Abstract
Background:
The mechanism underlying disease progression in progressive multiple sclerosis (MS) is uncertain. Pathological studies found widespread inflammation in progressive MS brains correlating with disease progression and axonal damage.
Objectives:
To study cerebrospinal fluid (CSF) biomarkers and clarify whether inflammation and axonal damage are associated in progressive MS.
Methods:
Using enzyme-linked immunosorbent assay (ELISA), we analysed CSF from 40 secondary progressive (SPMS), 21 primary progressive (PPMS), and 36 relapsing–remitting (RRMS) and 20 non-inflammatory neurological disease (NIND) patients. Twenty-two of the SPMS patients participated in an MBP8298 peptide clinical trial and had CSF follow-up after one year.
Results:
Compared to NIND patients, inflammatory biomarkers osteopontin and matrix metalloproteinase-9 (MMP9) were increased in all MS patients while CXCL13 was increased in RRMS and SPMS patients. Biomarkers of axonal damage (NFL) and demyelination (MBP) were increased in all MS patients. In progressive MS patients CSF levels of osteopontin and CXCL13 correlated with NFL while osteopontin and MMP9 correlated with MBP. MBP8298 treatment did not affect the levels of the biomarkers after one year of treatment. All biomarkers were continuously increased after one year of follow-up except MBP, which decreased.
Conclusion:
CSF biomarkers of inflammation, axonal damage and demyelination are continuously increased in progressive MS patients and correlate. These findings parallel pathology studies, emphasise a relationship between inflammation, axonal damage and demyelination and support the use of CSF biomarkers in progressive MS clinical trials.
Keywords
Introduction
Progressive multiple sclerosis (MS) is characterised by a slow but steady progression of neurological disability without remission, and entering the progressive disease phase is a major determinant for the long-term prognosis in MS. 1,2 A prevailing view has been that progressive disease activity is driven by neurodegenerative processes, occurring independent of inflammation. 3 This view has been challenged by pathology studies, which have found pronounced inflammation in progressive MS. 4,5 Other studies established that inflammation, demyelination and axonal damage are closely associated. 6 –8 Additionally, oxidative stress has been suggested to contribute to axonal damage and demyelination. 9,10
Magnetic resonance imaging (MRI) measures, which have been established as the primary surrogate biomarker for disease activity in relapsing–remitting multiple sclerosis (RRMS), do not reflect the pathology of progressive MS well, and conventional MRI measures show only modest associations with disease progression. 11,12 Thus, there is an unmet need for biomarkers reflecting the disease processes in progressive MS.
Cerebrospinal fluid (CSF) studies have confirmed the findings of increased inflammation, 13,14 axonal damage, 15,16 demyelination 17 and oxidative stress 18,19 in progressive MS. However, to our knowledge no studies have explored whether these CSF biomarkers are related in progressive MS. On this background we hypothesised that biomarkers of inflammation, axonal damage and demyelination are increased in secondary progressive MS (SPMS) and primary progressive MS (PPMS) and that these biomarkers correlate. In addition, we studied the effect of treatment with the peptide MBP8298 in repeated lumbar puncture samples from patients participating in a pivotal treatment trial of this compound in SPMS. 20
Materials and methods
Subjects
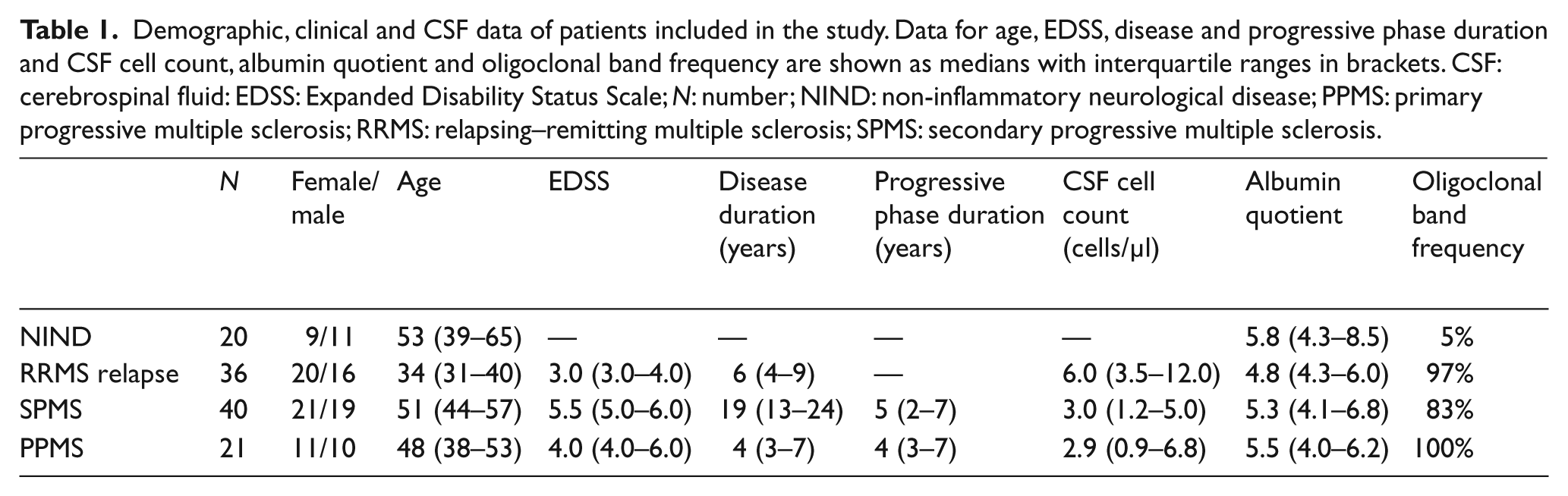
We included CSF samples from MS patients diagnosed and classified according to the McDonald criteria 21 (40 SPMS, 21 PPMS, and 36 RRMS patients in relapse) and 20 non-inflammatory neurological disease (NIND) (five herniated discs; five low back pain; three spinal stenosis; one seizure; one Charcot-Marie-Tooth disease; one headache; one amyotrophic lateral sclerosis; one psychosomatic symptoms; one stroke; one transient cerebral ischaemia) collected at Copenhagen University Hospitals Rigshospitalet and Glostrup; and Karolinska Hospital, Stockholm (Table 1). All MS patients were untreated at the time of sampling, and in patients in relapse lumbar puncture was performed prior to the initiation of steroid therapy. Lumbar puncture was repeated after one year in 22 of the SPMS patients, all of whom were enrolled in the MAESTRO study, nine receiving the MBP8298 peptide and 13 receiving placebo. 20 All patients had Expanded Disability Status Scale (EDSS) scores assigned by a neurologist.
Demographic, clinical and CSF data of patients included in the study. Data for age, EDSS, disease and progressive phase duration and CSF cell count, albumin quotient and oligoclonal band frequency are shown as medians with interquartile ranges in brackets. CSF: cerebrospinal fluid: EDSS: Expanded Disability Status Scale; N: number; NIND: non-inflammatory neurological disease; PPMS: primary progressive multiple sclerosis; RRMS: relapsing–remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis.
Written informed consents were obtained from all patients, and the studies were approved by the local scientific ethics committees.
CSF samples and analysis
CSF was cooled on an ice-bath during sampling, centrifuged at 4°C, and the cell-free CSF immediately frozen at −80°C until analysis. We used Human Osteopontin Quantikine enzyme-linked immunosorbent assay (ELISA) Kit, Human CXCL13 Quantikine ELISA Kit, Human MMP-9 Quantikine ELISA Kit, Total Nitric Oxide and Nitrite/Nitrate Assay (all from R&D Systems, USA), NF-Light Neurofilament ELISA (UmanDiagnostics, Sweden), Myelin Basic Protein ELISA (Beckman Coulter, USA) and Human glial fibrillary acidic protein (GFAP) ELISA (Biovendor, Czech Republic). We performed tests of stability after three freeze-thaw cycles, and found matrix metalloproteinase-9 (MMP9), total nitric oxide (NOx), neurofilament light (NFL) and myelin basic protein (MBP) to be stable under these conditions. Therefore, samples used in these assays were allowed to have passed up to three freeze-thaw cycles. In the CXCL13, MMP9 and MBP assays, some samples were below detection level, and we assigned the value of the lowest detection level to the sample (CXCL13 = 1.6 pg/ml; MMP9 = 0.16 ng/ml; MBP = 0.1 ng/ml). Samples were analysed in duplicates and mean intra-assay variability was 8.9% for osteopontin, 6.5% for CXCL13, 10.2% for MMP9, 7.7% for NOx, 3.5% for NFL, 12.9% for MBP and 9.1 % for GFAP.
Statistical analysis
Statistical analysis was performed using PASW 19 software (IBM, USA). Data did not fit to the normal distribution, and accordingly non-parametric analyses were applied.
Results
CSF biomarkers of inflammation are increased in progressive MS
As shown in Figure 1A, we found osteopontin to be significantly increased in all three subtypes of MS compared to NIND patients. The osteopontin level is in the same range across the three subtypes. CXCL13 levels are significantly increased in RRMS relapse and SPMS compared to NIND patients, and RRMS relapse patients have increased levels compared with SPMS patients. MMP9 is increased in all MS subtypes, compared to NIND patients, where MMP9 concentrations were below the detection level.
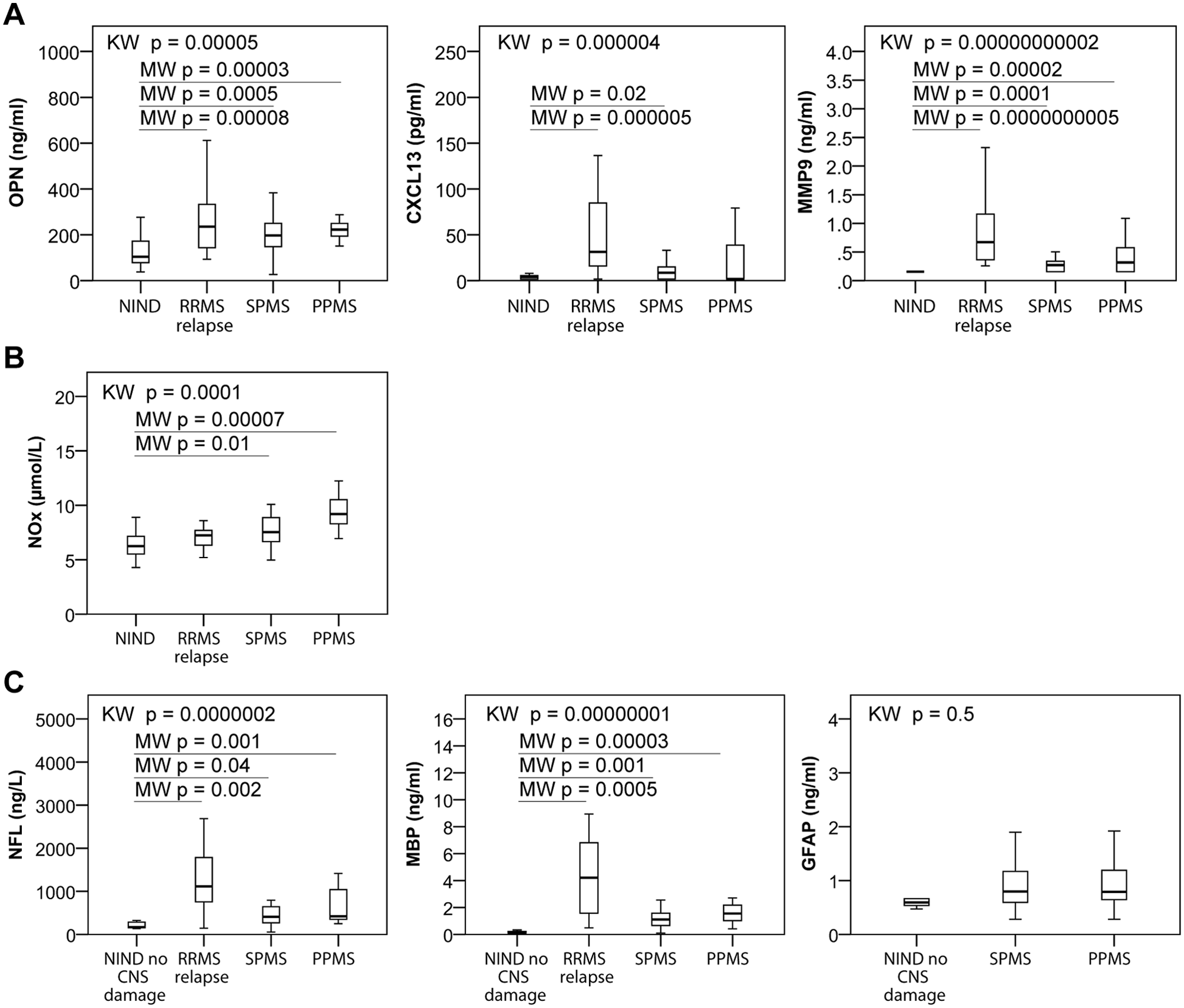
Levels of biomarkers in CSF from MS and NIND patients. Boxplots showing levels of the CSF biomarkers of (A) inflammation (OPN, CXCL13 and MMP9); (B) oxidative stress (NOx); and (C) axonal damage (NFL) and demyelination (MBP) in NIND, RRMS in clinical relapse, SPMS and PPMS patients. Kruskal-Wallis test (KW) was used for testing multiple groups and Mann-Whitney (MW) was used for post-hoc testing against the NIND group. For NFL, MBP and GFAP post-hoc tests were performed against NIND patients with no expected CNS damage (NIND no CNS damage). CNS: central nervous system; CSF: cerebrospinal fluid; GFAP: glial fibrillary acidic protein; MBP: myelin basic protein; MMP9: matrix metalloproteinase-9; MS: multiple sclerosis; NFL: neurofilament light chain; NIND: non-inflammatory neurological disease; NOx: total nitrite/nitrate; OPN: osteopontin; PPMS: primary progressive multiple sclerosis; RRMS: relapsing–remitting multiple sclerosis; SPMS: secondary progressive multiple sclerosis.
CSF biomarker of oxidative stress is increased in progressive MS
Since oxidative stress has been suggested to be a contributing factor in the pathogenesis of progressive MS, 10 we included NOx as a biomarker for oxidative stress. As Figure 1B shows, we found NOx to be significantly increased in SPMS and PPMS patients compared to NIND patients.
CSF biomarkers of axonal damage and demyelination are increased in progressive MS
Given that biomarkers of axonal damage and demyelination are expected to increase under conditions with central nervous system (CNS) damage, we chose to split the NIND group in two; one group with expected CNS damage based on the diagnosis (NIND CNS damage); and one group with no expected CNS damage based on the diagnosis (NIND no CNS damage; low back pain, headache and psychosomatic symptoms). NIND no CNS damage was used as the control group in the post-hoc tests. We found (Figure 1C) both NFL and MBP to be significantly increased in all MS subtypes, most markedly in RRMS relapse patients. We did not find any significant changes for GFAP.
Correlations between CSF biomarkers of inflammation and neuronal damage in progressive MS
Having established an association between progressive MS and CSF biomarkers of inflammation, oxidative stress, axonal damage and demyelination, we next sought to determine whether inflammation is related to axonal damage and demyelination. Accordingly, we performed Spearman’s correlation analysis on the data from progressive MS (SPMS and PPMS) patients. As shown in Figure 2, we observed significant correlations between osteopontin levels and NFL and MBP; CXCL13 and NFL; and MMP9 and MBP. NOx, the biomarker of oxidative stress, correlated significantly with MBP (Figure 2). These findings indicate that in progressive MS, there is a close relationship between inflammation and axonal and myelin damage, a relationship which is most pronounced and consistent for osteopontin.
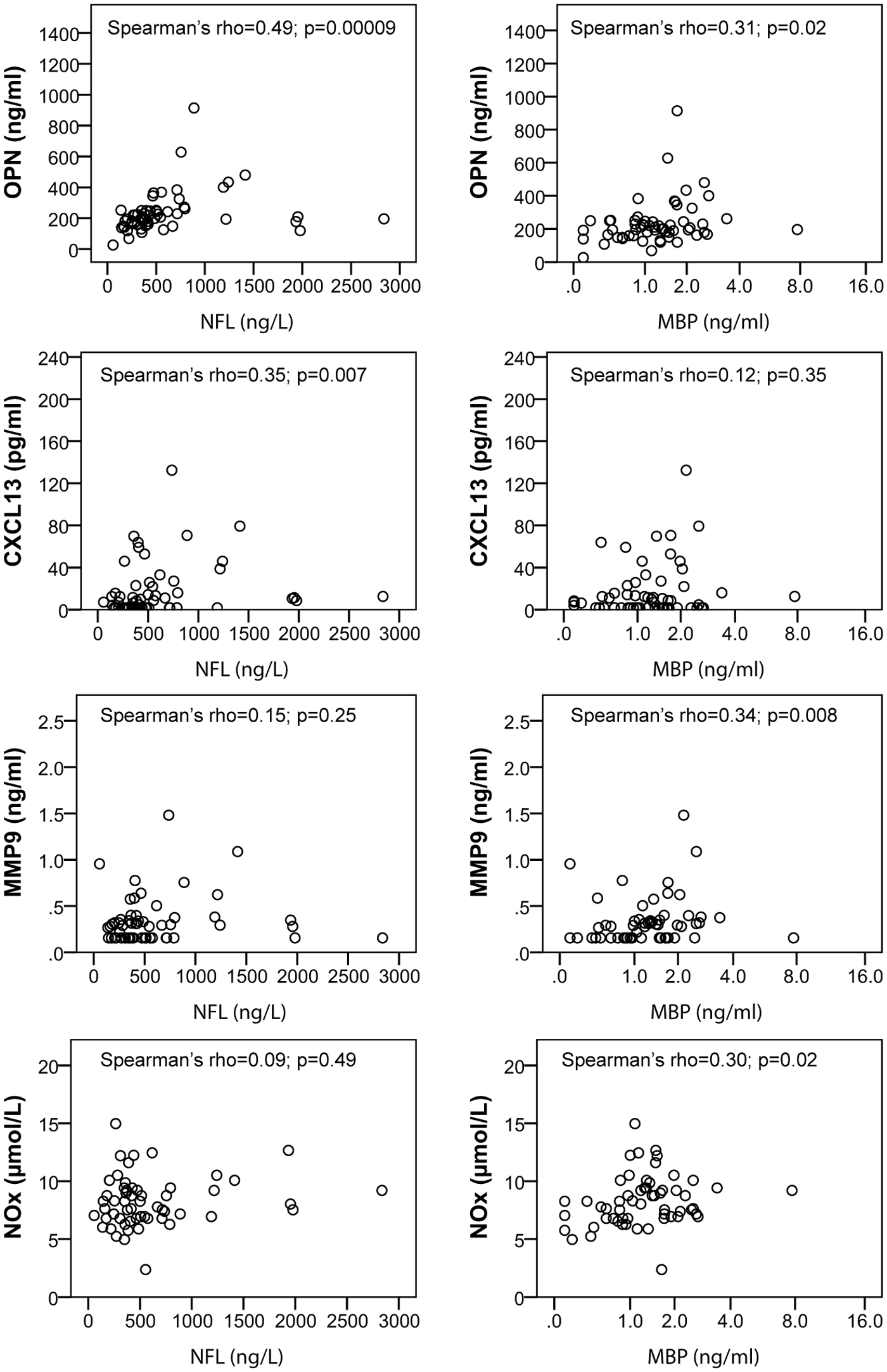
Correlation analyses of CSF biomarkers of inflammation and oxidative stress versus CSF biomarkers of axonal damage and demyelination in progressive MS patients. Dot plots with Spearman’s rank correlation coefficient (Spearman’s rho) showing the relationship between the biomarkers of inflammation (OPN, CXCL13 and MMP9), the biomarker of oxidative stress (NOx), and the axonal damage biomarker neurofilament light chain (NFL) and the demyelination biomarker myelin basic protein (MBP) in CSF from progressive MS patients. CSF: cerebrospinal fluid; MBP: myelin basic protein; MMP9: matrix metalloproteinase-9; MS: multiple sclerosis; NFL: neurofilament light chain; NOx: total nitrite/nitrate; OPN: osteopontin.
No effect of MBP8298 peptide treatment on CSF biomarkers
CSF from nine SPMS patients randomised to treatment with the MBP8298 peptide and 13 receiving placebo was analysed prior to treatment and after one year of treatment. We found no difference between MBP8298 peptide-and placebo-treated patients in absolute levels or in changes in CSF biomarkers over the one-year period studied (data not shown).
Changes in CSF biomarkers over time
The changes in the CSF biomarkers over time are interesting for two reasons. Firstly, pathological findings indicate that many features of progressive MS pathology are continuous over time, as opposed to the acute focal lesions characteristic of RRMS. 3 Secondly, since CSF biomarkers are candidate surrogate biomarkers for treatment effects in progressive MS, it is of interest whether these change substantially over time. Figure 3 shows the development in CSF biomarker levels from baseline to 12 months for each patient, and using the Wilcoxon signed-rank test we found no significant differences between baseline and 12 months levels for osteopontin, CXCL13, MMP9, NOx, NFL and GFAP. For MBP, there was a significant decrease from baseline (median 0.85 ng/ml) to month 12 (median 0.65 ng/ml).
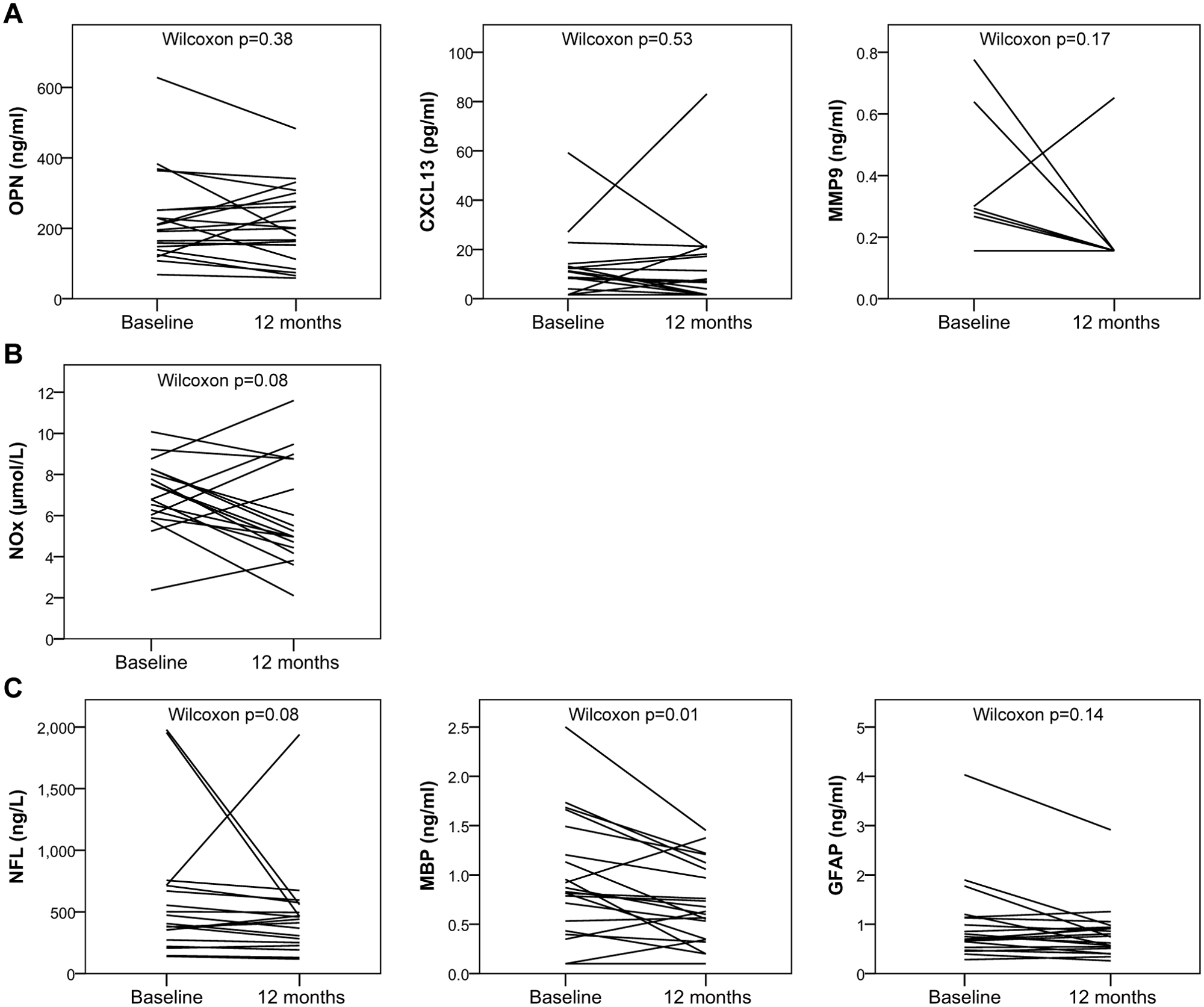
Development over time of CSF biomarkers in SPMS patients. Twenty-two SPMS patients participating in a clinical trial with MBP8298 peptide had lumbar puncture at baseline and after one year. No differences between MBP8298 peptide- and placebo-treated patients were observed after one year. Lines represent the development in CSF biomarker level for each patient during one year. Wilcoxon signed-rank test was used to analyse the paired samples. CSF: cerebrospinal fluid; GFAP: glial fibrillary acidic protein; MBP: myelin basic protein; MMP9: matrix metalloproteinase-9; MS: multiple sclerosis; NFL: neurofilament light chain; NOx: total nitrite/nitrate; OPN: osteopontin; SPMS: secondary progressive multiple sclerosis.
Discussion
The findings of the present study confirm that in CSF from progressive MS patients, there is an increase in biomarkers of inflammation, axonal damage, demyelination and oxidative stress and, importantly, correlation analyses of the CSF biomarkers emphasise the close relationship between inflammation, axonal damage and demyelination. Further, the study provides evidence that the CSF biomarkers studied generally are stable over time in SPMS, and thus could be suitable as surrogate biomarkers in clinical trials. Finally, we found that treatment with the MBP8298 peptide did not change the CSF concentrations of any of the biomarkers studied. This is in agreement with the lack of efficacy of this compound in a phase III randomised, placebo-controlled trial. 20
We used a cross-sectional approach to study the levels of the CSF biomarkers in SPMS and PPMS, and used RRMS relapse patients and a group of NINDs as control groups. This approach is usual given the limited availability of CSF samples, and suitable for establishing an involvement of the CSF biomarkers. However, cross-sectional studies have low sensitivity for detecting a difference if a biomarker fluctuates. Consequently, we included a longitudinal analysis of CSF samples from SPMS patients obtained one year apart to address whether the CSF biomarkers fluctuate over time.
Previous studies have shown that osteopontin is increased in lesional brain tissue from progressive MS patients, 22,23 that osteopontin has important pro-inflammatory functions, 24 that osteopontin is increased in CSF from MS patients 25 and that osteopontin decreases in CSF from RRMS patients during natalizumab treatment. 26 We found osteopontin to be increased in CSF from SPMS, PPMS and RRMS in relapse compared to NIND patients. A previous study from our group found significantly increased osteopontin in CSF from RRMS relapse and PPMS patients, whereas there was a non-significant increase in CSF osteopontin from SPMS patients. 14 This difference most likely represents the larger SPMS cohort in the present study. Importantly, we find osteopontin levels to be correlated with both NFL and MBP levels in CSF from SPMS and PPMS. Thus, osteopontin is the inflammatory CSF biomarker which correlates best with the markers for axonal damage and demyelination. Finally, osteopontin levels are stable and continuously increased over a one-year interval in SPMS.
CXCL13 has previously been shown to be increased in active lesions from progressive MS patients and linked to the frequency of B-cells in the CSF, 27 to be present in ectopic lymphoid follicles of SPMS patients, 28 to be increased in all subtypes of MS, but most pronounced during relapses, and to be decreased during natalizumab and methylprednisolone treatment. 13 In the present study we found significantly increased CXCL13 in SPMS and RRMS relapse patients; we could not reproduce the previous finding of increased CXCL13 in PPMS; 13 however, it is worth noting that 10 out 21 PPMS patients had CXCL13 above 10 pg/ml. CXCL13 correlated significantly with NFL in SPMS and PPMS patients, and was stable in the longitudinal study.
MMP9 has also been implicated in MS pathogenesis and is thought to be important for the transmigration of blood cells across the glia limitans 29 of the blood-brain barrier. In agreement with previous studies, 30 we found MMP9 to be significantly increased in progressive MS and more markedly in RRMS relapse. MMP9 correlated significantly with MBP levels in progressive MS patients and was stable in SPMS patients at one-year follow-up.
Since oxidative damage has been suggested to contribute to axonal damage and demyelination in progressive MS, we included NOx in our analyses. We found NOx to be increased in the progressive forms of MS, whereas in RRMS relapse there was only a trend (p = 0.08) toward increase. In general other studies have found increased NOx in MS, but with varying results for the subtypes. 18,19 In CSF from progressive MS, levels of NOx correlated with MBP, but not NFL. Consequently, these results do not support a major role of NOx in relation to the development of axonal damage.
The findings of increased inflammatory biomarkers in CSF of progressive MS underscore that inflammation is present in the CNS compartment in progressive MS, and thus corresponds very well to the findings in pathology studies. 7 Indeed osteopontin, 22,31 CXCL13 27,28 and MMP9 32 have previously been found to be highly expressed in lesions from progressive MS brains. Compared with RRMS in relapse, the degree of inflammation, especially for CXCL13, is less intense in progressive MS. Likewise; we find that biomarkers of axonal damage (NFL) and demyelination (MBP) are increased in all subtypes of MS, with a marked increase in RRMS relapse patients. These findings are in accordance with previous studies; 13 –15,17,30 however, in the interpretation one must emphasise the differences in dynamics between the relapsing and progressive disease phases. RRMS is characterised by transient attacks of focal inflammation, which is reflected by intense inflammation in the acute plaques, whereas progressive MS is characterised by a chronic, less intense inflammation, which is diffusely disseminated to white matter, grey matter, meninges and normal-appearing white matter (NAWM). 7 This is paralleled by studies showing that axonal damage is most intense in lesions from RRMS compared to progressive MS patients, whereas axonal damage is more widely distributed in progressive MS. 6,7 Thus, our findings of inflammation, axonal damage and demyelination in progressive MS are in line with recent pathology studies. A likely interpretation is that progressive disease activity occurs on a background of continuing inflammation and axonal damage, which can be measured in the CSF of progressive MS patients.
A central question in the understanding of the pathogenesis of progressive MS is whether inflammation and axonal damage are associated. In support of a relationship between levels of inflammation and axonal damage, we found that osteopontin and CXCL13 correlated significantly with NFL levels in progressive MS patients. To our knowledge, this is the first study demonstrating a relationship between inflammatory and axonal damage biomarkers in CSF from progressive MS patients. Again, this finding is in accordance with pathology studies 6,7 which have found an association between inflammation and axonal damage in progressive MS. An unexpected finding in the present study was that the CSF concentration of MBP decreased slightly but significantly during one year of follow-up. MBP has been established as a biomarker of demyelination in clinically isolated syndromes and RRMS, where there is a close relationship between concentrations of MBP and disease activity. 33 Furthermore, MBP in CSF is a response biomarker of the effect of treatment with methylprednisolone, and patients with high CSF concentrations of MBP have a better response to methylprednisolone. 34,35 It is tempting to speculate that the decreases in MBP concentrations could reflect the pathological observation of a shift from a white matter demyelinating pathology to a cortical-based pathology, where demyelination is present but less intense than in the white matter, but this hypothesis must be addressed in future studies.
With the appearance of more effective treatments for RRMS patients, studies have shown that CSF biomarkers of inflammation and axonal degeneration are useful as biomarkers for treatment effects. 13,14,26,36 As development of treatments for progressive MS is challenging, CSF biomarkers for inflammation and axonal degeneration could be valuable in future clinical trials. The observation of stability over a one-year period of the inflammatory and axonal degeneration biomarkers in CSF from SPMS patients is supportive of the use of these biomarkers in future studies. Future studies should address the extent to which these CSF biomarkers correlate with the long-term development of neurological disability in progressive MS.
In conclusion, we find that biomarkers of inflammation and axonal damage are continuously increased in the CSF of progressive MS patients and, importantly, inflammation and axonal damage biomarkers are significantly correlated. These findings are in accordance with recent pathology studies and indicate a relationship between inflammation, axonal damage and disease progression in MS.
Footnotes
Acknowledgements
We wish to thank laboratory technicians Joy Mendel-Hartvig, Michael Kolbjørn Jensen, Vibeke Fuglholt and Rikke Larsen for their skilful help with the ELISA experiments.
Funding
This work was supported by the Danish Council for Independent Research (grant 271-06-0246), the Danish Council for Strategic Research (grant 2142-08-0039), the Danish MS Society, the Warwara Larsen Foundation, the Johnsen Foundation, Brdr. Rønje Holding, Jeppe Juel Memorial Legacy, research grants from Biogen Idec, the Swedish Research council, the AFA foundation, the Swedish Brain Foundation and the Knut and Alice Wallenbergs Foundation.
Conflict of interest
J. Romme Christensen has received honoraria for lecturing from Biogen Idec and has had travel expenses covered by Merck Serono. L. Börnsen, M. Khademi and P.E. Jensen report no conflict of interest. T. Olsson has received honoraria for lectures, participation in advisory boards and unrestricted MS research grants from Biogen Idec, Novartis, Merck, Sanofi Aventis and Bayer. P.S. Sørensen has served on scientific advisory boards for Biogen Idec, Merck Serono, Novartis, Genmab, TEVA, Elan, and GSK; has been on steering committees or independent data monitoring boards in clinical trials sponsored by Merck Serono, Genmab, TEVA, GSK, and Bayer Schering; and he has received funding for travel for these activities; has served as editor-in-chief of the European Journal of Neurology, and is currently editorial board member for Multiple Sclerosis Journal, European Journal of Neurology, Therapeutic Advances in Neurological Disorders; and has received speaker honoraria from Biogen Idec, Merck Serono, TEVA, Bayer Schering, Sanofi Aventis, and Novartis. His department has received research support from Biogen Idec, Bayer Schering, Merck Serono, TEVA, Baxter, Sanofi-Aventis, BioMS, Novartis, Bayer, RoFAR, Roche, Genzyme, from the Danish Multiple Sclerosis Society, the Danish Medical Research Council, and the European Union Sixth Framework Programme: Life Sciences, Genomics and Biotechnology for Health. F. Sellebjerg has served on scientific advisory boards for Biogen Idec, Merck Serono, Novartis, Sanofi-Aventis and TEVA and as consultant for Biogen Idec and Novo Nordisk; has received support for congress participation from Biogen Idec and Sanofi Aventis; has received speaker honoraria from Biogen Idec, Merck Serono, Bayer Schering, Schering-Plough, Sanofi-Aventis and Novartis; and has received research support from Biogen Idec, Bayer Schering, Merck Serono, Sanofi-Aventis and Novartis.
