Abstract
Background:
Treatment with MD1003 (high-dose biotin) showed promising results in progressive multiple sclerosis (MS) in a pilot open-label study.
Objective:
To confirm the efficacy and safety of MD1003 in progressive MS in a double-blind, placebo-controlled study.
Methods:
Patients (
Results:
A total of 13 (12.6%) MD1003-treated patients achieved the primary endpoint versus none of the placebo-treated patients (
Conclusion:
MD1003 achieves sustained reversal of MS-related disability in a subset of patients with progressive MS and is well tolerated.
Keywords
Introduction
Multiple sclerosis (MS) is the most common disabling neurological disease of young adults. 1 Both primary progressive multiple sclerosis (PPMS) and secondary progressive multiple sclerosis (SPMS) diseases are part of the same progressive disease spectrum which can be further split into ‘active’ and ‘not-active’ progressive disease based on the presence or absence of inflammatory activity demonstrated either clinically or radiologically.2,3 Disability worsening in not-active progressive MS is mainly due to chronic demyelination and mitochondrial dysfunction, both of which result in virtual hypoxia leading to axonal degeneration.4 –6 Although anti-CD20, mitoxantrone and interferons have shown some efficacy in patients with active progressive disease,7 –9 there is still no approved disease-modifying therapy (DMT) in patients with not-active progressive MS, which represents a significant unmet medical need. 10
MD1003 is an oral formulation of high-dose pharmaceutical-grade biotin (10,000 times the recommended daily intake) that recently demonstrated promising efficacy in patients with not-active progressive MS.
11
Over 90% of the patients (
Methods
Study design
Study MS-SPI was a 12-month randomised, double-blind, placebo-controlled trial followed by an open-label 12-month extension phase where all patients received MD1003. The study was conducted at 16 French MS reference centres in accordance with Good Clinical Practice. The study was approved by an independent ethical committee (Comité de Protection des Personnes Est 1, Dijon, France; approval number: CPP EST I: 2013/36) and was overseen by an independent data monitoring committee.
Participants
Eligible patients were 18–75 years old with PPMS or SPMS that fulfilled revised McDonald and Lublin criteria with clinical evidence of spastic paraparesis. Patients with clinical or radiological evidence of inflammatory activity within the previous year were excluded. Eligible patients had a baseline EDSS of 4.5–7 with evidence of disease progression during the previous 2 years (an increase of ⩾1 point if EDSS was 4.5–5.5 and ⩾0.5 point if EDSS was 6–7). Detailed eligibility criteria are provided in the supplementary materials. All participants provided written informed consent at enrolment.
Intervention
During a 12-month placebo-controlled phase, patients were randomised (2:1) to receive MD1003 (biotin 100 mg) or placebo orally thrice daily, stratified by study centre only. This was followed by MD1003 100 mg thrice daily for all patients for a further 12 months (extension phase). Treatments were provided in identical capsules containing the same quantity of tasteless white powder. Patients and investigators were masked to study treatment during the initial 12 months and remained blinded during the extension phase as to which treatment was administered during the first phase. As it was unlikely that the treating neurologist could differentiate active treatment from placebo, the treating and examining neurologist could be the same individual.
All concomitant medications were allowed throughout the study, including immune modulators and immunosuppressive drugs provided they were introduced at least 3 months before inclusion (⩾1 month for fampridine). Intravenous methylprednisolone without oral taper was allowed for MS relapse. Physical therapy was permitted, but an in-patient intensive physical therapy programme was not allowed in the 3 months prior to inclusion or during the trial because this could interfere with disability evaluation.
Outcomes
Neurological assessments were conducted by an examining neurologist every 12 weeks in the placebo-controlled phase. All evaluation scales used in the study are described in detail in the supplementary materials, and Figure S1 shows the timing of study assessments.
The primary endpoint was the proportion of patients with improvement of MS-related disability at month 9, confirmed at month 12. Improvement was defined as a decrease of ⩾0.5 point or ⩾1 point in EDSS (if baseline score was 6–7 or 4.5–5.5, respectively) or a ⩾20% decrease in timed 25-foot walk (TW25) time, compared with the best EDSS or TW25 value recorded at either the screening or the randomisation visit. Since the TW25 was performed twice per visit, the baseline value was the best out of four values obtained at screening or randomisation. EDSS was assessed using the Neurostatus EDSS (www.neurostatus.net) by EDSS raters qualified to Neurostatus level C. In line with Multiple Sclerosis Functional Composite guidelines, TW25 times were truncated to 180 seconds for times above 180 seconds or when the patient could not perform the test because of MS-related disability. The best of the two TW25 values achieved at each visit was recorded.
Secondary endpoints comprised mean change in EDSS from randomisation to month 12; mean clinical global impression of change scored by the clinician (clinician-assessed Clinical Global Impression Scale (CGI)) and patient (subject-assessed Clinical Global Impression Scale (SGI)) at month 12; and mean changes from randomisation in the 12-item Multiple Sclerosis Walking Scale (MSWS), Short Form 36 Health Survey (SF-36) subscores, TW25, modified Fatigue Impact Scale (MFIS), nine-hole peg test (9-HPT) and Kurtzke EDSS functional subscores. The proportion of patients with improvement of EDSS and TW25 values, stable EDSS or EDSS progression (⩾0.5 point or ⩾1 point if value at randomisation was 4.5–5.5) at month 9 (confirmed at month 12) was also assessed. Safety was investigated by comparing the incidence of adverse events (AEs) and laboratory or electrocardiogram (ECG) findings between study arms. Magnetic resonance imaging (MRI) investigations (3DT1, T2-spin–echo, T2-fluid attenuated inversion recovery (FLAIR) and post-gadolinium T1 sequences) were conducted in 74 patients from six centres who are participating in a long-term ancillary study using non-conventional MRI sequences.
Statistical analysis
No controlled data for clinical outcomes with MD1003 were available at the time of study design for sample size calculation. Data from seven MS patients treated with MD1003 for >3 months were used to estimate that >40% of MD1003-treated patients might show a reduction in disability. While progressive MS patients typically do not exhibit a confirmed decrease in EDSS scores, 13 and as a conservative approach, it was estimated that 10% of the placebo-treated patients might improve. It was estimated that at least 105 patients (70 in the MD1003 arm and 35 in the placebo arm) would be required to detect a difference in the proportion of patients demonstrating clinical improvement with 90% power at a 5% two-sided significance level.
Differences in proportions were compared between arms with Fisher’s exact test and differences in means with the non-parametric Mann–Whitney
No imputation method was used to handle missing data for the primary endpoint as this could have overestimated the number of responding patients. The ‘last observation carried forward’ approach was used to handle missing data for all supportive analyses, except for the EDSS change, EDSS progression and TW25 change where no imputation was used.
Results
Between October 2013 and January 2014, 166 patients were screened and 154 were randomised to MD1003 (
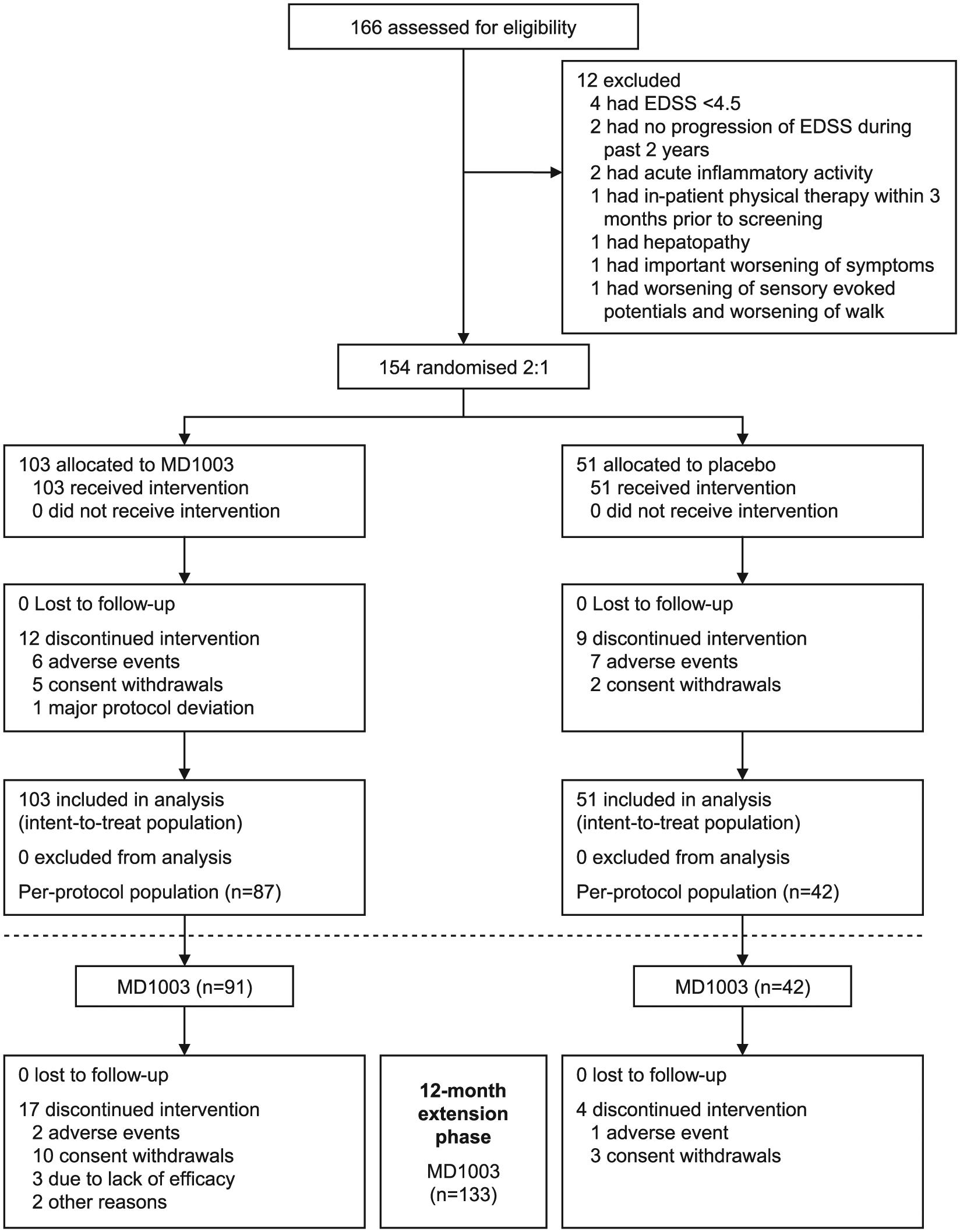
Screening, enrolment, randomisation and follow-up of study patients. The ITT population was defined as all patients who were assigned to a treatment arm. The per-protocol population was defined as all patients of the ITT population with assessments of EDSS or TW25 at screening, baseline, month 9 and month 12 without major protocol deviations.
Baseline demographic characteristics of the intention-to-treat population.
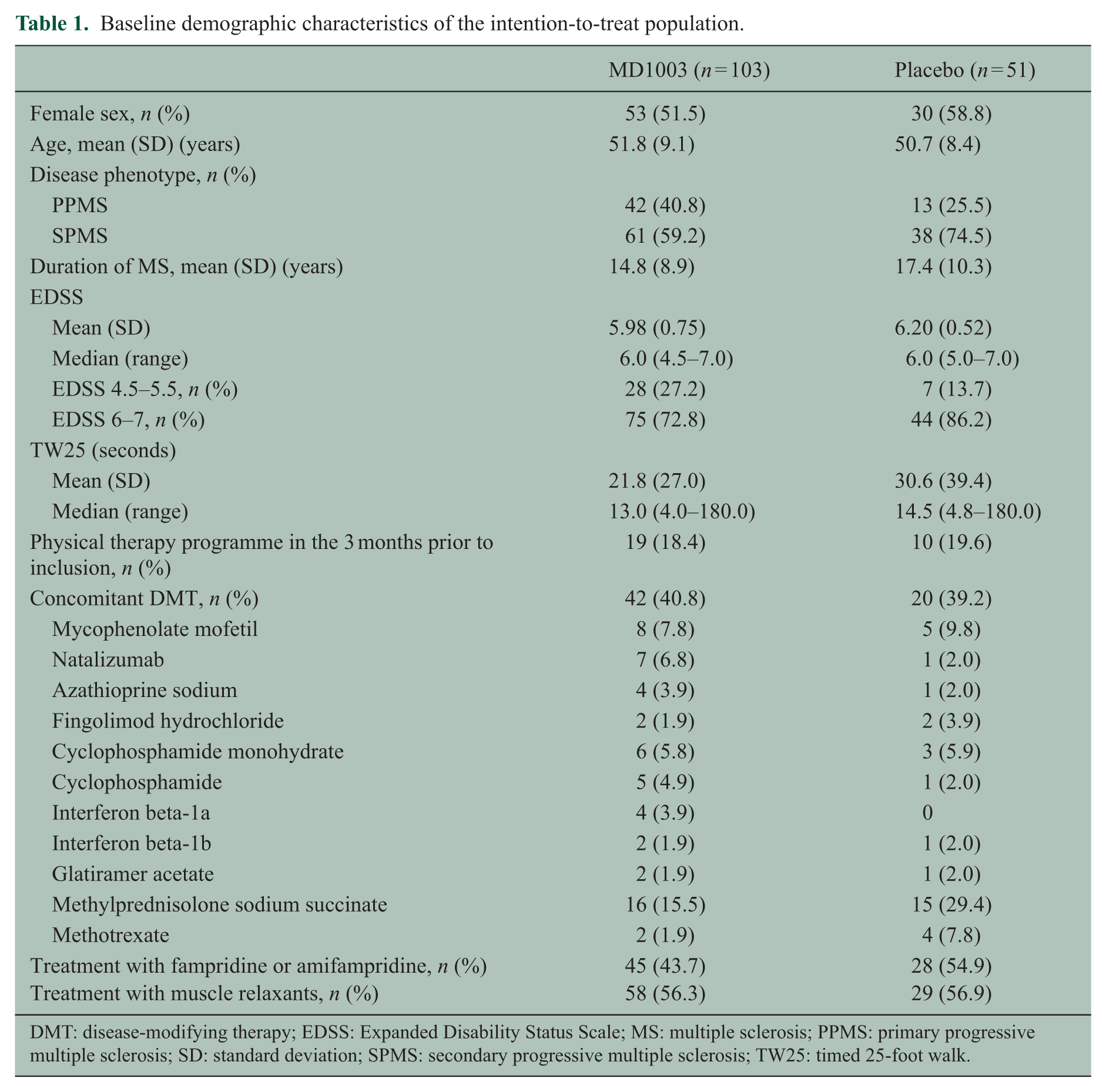
DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; MS: multiple sclerosis; PPMS: primary progressive multiple sclerosis; SD: standard deviation; SPMS: secondary progressive multiple sclerosis; TW25: timed 25-foot walk.
During the placebo-controlled phase, 12 (11.7%) MD1003-treated patients discontinued treatment (Figure 1) including six patients withdrawn due to AEs (suicide, mucocutaneous rash, asthenia, muscle spasms, abdominal pain and libido disorder). Nine (17.6%) placebo-treated patients discontinued treatment including seven due to AEs (overdose, dry mouth, intracranial haemorrhage, mental disorder, extrasystoles, muscle spasticity and pregnancy). During the extension phase, 17 of the 91 (18.6%) patients in the MD1003 > MD1003 arm discontinued treatment (2 due to AEs: weight loss and breast cancer) as did 4 of the 42 (9.5%) patients in the placebo > MD1003 arm (1 due to AE: myopathy).
Primary endpoint
A total of 13 (12.6%; 95% confidence interval (CI): 6.9%–20.6%) patients treated with MD1003 had a reduction in MS-related disability at month 9, confirmed at month 12, compared with none in the placebo arm (Figure 2;
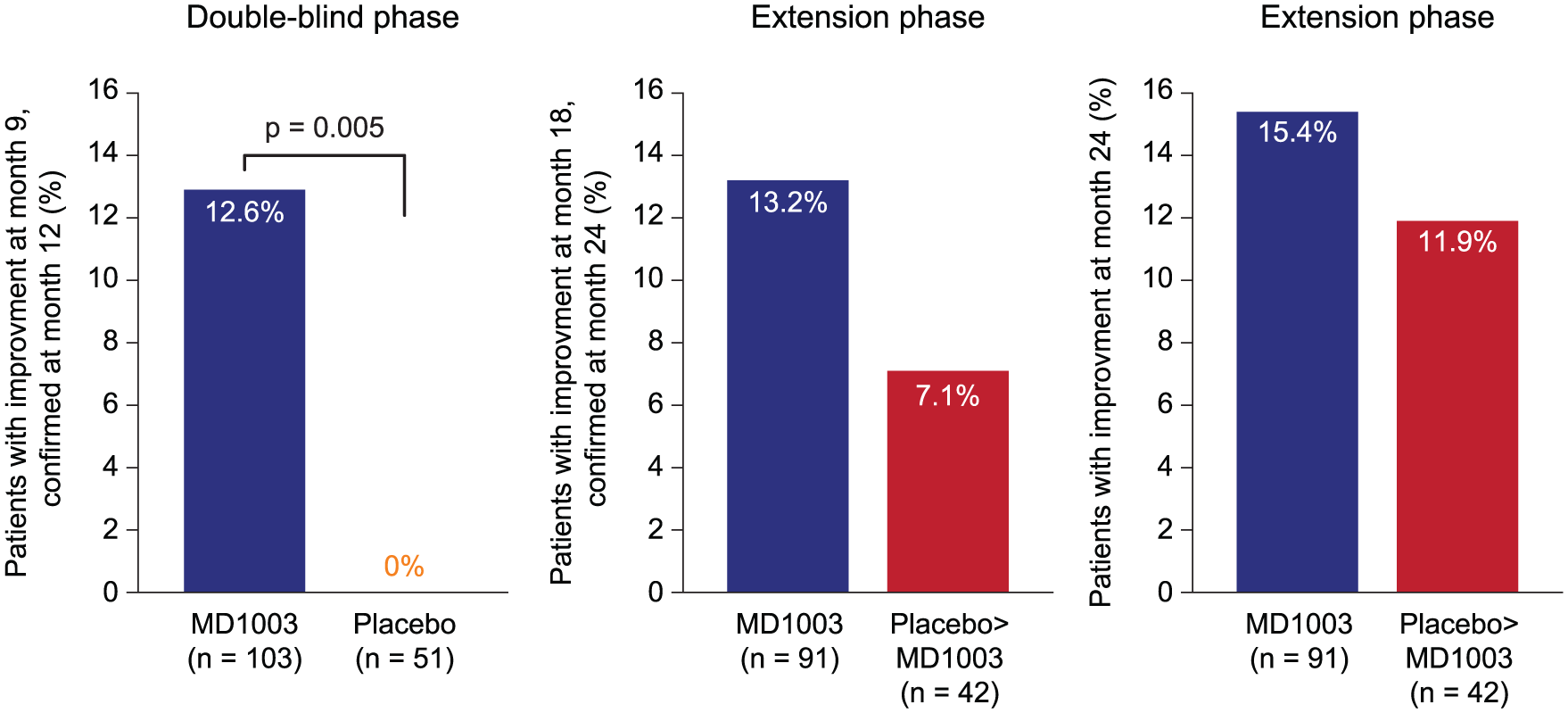
Proportion of patients with reversal of MS-related disability. Reversal of disability was defined as improvement of EDSS or TW25 values confirmed at the next visit (except for month 24 where no subsequent visit was available) compared with best respective values recorded at either the screening or the randomisation visits.
Baseline characteristics of the 13 patients who achieved the primary endpoint of the study.
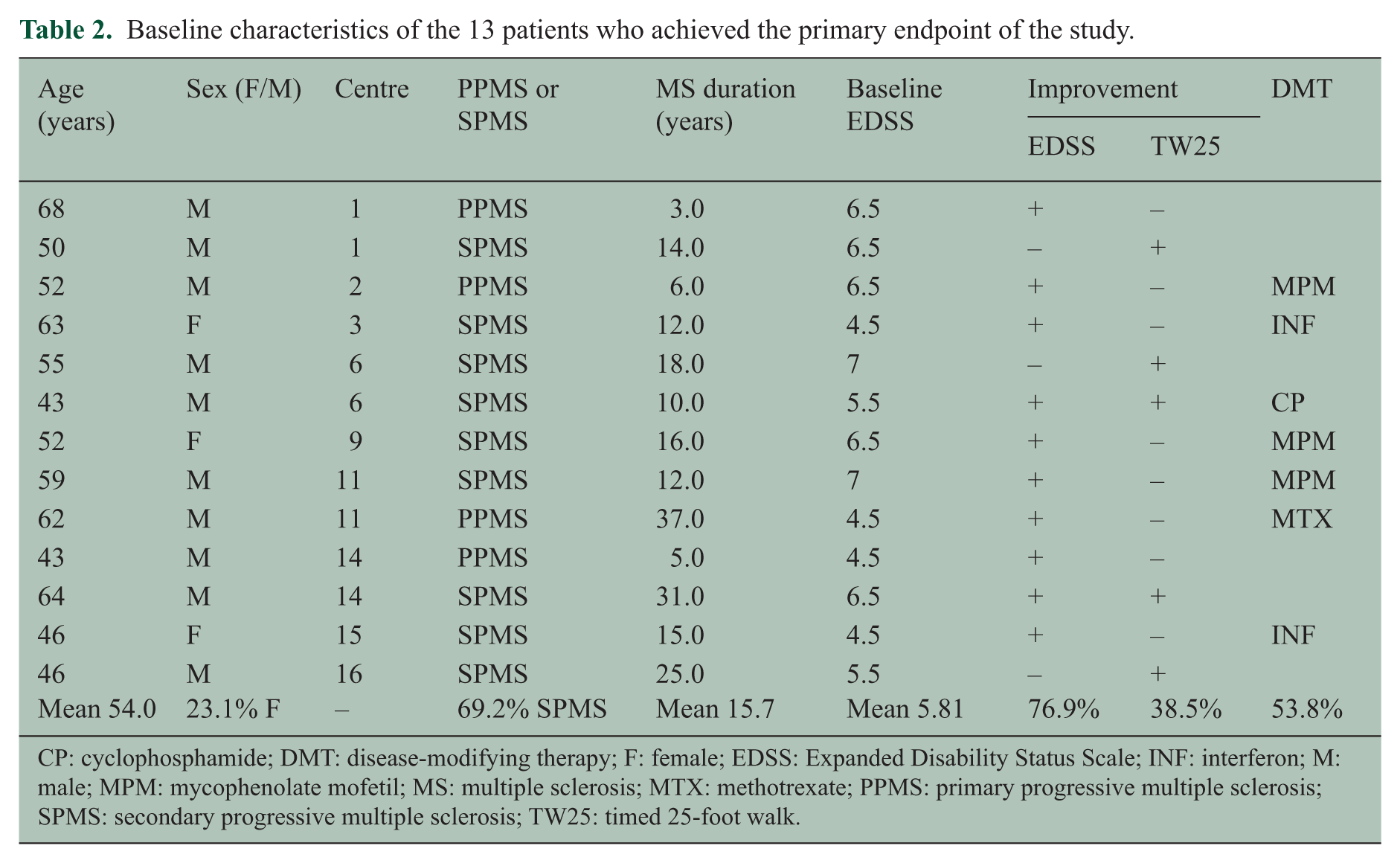
CP: cyclophosphamide; DMT: disease-modifying therapy; F: female; EDSS: Expanded Disability Status Scale; INF: interferon; M: male; MPM: mycophenolate mofetil; MS: multiple sclerosis; MTX: methotrexate; PPMS: primary progressive multiple sclerosis; SPMS: secondary progressive multiple sclerosis; TW25: timed 25-foot walk.
A total of 12 of the 91 (13.2%; 95% CI: 7.0%–21.9%) patients initially treated with MD1003 had reduced MS-related disability at 18 months (confirmed at 24 months; Figure 2), including 10 of the 13 (77%) patients who responded during the placebo-controlled phase. After switching to MD1003, 3 of the 42 (7.1%; 95% CI: 1.5%–19.5%) patients in the placebo > MD1003 arm had reduced disability at 18 months (confirmed at 24 months). At month 24, 14 of the 91 (15.4%) patients in the MD1003 > MD1003 arm and 5 of the 42 (11.9%) patients in the placebo > MD1003 arm had reduced MS-related disability.
Secondary endpoints
Table 3 shows the results of secondary endpoints. The proportion of patients with EDSS progression at month 9 (confirmed at month 12) was 13.6% in the placebo arm and 4.2% in the MD1003 arm (
Secondary endpoints (according to the protocol pre-defined rank).
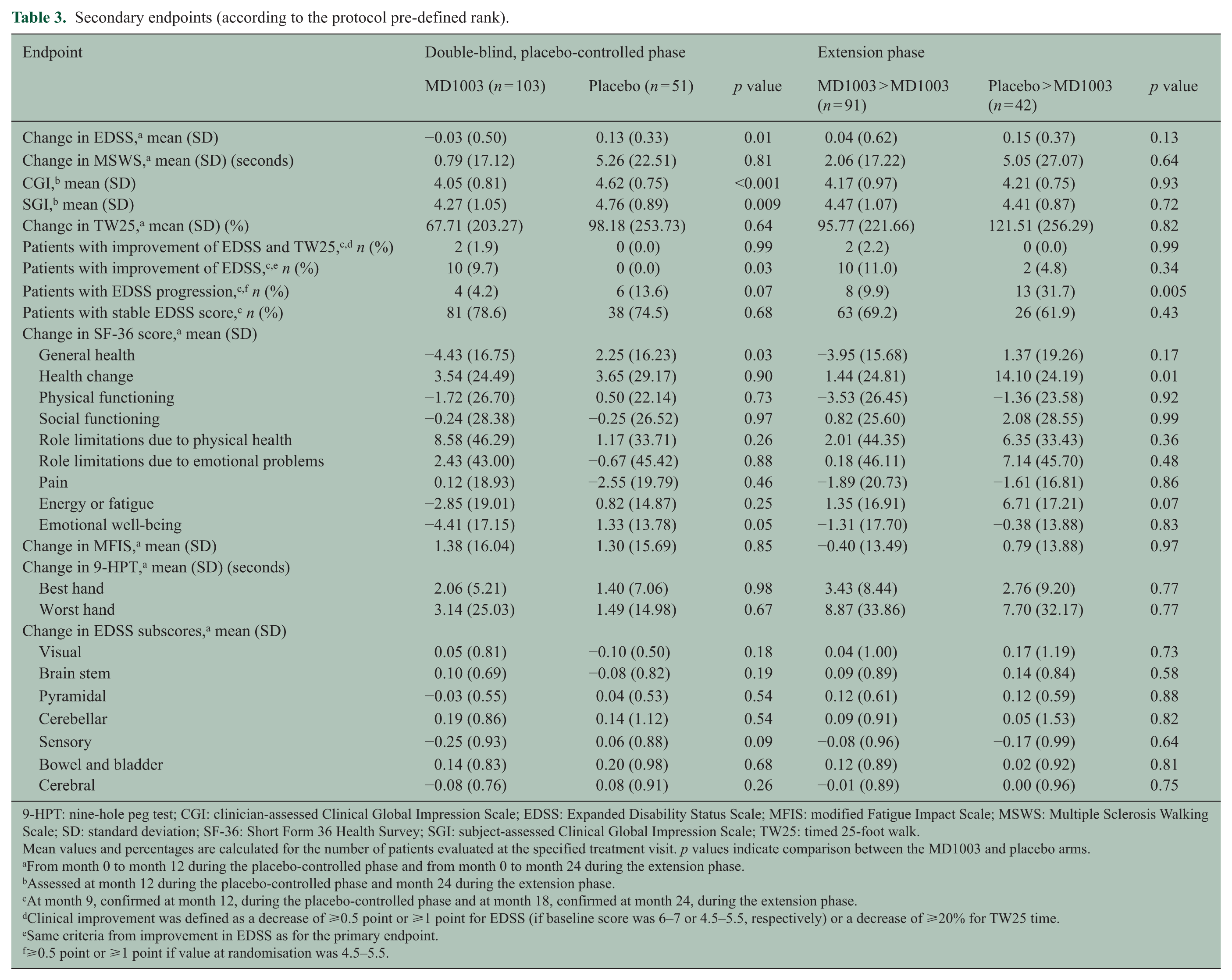
9-HPT: nine-hole peg test; CGI: clinician-assessed Clinical Global Impression Scale; EDSS: Expanded Disability Status Scale; MFIS: modified Fatigue Impact Scale; MSWS: Multiple Sclerosis Walking Scale; SD: standard deviation; SF-36: Short Form 36 Health Survey; SGI: subject-assessed Clinical Global Impression Scale; TW25: timed 25-foot walk.
Mean values and percentages are calculated for the number of patients evaluated at the specified treatment visit.
From month 0 to month 12 during the placebo-controlled phase and from month 0 to month 24 during the extension phase.
Assessed at month 12 during the placebo-controlled phase and month 24 during the extension phase.
At month 9, confirmed at month 12, during the placebo-controlled phase and at month 18, confirmed at month 24, during the extension phase.
Clinical improvement was defined as a decrease of ⩾0.5 point or ⩾1 point for EDSS (if baseline score was 6–7 or 4.5–5.5, respectively) or a decrease of ⩾20% for TW25 time.
Same criteria from improvement in EDSS as for the primary endpoint.
⩾0.5 point or ⩾1 point if value at randomisation was 4.5–5.5.
At month 12, the mean (±standard deviation (SD)) EDSS decreased from baseline in the MD1003 arm (−0.03 ± 0.50) but increased at the expected rate in the placebo arm (+0.13 ± 0.33,
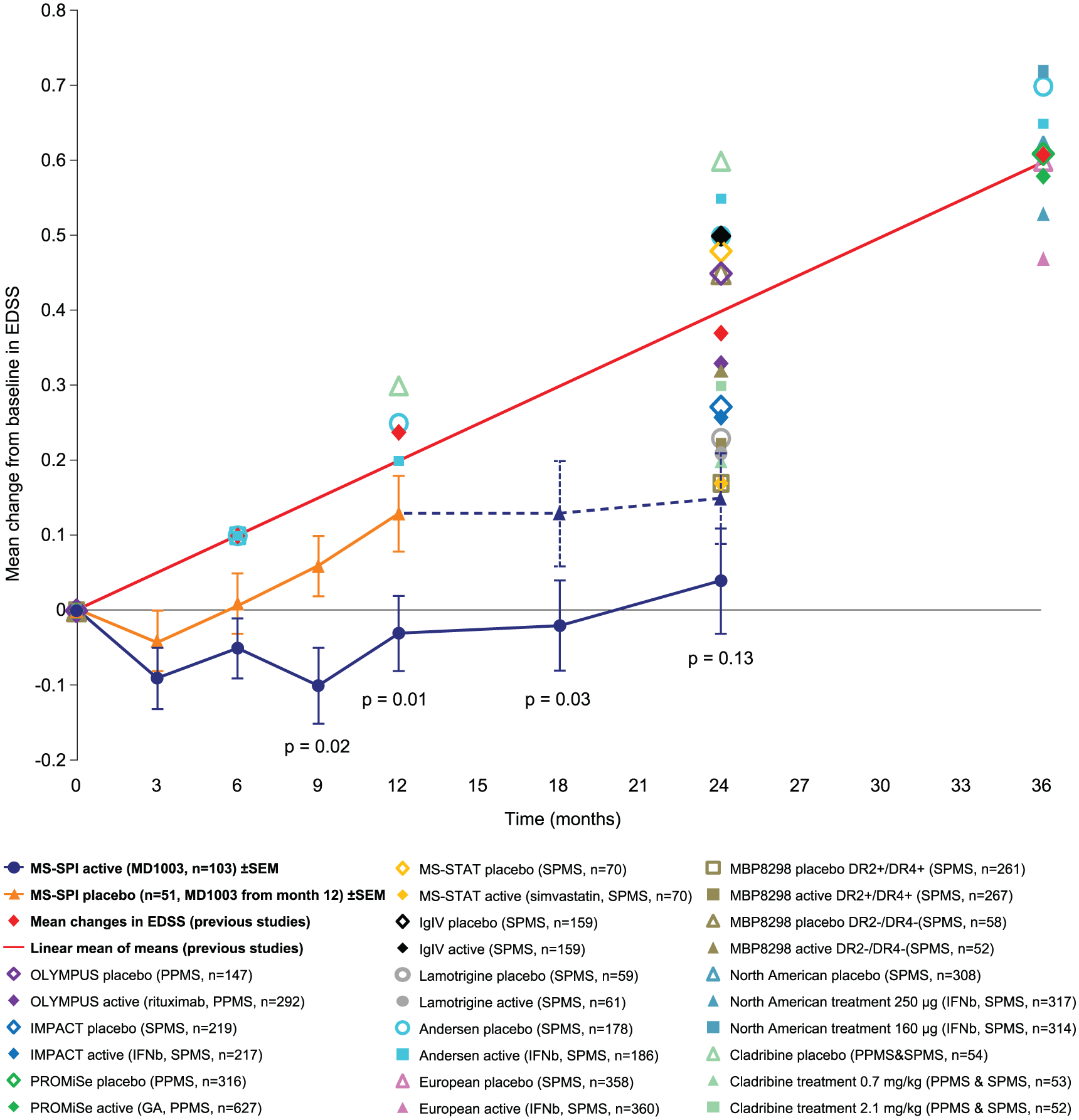
Mean change from baseline in EDSS during the 12-month double-blind placebo-controlled phase and 12-month extension phase. This figure also shows the mean change from baseline in EDSS (represented as the mean of means) as reported in other published placebo-controlled studies of pharmacological agents in PPMS or SPMS (>6000 patients in total).7,9,14 –22
Mean TW25 times increased more in the placebo arm than in the MD1003 arm, but this difference was not significant (Table 3). The median TW25 time remained relatively constant in MD1003-treated patients throughout the study (Figure S3). Other post hoc analyses showed that in the MD1003 group, nine (8.7%) patients had >20% improvement in TW25 times at month 9, confirmed at month 12, compared with the last pre-treatment visit versus none in the placebo group (
MD1003-treated patients had significantly lower CGI scores (mean = 4.05 ± 0.81 vs 4.62 ± 0.75;
Safety
The incidence and distribution of AEs during the placebo-controlled phase were similar between arms (Table 4 and Table S2). Most reported AEs were mild or moderate. The only serious AE reported in more than one patient was MS relapse (five (4.9%) MD1003-treated patients and four (7.8%) placebo-treated patients). One serious AE was reported to be possibly related to MD1003 (mucocutaneous rash), although subsequent chamber patch testing was negative for biotin. One death by suicide occurred during the study in the MD1003 arm and was not considered treatment related. AEs reported during the extension phase included MS relapses in 7 of the 91 (7.7%) patients who initially received MD1003 and in 2 of the 42 (4.8%) patients who initially received placebo. A total of two neoplasms not considered treatment related were found in two patients initially receiving MD1003.
Adverse events reported during the study.
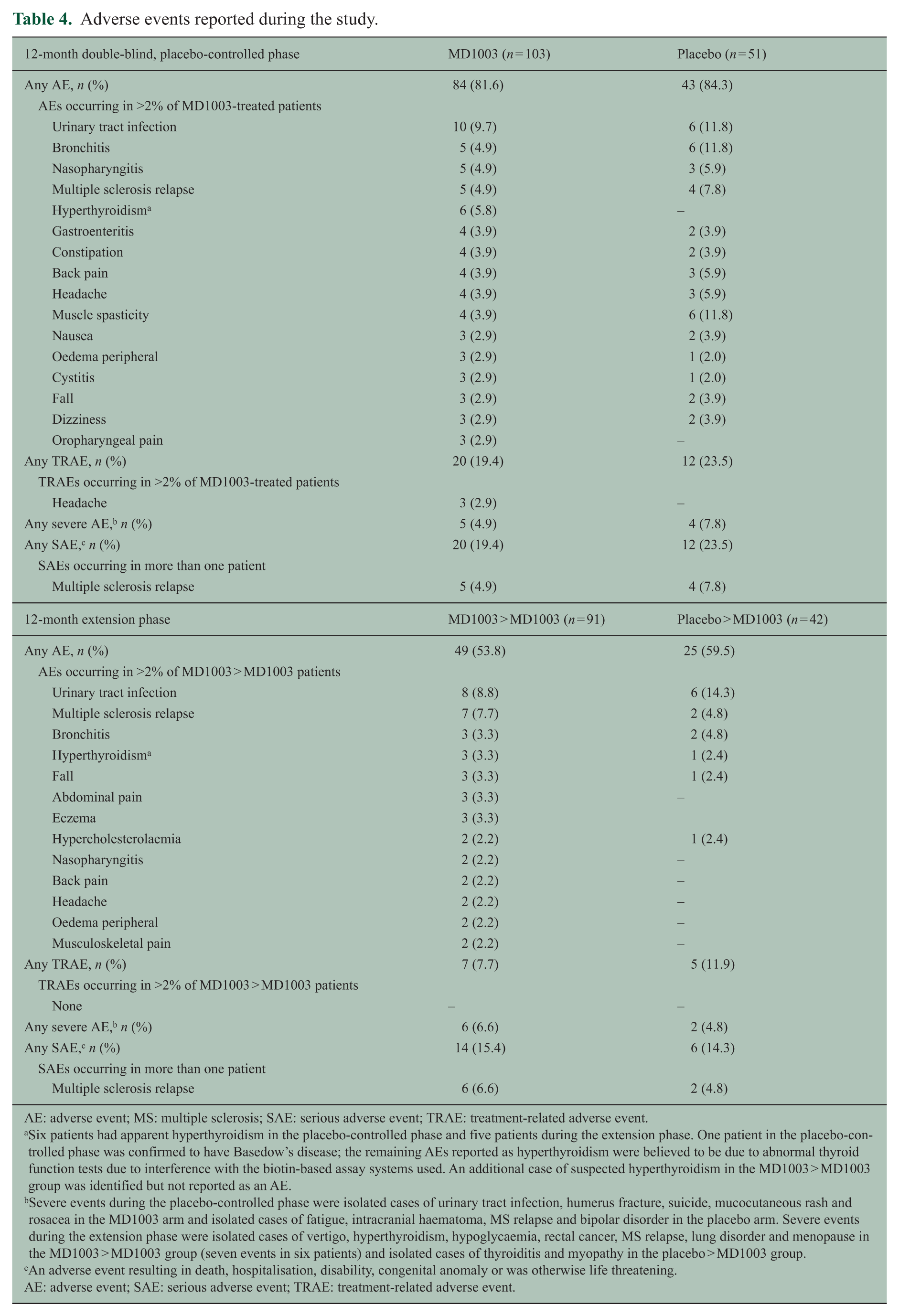
AE: adverse event; MS: multiple sclerosis; SAE: serious adverse event; TRAE: treatment-related adverse event.
Six patients had apparent hyperthyroidism in the placebo-controlled phase and five patients during the extension phase. One patient in the placebo-controlled phase was confirmed to have Basedow’s disease; the remaining AEs reported as hyperthyroidism were believed to be due to abnormal thyroid function tests due to interference with the biotin-based assay systems used. An additional case of suspected hyperthyroidism in the MD1003 > MD1003 group was identified but not reported as an AE.
Severe events during the placebo-controlled phase were isolated cases of urinary tract infection, humerus fracture, suicide, mucocutaneous rash and rosacea in the MD1003 arm and isolated cases of fatigue, intracranial haematoma, MS relapse and bipolar disorder in the placebo arm. Severe events during the extension phase were isolated cases of vertigo, hyperthyroidism, hypoglycaemia, rectal cancer, MS relapse, lung disorder and menopause in the MD1003 > MD1003 group (seven events in six patients) and isolated cases of thyroiditis and myopathy in the placebo > MD1003 group.
An adverse event resulting in death, hospitalisation, disability, congenital anomaly or was otherwise life threatening.
AE: adverse event; SAE: serious adverse event; TRAE: treatment-related adverse event.
Six cases of apparent hyperthyroidism (low thyroid-stimulating hormone (TSH) and high triiodothyronine or thyroxine) were recorded as AEs in the MD1003 arm during the placebo-controlled phase and in five additional patients during the extension phase (including one patient who initially received placebo). This was determined to be due to biotin interference with the thyroid function laboratory tests that used a biotinylated antibody. 23 Only one of the six patients with apparent biological hyperthyroidism during the double-blind phase was confirmed to have Basedow’s disease based on histological examination after thyroidectomy.
At month 12, MRI examination identified new MS-specific lesions in 11 of the 47 (23.4%) MD1003-treated patients and 3 of the 23 (13.0%) placebo-treated patients (
Discussion
The primary endpoint of most trials in progressive MS is disability progression. As MD1003 is believed to target the nervous system, and not the immune system, MS-SPI was instead designed and powered with disability improvement as the primary criterion. Results show that MD1003 can reverse MS-related disability in 12.6% of the patients, while this was observed in none of the placebo patients (
MD1003 also reduced the proportion of patients with confirmed EDSS progression. As a result of both disability improvement and decreased progression, the mean EDSS in the active treatment arm was almost unchanged over 24 months compared with baseline. Importantly, the mean EDSS increase observed under placebo was halted after patients were switched to MD1003 during the extension phase, with reversal of MS-related disability observed in some patients. However, the higher level of disability reached by these patients compared with those in the active treatment arm suggests that delaying treatment initiation could lead to a loss of chance. This observation would favour a DMT effect of MD1003 rather than a simple symptomatic effect. On the other hand, the early improvement already observed after 3 months would favour a symptomatic effect. This early response was also observed in the placebo group (not confirmed at subsequent visits), which could also indicate an initial placebo effect.
EDSS progression observed in the placebo arm of MS-SPI was consistent with previous placebo data in progressive MS trials (Figure 3).7,9,14 –22 The effect of MD1003 on EDSS change is strikingly different from observations in published placebo-controlled studies in progressive MS (Figure 3)7,9,14 –22 which showed that mean EDSS scores under both active drug and placebo increased over the treatment period, irrespective of the differences in design and treatment interventions.
The improvements in CGI and SGI scores with MD1003 compared with placebo support the clinical meaningfulness of the results observed on EDSS. The low responsiveness of the TW25 endpoint was surprising, as this parameter is usually sensitive to change. 24 Other post hoc analyses support a superiority of MD1003 over placebo on the TW25 measure which is consistent with results seen for EDSS.
MD1003 was well tolerated over 24-month treatment, consistent with data from the pilot study. 11 The suicide in the MD1003 arm was judged not to be treatment related. This individual had severe and progressing MS and a past history of anxiety disorder treated with an anxiolytic. Individuals with MS have a higher rate of suicide than the general population. 25
More new or enlarging MRI lesions occurred in the MD1003 arm than in the placebo arm during the double-blind phase. Future trials should include careful assessment of MRI activity to rule out an unwanted pro-inflammatory effect of biotin. There are no published data on animal models concerning a potential pro-inflammatory effect of high-dose biotin. Of note, MRI was not used to assess efficacy because of the relatively short trial duration which was not powered to see an effect on brain volume.
One important safety observation from this study was that MD1003 interferes with biotin-based laboratory tests. This results in falsely low values with sandwich immunoassays and false elevations with competitive immunoassays. 23 This may potentially affect a wide range of laboratory assays, particularly the thyroid panel. This finding led to the recommendation to use non-biotin-based laboratory assays in patients taking MD1003.
Our study has some methodological limitations. The relative short follow-up of the placebo-controlled phase did not allow us to fully assess the potential disease-modifying effect of high-dose biotin. The number of responding patients was lower than our initial expectations, possibly because of the very stringent primary endpoint used in this study. The absence of separate examining and treating physicians could have theoretically perturbed the study blinding (although biotin has no recognisable flavour or AEs). Of note, the success of blinding was not specifically assessed in this study. There were also some potential imbalances between the two treatment groups at baseline.
In conclusion, our data suggest that targeting neuron or oligodendrocyte metabolism with high-dose biotin may represent an effective and safe treatment for patients with progressive MS.
Footnotes
Acknowledgements
We thank Yann Poulouin, MSc, the statistician at Biotrial, Rennes, France, who was responsible for the statistical analysis of study data and Jacques Ropers, PharmD, URC Ambroise-Paré, Boulogne, France, for his help with the methodology and interpretation of data. Editorial support for the preparation of this manuscript was provided by Jamie Ashman, PhD, of Prism Ideas GmbH, Basel, Switzerland, and funded by MedDay Pharmaceuticals. We also thank the patients and their families, the MS-SPI study coordinators, and all the staff at the study sites. Trial registration: EudraCT:2013-002113-35.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Prof. Ayman Tourbah has received consulting fees, travel grants and reports institutional financial compensation for patient visits during the trial from MedDay Pharmaceuticals and consulting and lecturing fees, travel grants and research support from Biogen Idec, Sanofi-Genzyme, Novartis, Merck Serono, Teva Pharmaceuticals and Roche. Prof. Gilles Edan reports personal fees from Biogen Idec and Novartis and grants and personal fees from Merck Serono, Teva, Sanofi and Bayer, outside the submitted work. Prof. Sandra Vukusic reports clinical trial support from MedDay Pharmaceuticals, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi-Aventis, Teva Pharmaceuticals and Genzyme; advisory or consultancy fees from Biogen Idec, Genzyme, Merck Serono, Novartis, Roche, Sanofi-Aventis and Teva Pharmaceuticals; and invited congress support from Biogen Idec, Merck Serono, Novartis and Genzyme. Prof. Jérôme De Sèze reports clinical trial support from MedDay Pharmaceuticals, Biogen Idec, Merck Serono, Bayer, LFB, UCB, AB Sciences, Sanofi-Aventis, Teva Pharmaceuticals and Genzyme; advisory or consultancy fees from Biogen Idec, Merck Serono, Bayer, LFB, Sanofi-Aventis, Teva Pharmaceuticals, Almirall and Allergan; and invited congress support from Biogen Idec, Merck Serono, Bayer, LFB, Sanofi-Aventis, Teva Pharmaceuticals, Genzyme and Allergan. Dr Olivier Gout reports personal fees and non-financial support from Biogen Idec, Teva Pharmaceuticals, Genzyme and Novartis and personal fees from Merck, outside the submitted work. Prof. Gilles Defer reports personal fees from Biogen Idec, Novartis, Sanofi-Aventis, Genzyme and Teva Pharmaceuticals; grants from Novartis, Merck Serono and Teva Pharmaceuticals; and funding for travel from Merck Serono, Biogen Idec, Guerbet, Sanofi-Aventis, Novartis, Genzyme and Teva Pharmaceuticals, outside the submitted work. Prof. David-Axel Laplaud reports personal fees from Biogen and Teva Pharmaceuticals and grants and personal fees from Novartis and Genzyme, outside the submitted work. Prof. Thibault Moreau reports personal fees and non-financial support from Biogen, Novartis, Sanofi-Genzyme, Bayer, Merck, Teva Pharmaceuticals and Almirall, outside the submitted work. Prof. Bruno Brochet received grants from MedDay Pharmaceuticals during the conduct of the study; personal fees and non-financial support from Biogen Idec, Novartis and Genzyme; grants from Merck Serono; grants, personal fees and non-financial support from Teva Pharmaceuticals; and grants and non-financial support from Bayer, outside the submitted work. Dr Frédéric Sedel is CEO and a shareholder at MedDay Pharmaceuticals (the study sponsor). Prof. Jean Pelletier has received consulting and lecturing fees, travel grants and unconditional research support from Biogen Idec, Sanofi-Genzyme, Novartis, Merck Serono and Teva Pharmaceuticals. Dr Christine Lebrun-Frenay, Prof. Michel Clanet, Dr Caroline Papeix, Prof. Marc Debouverie, Prof. Pierre Clavelou and Prof. Pierre Labauge have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by MedDay Pharmaceuticals which also provided MD1003.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
