Abstract
Background:
Daclizumab high-yield process (DAC HYP) is a humanized anti-CD25 monoclonal antibody that inhibits high-affinity interleukin-2 receptor signaling.
Objective:
The objective of this paper is to assess the proportion of DAC HYP- versus placebo-treated patients who were free from disease activity.
Methods:
SELECT was a randomized, double-blind, multicenter study of DAC HYP 150 mg or 300 mg, or placebo, administered subcutaneously every four weeks for 52 weeks. In this post-hoc analysis of the SELECT trial, ‘disease-activity free’ was defined as completion through week 52 without relapses or confirmed three-month disability progression (clinical), with no new/newly enlarging T2-hyperintense lesions and no new gadolinium-enhancing lesions at the week 52 scan (radiological). Primary analyses were based on logistic regression controlling for baseline characteristics.
Results:
More DAC HYP-treated (39%, n = 156) versus placebo-treated patients (11%, n = 22) were disease-activity free (odds ratio (95% confidence interval), 6.18 (3.71–10.32); p < 0.0001). Furthermore, 77% and 48% of DAC HYP-treated patients were free from clinical or radiological disease activity, respectively, compared with 60% and 18% of placebo-treated patients.
Conclusion:
At one year, DAC HYP resulted in a meaningful increase in the proportion of relapsing–remitting MS patients who were disease-activity free versus placebo.
Keywords
Introduction
Multiple sclerosis (MS) is the most prevalent cause of nontraumatic disability in young adults in Europe and North America, with an estimated median global incidence of 2.5 per 100,000.1,2 The majority of patients with MS (~85%) present with the primary phase termed relapsing–remitting MS (RRMS). 3 Within 25 years of onset, approximately 90% of RRMS cases enter the secondary progressive phase (SPMS), which is characterized by an irreversible neurological decline, severe disability, and deterioration in quality of life.2–4 Prevention of the transition to SPMS and achievement of a disease-activity-free status is therefore a key therapeutic goal.3,5
The development of targeted biological therapies has made remission/freedom from disease activity, as defined by clinical MS activity and standard cranial magnetic resonance imaging (MRI),5–8 an achievable goal for MS.5,6 Thus, the therapeutic paradigm has shifted from managing symptoms to treating with mechanism-based disease-modifying therapies.5,6 Daclizumab is a humanized monoclonal antibody that binds the α-subunit (CD25) of the interleukin-2 receptor (IL-2R) and inhibits signaling through the high-affinity IL-2R isoform. 9 In the phase 2 CHOICE study, daclizumab used in combination with interferon beta (IFNβ) was well tolerated and reduced MS activity in patients previously refractory to IFNβ alone. 10
The subsequent SELECT study was conducted to determine the safety and efficacy of daclizumab high-yield process (DAC HYP) monotherapy in patients with RRMS. 11 The SELECT study randomized patients to treatment with subcutaneous DAC HYP 150 mg, DAC HYP 300 mg or placebo every four weeks for 52 weeks. DAC HYP monotherapy reduced the annualized relapse rate and new brain MRI lesion activity, and slowed disability progression, in patients with RRMS compared with placebo. 11 This paper reports an analysis of data from the SELECT study to determine if patients treated with DAC HYP are more likely to be free of MS disease activity at one year compared with placebo.
Materials and methods
Full details of the methods of SELECT have been published previously. 11 Briefly, 621 patients with RRMS (McDonald criteria, 2005), 12 aged 18–55 years and with a baseline Expanded Disability Status Scale (EDSS) 13 score of 0–5 were randomized to treatment. Patients were required to have had at least one confirmed MS relapse within 12 months prior to randomization or one new gadolinium-enhancing (Gd+) lesion on brain MRI performed within six weeks prior to randomization. Patients with MS of any disease courses other than RRMS were excluded.
The primary efficacy endpoint was the annualized relapse rate (ARR). The current post-hoc analysis evaluated the potential of DAC HYP to increase the proportion of patients with RRMS who were free of disease activity at one year versus placebo. MS disease-activity free was defined by a composite of completion of the study through week 52 without relapses or confirmed disability progression, no new or newly enlarging T2-hyperintense lesions at week 52, and no new Gd+ lesions at week 52.5,14–16 Patients who withdrew from the study prior to week 52 for any reason were not counted as being disease-activity free even when there was no disease activity documented prior to withdrawal. Confirmed disability progression was defined as a one-point increase in EDSS score for patients with baseline EDSS score ≥1.0 or a 1.5-point increase for patients with a baseline EDSS score of 0 that was sustained for 12 weeks. The proportion of patients who fulfilled this composite requirement, as well as the individual requirements of the composite or a clinical alone (no relapses and no confirmed disability progression) and radiological alone composite (no new Gd+ lesions and no new or newly enlarging T2-hyperintense lesions at week 52) were assessed.
A logistic regression model adjusting for baseline disease status and age was used to analyze the proportions of DAC HYP-treated and placebo-treated patients free from disease activity. Specifically, the covariates included in the model included age (≤35 or >35 years), baseline EDSS score (≤2.5 or >2.5), the number of baseline Gd+ lesions, the number of relapses in one year prior to study entry and the baseline number of T2 lesions. The effects of baseline characteristics were further explored by repeating the primary analysis on subgroups of patients based on the following dichotomous categories: age (≤35 or >35 years), baseline EDSS score (≤2.5 or >2.5), baseline Gd+ lesions (present or absent), number of relapses in one year prior to study entry (≤1 or >1), baseline T2 lesions (≤median or >median) and nonhighly active RRMS versus highly active RRMS (defined as two or more relapses in the year prior to randomization and one or more Gd+ lesion at baseline).
Logistic regression models were also used to analyze the radiological components of disease activity at week 52, adjusting for age (≤35 or >35 years), baseline EDSS score (≤2.5 or >2.5), the number of relapses in one year prior to study entry, and the baseline number of lesions. Age and EDSS cut-offs were the median values at baseline. The clinical components of disease activity were monitored on a frequent or ongoing basis throughout the study; therefore, Kaplan-Meier methods and a Cox proportional hazards model were used for the analysis of these outcomes. The proportion of patients free of clinical activity was estimated using the Kaplan-Meier product-limit estimator, as were the proportions of patients free of each component of clinical activity. The Cox model for disability progression adjusted for age (≤35 or >35 years) and baseline EDSS score (≤2.5 or >2.5). In addition to these covariates, the models for freedom from relapse and composite clinical activity adjusted for the number of relapses in one year prior to study entry. No statistical adjustments for multiple comparisons were made.
Disease activity was also analyzed by six-month time period (weeks 0–24 and weeks 24–52). Disease-activity-free status in Period 1 (weeks 0–24) was defined as no new Gd+ lesions and no new/newly enlarging T2 lesions on the week 24 scan, no relapses in weeks 0–24, and no disability progression prior to week 24. In Period 2 (weeks 24–52), disease-activity-free status was defined as no new Gd+ lesions at week 52, no new/newly enlarging T2 lesions at week 52 compared with week 24, and no relapses or disability progression during Period 2.
This post-hoc analysis of disease activity and its components was conducted in all 600 patients in the intention-to-treat (ITT) population, consisting of the 621 patients enrolled in the study, except for 21 patients from a single study site who were prospectively excluded from the ITT population because of systematic misdosing at the site. The analysis by six-month time period was restricted to patients in the ITT population who completed the treatment period of the study and had interpretable MRI evaluations at baseline, week 24, and week 52.
Results
Study population demographics and baseline characteristics have been previously reported. 11 Prior to enrollment, most patients were MS treatment naive and the mean baseline EDSS score was 2.7. The mean number of Gd+ lesions was 1.8 and the number of T2-hyperintense lesions was 40 at baseline.
A greater proportion of DAC HYP-treated patients were free of disease activity compared with placebo (Figure 1; 39% (n = 156) vs 11% (n = 22), respectively; adjusted odds ratio (OR) (95% confidence interval (CI)), 6.18 (3.71–10.32); p < 0.0001). Furthermore, each DAC HYP dose group individually demonstrated a significantly greater proportion of patients free of disease activity compared with placebo (Figure 1; DAC HYP 150 mg: 36% (n = 73), adjusted OR (95% CI) 6.31 (3.59–11.11), p < 0.0001; DAC HYP 300 mg: 41% (n = 83), adjusted OR (95% CI) 6.07 (3.49–10.56), p < 0.0001). There was no statistical difference between the proportion of patients achieving disease-activity-free status in the DAC HYP 150 mg group versus the DAC HYP 300 mg group (p = 0.8653).
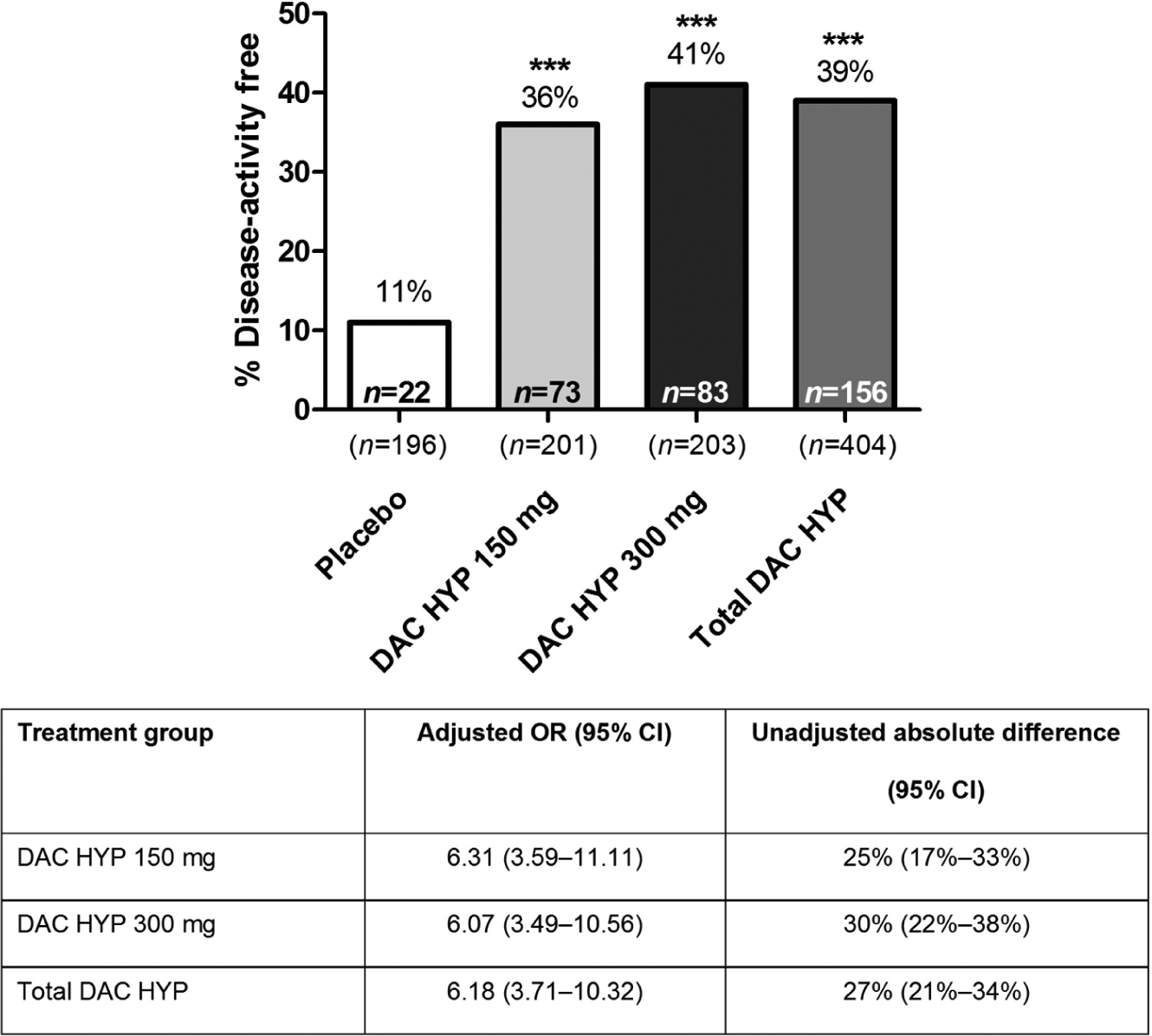
Proportion of patients disease-activity free in placebo and DAC HYP treatment groups.
Freedom from the composite measure of clinical disease activity was achieved by 77% of all DAC HYP-treated patients (Figure 2; adjusted risk reduction (RR) (95% CI) 51% (34%–64%); p < 0.0001) compared with 60% of placebo-treated patients. Additionally, equal proportions of patients (77%) in the individual DAC HYP dose groups met the clinical disease-activity-free criterion and achieved comparable reductions (Figure 2; DAC HYP 150 mg: adjusted RR (95% CI) 52% (30%–67%); DAC HYP 300 mg: 51% (29%–66%); p < 0.001 for both comparisons with placebo). Freedom from radiological disease activity was evident in 48% of DAC HYP patients (Figure 2; adjusted OR (95% CI) 5.35 (3.42–8.36); p < 0.0001) versus 18% of the placebo group. By individual DAC HYP group, 45% of DAC HYP 150 mg patients and 50% of DAC HYP 300 mg patients met the criteria for freedom from radiological activity, and the likelihood of achieving freedom from radiological disease activity was similar across DAC HYP groups (Figure 2; DAC HYP 150 mg: adjusted OR (95% CI) 5.60 (3.37–9.30) and DAC HYP 300 mg: 5.14 (3.14–8.42); p < 0.0001 for both comparisons with placebo).
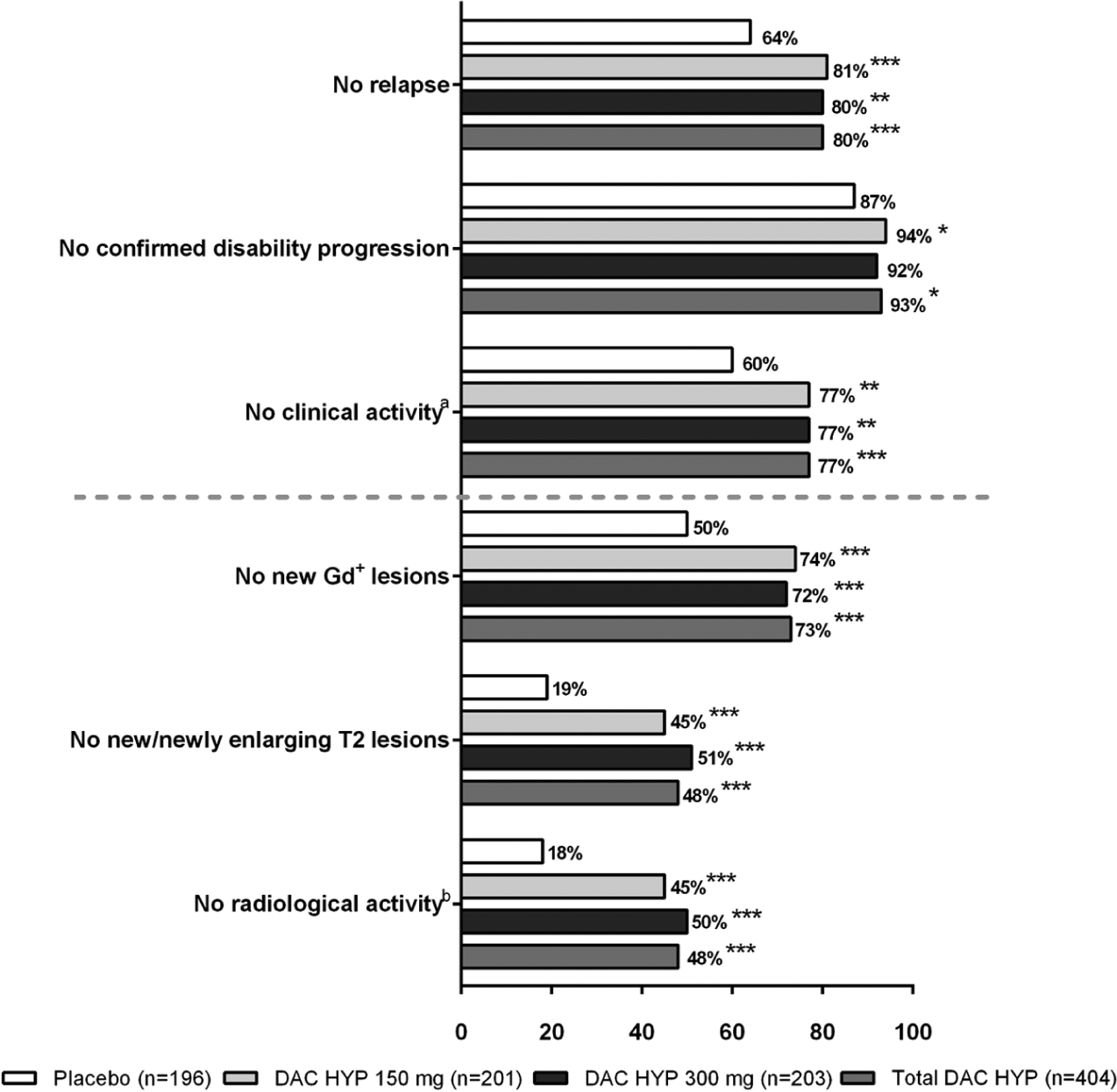
Proportion of patients free from each component of disease activity in the placebo and DAC HYP treatment groups.
DAC HYP efficacy over placebo was also evident on the individual elements of the composite measure of freedom from disease activity. A total of 80% of patients receiving DAC HYP were free of relapses compared with 64% of patients in the placebo group (Figure 2; p < 0.0001), representing a 53% adjusted RR of relapse (95% CI: 35%–66%). No significant differences in freedom from relapse were observed between the two DAC HYP dose groups (81% DAC HYP 150 mg vs 80% DAC HYP 300 mg, p = 0.7365). Furthermore, 7% of DAC HYP-treated patients experienced confirmed three-month disability progression (Figure 2; p = 0.015) compared with 13% in the placebo group, representing a 50% reduction (95% CI: 12%–71%) in confirmed disability progression. No significant differences in freedom from disability progression were observed between the two DAC HYP dose groups (94% DAC HYP 150 mg vs 92% DAC HYP 300 mg, p = 0.4799; RR (95% CI) 57% (12%–79%) vs 43% (–9% to 70%)).
MRI studies revealed that DAC HYP treatment significantly reduced the development of new brain lesions compared with placebo. As shown in Figure 2, 48% of DAC HYP-treated patients had no new or newly enlarging T2-hyperintense lesions, whereas only 19% of patients in the placebo group lacked new or newly enlarging T2-hyperintense lesions (adjusted OR (95% CI) 4.76 (3.10–7.33); p < 0.0001). Reduction in T2 lesion activity was also comparable across both individual DAC HYP dose groups (45% DAC HYP 150 mg vs 51% DAC HYP 300 mg, p = 0.5006; adjusted OR (95% CI) 4.42 (2.73–7.16) vs 5.12 (3.17–8.27)). Similarly, there was no evidence of new Gd+ lesions in 73% of DAC HYP-treated patients and 50% of patients in the placebo group (p < 0.0001; adjusted OR (95% CI) 3.10 (2.12–4.52)). No significant differences in freedom from Gd+-lesion activity were observed in the separate dose groups (74% DAC HYP 150 mg vs 72% DAC HYP 300 mg, p = 0.3334) compared with the total DAC HYP-treated population (Figure 2), and the likelihood of not developing new Gd+ lesions was similar between the DAC HYP 150-mg and 300-mg groups (adjusted OR (95% CI) 3.50 (2.22–5.51) vs 2.76 (1.78–4.29)).
Stratification of patients by demographics and disease characteristics at baseline revealed that a significantly higher proportion of DAC HYP-treated patients were free of disease activity versus placebo across all categories examined (Table 1). The dose of DAC HYP treatment received (150 mg or 300 mg) did not notably alter the proportion of patients achieving freedom from disease activity in any of the subgroups investigated (Table 1). When the analysis was stratified by time period, DAC HYP treatment was associated with being disease-activity free during weeks 0–24 (OR 3.75; 95% CI 2.39–5.89; p < 0.0001) and during weeks 24–52 (OR 5.83; 95% CI 3.71–9.17; p < 0.0001).
Number and proportion of patients free from disease activity over one year in patients stratified by baseline demographics and disease characteristics.
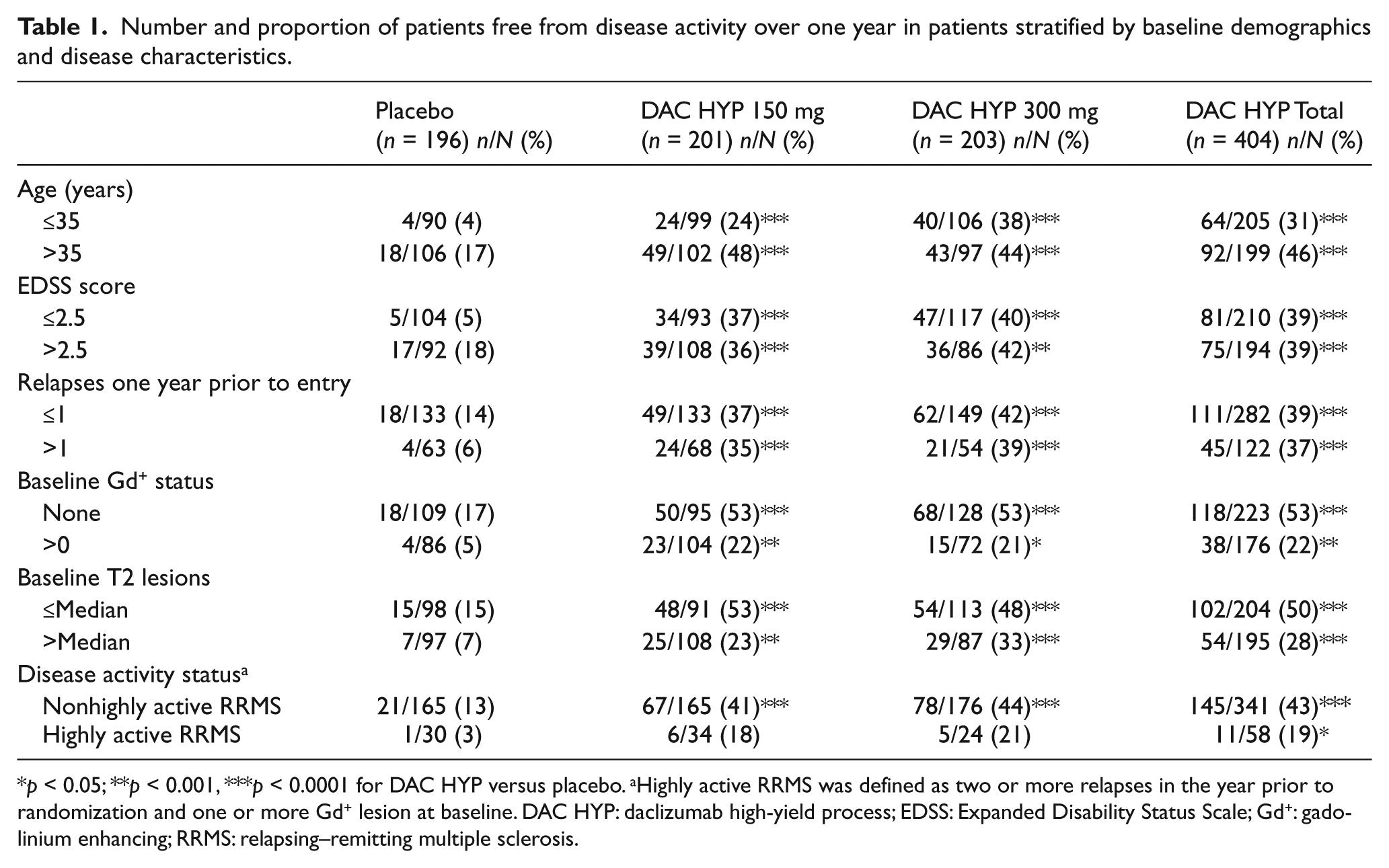
p < 0.05; **p < 0.001, ***p < 0.0001 for DAC HYP versus placebo. aHighly active RRMS was defined as two or more relapses in the year prior to randomization and one or more Gd+ lesion at baseline. DAC HYP: daclizumab high-yield process; EDSS: Expanded Disability Status Scale; Gd+: gadolinium enhancing; RRMS: relapsing–remitting multiple sclerosis.
Discussion
In this post-hoc analysis of the SELECT study, the proportion of patients who experienced freedom from disease activity was almost four-fold greater in the DAC HYP-treated patients compared with the placebo group over one year of treatment. The results were robust and consistent across the dose groups and baseline disease characteristics. The impact of DAC HYP on this composite endpoint reflected comparable effects of treatment on both clinical and radiological endpoints. 11
Composite measures of disease activity are sometimes preferred over individual disease activity measures when they are able to more accurately measure the broader disease state over time.17–19 In MS, clinical and MRI endpoints are frequently employed as measures of disease status, combining the sensitivity of MRI 5 with clinical assessments that provide a more direct assessment of the clinical impact of therapy. Studies evaluating the achievement of composite disease-activity-free status have become more common and may provide useful information for assessing the overall effectiveness of a treatment.5–8,14–16,20 Direct comparison of the current results with other analyses of treatment effects on the disease-activity-free endpoint is problematic because of differences in study populations, trial durations, study design, and in the level of MS disease activity in the control groups.14,15,21,22 Nevertheless, the current results are roughly comparable with the disease-activity-free findings reported at one year in the natalizumab AFFIRM trial, in which being disease-activity free at one year was approximately three times more likely in natalizumab-treated patients compared with placebo-treated patients (47% vs 15%). 8
One limitation of composite outcome measures in current MS trials is that results may sometimes be driven by an effect on only one of its individual components, particularly MRI-defined events. 23 As expected based on the higher sensitivity of MRI, absolute rates of disease-activity-free status in the current analysis were lower for both DAC HYP- and placebo-treated patients for MRI-defined measures compared with clinical measures of disease activity. In contrast, the relative impact of DAC HYP treatment on disease-activity-free status was higher for MRI measures (48% DAC HYP vs 18% placebo) compared to clinical measures (77% DAC HYP vs 60% placebo). This contrast between absolute rate of disease-activity freedom and the relative benefit of treatment is also apparent in the high disease activity subgroup. While a lower proportion of patients were disease-activity free on DAC HYP in the baseline highly active versus baseline nonhighly active subgroups (19% vs 43%), the relative impact of treatment was strongest in the highly active subgroup. DAC HYP-treated patients with highly active MS were more than six times as likely to have freedom from disease activity compared with placebo-treated patients with highly active MS (19% vs 3%).
Studies assessing long-term treatment efficacy have found that MRI-defined activity early in treatment is an important predictor of long-term response even in the absence of clinical activity. For example, in a 13-year longitudinal study of 30 patients, T2 lesions that appeared between baseline and year 2 strongly correlated with clinical disease severity at year 13. 24 Although the treatment effect of DAC HYP on becoming disease-activity free was stronger in months six to 12 compared with the first half of the treatment period, additional studies should be conducted to confirm the ability of DAC HYP to achieve disease-activity-free status in a wider MS population, and over a longer duration than SELECT.
The concept of freedom from disease activity is likely to evolve over time to encompass new clinical or radiological measures of disease, 8 , 15 such as gray matter atrophy, cortical lesion burden, and spinal cord pathology. Nevertheless, the clinical and radiological parameters used in the current definition are the most widely used by clinicians at present to define MS disease activity and assess therapeutic efficacy. The observed efficacy of DAC HYP on these measures will need to be weighed against its risks, which may include an increase in infections, cutaneous adverse events, and hepatotoxicity, 11 when considering the overall benefit/risk for an individual patient.
In conclusion, the current findings demonstrate that DAC HYP increases the numbers of MS patients who achieve freedom from disease activity as measured by a combination of lesion counts on serial cranial MRI and close clinical monitoring. These findings support further assessment of DAC HYP in MS.
Footnotes
Conflict of interest statement
Eva Havrdova has received speakers’ honoraria and research grant support from Bayer Schering Healthcare, Biogen Idec, Genzyme, Merck Serono, Novartis, and Teva, and compensation for advisory board activities from Biogen Idec, Genzyme, Merck Serono, Novartis, and Teva.
Gavin Giovannoni has received research grant support from Bayer Schering Healthcare, Biogen Idec, GW Pharma, Merck Serono, Merz, Novartis, Teva, and Sanofi-Aventis, and personal compensation for participating on advisory boards in relation to clinical trial design, trial steering committees, and data and safety monitoring committees from Bayer Schering Healthcare, Biogen Idec, Canbex Therapeutics, Eisai, Elan, Five Prime Therapeutics, Genzyme, Genentech, GSK, Ironwood Pharma, Merck Serono, Novartis, Pfizer, Roche, Sanofi-Aventis, Synthon BV, Teva, UCB Pharma, and Vertex Pharmaceuticals.
Dusan Stefoski has received research funding and support, and speaker bureau honoraria, from Biogen Idec, EMD Serono, Teva, Pfizer, Elan, and Novartis
Samantha Forster, who is an employee of Excel Scientific Solutions, provided medical writing and editorial support in the development of this manuscript, and this work was funded by Biogen Idec and AbbVie Biotherapeutics.
Kimberly Umans, Lahar Mehta, and Jacob Elkins are full-time employees of Biogen Idec.
Steven Greenberg is a full-time employee of AbbVie Biotherapeutics, a subsidiary of AbbVie Inc.
Funding
This work was supported by Biogen Idec and AbbVie Biotherapeutics. Biogen Idec and AbbVie Biotherapeutics had the opportunity to review and comment on the manuscript content; however, the authors had full editorial control of the manuscript and provided their final approval of all content.
Eva Havrdova was supported by the Czech Ministry of Education, VZ MSM 0021620849 and PRVOUK-P26/LF1/4.
Study investigators and other participants
SELECT study investigators: Czech Republic: Prof Zdeněk Ambler, Prof Ivan Rektor, Dr Radomir Talab, Prof Petr Kanovsky, Dr Pavel Stourac, Dr Denisa Zimova, Dr Marta Vachova. Germany: Prof Dr Bernd C. Kieseier, Dr Björn Tackenberg, Prof Dr Heinz Wiendl, Prof Dr Reinhard Hohlfeld, Prof Dr Klemens Angstwurm, Prof Dr Judith Haas, Prof Dr Uwe Zettl, Prof Dr Florian Stögbauer, Dr Ralf Linker, Prof Dr Andrew Chan, Prof Dr Patrick Oschmann. Hungary: Dr Attila Csányi, Dr Péter Diószeghy, Dr János Nikl, Dr Gyula Pánczel, Dr Béla Clemens, Dr Etelka Jófejű, Dr Attila Valikovics, Dr István Kondákor, Dr Dániel Bereczki, Dr Zsuzsanna Lohner, Prof Lászlo Csiba, Dr András Folyovich, Dr Péter Harcos, Dr Gabriella Kovács, Dr Mária Sátori. India: Dr Rajaram Agarwal, Dr Pahari Ghosh, Dr Sangeeta Ravat, Dr Subhash Mukherjee, Dr Rustom Wadia, Prof Kolichana Venkateswarlu, Dr Meenakshisundaram Umaiorubahan, Prof Medasari Padma, Dr Thomas Mathew, Dr AK Meena, Dr Suresh Kumar. Poland: Dr Wieslaw Drozdowski, Dr Waldemar Fryze, Dr Jan Kochanowicz, Prof Anna Kaminska, Dr Krzysztof Selmaj, Dr Andrzej Szczudlik, Dr Andrzej Wajgt, Dr Anna Czlonkowska, Dr Zbigniew Stelmasiak, Dr Gabriela Klodowska-Duda, Dr Janusz Zbrojkiewicz. Romania: Dr Ovidiu Bajenaru, Dr Dafin Fior Muresanu, Dr Mihaela Simu. Russia: Dr Olga Vorobyeva, Dr Leonid Zaslavs.ky, Dr Sergey Shvarkov, Dr Miroslav Odinak, Dr Anna Belova, Dr Irina Sokolova, Dr Farit Khabirov, Dr Natalia Nikolaevna Maslova, Dr Irina Poverennova, Dr Nikolay Spirin, Dr Nadezhda Malkova, Dr Semen Prokopenko, Dr Alexey Rozhdestvensky, Dr Alexei Boiko, Dr Rim Magzhanov. Turkey: Prof Sabahattin Saip, Prof Omer Faruk Turan, Ass. Prof Serhat Ozkan, Prof Ayse Sagduyu Kocaman. Ukraine: Dr Natalia Buchakchyys’ka, Dr Natalia Lytvynenko, Dr Borys Palamar, Dr Tatyana Nehrych, Dr Natalia Voloshina, Dr Larisa Sokolova, Prof Olexandr Kozyolkin, Dr Olena Moroz, Prof Valeriy Pashkovs.kyy, Dr Elena Statinova, Dr Tetjana Kobys, Dr Igor Pasyura. United Kingdom: Dr Clive Hawkins, Dr Basil Sharrack, Dr Cris Constantinescu, Dr John Zajicek, Dr David Bates, Dr Eli Silber.
Data Safety Monitoring Board: Dr Volker Limmroth (chair), Dr Richard Furie, Dr Daniel McQuillen, Dr Raymond Chung, and Dr Richard Kay.
Relapse Adjudication Committee: Dr Chris Polman, Dr Ted Phillips, Dr Paul O’Connor, Dr Ari Green, and Dr Oliver Lyon-Caen.
