Abstract
Alemtuzumab is a humanized monoclonal antibody directed against CD52 to deplete circulating T and B lymphocytes; lymphocyte depletion is followed by a distinctive pattern of T- and B-cell repopulation, changing the balance of the immune system. This review reports the efficacy and safety findings of the phase 2 CAMMS223 trial and the phase 3 CARE-MS I and II trials investigating alemtuzumab for the treatment of active relapsing–remitting MS. Alemtuzumab, administered intravenously, was shown to improve relapse rate versus subcutaneous interferon beta-1a in patients who were treatment-naive (CAMMS223 and CARE-MS I) or had relapsed on prior therapy (CARE-MS II), and to reduce sustained accumulation of disability (CAMMS223 and CARE-MS II). Important adverse events were infusion-associated reactions, serious infections and autoimmune events. A safety monitoring program allowed for early detection and management of autoimmune events. Recommendations for the monitoring of adverse events are made. Alemtuzumab’s mechanism of action, pharmacodynamics and opportunities for future research are discussed.
Introduction
Multiple sclerosis is a chronic and degenerative autoimmune inflammatory disease leading to demyelination and damage of axons in the brain and spinal cord.1,2 Alemtuzumab is a humanized (immunoglobulin G1 (IgG1) isotype) monoclonal antibody that was recently approved in the European Union (EU), Australia and Latin America for adult patients with relapsing–remitting multiple sclerosis (RRMS) with active disease defined by clinical or imaging features, and is also approved in Canada for patients who had an inadequate response to interferon-beta or other disease-modifying agents. It is directed against the glycoprotein CD52, a protein present on the surface of mature lymphocytes, and it depletes circulating T and B lymphocytes, thought to be critical mediators of MS.3,4 After lymphocyte depletion, a distinctive pattern of T- and B-lymphocyte repopulation occurs over time, changing the balance of the immune system.5,6
In this review, trial results that are pertinent to its approval for the treatment of active RRMS are summarized. Recommendations for the monitoring of adverse events are made. Additionally, alemtuzumab’s mechanism of action, pharmacodynamics and opportunities for future research are discussed.
Clinical findings
Phase 3 trials
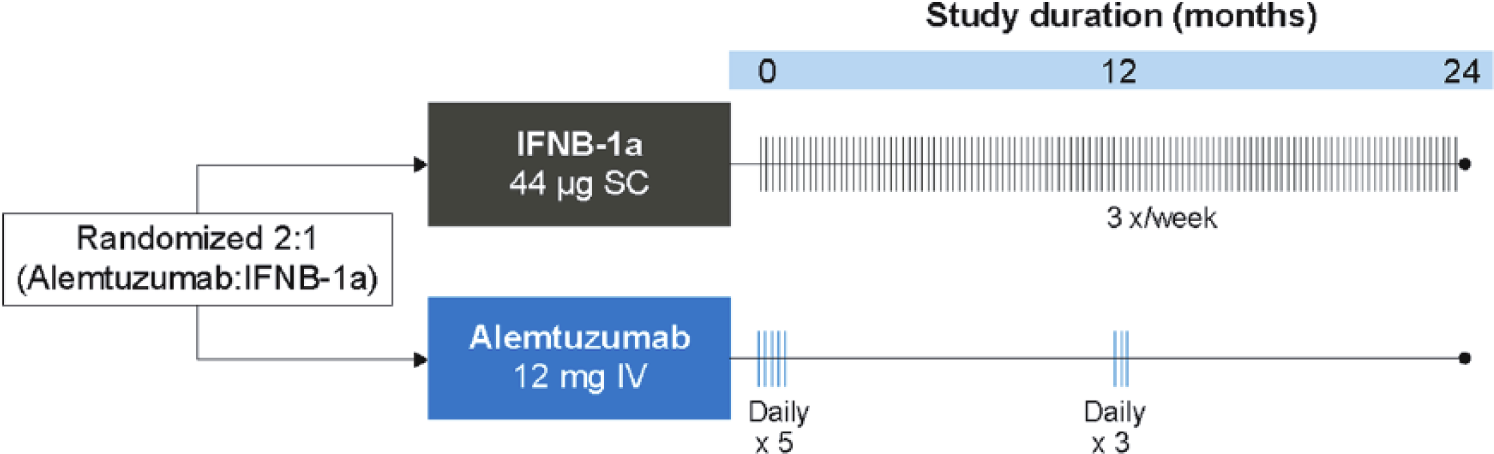
The efficacy and safety of alemtuzumab in patients with RRMS was evaluated in two phase 3 studies, the Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis (CARE-MS) I (ClinicalTrials.gov number NCT00530348) and II (ClinicalTrials.gov number NCT00548405) studies (for an overview, see Table 1).7,8 In both studies, patients were randomized to receive alemtuzumab (12 mg/day intravenous (IV) once daily for five consecutive days at baseline and for three consecutive days at 12 months) or a high-dose active comparator, subcutaneous interferon beta-1a (SC IFNβ-1a; Rebif®) 44 μg three times weekly (Figure 1). Efficacy assessments (clinical and magnetic resonance imaging (MRI)) were rater-blinded. While a double-blind trial model would be ideal in comparing therapies, it was decided at the design stage that patient unblinding due to the distinct mode of administration and established side effect profile of the two therapies was inevitable in practice, and there were expressed concerns about the ethics and practicality of maintaining patients, well aware they had the active infused therapy, on over two years of thrice weekly subcutaneous injections.
Overview of key clinical trials investigating alemtuzumab vs an active comparator for the treatment of RRMS.
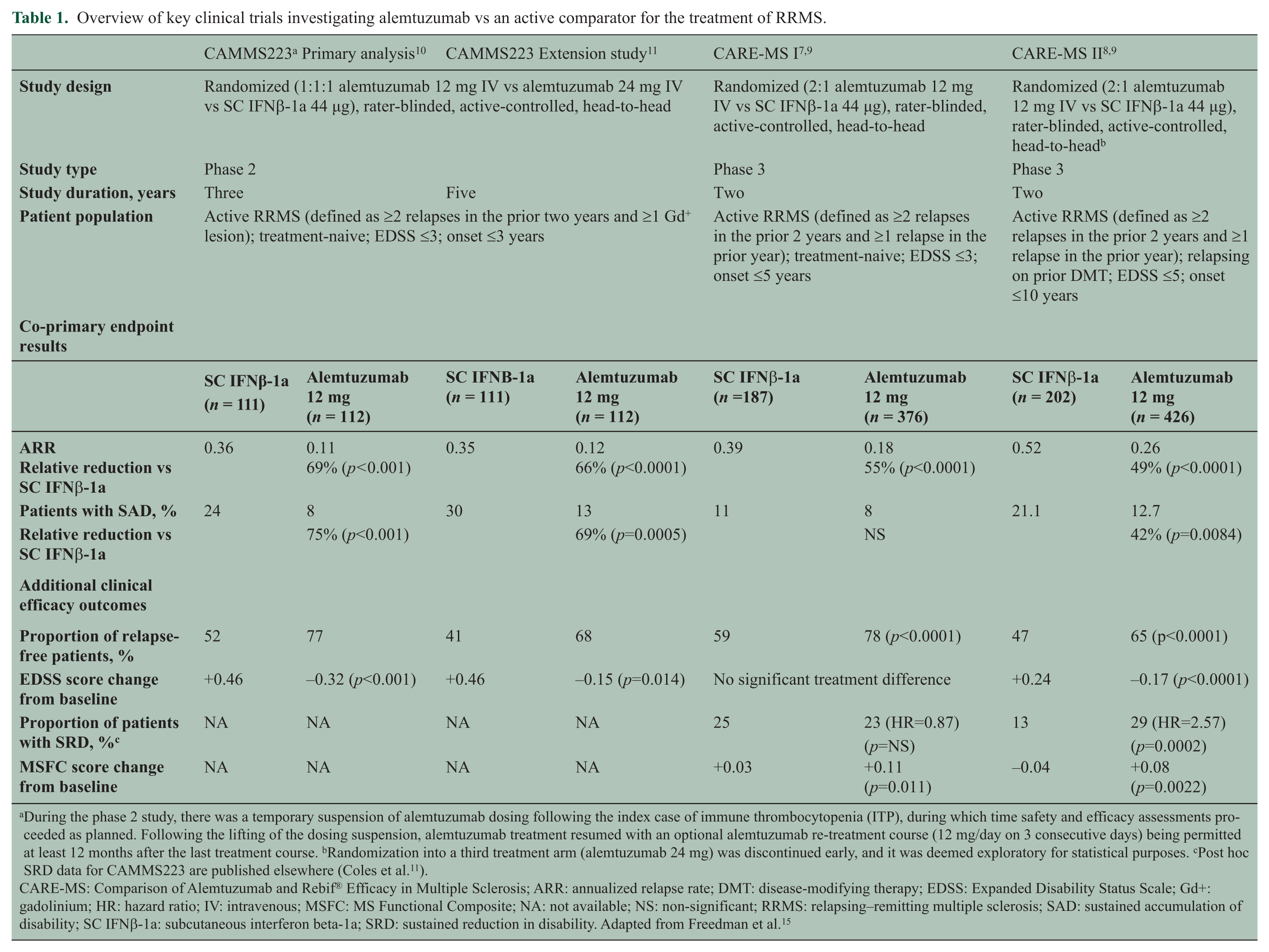
During the phase 2 study, there was a temporary suspension of alemtuzumab dosing following the index case of immune thrombocytopenia (ITP), during which time safety and efficacy assessments proceeded as planned. Following the lifting of the dosing suspension, alemtuzumab treatment resumed with an optional alemtuzumab re-treatment course (12 mg/day on 3 consecutive days) being permitted at least 12 months after the last treatment course. bRandomization into a third treatment arm (alemtuzumab 24 mg) was discontinued early, and it was deemed exploratory for statistical purposes. cPost hoc SRD data for CAMMS223 are published elsewhere (Coles et al. 11 ).
CARE-MS: Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis; ARR: annualized relapse rate; DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; Gd+: gadolinium; HR: hazard ratio; IV: intravenous; MSFC: MS Functional Composite; NA: not available; NS: non-significant; RRMS: relapsing–remitting multiple sclerosis; SAD: sustained accumulation of disability; SC IFNβ-1a: subcutaneous interferon beta-1a; SRD: sustained reduction in disability. Adapted from Freedman et al. 15
All primary outcomes for the treatment of RRMS were reported versus SC IFNβ-1a; there were no placebo arms. The active comparator for these clinical trials, SC IFNβ-1a, has been shown to provide clinically meaningful benefit in patients with RRMS. In the SC IFNβ-1a pivotal clinical trials, SC IFNβ-1a demonstrated a reduction in relapse rate (33%), active T2 lesions (78%), and disability progression (30%) compared with placebo. This was among the most effective approved therapies at the time of the alemtuzumab clinical trial program.9,12
Eligibility rules for both studies required patients to have active disease defined as having experienced at least two relapses in the past two years, with at least one in the past year. In CARE-MS I, treatment-naive RRMS patients with an Expanded Disability Status Scale (EDSS) score ≤3.0 at baseline were included in the trial. Baseline characteristics included a mean age of 33 years, a mean time of two years since first symptoms and a mean EDSS score of 2.0. 7 CARE-MS II enrolled RRMS patients who had relapsed on prior therapy and had an EDSS score ≤5.0 at baseline. 8 At baseline, patients had a mean age of 35 years, a mean time since first symptoms of 4.5 years and a mean EDSS score of 2.7; the mean duration of exposure to prior MS therapies (one or more drug used) was 35 months and 30% had received ≥2 prior MS therapies. An additional third arm in CARE-MS II using double-dose alemtuzumab was terminated early because of slow recruitment. Efficacy findings were broadly in line with the 12 mg arm with no new safety signals and, as this dose has not been considered for approval, the results will not be discussed further in this review.
The primary outcome measures for CARE-MS I and II were the annualized relapse rate (ARR) over two years and the time to onset of sustained accumulation of disability (SAD) compared with SC IFNβ-1a, defined as an increase of at least one point on the EDSS from a baseline score ≥1.0 (≥1.5-point increase for patients with baseline EDSS score of 0) that was sustained for six months.
In CARE-MS I, alemtuzumab reduced the ARR by 55% (p < 0.0001) compared with SC IFNβ-1a (Table 1).7,9 There was a 30% reduction in six-month SAD that did not reach statistical significance (alemtuzumab, 8% vs SC IFNβ-1a, 11%; p = 0.22), with the mean EDSS score change from baseline being −0.14 in both the alemtuzumab and the SC IFNβ-1a arms (p = 0.97). One potential contributor to this statistically non-significant finding may have been the lower-than-expected proportion of patients in the SC IFNβ-1a group who met the six-month SAD endpoint in CARE-MS I (i.e. 11%) compared with 20% at 24 months in the phase 2 study 11 (described in further detail below) on which the power calculations for CARE-MS I were in part based. One might also speculate that given the lower MRI T2 lesion load at baseline in CARE-MS I (median lesion volume 4.2 vs 8.5 cm3 in the phase 2 study), these patients had a lesser probability of developing disability progression than patients in the phase 2 study.
In CARE-MS II, alemtuzumab reduced the ARR by 49% (p < 0.0001) and six-month SAD by 42% (alemtuzumab, 13% vs SC IFNβ-1a, 21%; p = 0.0084) over two years (Table 1).8,9 The mean EDSS score in alemtuzumab-treated patients was significantly reduced over two years, indicating an improvement in disability score, whereas the mean EDSS score for patients treated with SC IFNß-1a was significantly increased from baseline (alemtuzumab, −0.17 vs IFNβ-1a, +0.24; p < 0.0001). Compared with SC IFNß-1a-treated patients, alemtuzumab-treated patients were 2.6 times more likely to demonstrate a sustained reduction in preexisting disability (SRD) over six months (Kaplan-Meier estimate: 28.8% vs 12.9% (hazard ratio (HR) 2.57; p = 0.0002)). In both CARE-MS I and II, treatment effects on clinical endpoints were associated with significant effects on MRI measures of inflammation and disease progression. Alemtuzumab significantly reduced the proportion of patients with new or enlarging T2-hyperintense lesions and gadolinium-enhancing lesions, and also slowed the parenchymal brain volume loss (a measure of brain atrophy) compared with SC IFNβ-1a (Table 2). No treatment differences were observed in median volume change of T2-hyperintense lesions.7–9
MRI and disease-free survival endpoints from CARE-MS I and II.
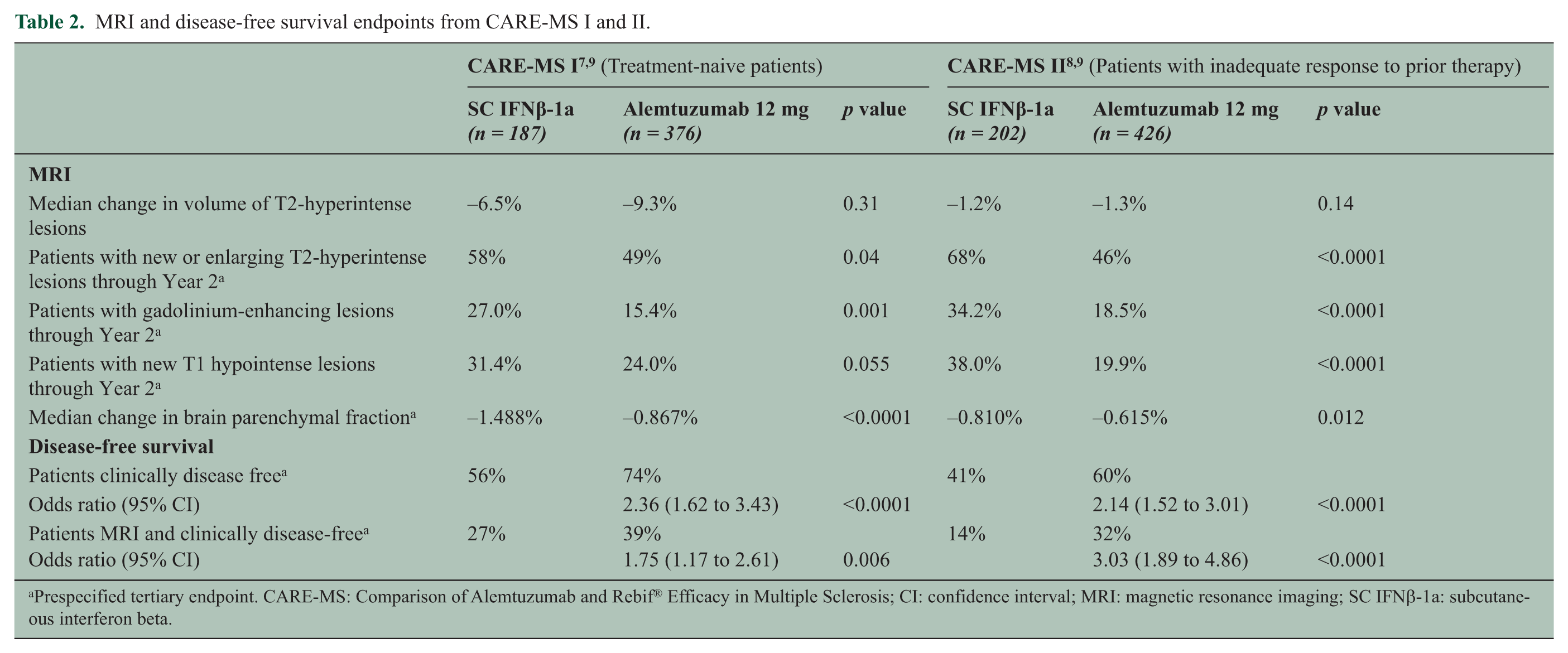
Prespecified tertiary endpoint. CARE-MS: Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis; CI: confidence interval; MRI: magnetic resonance imaging; SC IFNβ-1a: subcutaneous interferon beta.
Supportive analyses of the CARE-MS I and II datasets showed that fewer alemtuzumab-treated patients experienced severe relapses (CARE-MS I: 61% risk reduction, p = 0.0056; CARE-MS II: 48% reduction, p = 0.0121), or relapses that led to steroid treatment (CARE-MS I: 58% reduction, p < 0.0001; CARE-MS II: 56% reduction, p < 0.0001). Moreover, among alemtuzumab-treated patients experiencing relapses, there was a trend for reduced hospitalizations in the CARE-MS I study (29% reduction, p = 0.34) and a significant reduction in hospitalizations in the CARE-MS II study (55% reduction, p = 0.0045) compared with SC IFNβ-1a.7,8
An extension study (ClinicalTrials.gov number NCT00930553) investigating the long-term efficacy and safety of alemtuzumab in patients who completed either CARE-MS I or II is currently ongoing.
Phase 2 trial
In the phase 2 study CAMMS223 (ClinicalTrials.gov number NCT00050778), the efficacy of alemtuzumab was evaluated in treatment-naive patients with active RRMS, with patients being treated with either alemtuzumab 12 mg/day (n = 108) or 24 mg/day (n = 108) (administered once per day on five consecutive days at baseline and on three consecutive days at 12 months, and for some patients at 24 months or later as needed) or SC IFNβ-1a 44 µg (n = 107) administered three times per week. 10 Patients in the three-year CAMMS223 study then had the option to continue in an extension phase 11 (for an overview, see Table 1). Forty-one patients received three or more courses of alemtuzumab, of which 37 patients received three courses and four patients received four courses; 31 of the 41 patients received three or more courses during the re-treatment phase (37 to 58 months after last alemtuzumab course). At baseline, patients had an EDSS score ≤3.0, a time since first symptoms of ≤3 years, at least two clinical episodes of MS in the two years prior to the study, and one or more gadolinium-enhancing lesion. At three years, alemtuzumab 12 mg reduced the ARR by 67% (HR, 0.33 (95% confidence interval (CI): 0.20–0.55), p < 0.0001) and the six-month SAD by 76% (HR, 0.24 (95% CI: 0.11–0.55), p < 0.001) compared with SC IFNβ-1a. These significant reductions in ARR and SAD were maintained after five years of follow-up. 11 During the five-year follow-up period, 41.7% of patients received re-treatment with three or more courses of alemtuzumab.
Considering freedom from clinical disease activity, 13 an increasingly reported efficacy criterion in MS trials defined as absence of both relapse and SAD (confirmed at six months in this study), a post hoc analysis of the CAMMS223 study showed that at three years, 73% of patients treated with alemtuzumab 12 mg were free from clinical disease activity compared to only 43% of patients treated with SC IFNβ-1a (p > 0.0001). 14
Pooled safety analysis
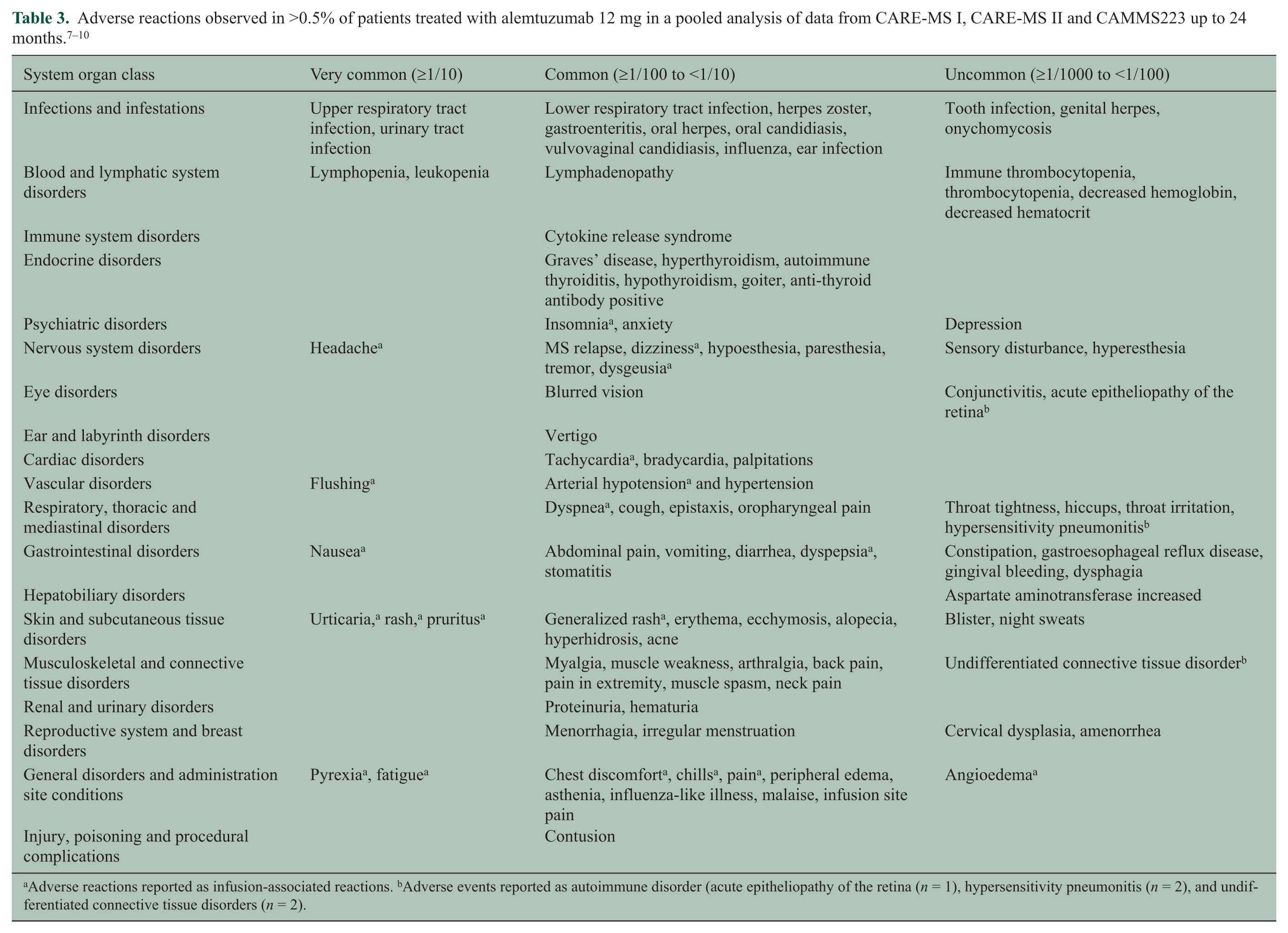
A total of 1188 patients with RRMS treated with alemtuzumab (12 mg (n = 919) or 24 mg (n = 269)) constituted the safety population in a pooled analysis of data from CARE-MS I, CARE-MS II and CAMMS223, resulting in 2363 patient-years of follow-up and a median follow-up of 24 months (Table 3). Important adverse events were infusion-associated reactions (IARs), serious infections and autoimmune adverse events (thyroid disorders (including thyroid cancers), immune thrombocytopenia (ITP), nephropathies and cytopenias) and these are discussed in more detail in the Management section below.
Adverse reactions reported as infusion-associated reactions. bAdverse events reported as autoimmune disorder (acute epitheliopathy of the retina (n = 1), hypersensitivity pneumonitis (n = 2), and undifferentiated connective tissue disorders (n = 2).
Mechanism of action of alemtuzumab
The mechanism by which alemtuzumab exerts its clinical effects in MS has not been fully elucidated. Alemtuzumab selectively targets CD52, a cell surface antigen present at high levels on T (CD3+ and CD4+ or CD8+), B (CD19+) lymphocytes and monocytes (Figure 2), whose function remains unknown. In contrast, little or no CD52 is detected on cells of the innate immune system, e.g. neutrophils and natural killer cells.15–17
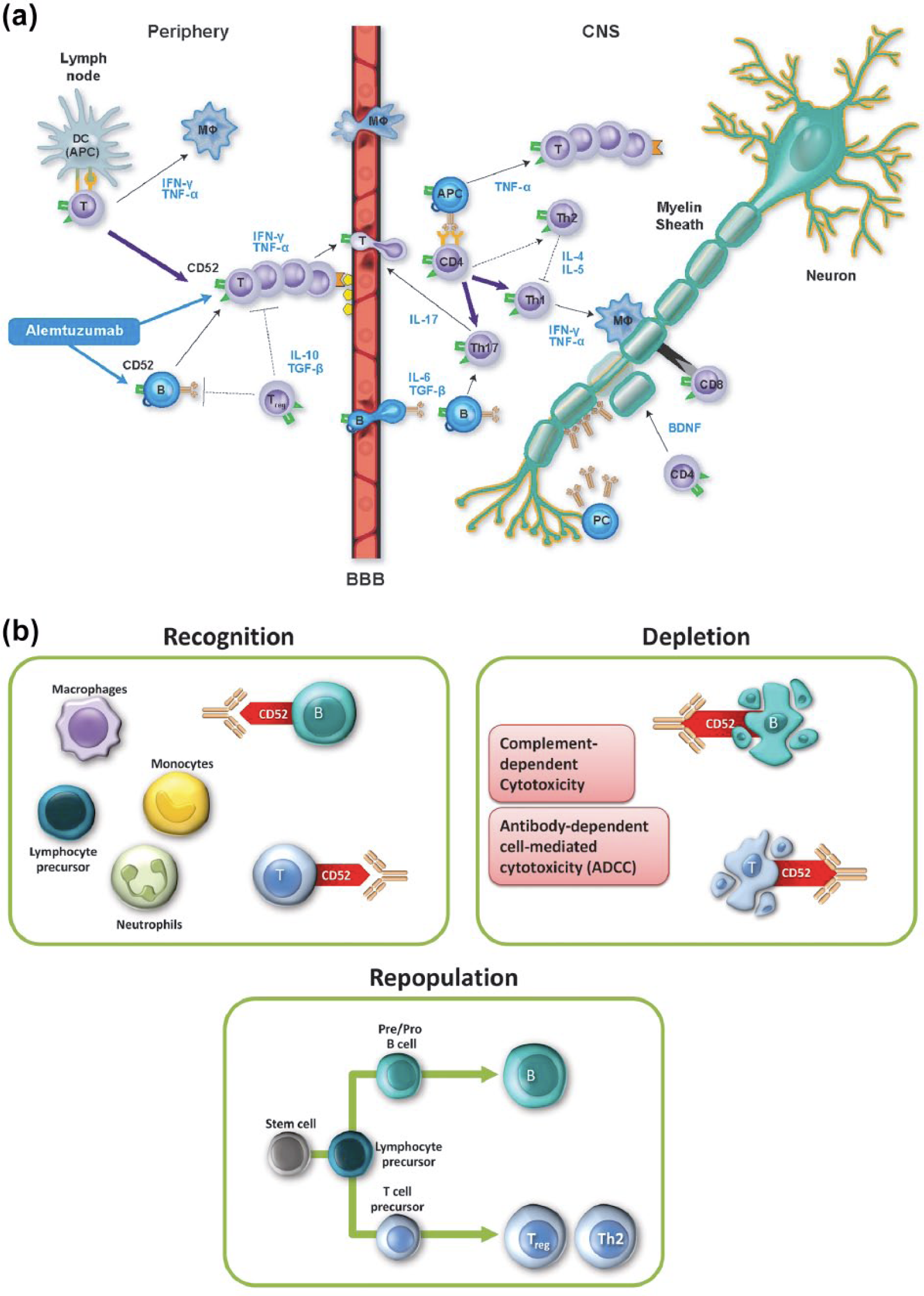
Mechanism of action of alemtuzumab.
Alemtuzumab causes antibody-dependent cellular cytolysis and complement-mediated lysis following cell binding to the surface of T and B lymphocytes. Current evidence suggests immunomodulatory effects through the depletion and repopulation of lymphocytes, 6 including alterations in the number, proportion, and properties of certain lymphocyte subsets;18,19 increased representation of regulatory T-cell subsets; 5 an increased representation of memory T lymphocytes; and rapid cell cycling of repopulating T cells with enhanced T-cell apoptosis. 15
The initial decrease in the level of circulating T and B lymphocytes by alemtuzumab, coupled with a distinctive temporal (early B-cell and monocyte recovery, delayed T-cell recovery) and qualitative pattern of repopulation, is believed to have an anti-inflammatory effect and induces a change in the disturbed balance of the immune system that diminishes the potential for future relapse and disease progression. 19
Pharmacodynamics of alemtuzumab
In an analysis investigating a population of 216 patients with RRMS, alemtuzumab concentrations decreased to low or undetectable levels within one month after dosing; however, its pharmacodynamic effect lasted considerably longer. 20 Each treatment course of alemtuzumab depletes circulating T and B lymphocytes, with the lowest observed values occurring at the earliest post-treatment time point (one month after a course of treatment in the above-mentioned phase 3 studies). Lymphocytes repopulate over time, with B-cell recovery being usually complete within six months, whereas T-lymphocyte counts rise more slowly and generally do not return to baseline by 12 months post-treatment. In the CARE-MS studies, approximately 40% of patients had total lymphocyte counts reaching the lower limit of normal (LLN) by six months after each treatment course, and approximately 80% of patients had total lymphocyte counts reaching the LLN by 12 months after each course.7,8
Future research
There is a conjecture that the clinical improvement seen is beyond a simple anti-inflammatory effect, 18 as is suggested by a post hoc analysis of CAMMS223 that demonstrated a sustained reduction of preexisting disability in a subgroup of patients who had no clinical disease activity immediately prior (<3 months) to alemtuzumab treatment. Furthermore, in a prespecified analysis of the CARE-MS II trial data, significant improvement in preexisting disability was seen in patient cohorts both with and without recent pretreatment relapses (<3 months) prior to alemtuzumab therapy. 21
Additional evidence supporting the notion that alemtuzumab may confer neuroprotective effects 18 comes from brain MRI studies in CARE-MS patients, in which the decrease of the brain parenchymal fraction, as a measure of brain atrophy, was reduced by 42% (CARE-MS I) and 23% (CARE-MS II) in alemtuzumab-treated patients vs SC IFNβ-1a over two years (p < 0.0001 and p = 0.012, respectively).7–9 It remains to be determined whether this effect reflects a strong anti-inflammatory action that limits the collateral damage to neurons or is indicative of an independent primary neuroprotective activity. 18
In this context brain MRI studies investigating alemtuzumab’s regional and temporal effects on gray and white matter atrophy as outcome measures may yield important further insights.
Management
Alemtuzumab has a well-characterized safety and tolerability profile, with a safety monitoring program that allows for early detection and management of adverse events.7,8,10 Adverse events of interest include IARs, serious infections and autoimmune adverse events, including thyroid disorders and, less frequently, ITP and nephropathies.7,8,10
IARs
In the CARE-MS studies described above, IARs were defined as any adverse event occurring during or within 24 hours of alemtuzumab infusion. Most patients treated with the monoclonal antibody in this trial setting experienced mild to moderate IARs following administration of alemtuzumab 12 mg, most commonly headache, rash, nausea and pyrexia. Serious reactions occurred in 3% of patients.7–9 Cardiac alterations such as tachycardia, bradycardia and palpitations have been reported among alemtuzumab-treated patients with MS (Table 3), of which serious adverse events were uncommon, symptoms resolved, and did not preclude alemtuzumab treatment.
It is recommended that patients be premedicated with corticosteroids immediately prior to the administration of alemtuzumab on each of the first three days of any treatment course to diminish IARs (Table 4).7–9 In the CARE-MS trials, IARs were effectively managed with methylprednisolone, antipyretics and antihistamines (Table 4).
Risk-management strategies adopted in core alemtuzumab clinical studies and recommended for clinical practice. 9 .
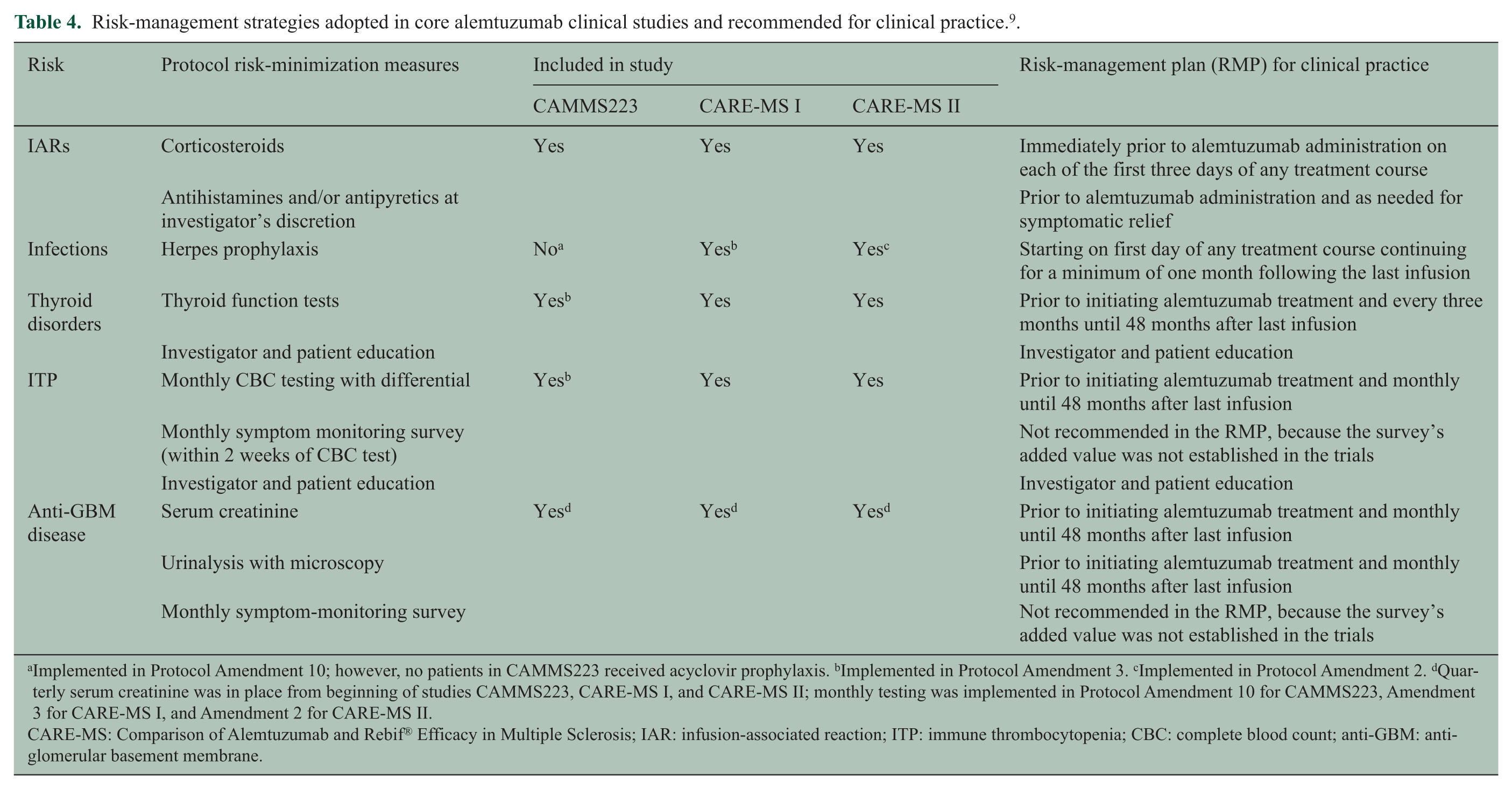
Implemented in Protocol Amendment 10; however, no patients in CAMMS223 received acyclovir prophylaxis. bImplemented in Protocol Amendment 3. cImplemented in Protocol Amendment 2. dQuarterly serum creatinine was in place from beginning of studies CAMMS223, CARE-MS I, and CARE-MS II; monthly testing was implemented in Protocol Amendment 10 for CAMMS223, Amendment 3 for CARE-MS I, and Amendment 2 for CARE-MS II.
CARE-MS: Comparison of Alemtuzumab and Rebif® Efficacy in Multiple Sclerosis; IAR: infusion-associated reaction; ITP: immune thrombocytopenia; CBC: complete blood count; anti-GBM: anti-glomerular basement membrane.
Infections
As might be expected from alemtuzumab’s mechanism of action, infections occurred more frequently with alemtuzumab 12 mg compared with SC IFNβ-1a in the CARE-MS trials (71% vs 53%, respectively), but were predominantly mild to moderate in severity.7,8 Infections that occurred more often in alemtuzumab-treated patients than SC IFNβ-1a–treated patients included oral herpes, herpes zoster, nasopharyngitis, urinary tract infection, upper respiratory tract infection, sinusitis, influenza, bronchitis, and localized superficial (noninvasive) fungal infections. One case of esophageal candidiasis was reported as a serious adverse event and resolved with conventional treatment. 9 Two cases of active tuberculosis occurred with alemtuzumab. Both tuberculosis cases were from regions of known endemicity, and resolved with conventional antituberculosis treatment. 8 One patient with listeria meningitis responded to parenteral antibiotics and was discharged from hospital 15 days later, without sequelae, except fatigue. The patient received a second course of alemtuzumab 24 mg without infectious complication. 10 The relatively rare occurrence of serious infections may be consequent on the preservation of alemtuzumab-treated patients to mount protective B- and T-cell responses. This was shown in a case-control study of 24 patients in whom T-cell-dependent and T-cell-independent and antibody responses were recorded following vaccination with multiple bacterial and viral antigens. 22
Prophylaxis with an oral anti-herpes agent should be initiated starting on the first day of alemtuzumab treatment and continuing for a minimum of one month following each course of treatment. In the CARE-MS program, patients received oral acyclovir 200 mg twice daily during alemtuzumab infusion and for 28 days thereafter as prophylaxis against herpes infections (Table 4).7,8 Supportive data suggest that this prophylactic treatment following each alemtuzumab course is effective in reducing the risk of herpetic infections. 23
Autoimmune adverse events
Treatment with alemtuzumab has been shown to increase the risk of autoimmune-mediated conditions including thyroid disorders, ITP or, rarely, nephropathies such as Goodpasture disease with anti-glomerular basement membrane (anti-GBM) antibodies. One potential mechanism for this may be a link between homeostatic T-cell proliferation following alemtuzumab-mediated lymphocyte depletion and lymphopenia-associated autoimmunity.24,25 As autoimmune adverse events may occur months to years after alemtuzumab treatment, monitoring for these side effects until 48 months after the last alemtuzumab infusion is imperative.
Autoimmune thyroid disorders have been observed in 34.2% of RRMS patients treated with alemtuzumab in the phase 2 CAMMS223 study with a median follow-up of 57.3 months following first alemtuzumab exposure (including hyperthyroidism or hypothyroidism). 26 Extended follow-up revealed an onset of thyroid disease occurring between six and 61 months after the first treatment course, peaking in the third year post-treatment and declining thereafter. 12 In three years of follow-up following the first course of alemtuzumab in the phase 3 program, the incidence of first thyroid adverse events was higher in Year 3 (20.5%) than in the first two years of the core studies (16.9%), consistent with experience in the CAMMS223 study. 27 Most events were mild to moderate in severity. At three-year follow-up, the cumulative incidence of serious thyroid events (cancer, Grave’s requiring radioiodine treatment) was 2.5%. Most thyroid events were managed with conventional medical therapy. However, some patients required surgical intervention (in the CARE-MS trials, three patients underwent thyroidectomy) and two patients received radioiodine ablation. 27 Thyroid function tests, such as the measurement of thyroid-stimulating hormone levels, should be obtained prior to initiation of treatment and every three months thereafter until 48 months following the last infusion (Table 4).
Based on the full safety experience up to November 26, 2012, the most common malignancies reported in alemtuzumab patients were thyroid cancer, breast cancer, basal cell carcinoma, and melanoma. Five cases of thyroid malignancies have been reported in alemtuzumab-treated patients.7,8 All five thyroid malignancies occurred in patients who developed a thyroid disorder during the study (entry into which may itself be a risk factor for ascertainment of thyroid malignancies) and were discovered as incidental findings based on ultrasound examinations (a potential ascertainment bias). Although there was no evidence of increased risk of malignancy in alemtuzumab-treated MS patients, further study is needed to determine whether alemtuzumab raises the risk of malignancy.
Serious events of ITP have been observed in approximately 1% of patients treated with alemtuzumab in the CARE-MS program, between 14 and 36 months after first exposure to alemtuzumab.7,8 The first case of ITP, in the phase 2 CAMMS223 trial, went unrecognized and the patient died from intracerebral hemorrhage. 28 Following this index case, a monitoring program was implemented to identify and manage ITP systematically, including education on signs and symptoms for patients and physicians and monthly blood monitoring. Other autoimmune cytopenias, such as neutropenia, hemolytic anemia, agranulocytosis and pancytopenia, have been reported in the CARE-MS trials with lower incidence than ITP.7,8 One patient experienced a recurrence of pancytopenia, which was associated with lack of compliance with corticosteroid therapy, resulting in fatal sepsis 20 months post-alemtuzumab treatment. It is recommended that complete blood counts with differential be obtained prior to initiation of treatment and at monthly intervals thereafter until 48 months after the last infusion (Table 4); if ITP or other cytopenia is suspected, a complete blood count should be obtained immediately, and if confirmed, the patient should be transferred for urgent care by a specialist.
Nephropathies, including anti-GBM disease, have been observed in 0.3% of patients in the clinical trial program and generally occurred within the recommended 48-month monitoring period following the last administration of alemtuzumab. In clinical trials, there were two cases of anti-GBM disease and two cases of membranous glomerulonephritis, identified early through clinical and laboratory monitoring.29–31 Although these cases were serious, there was a positive outcome in renal function after treatment (normalization of serum creatinine and/or decreased proteinuria). Improvements in renal function were observed in both cases of anti-GBM disease after treatment with plasmapheresis, cyclophosphamide and glucocorticosteroids, and in the two cases of membranous glomerulonephritis after treatment with diuretics and/or lisinopril.28,30
Clinical manifestations of nephropathy may include hematuria and/or proteinuria and elevation in serum creatinine. Serum creatinine levels and urinalysis with cell counts should be obtained prior to initiation of treatment and at monthly intervals thereafter until 48 months after the last infusion (Table 4). The observation of unexplained hematuria and/or proteinuria or clinically significant changes from baseline in serum creatinine should prompt urgent evaluation for possible nephropathy, since untreated anti-GBM can rapidly progress to renal failure. Observation of isolated proteinuria should prompt further evaluation for other types of nephropathy.
A recently published single-center observational study of 87 patients followed for a mean of seven years did not show any additional safety signals. 32
Further research is required to find a predictive biomarker that would reliably find patients at risk to develop a secondary autoimmune disease. Earlier evidence that interleukin-21 could serve as such a marker have not been substantiated. 33 A search for immune signatures and/or genetic factors with predictive properties should continue.
As with all drugs that affect the immune system, some rare but potentially serious risks such as opportunistic infections or malignancies may emerge or their dimension may become apparent only after exposure of a larger number of patients to the drug following market approval. The example of progressive multifocal leukoencephalopathy following natalizumab treatment has been informative. 34
Apparently, absolute lymphocyte counts do not reliably indicate treatment response as shown in a recent study in a cohort of 106 patients who were followed for a median of 99 months. 32 A total of 52% of alemtuzumab-treated (at least two cycles) patients relapsed at some point. Recovery of CD4, CD8 and CD19 cell populations was not different in patients with or without disease activity or accumulation of disability. This emphasizes the need for continued research into the nature and dynamics of the restoration of the immune system and overall immunocompetence. 35
An open clinical issue needs to be addressed: How many cycles of alemtuzumab can be safely administered and what would trigger repeat infusions? In the most recent account of the Cambridge, United Kingdom (UK) cohort followed for up to 144 months, relapses prompted re-treatment to a total of three cycles in 36%, four cycles in 8% and five cycles in 1% of the 87 patients studied. 32
Given its mode of action, which appears to effectively re-configure a disturbed immune system, it would also be interesting to assess its potential and safety as induction therapy.
Conclusions
Alemtuzumab is a recent addition to the therapeutic arsenal for relapsing–remitting disease. It has consistently been shown to provide superior efficacy when compared with the baseline disease-modifying agent IFNβ-1a.
Unlike other labeling classifications of RRMS such as “highly active” or “rapidly evolving severe,” which require the presence of both clinical and imaging features with very specific criteria in each category, active RRMS in the alemtuzumab EU Summary of Product Characteristics (SmPC) is defined based on either clinical or imaging features. 11 This provides the treating neurologist the freedom to select appropriate patients for treatment based on clinical assessment of an individual patient and whether a patient has the ability to comply with monitoring requirements. Long-term monthly monitoring through 48 months after the last alemtuzumab infusion poses a challenge to patient adherence and requires appropriate education both of physicians and patients.
It needs to be determined if and when after the first two or three annual cycles alemtuzumab treatment should be repeated with recrudescence of disease activity.
As with any other newly developed agents, in particular those that exert a powerful impact on the immune system, a thorough assessment of benefits and risks, adherence to long-term monitoring requirements and pharmacovigilance are all mandatory. Alemtuzumab represents a new option for the treatment of active RRMS.
Footnotes
Acknowledgements
Editorial support was provided by Fiona Nitsche, PhD, and Susan M Kaup, PhD, Evidence Scientific Solutions, Philadelphia, PA, USA. Funding for this editorial support was provided by Genzyme, a Sanofi company.
Conflicts of interest
H-P Hartung has received honoraria for consulting and speaking at symposia funded by Bayer Healthcare, Biogen Idec, GeNeuro, Genzyme, MedImmune, Merck Serono, Novartis, Roche, Teva, and Sanofi-Aventis, with approval by the Rector of Heinrich-Heine University. O Aktas reports receiving grants from the German Research Foundation (DFG), the German Ministry for Education and Research, and the Schaufler Foundation; honoraria for lectures funded by Bayer Healthcare, Biogen Idec, Genzyme, Novartis, Merck Serono, and Teva; and travel funds from Bayer Healthcare, Novartis, and Merck Serono. A Boyko reports receiving honoraria for lectures and is a member of the advisory boards of Merck Serono, Teva, Genzyme/Sanofi, Biogen/Takeda, Bayer, and Nycomed.
Funding
The MS Center at the Department of Neurology, HHU Düsseldorf, is supported in part by the Walter-und Ilse-Rose-Stiftung.