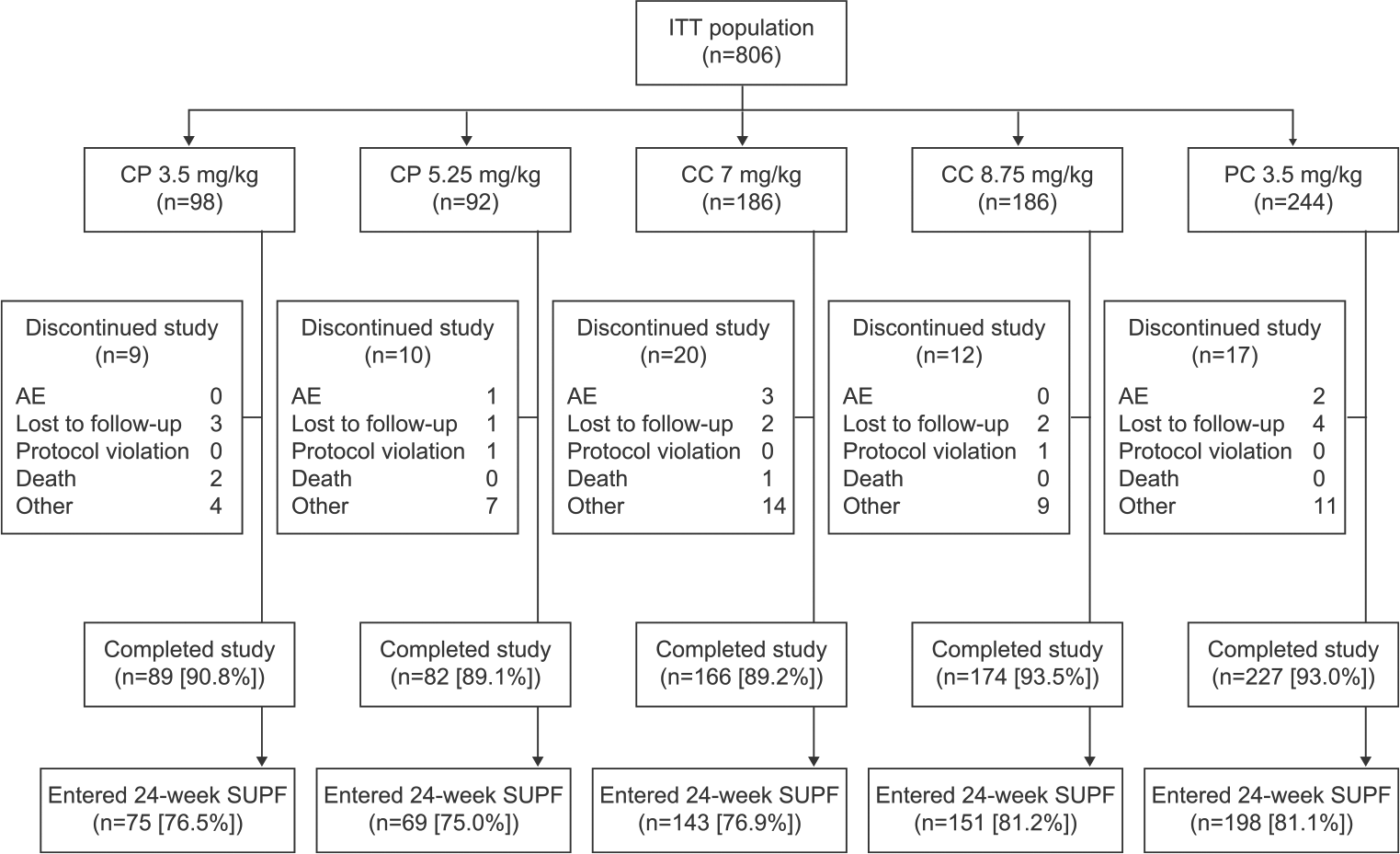
Patient disposition during the CLARITY Extension study. Seven patients received rescue therapy during the 2-year treatment period (CP 3.5 mg/kg, n = 3 (3.1%); CP 5.25 mg/kg, n = 2 (2.2%); CC 7 mg/kg, n = 1 (0.5%); PC 3.5 mg/kg, n = 1 (0.4%)).
Baseline demographics and disease characteristics of treatment groups in CLARITY Extension (ITT population).
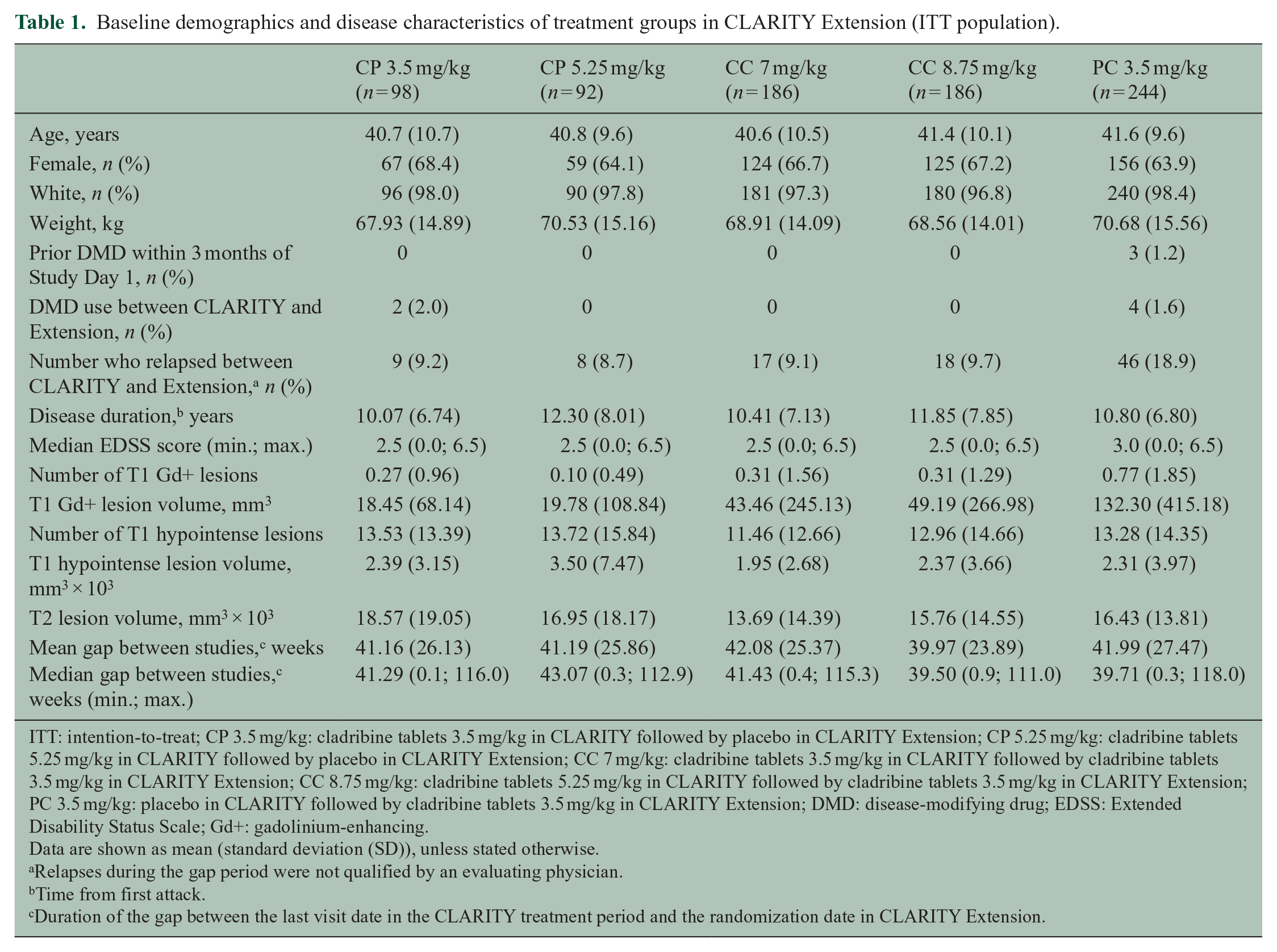
ITT: intention-to-treat; CP 3.5 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by placebo in CLARITY Extension; CP 5.25 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by placebo in CLARITY Extension; CC 7 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CC 8.75 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; PC 3.5 mg/kg: placebo in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; DMD: disease-modifying drug; EDSS: Extended Disability Status Scale; Gd+: gadolinium-enhancing.
Data are shown as mean (standard deviation (SD)), unless stated otherwise.
Relapses during the gap period were not qualified by an evaluating physician.
Time from first attack.
Duration of the gap between the last visit date in the CLARITY treatment period and the randomization date in CLARITY Extension.
Safety outcomes
The incidence of AEs was generally similar between groups (Table 2), and most AEs were classified as mild or moderate. There were 89 (11.0%) treatment discontinuations due to AEs (Table 2 and Supplementary Table 2) but only 11 (1.4%) patients withdrew from the study due to AEs (not shown). During the Extension, the frequency of lymphopenia and leukopenia was higher in patients treated with cladribine tablets compared with placebo recipients. For patients who received cladribine tablets 3.5 mg/kg in both CLARITY and the Extension, the treatment discontinuation rate due to lymphopenia and the incidence of lymphopenia AEs was somewhat elevated compared to patients who had received placebo in CLARITY and cladribine tablets in the Extension (Table 2). The majority of patients in each treatment group had no platelet toxicity, and among patients with decreased platelet count, most cases were Grade 1 or 2 in severity. One patient in each of the CC 7 mg/kg and CC 8.75 mg/kg groups had ⩾Grade 3 platelet toxicity, but there were none in any other group (not shown). A total of 94 (11.7%) patients experienced ⩾one serious adverse event (SAE), including infections and infestations (n = 16, 2.0%), and gastrointestinal disorders (n = 8, 1.0%).
Overview of safety outcomes, including treatment-emergent adverse events reported in 5% or more of patients.
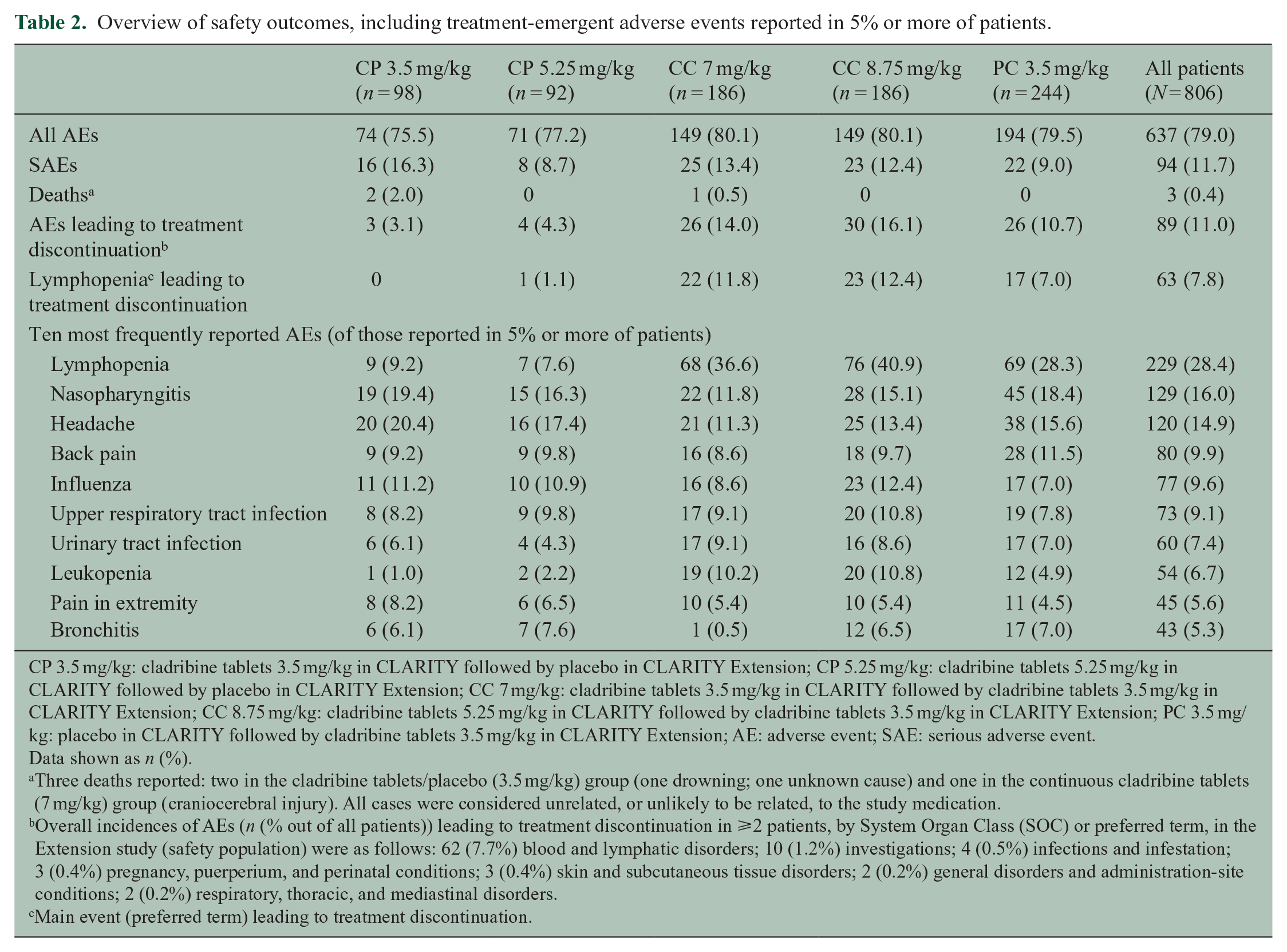
CP 3.5 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by placebo in CLARITY Extension; CP 5.25 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by placebo in CLARITY Extension; CC 7 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CC 8.75 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; PC 3.5 mg/kg: placebo in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; AE: adverse event; SAE: serious adverse event.
Data shown as n (%).
Three deaths reported: two in the cladribine tablets/placebo (3.5 mg/kg) group (one drowning; one unknown cause) and one in the continuous cladribine tablets (7 mg/kg) group (craniocerebral injury). All cases were considered unrelated, or unlikely to be related, to the study medication.
Overall incidences of AEs (n (% out of all patients)) leading to treatment discontinuation in ⩾2 patients, by System Organ Class (SOC) or preferred term, in the Extension study (safety population) were as follows: 62 (7.7%) blood and lymphatic disorders; 10 (1.2%) investigations; 4 (0.5%) infections and infestation; 3 (0.4%) pregnancy, puerperium, and perinatal conditions; 3 (0.4%) skin and subcutaneous tissue disorders; 2 (0.2%) general disorders and administration-site conditions; 2 (0.2%) respiratory, thoracic, and mediastinal disorders.
Main event (preferred term) leading to treatment discontinuation.
AEs of special interest
Lymphopenia events of Grade ⩾ 3 were reported in all treatment groups, including approximately 6% of patients treated with placebo (Table 3). No patients in either the CP 3.5 or 5.25 mg/kg group experienced Grade 4 lymphopenia. In the CP 3.5 mg/kg group, five patients experienced Grade ⩾ 3 lymphopenia, and all recovered to Grade 0–1 lymphocyte counts during the Extension, with a mean (standard deviation (SD)) time to recovery of 41.0 (33.5) days (min.–max.: 14–85 days). In the CP 5.25 mg/kg group, four of the six patients who had Grade ⩾ 3 lymphopenia recovered, with a mean time to Grade 0–1 recovery of 34.9 (11.7) days (min.–max.: 26–51 days); lymphocyte count data were incomplete for the other two patients. The two groups treated with cladribine tablets in both CLARITY and the Extension (CC 7 and CC 8.75 mg/kg) showed the highest incidence of Grade ⩾ 3 lymphopenia (40.9% and 53.2%, respectively) and longest median time to recovery to Grade 0–1 lymphopenia (212 and 168 days, respectively) during the Extension. The incidence rates of infections and infestations showed no clear relation to total dose received (Table 3), with the exception of the herpes zoster infection rate which was higher in the CC 8.75 mg/kg group (4.8%) than the other groups (CC 7 mg/kg = 1.1%, CP 3.5 mg/kg = 2.0%, CP 5.25 mg/kg = 1.1%, and PC 3.5 mg/kg = 2.0%; Table 3). The single case of “pulmonary mycosis” resulted from an erroneous coding of the reported term “pulmonary mycoplasmosis”; this event was non-serious, mild, and considered unrelated to study medication by the investigator. There were no cases of aspergillus in CLARITY Extension. The “fungal infection” was considered non-serious, and no specific location was reported.
Overview of AEs of special interest (safety population).
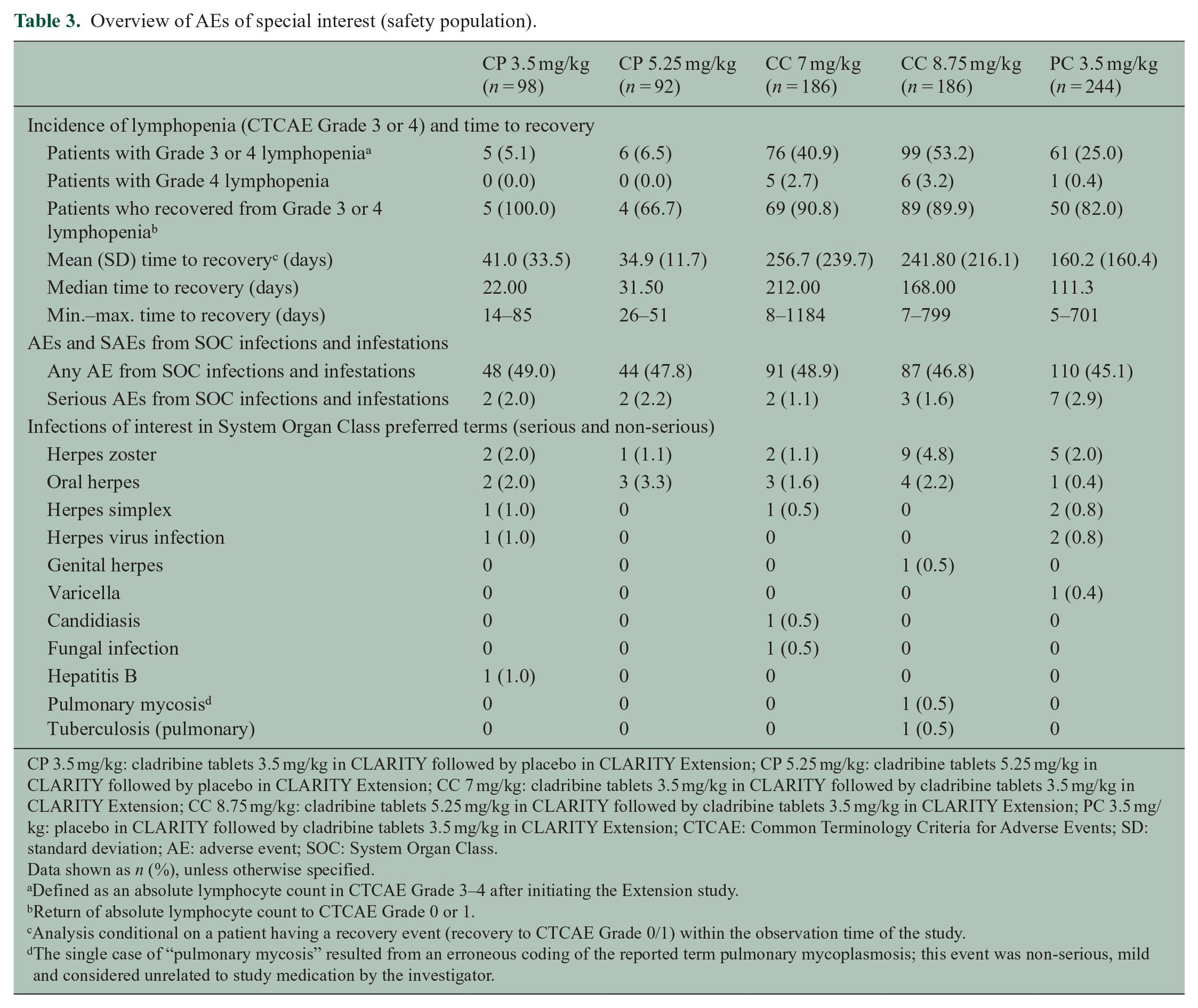
CP 3.5 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by placebo in CLARITY Extension; CP 5.25 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by placebo in CLARITY Extension; CC 7 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CC 8.75 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; PC 3.5 mg/kg: placebo in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CTCAE: Common Terminology Criteria for Adverse Events; SD: standard deviation; AE: adverse event; SOC: System Organ Class.
Data shown as n (%), unless otherwise specified.
Defined as an absolute lymphocyte count in CTCAE Grade 3–4 after initiating the Extension study.
Return of absolute lymphocyte count to CTCAE Grade 0 or 1.
Analysis conditional on a patient having a recovery event (recovery to CTCAE Grade 0/1) within the observation time of the study.
The single case of “pulmonary mycosis” resulted from an erroneous coding of the reported term pulmonary mycoplasmosis; this event was non-serious, mild and considered unrelated to study medication by the investigator.
Overall, 11/806 (1.4%) patients from the safety population developed a malignancy or unspecified tumor (see Supplementary Table 3). No cases of progressive multifocal leukoencephalopathy (PML) occurred during the Extension. Three deaths occurred during the Extension: two in the CP 3.5 mg/kg group (one drowning and one unknown cause) and one in the CC 7 mg/kg group (craniocerebral injury). All were considered unrelated or unlikely to be related to treatment with cladribine tablets (Table 2).
Efficacy outcomes
During the Extension, ARR were broadly similar across treatment groups (Table 4), and between-group comparisons confirmed that there were no significant differences (Table 5). Proportions of relapse-free patients were also comparable across treatment groups. At the end of the CLARITY study, approximately 80% of patients who received cladribine tablets 3.5 mg/kg were relapse-free. 2 In patients from the group that received placebo in the Extension (i.e. the CP 3.5 mg/kg group), approximately 75% remained relapse-free at 120 weeks (Table 4). No significant differences in time to first qualifying relapse (relative to first dose) were seen between treatment groups during the Extension (Figure 3). However, there was a consequence to delaying treatment with cladribine tablets; when CLARITY and the Extension phase were considered together, the shortest time to first qualifying relapse was seen in patients treated with placebo in CLARITY and cladribine tablets 3.5 mg/kg in the Extension (PC 3.5 mg/kg, Supplementary Figure 1). The proportions of patients who did not experience 3-month confirmed EDSS progression ranged from 72.4% (CP 3.5 mg/kg group) to 78.3% (CP 5.25 mg/kg group), with no clear pattern of differences between treatment groups (Table 4). Median EDSS scores by visit and treatment groups during CLARITY and CLARITY Extension are shown in Supplementary Figure 2.
Summary of clinical efficacy endpoints.
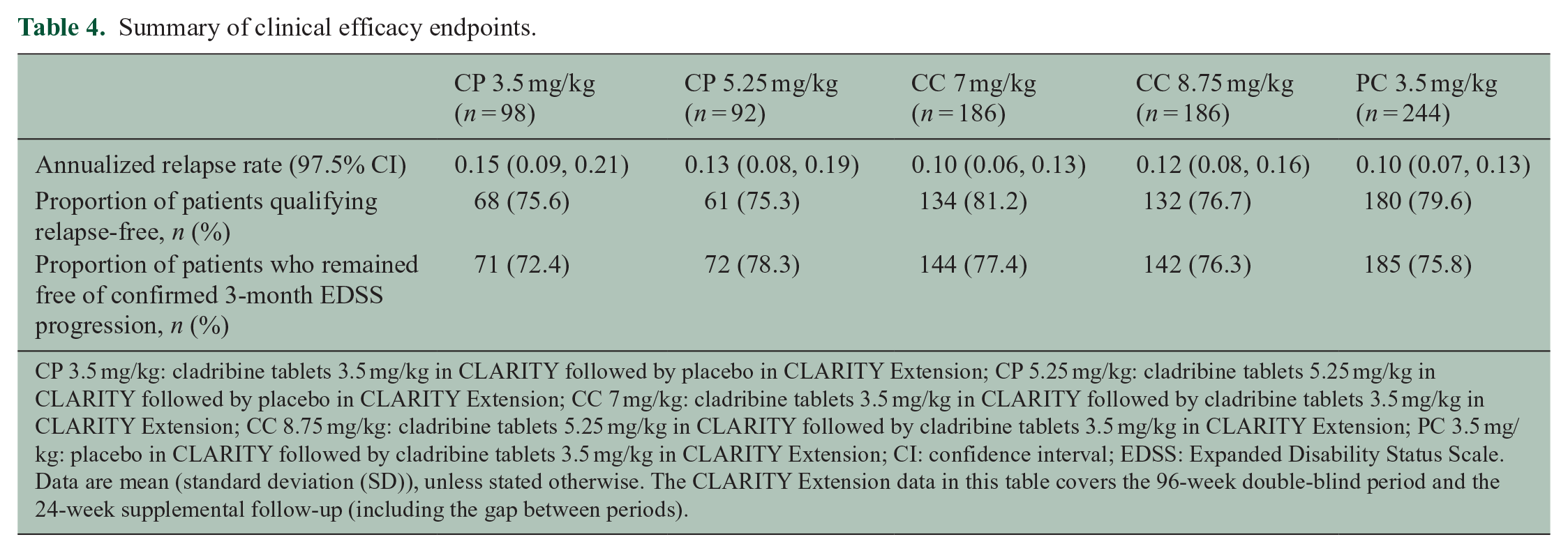
CP 3.5 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by placebo in CLARITY Extension; CP 5.25 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by placebo in CLARITY Extension; CC 7 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CC 8.75 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; PC 3.5 mg/kg: placebo in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CI: confidence interval; EDSS: Expanded Disability Status Scale.
Data are mean (standard deviation (SD)), unless stated otherwise. The CLARITY Extension data in this table covers the 96-week double-blind period and the 24-week supplemental follow-up (including the gap between periods).
Between-group analyses of clinical efficacy endpoints.
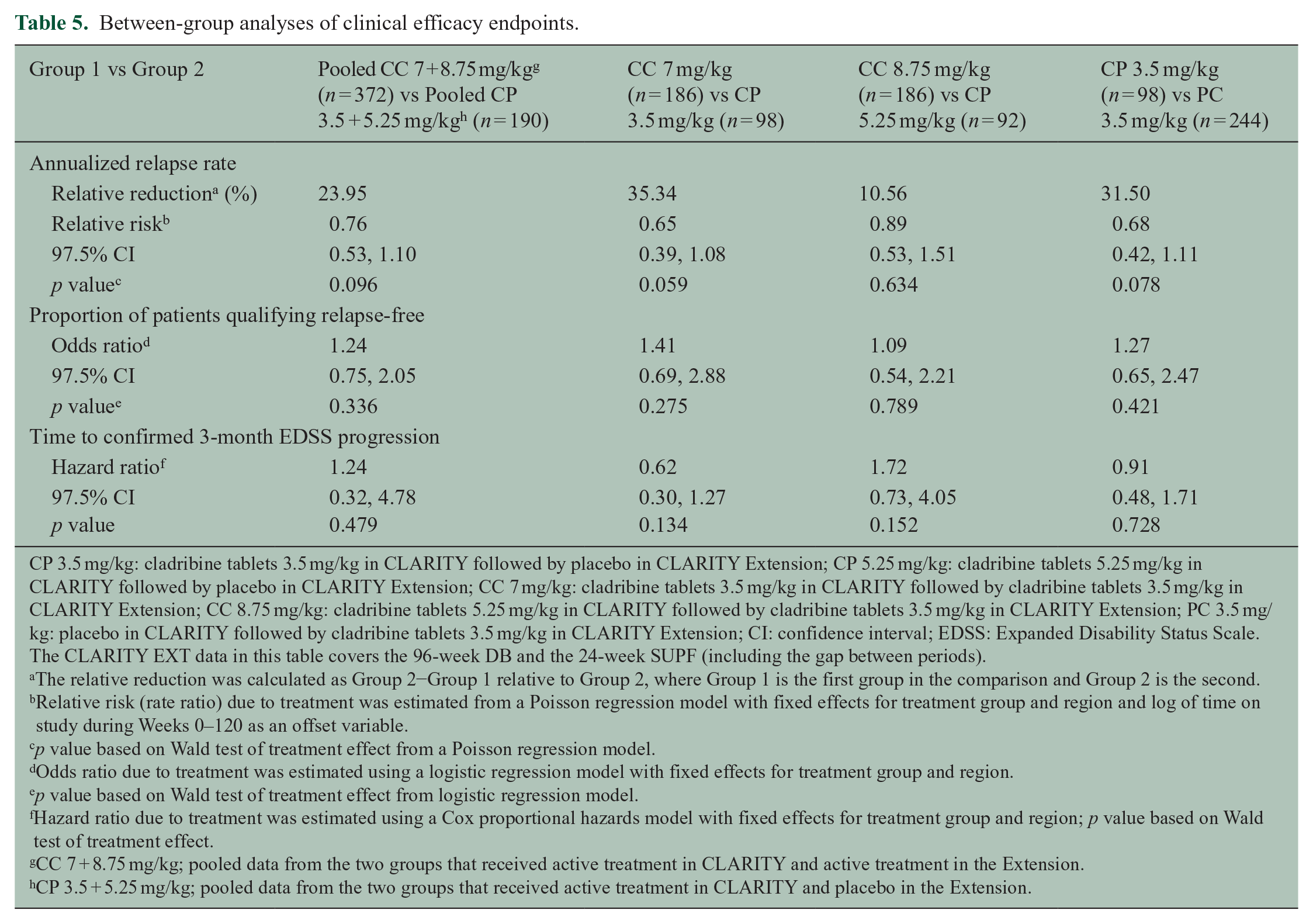
CP 3.5 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by placebo in CLARITY Extension; CP 5.25 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by placebo in CLARITY Extension; CC 7 mg/kg: cladribine tablets 3.5 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CC 8.75 mg/kg: cladribine tablets 5.25 mg/kg in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; PC 3.5 mg/kg: placebo in CLARITY followed by cladribine tablets 3.5 mg/kg in CLARITY Extension; CI: confidence interval; EDSS: Expanded Disability Status Scale. The CLARITY EXT data in this table covers the 96-week DB and the 24-week SUPF (including the gap between periods).
The relative reduction was calculated as Group 2−Group 1 relative to Group 2, where Group 1 is the first group in the comparison and Group 2 is the second.
Relative risk (rate ratio) due to treatment was estimated from a Poisson regression model with fixed effects for treatment group and region and log of time on study during Weeks 0–120 as an offset variable.
p value based on Wald test of treatment effect from a Poisson regression model.
Odds ratio due to treatment was estimated using a logistic regression model with fixed effects for treatment group and region.
p value based on Wald test of treatment effect from logistic regression model.
Hazard ratio due to treatment was estimated using a Cox proportional hazards model with fixed effects for treatment group and region; p value based on Wald test of treatment effect.
CC 7 + 8.75 mg/kg; pooled data from the two groups that received active treatment in CLARITY and active treatment in the Extension.
CP 3.5 + 5.25 mg/kg; pooled data from the two groups that received active treatment in CLARITY and placebo in the Extension.
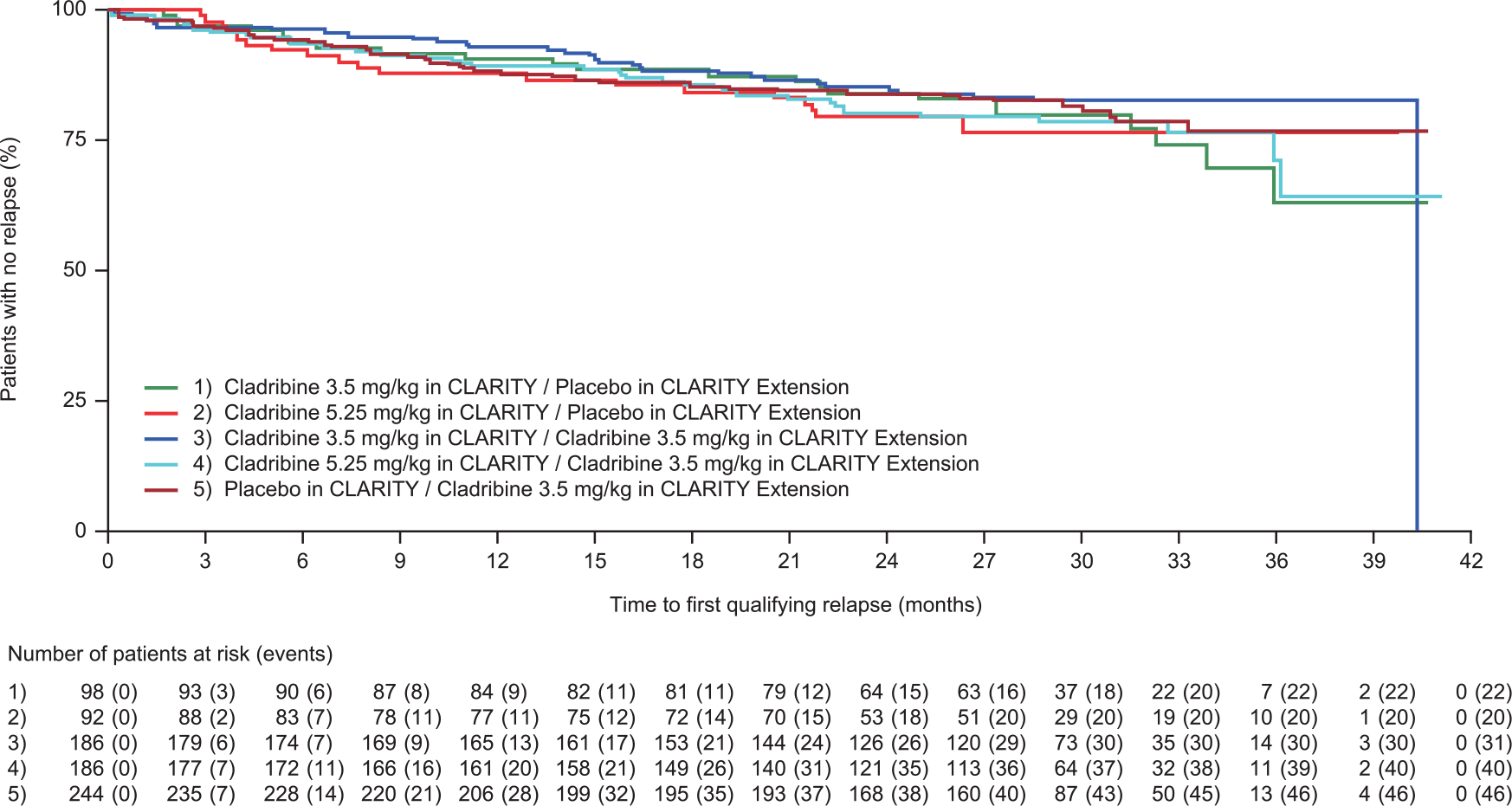
Time to first qualifying relapse during CLARITY Extension. Time in months shown in 30-day increments.
In an effort to evaluate the longitudinal effect of cladribine on ARR, we compared the same groups of patients from the end of CLARITY to the end of CLARITY Extension. The only difference was observed in the PC 3.5 mg/kg group. Compared with placebo treatment during CLARITY (ARR 0.26), a significant reduction was seen when these patients received cladribine tablets 3.5 mg/kg during the Extension (ARR 0.10; 60.7% relative reduction, p < 0.0001). This difference in ARR was comparable to that seen between patients who received placebo and the group treated with cladribine tablets 3.5 mg/kg during the CLARITY study (57.6% relative reduction, p < 0.001). 2 There were no clear relationships between the length of the gap period and the following: ARR, proportion of relapse-free patients, time to first qualifying relapse, or time to 3-month confirmed EDSS progression (data not shown). Data relating to MRI outcomes are the subject of a separate report.
Discussion
Overall, the safety and tolerability in CLARITY Extension were comparable with CLARITY, with lymphopenia the most frequently reported AE and the most frequent reason for treatment discontinuation, consistent with previous studies of cladribine tablets in RRMS. 2 In CLARITY Extension, the majority of lymphopenia events were classified as mild or moderate, and the majority of patients who experienced lymphopenia Grade ⩾ 3 actually experienced Grade 3 only. Furthermore, the majority of patients who experienced lymphopenia Grade ⩾ 3 recovered to Grade 0–1 lymphopenia by the end of the Extension. With the higher cumulative doses, the percentage of patients with Grade ⩾ 3 lymphopenia increased along with the time to recovery. Post-hoc analysis showed that in patients treated with cladribine tablets 3.5 mg/kg in CLARITY and cladribine tablets 3.5 mg/kg in CLARITY Extension according to the treatment guidelines (i.e. Grade 0 lymphocyte count at the start of CLARITY and Grade 0–1 at the start of Years 2, 3, and 4), no cases of Grade 4 lymphopenia were seen at the end of any treatment year. Furthermore, >85% of patients had recovered to Grade 0–1 lymphopenia by the end of each treatment year (not shown). The incidence of thrombocytopenia with cladribine tablets in CLARITY Extension was rare and similar to that in CLARITY, in which the only Grade 3 event was observed in the placebo group, in contrast to experience with higher relative doses of intravenous cladribine.2–4
No formal analysis of the association of infections with severity of lymphopenia was conducted for CLARITY Extension because of the potentially confounding effect from baseline status of study participants (i.e. pre-treated patients may have been lymphopenic for differing durations) and the overall small numbers of events available for the analysis. Although the rate of herpes zoster infections was highest (4.8%) in the group that received the greatest cumulative dose of cladribine (CC 8.75 mg/kg), the group that received the next highest cumulative dose (CC 7 mg/kg) showed a rate of herpes zoster infections (1.1%) similar to or lower than the groups that received placebo in the Extension (CP 3.5 and 5.25 mg/kg, 2.0% and 1.1%, respectively). In an integrated analysis of safety data from the development program of cladribine in MS focusing on infections in association with severe lymphopenia, the incidence of herpes zoster (reported as an AE of special interest) was higher in the period of treatment in which Grade 3 or 4 lymphopenia occurred until recovery to Grade 1, compared to the time when the patients were not experiencing Grade 3 or 4 lymphopenia. 5 Patients receiving cladribine tablets 3.5 mg/kg had adjusted AEs incidences of herpes zoster per 100 patient years (95% confidence interval (CI)) of 2.16 (0.90–5.19) during the period with Grade 3/4 lymphopenia, compared to 0.75 (0.50–1.12) without lymphopenia. 5 There were no reports of PML during this study. It should be noted that despite observed cases of PML in patients treated with other formulations of cladribine for non-MS conditions, 6 no cases of PML have been reported in clinical studies of patients with MS treated with cladribine tablets.
In this study, there was no clustering of particular malignancies with a common etiology, and no hematological malignancies commonly associated with immunosuppression were seen. Overall, 1.4% of patients from the safety population developed a malignancy or unspecified tumor. Of interest, a recent independent meta-analysis of Phase 3 clinical trials showed that there was no significant difference in the malignancy rate observed with cladribine tablets in the CLARITY study compared with other studies of DMDs in MS and did not support an increased risk of malignancy with doses of cladribine used in the CLARITY study. 7 In addition, Pakpoor et al. 7 noted that the incidence in the CLARITY study placebo group was exceptionally low. In a separate analysis, malignancy incidences in the cladribine program were compared to data from a reference general population as included in the GLOBOCAN epidemiological database (for method used and information on non-melanoma skin cancer, which is not included in GLOBOCAN, please see Supplementary Materials). The analysis of the standardized incidence ratio (SIR) resulted in an overall malignancy rate in cladribine-exposed subjects compared to a matched reference population of 1.06 (95% CI: 0.70; 1.55) indicating that the rate of malignancies in cladribine-treated patients was not higher than would have been expected in the general population. These findings support the need for more extensive registry studies to avoid potential confounding by low placebo group events and to more precisely determine the risk of malignancies with DMDs. It is reassuring that this study showed no evidence of increased incidence of malignancy over time (Supplementary Table 3).
Patients who received placebo in CLARITY had an increased ARR, a lower relapse-free rate, and increased risk of 3-month confirmed EDSS progression when compared with patients receiving cladribine tablets during CLARITY, 2 underlining the rationale for earlier treatment with cladribine tablets. CLARITY Extension indicates that treatment with cladribine tablets over 2 years, followed by 2 years of placebo, produced a durable clinical response (as measured from the end of CLARITY to the end of the Extension period): the response achieved at the end of CLARITY in the proportion of patients free from relapses or sustained disability progression was maintained in the Extension. Importantly, there was no evidence of increased clinical activity (i.e. disease rebound) after treatment discontinuation (e.g. in the CP 3.5 mg/kg group), as reported for other therapies in MS with a direct effect on lymphocytes.8,9 The gap period between the CLARITY and Extension treatment periods (median duration 40.3 weeks) along with the SUPF indicate that efficacy was maintained for prolonged periods for some patients (up to 6.5 years since the beginning of the CLARITY study and almost 5.5 years since the last administration of cladribine tablets), which is a clear indicator of the treatment effects based on cladribine’s mode of action.
One limitation of the Extension study is that patient numbers were restricted by enrolling patients from CLARITY; consequently, it was not powered for efficacy analyses. Also, the gap between CLARITY and the Extension was variable. However, since the gap was similar between treatment groups, the duration is expected to have had a minor influence on comparative outcomes. Because treatment with cladribine tablets leads to lymphocyte reduction and/or other noticeable differences in hematological parameters, it was essential that the treating physician but not the evaluating physician had access to such laboratory data in order to make safety assessments. In this case, although treating physicians may become functionally unblinded to treatment assignment (a challenge that applies in clinical trials of other DMDs), the blinding of evaluating physicians was maintained.
Overall, the safety and tolerability profile of cladribine tablets seen in this and previous studies,2,10,11 and the clinical benefits observed in CLARITY Extension, indicate a favorable risk–benefit ratio in RRMS. The short-duration dosing of cladribine tablets in Years 1 and 2 resulted in a durable clinical response and provided substantial “treatment-free” periods for at least two additional years with no requirement for a DMD; various efficacy analyses indicate that the clinical benefits of cladribine tablets may persist beyond 2 years of observation. On average, there was no significant incremental benefit from additional treatment with cladribine tablets in Years 3 and 4, following the initial courses received in Years 1 and 2 of CLARITY. The short-duration posology and favorable risk–benefit profile of cladribine tablets treatment may facilitate treatment adherence, a significant challenge for patients with MS. 12
Footnotes
Acknowledgements
The authors would like to thank the patients, investigators, co-investigators involved in the trial, as well as study teams at the participating centers, at Merck KGaA, Darmstadt, Germany, and Merck Switzerland. The authors also thank Bettina Stubinski for study coordination; Nathalie Lachenal (previously of Merck Switzerland, Geneva, Switzerland), Sanjeev Anand (Merck KGaA, Darmstadt, Germany) for monitoring and assessing safety data; and Emily Martin (EMD Serono, Inc., Billerica, MA, USA) and Patrick Engrand (Cytel, Inc., Geneva, Switzerland) for data statistical analyses. In addition, the authors thank Phil Jones of inScience Communications, Chester, UK, for medical writing support, funded by Merck KGaA, Darmstadt, Germany. Clinical trial registration: NCT00641537.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.G. has received speaker honoraria and consulting fees from Abbvie, Atara Bio, Almirall, Bayer Schering Pharma, Biogen Idec FivePrime, GlaxoSmithKline, GW Pharma, Merck, Pfizer, Inc., Protein Discovery Laboratories, Teva Pharmaceutical Industries, Ltd, Sanofi Genzyme, UCB, Vertex Pharmaceuticals, Ironwood, and Novartis; and has received research support unrelated to this study from Biogen Idec, Merck, Novartis, and Ironwood. P.S.S. has served on advisory boards for Biogen, Merck, Novartis, Teva, MedDay Pharmaceuticals, and GSK; on steering committees or independent data monitoring boards in trials sponsored by Merck, Teva, GSK, and Novartis; has received speaker honoraria from Biogen Idec, Merck, Teva, Sanofi-Aventis, Genzyme, and Novartis. His department has received research support from Biogen, Merck, Teva, Novartis, Roche, and Genzyme. S.C. has received honoraria for lectures/consultations from Merck, Bayer HealthCare, Sanofi-Aventis, Neurology Reviews, Biogen Idec, Teva Pharmaceuticals, and Actinobac Biomed, Inc.; and has served on advisory boards for Bayer HealthCare, Merck, Actinobac Biomed, Teva Pharmaceuticals, and Biogen Idec; and received grant support from Bayer HealthCare. K.R. has received honoraria for lectures and steering committee meetings from EMD Serono, Biogen Idec, Sanofi-Aventis, Genzyme, Novartis, Teva Neurosciences, Acorda, and Roche/Genentech. P.R. has received honoraria for lectures/steering committee meetings from Merck, Biogen Idec, Bayer Schering Pharma, Boehringer Ingelheim, Sanofi-Aventis, Genzyme, Novartis, Teva Pharmaceutical Industries, and Serono Symposia International Foundation. G.C. has received consulting fees from Novartis, Teva Pharmaceutical Industries, Ltd, Sanofi-Aventis, Merck, Receptos, Biogen Idec, Genentech–Roche, and Bayer Schering; lecture fees from Novartis, Teva Pharmaceutical Ind., Ltd, Sanofi-Aventis, Merck, Biogen Dompè, Bayer Schering, and Serono Symposia International Foundation; and trial grant support from Novartis, Teva Pharmaceutical Ind., Ltd, Sanofi-Aventis, Receptos, Biogen Idec, Genentech–Roche, Merck, Biogen Dompè, and Bayer Schering. F.D. and A.K.A. are employees of EMD Serono, Inc., a business of Merck KGaA, Billerica, MA, USA. P.V. has received honoraria or consulting fees from Biogen, Sanofi Genzyme, Bayer, Novartis, Merck, GSK, Roche, and Almirall; and research support from Biogen, Sanofi Genzyme, Bayer, and Merck.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by EMD Serono, Inc., a business of Merck KGaA, Darmstadt, Germany (in the United States), and Merck Serono SA, Geneva, an affiliate of Merck KGaA Darmstadt, Germany (rest of the world).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
