Abstract
Background:
Natalizumab is an effective treatment in relapsing-remitting multiple sclerosis (MS). Mainly because of the risk of progressive multifocal leukoencephalopathy (PML), a substantial proportion of John Cunningham (JC) virus–positive patients switch to fingolimod. Previous reports show a clear benefit when the duration of a washout (WO) period of natalizumab is 0–3 months in comparison to longer WO periods. However, there is no consensus regarding the optimal duration of a WO period under 3 months.
Objective:
We compared MS disease activity after different WO periods. In addition, we investigated several factors that possibly influence recurrence of disease activity, including serum natalizumab concentration and lymphocyte counts.
Methods:
From a prospective observational cohort study of natalizumab-treated patients, we selected 52 patients who switched to fingolimod. We divided the patients in three groups (<6 weeks, 6–8 weeks, >8 weeks WO). Serum natalizumab concentration and lymphocyte count were assessed during and after natalizumab treatment.
Results:
Patients with a WO period of >8 weeks had a significant higher recurrence of disease activity (odds ratio, 6.8; 95% confidence interval, 1.4–32.8) compared to patients with a WO period of <6 weeks. Serum natalizumab concentration and lymphocyte count did not predict recurrence of disease activity.
Interpretation:
A short WO period decreases the risk of recurrence of disease activity. The possible impact of a short WO period on the risk of carry-over PML in JC virus–positive patients remains uncertain.
Introduction
Natalizumab, a humanized monoclonal antibody targeting the α-4-integrin receptor, is an effective treatment in relapsing-remitting multiple sclerosis (RRMS). 1 Unfortunately, the risk of progressive multifocal leukoencephalopathy (PML), an opportunistic brain infection caused by the John Cunningham (JC) virus, complicates the long-term use of natalizumab in JC virus–positive patients. 2 Because of the risk of PML, patients often switch to an alternative disease-modifying therapy like fingolimod, a sphingosine 1-phosphatereceptor modulator. Natalizumab limits lymphocyte transport over the blood–brain barrier, whereas fingolimod prevents recirculation of lymphocytes from the lymph nodes. The different immunosuppressive pathways and the fact that natalizumab may be detectable in the serum up to 6 months after discontinuation, may theoretically increase the risk of opportunistic infections like PML. 3 Therefore, it has been suggested to consider a washout (WO) period between natalizumab discontinuation and fingolimod initiation. However, the length of the WO period remains a point of discussion as the increase of disease activity after natalizumab discontinuation is a well-known and clinically relevant issue.4,5 Recent publications have reported an increase of disease activity with longer WO periods, resulting in an advised WO period shorter than 3 months.6,7 However, even though current evidence is pointing towards the superiority of very short WO periods,8,9 uniformity in how short the WO period should be within the timeframe of 2–3 months is still lacking.
Apart from the WO period, disease breakthrough after discontinuation of natalizumab may also depend on natalizumab serum concentration which may vary largely between patients and are shown to correlate with disease activity.10,11 Another important factor may be the extent and swiftness by which fingolimod succeeds to reduce the number of circulating lymphocytes. Whereas natalizumab is associated with a stable increase of peripheral lymphocytes,12 –14 sphingosine 1-phosphatereceptor modulation caused by fingolimod aims to lower amounts of circulating lymphocytes.15,16 The interplay between drug-induced increased and subsequently decreased lymphocyte count may have impact on the recurrence of disease activity.
The aim of our study was to investigate the correlation between the recurrence of disease activity within 6 months of natalizumab discontinuation with different WO periods (<6 weeks, 6–8 weeks, and >8 weeks) and the influence of serum natalizumab concentration and lymphocyte count during treatment in RRMS patients switching from natalizumab to fingolimod.
Methods
Patients
This study is part of an ongoing prospective observational cohort study conducted at the VU University Medical Center including all RRMS patients treated with natalizumab. Patients in this cohort are at least annually subjected to a brain magnetic resonance imaging (MRI) and clinical testing including Expanded Disability Status Scale (EDSS). 17
We included all patients who switched from natalizumab to fingolimod between February 2013 and August 2016. We excluded patients who did not start fingolimod within 6 months after discontinuation of natalizumab, or without a brain MRI within 6 months. All patients in the observational cohort gave written informed consent for the use of the clinical, laboratory, and imaging data for research and teaching purposes. A waiver from our local institutional review board (IRB) stating that the requirements of the Medical Research Involving Human Subjects Act did not apply and that official IRB approval was not mandatory for the use of these data was obtained.
Measurement of clinical disease activity
The main outcome of this study was disease activity within 6 months of natalizumab discontinuation. We assessed disease activity according to the 2013 Lublin criteria. 18 Following these criteria, disease activity was defined as “active” when the patient had a clinical relapse and/or the occurrence of contrast-enhancing T1 and/or new or enlarging T2 lesions on brain MRI.
We collected data of the patients’ clinical files regarding relapses 1 year before, during, and 1 year after natalizumab use. A relapse was defined as a period of new neurological deficit, existing longer than 24 hours, and not attributable to another cause than multiple sclerosis (MS).
Clinical disability was defined by the Expanded Disability Status Scale (EDSS). We collected the EDSS scores at time of natalizumab initiation and at time of switch (initiation of fingolimod) from the clinical database of the observational cohort. The EDSS was assessed by certified raters.
MRI acquisition
The MRI acquisition and the scan interval was performed according to the magnetic resonance imaging in multiple sclerosis (MAGNIMS) guidelines on MS diagnosis and patient monitoring.19,20 MRI examination was performed annually during natalizumab usage in patients with a negative JC virus serostatus. In JC virus-seropositive patients (all patients described in this study), the frequency of brain MRI increased to every 3 months, which is our current pharmacovigilance protocol suggested by different international panel guidelines.20,21 Following natalizumab discontinuation, a follow-up scan was performed after 3 and 6 months because of the risk of carry-over PML. The scan performed within the first 3 months of fingolimod treatment was used as the baseline scan of fingolimod treatment. MRI scanning was performed at a 1.5 T or 3 T scanner (Siemens Magnetom Impact, Erlangen, Germany) including three-dimensional (3D) fluid-attenuated inversion recovery and axial proton density (PD)/T2-weighted sequences. We assessed brain MRI for active new or enlarging T2 lesions (new or enlarging) and contrast-enhancing lesions in case gadolinium was administered.
Blood sampling
Blood samples were routinely obtained every 3 months before natalizumab infusion. Blood samples during fingolimod treatment were also routinely obtained following our protocol: at baseline, month 1 and 3-monthly from month 3 during the first year. Hematological count including lymphocytes were measured in these samples in the clinical chemistry laboratory at the VU University medical center. Serum was subsequently stored at −80°C at the VU University medical center Biobank. In order to assess clearance of natalizumab, we measured natalizumab concentration before discontinuation of natalizumab and within 6 months after discontinuation. Natalizumab concentration before discontinuation was measured right before a new infusion (trough concentration). Intra-individual natalizumab trough concentration are stable in a 4-week-interval regimen. 22 Sera were sent to Sanquin Laboratory to measure natalizumab concentration using a cross-linking assay using polyclonal rabbit anti-natalizumab F(ab)2 fragments for capture and a mouse anti-IgG4 monoclonal antibody for detection. This method is described in more detail elsewhere. 11 Serum JC virus status and indexes were obtained every 6 months via Unilabs, Copenhagen, Denmark.
Statistical analysis
In order to assess the association between disease activity and WO period, we divided the WO period into three groups: a WO period <6 weeks, 6–8 weeks, and >8 weeks. Differences between the distinct groups were analyzed using a logistic regression model and potential confounders (sex and age) were analyzed via assessing the change of the regression coefficient of the risk factor of interest, before and after adjusting for the potential confounder. If the regression coefficient changed more than 10%, the confounder was considered relevant and will be adjusted for. No confounders were found and the statistical results did not change when comparing adjusted regression models with unadjusted regression models.
The association between disease activity and concentration of natalizumab was studied using a multivariable logistic regression model. Results are displayed as an odds ratio (OR) with the confidence interval (CI) per unit decrease, adjusted for WO period.
For comparing mean natalizumab concentration at certain time points we used the independent sample T test, as concentrations were normally distributed. For correlation analysis between lymphocyte count and disease activity, a logistic regression model was used. Potential confounders were analyzed, but not found. In this analysis, the mean lymphocyte count over the last year natalizumab treatment was used as continuous variable. Calculations were performed using SPSS version 22.0 (Windows). A p-value <0.05 was considered significant for main effects.
Results
Demographic characteristics
In our observational cohort, 60 patients switched from natalizumab to fingolimod because of the risk of developing PML. Six patients were excluded because of a WO period longer than 6 months. Two patients were excluded because of incomplete imaging during follow-up.
Of the remaining 52 patients, baseline demographics are described in Table 1.
Demographic characteristics.
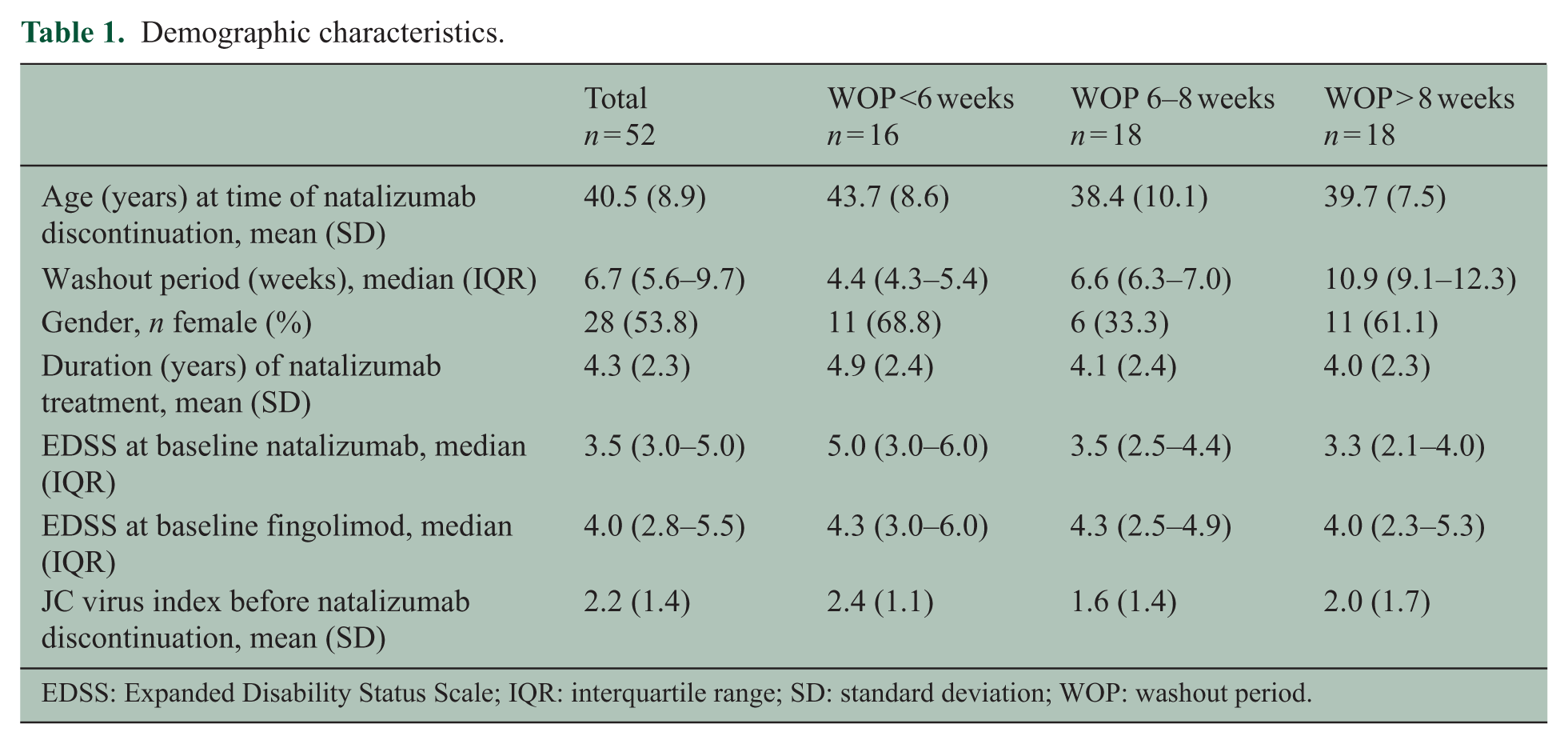
EDSS: Expanded Disability Status Scale; IQR: interquartile range; SD: standard deviation; WOP: washout period.
Disease activity
A total of 20 patients (38.5%) experienced disease activity within 6 months of natalizumab withdrawal. In total, 17 patients (32.7%) had activity on brain MRI (active T2 lesions and/or gadolinium-enhancing lesions) and 6 patients (11.5%) experienced a clinical relapse within 6 months of natalizumab discontinuation. No more than one relapse was reported per patient. All relapses occurred after at least 3 months of natalizumab discontinuation with a median delay of 3.9 months (interquartile range (IQR), 3.7–4.6 months). None of the patients developed PML.
Disease activity increased with longer WO periods compared to shorter periods (see Figure 1 and Table 2). The patients with a WO period >8 weeks showed a significant increase in disease activity with an OR of 6.8 (95% CI, 1.4–32.8; p = 0.02) when compared to the group of patients with a <6 weeks WO period. Comparing the patients with a WO period >8 weeks to a WO period 6–8 weeks, there was no significant difference in disease activity with an OR of 3.1 (95% CI, 0.8–12.5; p = 0.1).
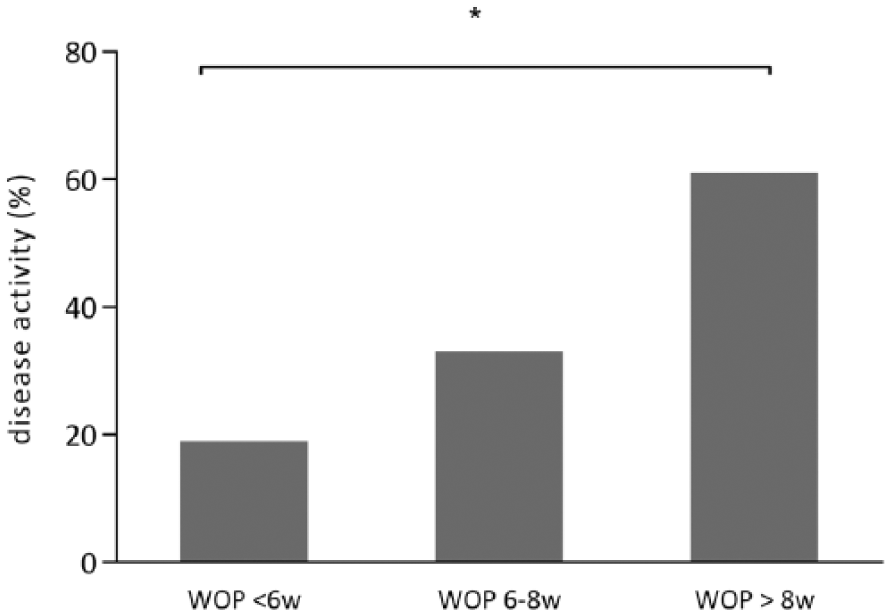
Disease activity and washout period.
Disease activity and washout period.
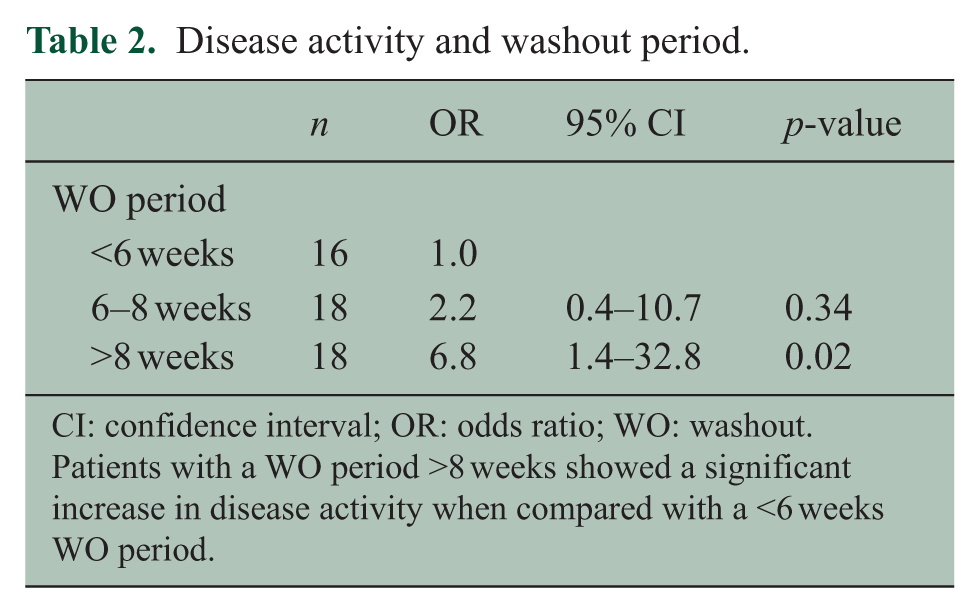
CI: confidence interval; OR: odds ratio; WO: washout.
Patients with a WO period >8 weeks showed a significant increase in disease activity when compared with a <6 weeks WO period.
In the group with a WO period >8 weeks (n = 18), four patients had a WO of 12–24 weeks. When comparing <6 weeks WO to a WO of 8–12 weeks (n = 14), disease activity remained significantly correlated with a longer WO period (OR, 7.8; 95% CI, 1.5–41.2; p = 0.02).
Subgroup analysis comparing disease activity in the group with the shortest WO (<4 weeks) to 8–12 weeks WO shows comparable results in favor of the shortest washout (OR, 7.2; 95% CI, 1.1–47.9; p = 0.04).
Of the six patients experiencing a relapse, three patients had a WO period >8 weeks, one patient had a WO period of 6–8 weeks, and two had a WO period of <6 weeks. In total, 17 patients experienced radiological activity. The percentage of patients with radiological disease activity increased with longer WO periods, that is, 11.1% in WO period <6 weeks, 33.3% in WO period 6–8 weeks, and 50.0% in WO period of >8 weeks. Comparable with disease activity, radiological activity only appeared statistically significant when comparing the group with a WO of <6 weeks with a WO of >8 weeks (OR, 7.0; 95% CI, 1.2–40.1; p = 0.03). In the 17 patients experiencing radiological activity, the median number of new T2 lesions was 3 (range, 1–24). Seven patients had a T2 increase of ⩾5, five of these patients (71.4%) had a WO period of >8 weeks. The remaining two had a WO of <6 weeks and 6–8 weeks.
Before natalizumab treatment, the annual relapse rate (ARR) was 1.5 ± 1.0, whereas the ARR under natalizumab treatment was 0.05 ± 0.1 and for the year after natalizumab discontinuation 0.20 ± 0.5. The ARR before natalizumab treatment did not correlate with disease activity after 6 months of natalizumab discontinuation (OR per unit increase in ARR, 1.1; 95% CI, 0.6–1.8; p = 0.9). No correlation was found between the duration of natalizumab use and disease activity (OR per additional year of natalizumab use, 1.0; 95% CI, 1.0–1.0; p = 0.1).
Natalizumab concentration
The mean natalizumab trough serum concentration during natalizumab treatment was 21.6 ± 12.1 µg/mL. Natalizumab concentration was not associated with disease activity within 6 months after discontinuation (OR, 1.0 after adjustment for the duration of the WO period; 95% CI, 1.0–1.1; p = 0.9). The 20 patients who experienced disease activity had similar natalizumab concentration during natalizumab treatment compared to patients who did not experience disease activity (mean concentration of, respectively, 21.1 ± 10.4 µg/mL and 21.9 ± 13.2 µg/mL, see Figure 2).
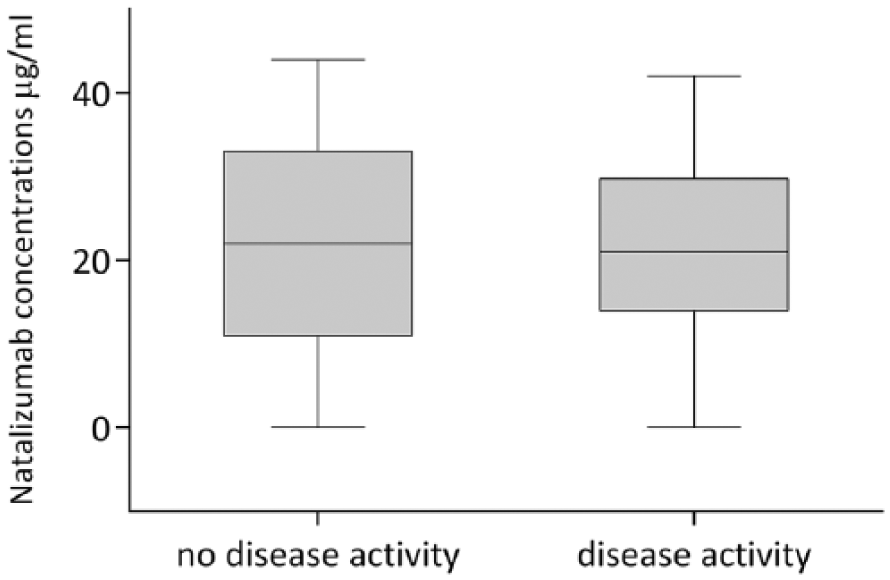
Natalizumab concentration and disease activity.
Of 42 patients (80.8%), a follow-up natalizumab concentration 4 weeks to 6 months after the last natalizumab infusion was available. When the mean concentration was assessed longitudinally, we observed an exponential decline of natalizumab concentration as expected. On average, 3 months after the last natalizumab infusion, the natalizumab concentration drops below therapeutic levels of 1 µg/mL. 23 In the six patients of whom we had a natalizumab concentration available 5–6 months after discontinuation, natalizumab was still measureable, although at very low values.
From 16 out of 20 patients who experienced disease activity, follow-up data on natalizumab concentration was available. Three months after natalizumab discontinuation, the patients with disease activity had a mean concentration of 0.9 µg/mL (n = 6) versus 4.2 µg/mL in the patients (n = 18) without disease activity (p = 0.3, Figure 3).
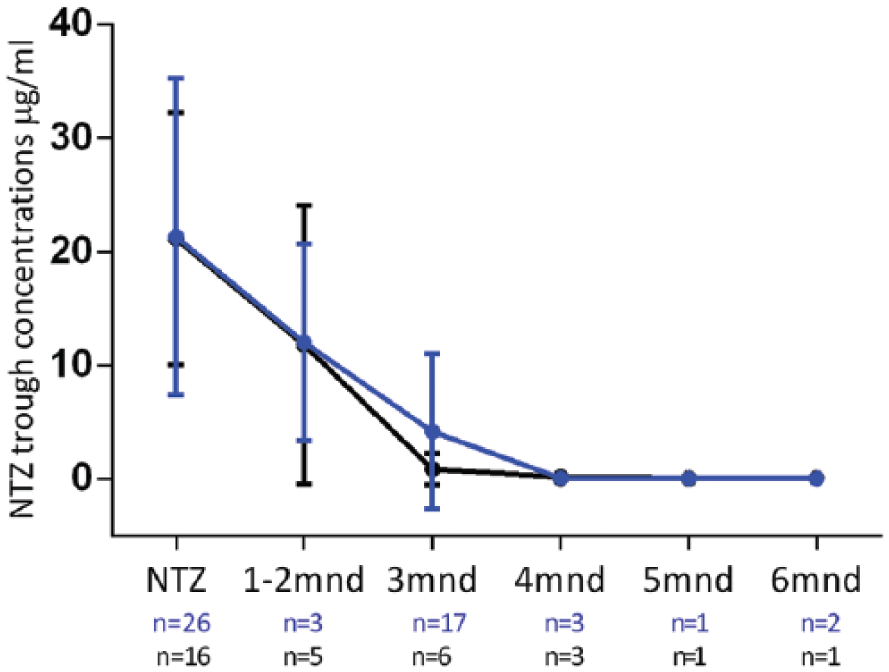
Longitudinal natalizumab concentration in patients with disease activity versus patients without disease activity.
Lymphocyte count and disease activity
Based on the last measured lymphocyte count during natalizumab, the patients were divided into two groups, one group with normal lymphocyte count (1.0–3.5 × 10E9/L) and the other with increased lymphocyte count (>3.5 × 10E9/L). Correcting for the WO period, we found no significant correlation for disease activity (OR, 1.0; 95% CI, 0.3–3.2; p = 0.9).
All patients showed an expected decrease in lymphocyte count at the moment of 3 months fingolimod use (mean difference of 2.8 ± 0.7 × 10E9/L, p < 0.01). In the two groups (normal and increased lymphocyte count), we investigated the relative difference of lymphocyte count between the last measured lymphocyte count under natalizumab and the lymphocyte count at approximately 3 months of fingolimod use. We found no significant difference in the relative change of lymphocyte count between the two groups (β = 0.03, p = 0.1, Figure 4(a) and (b)).
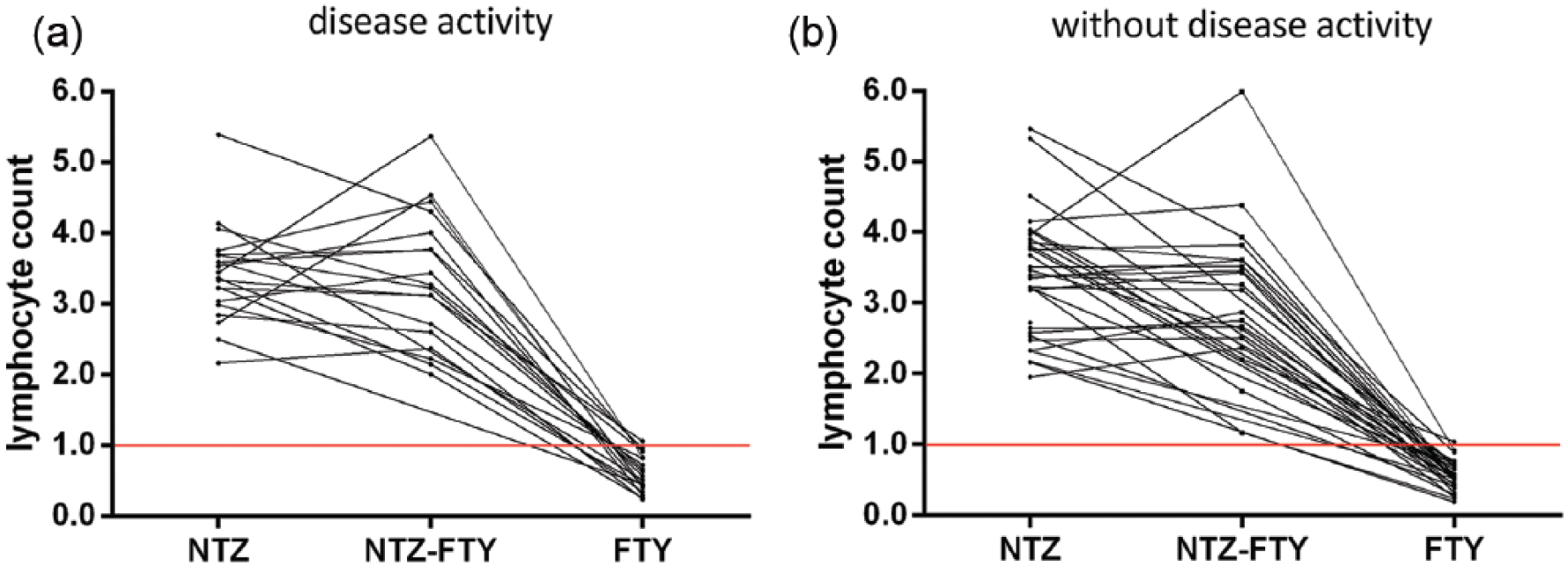
Lymphocyte count in patients (a) with disease activity versus (b) without disease activity.
Discussion
The main finding of this study is that switching patients from natalizumab to fingolimod with a WO period of more than 8 weeks increases the likelihood of recurrent disease activity almost sevenfold compared to patients with a WO period of less than 6 weeks.
Our results confirm earlier studies that report an increase of disease activity after 2–4 months of WO period in comparison to shorter WO periods.5–9,24 Ideally, to limit the risk of recurrence of disease activity, the WO period should be short enough to allow fingolimod to be clinically effective before natalizumab concentration drops under therapeutic levels. The downside of such a short WO period is the ongoing risk of PML in JC virus–positive patients after discontinuation of natalizumab and how fingolimod could possibly increase the risk of PML and negatively influence the course of this serious complication.
PML after discontinuation of natalizumab (carry-over PML) is rare with 15 reported cases to date. 25 PML under fingolimod monotherapy is also described but even rarer, with nine reported cases so far (Novartis safety update October 2016). All current literature regarding WO periods when switching from natalizumab to fingolimod recommend a delay of less than 3 months because of the risk of recurrence of disease activity.5–9,25 We estimate that the possible increased risk of PML with a decrease of 1–2 months of WO period does not outweigh the significant reduction of disease recurrence. Therefore, we recommend a WO period of less than 6 weeks when switching from natalizumab to fingolimod. Extra precautions regarding the risk of PML in JC virus–positive patients may be taken, such as a baseline MRI and repeated scans 3 and 6 months after cessation of natalizumab and timely testing of JC virus DNA in the cerebrospinal fluid in case of suspicious magnetic resonance (MR) activity. 25
Although intra-individual natalizumab trough concentration during natalizumab treatment is stable, concentrations vary widely between patients, presumably because of the difference in body mass index and metabolism. 22 In agreement with an earlier report, we found a large inter-individual variability of natalizumab trough concentration under natalizumab treatment. 22 We hypothesized that lower concentration would predispose to disease recurrence after natalizumab discontinuation. However, we found no significant difference in concentration under natalizumab treatment when comparing patients with disease activity and patients without disease activity, which is in agreement with a recently published study of 12 patients switching from natalizumab to fingolimod. 26 In our study, the possible explanation could be that the clinical disease activity appeared after 3 months of natalizumab discontinuation when concentration of the drug had already decreased under therapeutic levels. 5 When comparing longitudinal natalizumab concentration in patients with and without disease activity, the mean concentration at 3 months was lower in patients who did not experience disease activity (0.8 vs 4.2 µg/mL). However, no definite conclusions can be made because of limited sample size of the longitudinal concentrations. The α-4-integrin receptor on which natalizumab binds desaturates when natalizumab concentration decreases below 1 µg/mL. 23 Our longitudinal results show that on average, natalizumab concentration decreases under the therapeutic level 3–4 months after the last infusion.
Natalizumab inhibits the adhesion of lymphocytes to the endothelium and is associated with a stable increase of peripheral lymphocytes. 12 Yet increasing evidence shows that natalizumab also plays a role in attachment of hematopoietic precursor cells to bone marrow stromal cells and on that account facilitates the release of lymphocytes from the bone marrow.27,28 The return of disease activity after natalizumab discontinuation most likely reflects resumption of lymphocyte migration across endothelial membranes of the blood–brain barrier as natalizumab is cleared from the circulation. 29 Fingolimod is probably mainly clinically effective because it induces a systemic lymphopenia. When switching from natalizumab to fingolimod, we hypothesized that clinical effect of fingolimod might be delayed due to the relative lymphocytosis natalizumab causes in a proportion of patients. However, this study shows no correlation between lymphocyte count and disease activity. T cell subsets are unknown in this study, so describing the fingolimod-mediated changes on CD4+ and CD8+ T cells was not possible. The lack of correlation between an overall lymphocyte count and disease activity will not rule out the probability that the changes in subpopulations of lymphocytes (including CD4+/CD8+ ratio) may have impact on the recurrence of disease activity.
Our relatively small sample size warrants cautious interpretation of our data. To determine the optimal WO period in case of switching from natalizumab to fingolimod, larger randomized trials are needed, preferably comparing different WO periods, including one arm starting fingolimod immediately after the final natalizumab infusion. However, in future studies the influence of the WO period on the risk of PML will still be difficult to establish given the rarity of this serious complication.
In conclusion, we found that a WO period of less than 6 weeks is associated with a reduced recurrence of disease activity when compared to a WO period of 8 weeks. Natalizumab concentration and lymphocyte count during natalizumab treatment did not predict disease activity after natalizumab discontinuation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.E.L. reports a grant from Stichting MS research. Z.L.E.v.K. has nothing to disclose. I.D. reports a grant from Stichting MS research. L.J.B. has nothing to disclose. M.P.W. reports personal fees from Biogen, Novartis, Roche, and Genzyme. T.R. reports payment to institute for lectures from AbbVie, Pfizer, Regeneron, and a grant from Genmab. B.M.J.U. has received personal compensation for consulting from Biogen Idec, Genzyme, Merck Serono, Novartis, Roche, and Teva. J.K. has accepted speaker and consulting fees from Merck Serono, Biogen Idec, Teva, Genzyme, and Novartis (everything outside the submitted work).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
