Abstract
Bio-based materials have attracted great attention from industry and academia in tribology because they are renewable, bio-degradable and easily accessible. This study aims to evaluate the efficiency of bio-based materials used both as neat lubricants and additives on friction and wear performance. The evaluated lubricants include oleic acid, a mixture containing oleic acid and dodecyl amine in hexadecane and a neat ionic liquid composed of oleic acid and dodecyl amine. The influence of the additive concentration, ratio between acid and amine, and oscillation frequency under reciprocating contacts has been investigated. Surface analysis was conducted by optical microscope, scanning electron microscopy and energy-dispersive X-ray spectroscopy, and time-of-flight secondary ion mass spectrometry. The results confirm that a small amount of oleic acid as an additive can provide limited friction reduction. Bio-based ionic liquid shows the lowest friction and wear coefficient in all testing conditions. A synergistic effect between oleic acid and dodecyl amine as an additive mixture has been observed when the two components are equimolar and the oscillating frequency is high. Time-of-flight secondary ion mass spectrometry analysis of the wear track indicates that the tribological process enhances the ionic character of the additive mixture, improving the lubrication performance.
Introduction
Lubricant additives have a key role in tribology performance, especially under boundary lubrication regime, where direct contact occurs causing the highest friction and wear among all lubrication regimes. In current engineering applications, lubricant additives, such as zinc dialkyl-dithiophosphate (ZDDP) and molybdenum dialkyl-dithiocarbamate (MoDTC), are applied to improve the tribological performance under boundary lubrication regime. 1 The research of ZDDP has been going on for several decades 2 and a synergistic phenomenon between ZDDP and MoDTC was investigated in studies.3,4 Studies on in-situ experimental methods have also improved our understanding of the formation kinetics and properties of ZDDP tribo-films.5–8 These conventional metallic additives, however, contain sulphur and phosphorus, which can accumulate on the surfaces of mechanical components, suppressing the effectiveness of other additives and contaminating exhausting gas catalytic converters. These additives also contain heavy metals, causing sulphate ash. Therefore, it is important to reduce the sulphur, phosphorus and heavy metal concentrations in lubricants by substituting conventional additives with ‘greener’ alternatives.
One promising substitute for conventional additives is bio-based materials such as fatty acids and esters, amides and amines, which have been classified as organic friction modifiers. 9 This is also in correspondence with the principles of green tribology concept that the application of bio-based materials in lubricants should be as much as possible, as proposed in the literature. 10 Bio-based materials can be obtained from renewable resources and easily manufactured through refineries and purification from natural products without the necessity of complex synthesis processes as those for conventional metallic additives. 1 Some efforts11–14 can be found to combine certain bio-based materials with a low-concentration ZDDP to reduce its consumption and improve tribological performance at the same time.
Bio-based additives have a common structure: A polar head and a hydrophobic carbon chain. Carboxylic acid is very effective among different types of polar heads in reducing friction and wear.15–17 At the same time, fatty acids are accessible with abundant sources from plants, nuts and animal fats.18,19 Hence, they have been widely studied among different bio-based additives. As summarised in the literary works,9,20 the tribological improvement originates from the adsorption of additives on contact surfaces through both physical and chemical processes and this adsorption can be reflected by adsorption rate and surface coverage, which are related to the tribology performance. A general conclusion is that a higher adsorption rate and surface coverage generate better surface protection with lower friction and wear.21–23 The chemical adsorption of fatty acid on metals has been further studied both experimentally24,25 and theoretically. 26 The results confirm that chemical adsorption shows a much stronger interaction than physical adsorption. A similar conclusion can also be found in, 27 validated through experiments with quartz-crystal microbalance and molecular tribometer. The authors concluded that the stronger interaction between the carboxylic groups and the surface resulted in a higher order and thinner adsorption film than that for the amine group. The boundary film for the carboxylic head additive has a lower tangential stiffness than the amine head, hence lower friction. 27
There are many other factors that influence the tribology performance of bio-based additives. The concentration of the additive in base oils is first considered and a minimum value must be reached to witness friction deduction.9,28 The friction reduction continues with the increase of the additive concentration until a plateau is reached. Further increase of the concentration provides a marginal benefit to tribology performance and may even lead to additional cost. 29 The chain length also has a great influence on the additive effectiveness, with a similar trend as the concentration. 28 A fatty acid with 8 carbons in the chain shows obvious friction reduction, and the longer the chain, the lower the friction is. 30 One explanation for this phenomenon is that a longer carbon chain can lower the free energy of adsorption and facilitate the adsorption, 31 resulting in a higher surface coverage. 32 Another explanation is that a longer chain leads to larger interaction forces between additives molecules, forming condensed protecting films that produce lower friction and wear than those from shorter chain additives. 33 Apart from the chain length, the chain structure also plays a crucial role in the lubricity of the organic additives. As an example, isostearic acid with more branched methyl group structures presents a lower viscosity under higher confinement than un-branched isostearic acid. This lower shear force is attributed to the branched methyl groups preventing the ordering under nanoconfinement. 34 The unsaturation of additives can affect the tribology performance. The previous works conclude that saturated additives have better performance than unsaturated ones35,36 since the straight chain of saturated additives helps form well-organised layers.37,38 It is also confirmed that saturated additives show higher surface coverage and adsorption kinetics than unsaturated acids.39,40 A more recent work studied the influence of fatty acid saturation in terms of friction laws with a molecular tribo-meter. 41 It is found that elaidic acid (the trans-configurated isomer of C18 oleic acid) forms a dense and rigid boundary layer due to the Van der Waals force between double bonds, leading to a larger friction reduction than oleic acid with cis-configuration. The stearic acid, without saturation, performs between the above two unsaturated C18 fatty acids. The above factors are summarised from the prospects of the characters for the additives. Meanwhile, the tribology response of bio-based additives varies notably under different operating scenarios, such as temperature, contact pressure, sliding speed, and surface roughness.
The progress mentioned above has been achieved in a single-additive system. The continuous effort to improve the performance leads to another direction in which fatty acids and amines are synthesised into protic ionic liquids (ILs) that can be used as either neat lubricants or lubricant additives. These types of ILs are free of sulphur or phosphorus, bio-degradable, non-corrosive and non-toxic, and show good tribology performance. Due to the abundant source of oleic acid in nature, it is widely used as an anion to form these protic ILs. In the work of Shi and Larsson, 42 oleic acid and oleyl amine form an IL, producing excellent lubricating performance in both boundary and EHL conditions. In the study of Kreivaitis et al., 43 three types of amines were combined with oleic acid into ILs and investigated as neat lubricants, showing a friction coefficient as low as 0.05 under reciprocating contacts. These ILs were also tested under different contact pairs, where steel/steel contact had the lowest wear volume among all testing conditions. 44 Oleate ILs were used as additives in water with 1 wt% to reduce friction and wear, 45 and both diethanolamine oleate and tert-octylamine oleate can significantly decrease friction and wear for steel-steel contact pairs; however, not as efficiently as neat lubricants. In addition, under aluminium-steel contact, the friction is always higher than that for steel/steel contact. 45 The IL containing trioctylphosphine and oleic acid gave a better tribology response than the ILs with a carbon number less than 10 in the anion, when used as additives in polyethylene glycol (PEG200). 46 Oleic acid and three short-chain amines with hydroxyl groups were prepared into ILs and tested as neat lubricants under an aluminium/aluminium contact. 47 An improvement in tribology performance was witnessed for all three samples, among which Bis-2-hydroxy ethanol ammonium oleate performed the best in friction reduction, and the other two showed better wear performance. 47 The tribological performance of the IL, 2-(hydroxyethyl) ammonium oleate was examined under a copper/copper contact pair, confirming that IL has superior lubricity over the PAO base oil. 48 Three different fatty acids were combined with methyltrioctyl amine, respectively, to form three ILs and all these ILs showed good thermal stabilities and traction performance. 49 Two wt% of the IL, made from oleic acid and tetrabutylammonium in pentaerythritol tetra oleate (polyol) showed the best friction reduction, compared to ILs containing stearic and linoleic acid. 50
The studies above underline an interesting and important direction where a combination of oleic acid with other organic amines can boost the lubrication performance of bio-based materials. More recently, the research on the combination of bio-based materials has been further promoted and results illustrate that through mixing very low concentration (0.5 mM) of oleic acid and oleyl amine in hexadecane, 51 a similar friction coefficient to the neat IL 42 can be achieved. This is explained by the fact that the pre-formed IL in the lubricant mixture functions as effectively as the neat IL. 42 Hence, this work manifests that the tribology performance of fatty acid can be enhanced with fatty amine through not only the synthesis of ILs but also a simple additive mixture. 51 Then whether this finding in 51 is generic for other existing bio-based ILs made from fatty acid and amine can be an interesting question. Meanwhile, a direct tribological comparison between an additive mixture and an IL containing the same components is still missing. The lubrication mechanism of neat protic ILs and the combination of its components in a solvent has yet to be revealed. Moreover, how the testing scenarios including total additive concentration, the ratio between acid and amine and reciprocating frequency affect the synergism between fatty acid and amine has not been discussed. To further probe the potential of bio-based materials as lubricant additives, in the current study, the tribology of oleic acid, a combination of oleic acid and dodecyl amine in hexadecane and neat IL, has been experimentally evaluated. The worn surface of pins and plates was characterised by scanning electron microscopy and energy-dispersive X-ray spectroscopy (SEM-EDX) and time-of-flight secondary ion mass spectrometry (ToF-SIMS) techniques, respectively.
Experimental
Lubricants
The hexadecane, oleic acid and dodecyl amine were supplied by Sigma-Aldrich, with a purity of all three components over 98%. The chemical structures of oleic acid and dodecyl amine are shown in Figure 1.
Chemical structure of oleic acid and dodecyl amine.
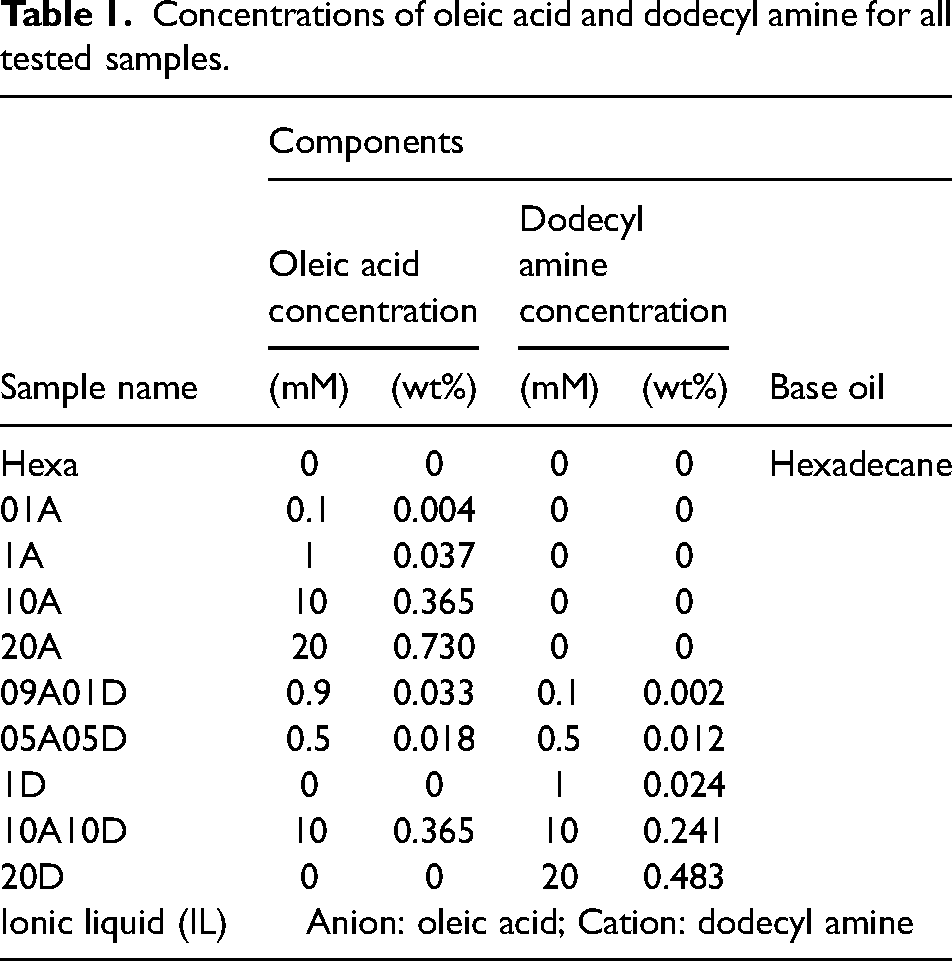
Hexadecane was used as a based oil and both acid and amine with designed concentrations are listed in Table 1. The sample names include both numbers and letters. The letter A denotes oleic acid and D denotes dodecyl amine. The numbers after each letter indicate the concentration of the corresponding additive, with unit 10−3 mol/L (mM). For instance, the lubricant 09A01D means this sample contains 0.9*10−3 mol/L oleic acid and 0.1*10−3 mol/L dodecyl amine with hexadecane as the base oil. All samples were mixed by magnetic stirring under 50 °C for about 1 h to ensure that samples were in a homogeneous state. The IL was prepared by carefully adding oleic acid into the dodecyl amine, with the reactants being stirred. To ensure that synthesis was properly completed, the product was examined with the Fourier transform infrared spectroscopy (FT-IR) method. The dynamic viscosity of samples, neat hexadecane, 20A, 10A10D and IL was measured with a rheometer and the results, provided in the supplementary materials, were used to estimate the lubrication regime.
Concentrations of oleic acid and dodecyl amine for all tested samples.
Methods
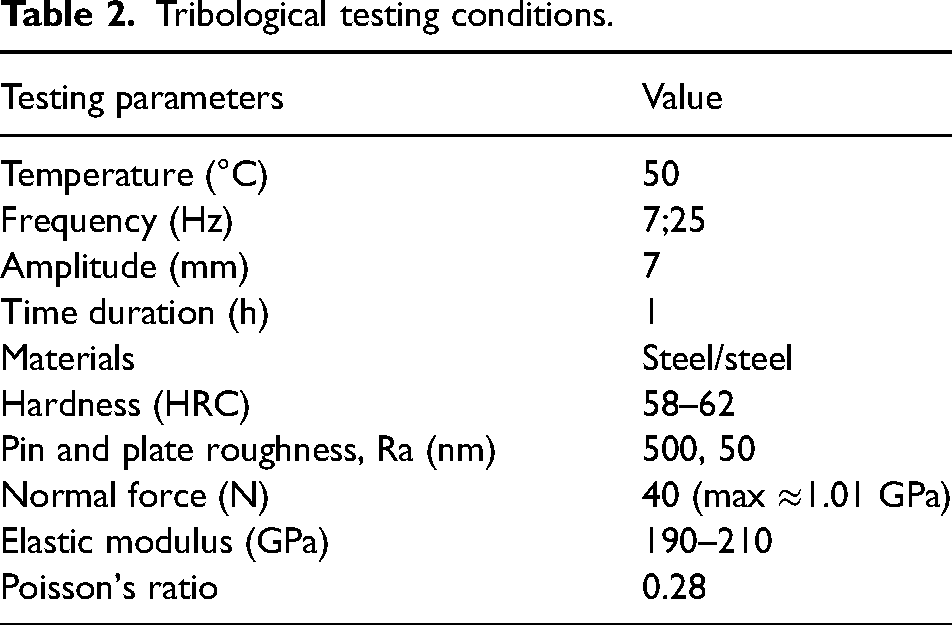
The tribology study was conducted with a reciprocating test rig, Cameron-Plint TE77. A pin-on-plate configuration was applied with a radius of 10 mm for the pin. Both pins and plates were made of steel EN31 with a hardness value of 58-62 HRC. The surface roughness, Ra, is about 50 nm for the plate and 500 nm for the pin. Before the tests, all pins and plates were immersed in heptane and cleaned in an ultrasonic bath. The stroke length was 7 mm with two different oscillating frequencies, 7 and 25 Hz, which gives two sliding velocities, 0.1 and 0.35 m/s, respectively. A normal load of 40 N produced an initial maximum contact pressure of 1.01 GPa. Each test lasted for 1 h which was long enough to reach a stable state, under a temperature of 50 °C. The above testing conditions are summarised in Table 2. With all the above parameters, the lubrication regime was estimated in a conservative condition and the detailed calculation procedure was illustrated in the supplementary material. With the lambda ratios much lower than 1, all tests were conducted under boundary lubrication conditions. The friction coefficient for each test was obtained by averaging the value of the last 20 min. An average of at least three tests per condition was regarded as the final friction value for each testing condition.
Tribological testing conditions.
Following the tribology tests, the wear scar diameter on pins was measured with an optical microscope (Leica Microsystems), then the wear coefficient was calculated by dividing wear volume by the product of normal load and sliding distance. The wear mechanisms were obtained by examining the wear scar on pins using scanning electron microscopy (SEM) with a Carl Zeiss EVO MA 15 (Carl Zeiss AG), energy-dispersive X-ray spectroscopy (EDX) from Oxford Instruments was then applied to detect the element type and concentration. ToF-SIMS analysis was conducted on the wear tracks from plates with a ToF-SIMS IV instrument (ION-TOF GmbH., Münster, Germany). The bombarding beam was from a bismuth liquid ion gun under an accelerating voltage at 25 kV, with a pulsed target current at about 1 pA. For each wear track, three different sites with an area of 500 × 500 µm2 were scanned to ensure repeatability. The resolution is 256 × 256 pixels with 20 scans for each site. ToF-SIMS spectra for both positive and negative ions were extracted from an area size of 300 × 300 µm2 within the wear tracks. The normalised intensity of relevant species (anion from oleic acid and cation from dodecyl amine) was obtained by dividing the intensity of each relevant ion by the total ion intensity and these normalised values represent the content of each ion on the wear tracks. The data was analysed with the software, SurfaceLab 7 (IONTOF GmbH).
Results
FT-IR analysis
FT-IR was applied to analyse the vibration peaks for pure oleic acid, dodecyl amine and IL. Results in Figure 2 show that the peak at 1708 cm−1, which belongs to oleic acid and is assigned to the stretching mode of C = O, disappeared from the spectrum for the IL shown as the black curve. Two representative peaks at 1569 and 1399 cm−1 positions were identified for the IL, corresponding to asymmetric and symmetric vibrations of COO−, formed by proton transfer from oleic acid to dodecyl amine. These three peaks agree with the published FT-IR data in the studies,43,52 indicating that the IL was properly prepared.
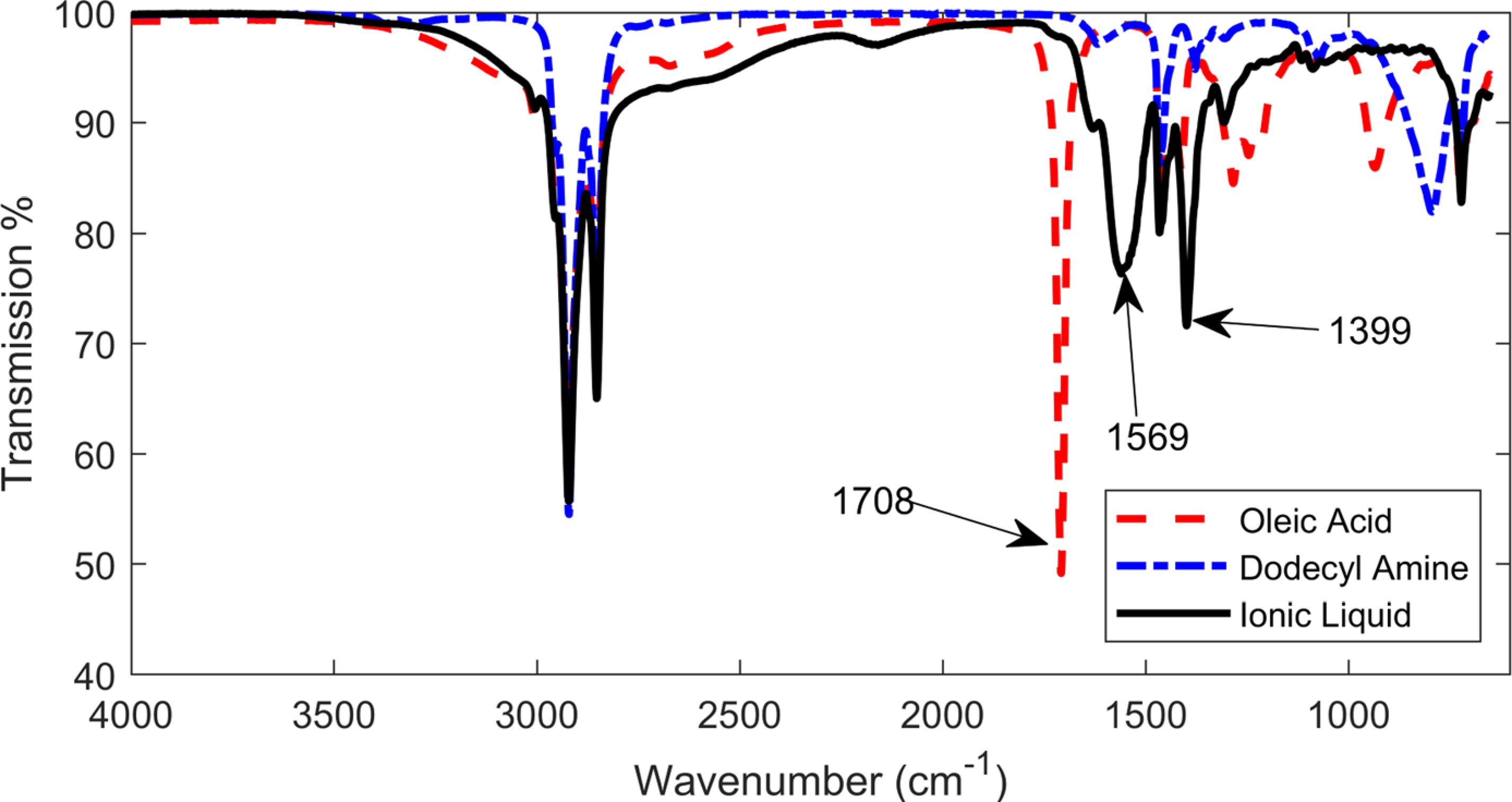
Fourier transform infrared spectroscopy (FT-IR) spectra for oleic acid, dodecyl amine and ionic liquid.
Tribological performance
Tribology for oleic acid as additive
Figure 3 shows the friction and wear results of the tested lubricants. As shown in Figure 3(a), the friction coefficient for only hexadecane as a lubricant was about 0.12. Friction decreased when the oleic acid was added. Friction stabilised at about 0.08 when the acid concentration was 1 mM, and no improvement was seen with further increase of the acid concentration. A similar trend can be seen for the wear coefficient in Figure 3(b). At the 20 mM concentration, the value remained almost the same as that for 1 mM, about 8*10−9 mm3/(Nm). These results clearly show that sample 1A has already reached the optimal performance for oleic acid as the single additive.
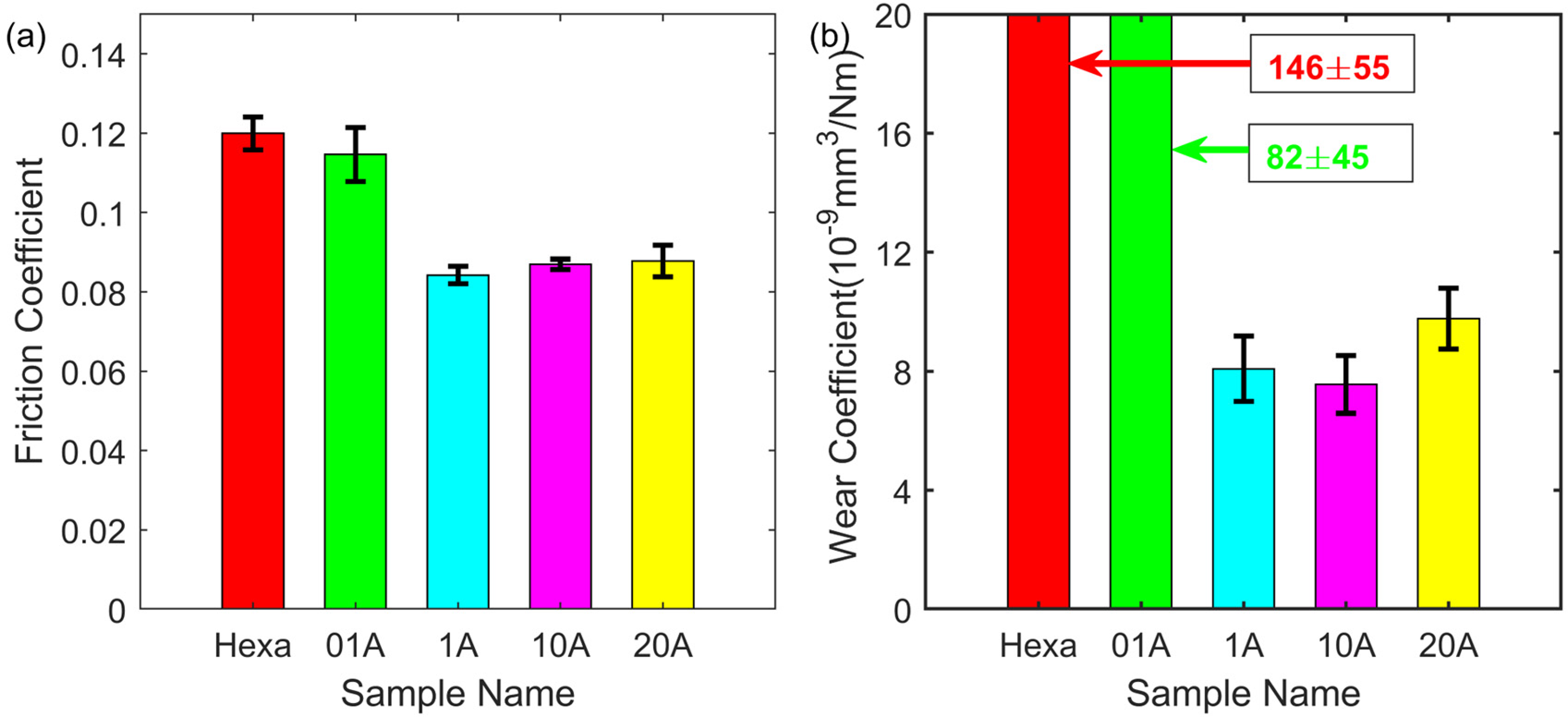
Friction (a) and wear coefficient (b) of oleic acid in hexadecane under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 Hz frequency and 7 mm stroke length.
Tribology for the additive mixture
To boost the tribological performance, additional amine was applied with a total concentration of 1 mM. Figure 4 shows the friction and wear results from the oleic acid/amine lubricant mixtures. Although 1 mM dodecyl amine reduced the friction to about 0.105 under 7 Hz reciprocation; its friction was not as low as the 1 mM oleic acid as the additive. These observations agree with the results found in literary works.51,53 The higher the proportion of amine in the additive mixture, the higher the friction. This phenomenon indicates that these two additives functioned individually and no interaction between them or synergistic effect has been found. To further explore the potential of the additive mixture, the reciprocating frequency was increased to 25 Hz. The green bar for 1A in Figure 4(a) reflected that the increase in frequency provided little benefit to the friction reduction compared to 7 Hz, with the coefficient staying at about 0.08. For 09A01D, there was no synergistic effect under 25 Hz as the friction was close to that for 1A. Nevertheless, for 05A05D with an acid and amine ratio of 1:1, the friction dropped significantly from 0.10 to 0.06. Under 25 Hz, the low friction for 05A05D highlights the potential of improved tribological performance through simply mixing oleic acid and dodecyl amine in hexadecane. In Figure 4(b), the wear coefficient for all samples including 1A under 7 Hz kept steady at about 8*10−9 mm3/(Nm) and descended when the frequency was increased to 25 Hz. It is worth emphasising that the wear coefficient for 05A05D was slightly lower than that for 1A, which is also a sign that the additive mixture with a 1:1 ratio possesses a better tribological performance than only oleic acid as an additive.
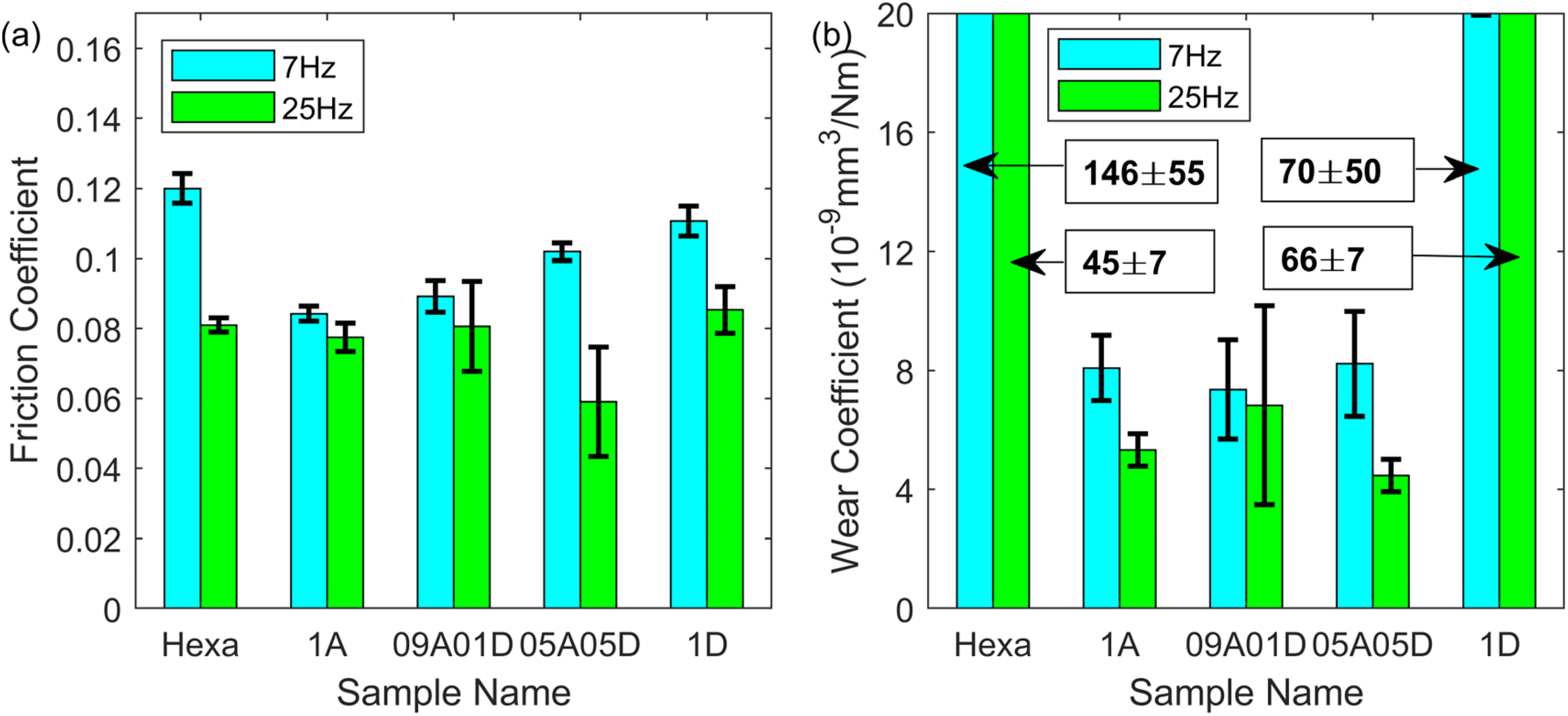
Friction (a) and wear coefficient (b) of lubricant samples with 1 mM total concentration in hexadecane under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 and 25 Hz frequency and 7 mm stroke length.
In Figure 5, the total additive concentration for lubricants was increased to 20 mM. Under 7 Hz, the friction for all samples with 20 mM additives (20A, 10A10D and 20D) ranged between 0.08 and 0.10. However, friction exhibited a striking divergence when the frequency was 25 Hz. 10A10D provided a coefficient as low as 0.045 which was about 60% of that under 7 Hz, while for 20A and 20D, the friction decreased only slightly with the frequency increased. It is interesting to mention that for pure hexadecane (Hexa), the coefficient was almost the same as that for 20A and 20D under 25 Hz. Considering the wear performance in Figure 5(b), the wear coefficient for all samples dropped significantly except for 20A which may be because that high concentration of acid dominated the reaction between additive and metal surface, a higher sliding speed imposed little influence on the wear loss. Similar to the friction coefficient, the wear coefficient for 10A10D was about 25% of the value for 20A under 25 Hz. Generally, with the total additive concentration at 20 mM, there was considerable improvement in the tribological performance of the 10A10D additive mixture compared to 20 mM oleic acid as the lubricant additive when the reciprocating frequency was 25 Hz.
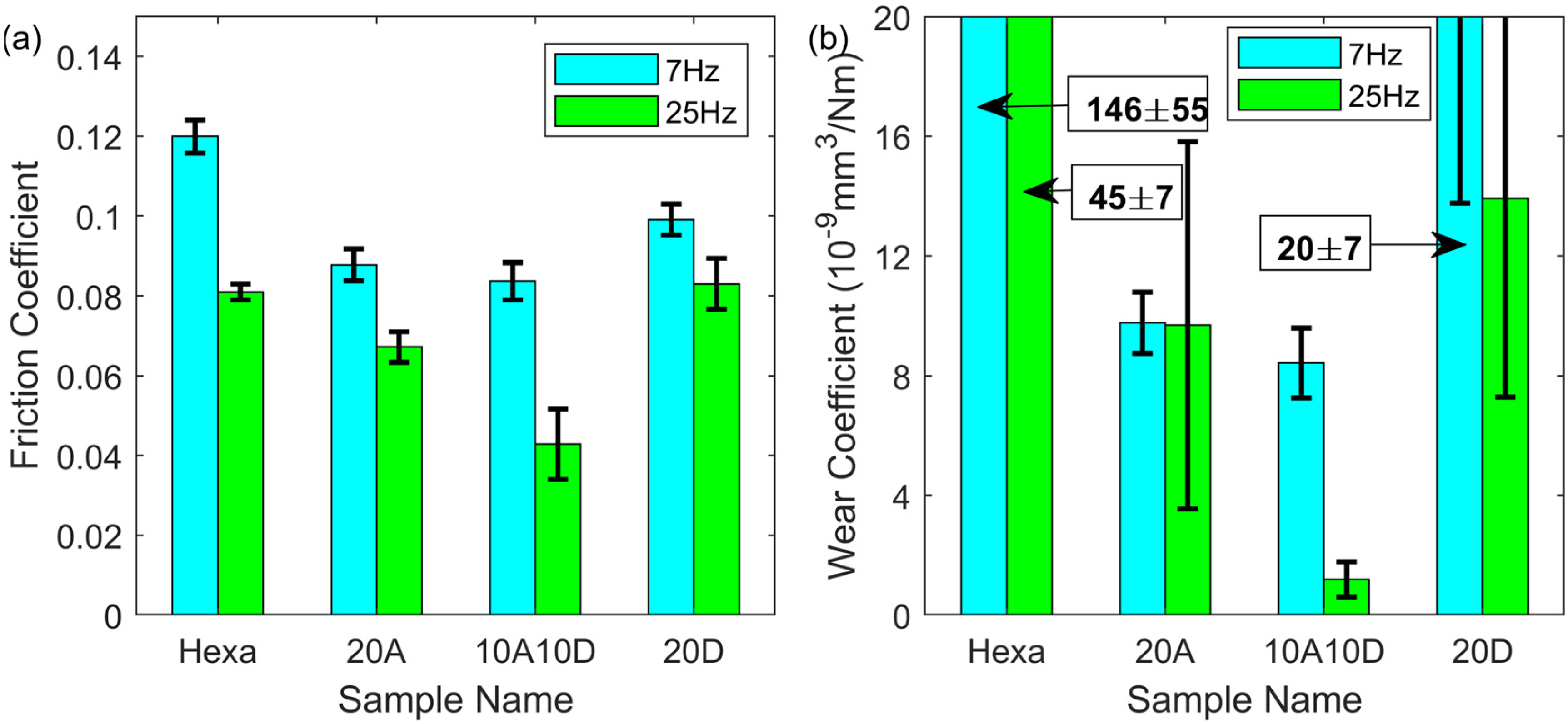
Friction (a) and wear coefficient (b) of additive mixture with 20 mM total concentration in hexadecane under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 and 25 Hz frequency and 7 mm stroke length.
Tribology of the acid, additive mixture and IL
To compare the tribology performance for only acid, additive mixtures and neat IL, the friction and wear results are summarised in Figure 6. When the reciprocating frequency was 7 Hz, the friction was dominated by the oleic acid, staying at about 0.08. Replacing half of the oleic acid with dodecyl amine in the system had a negative effect on the friction performance. The neat IL provided the lowest friction among all samples due to both its viscous separating film and carboxylic soap low-shear film, similar to the performance in. 43 Under a frequency of 25 Hz, for both low and high total additive concentrations, the acid and amine mixture greatly reduced the friction. The higher the total concentration of the additives, the lower the friction coefficient for the additive mixture. The lowest friction came from the IL, as low as about 0.04. Regarding the wear coefficient in Figure 6(b), whether only acid (1A and 20A) or additive mixture (05A05D and 10A10D) had little influence on the wear performance with a wear coefficient in the range of 8 to 10 *10−9 mm3/(Nm) when the frequency was low. The neat IL greatly reduced the wear compared to the other samples under 7 Hz oscillation. From the tribological results for the higher frequency, 25 Hz, 10A10D possessed much better wear protection ability than 05A05D and its wear coefficient was almost close to that for neat IL, which was below 2 *10−9 mm3/(Nm).
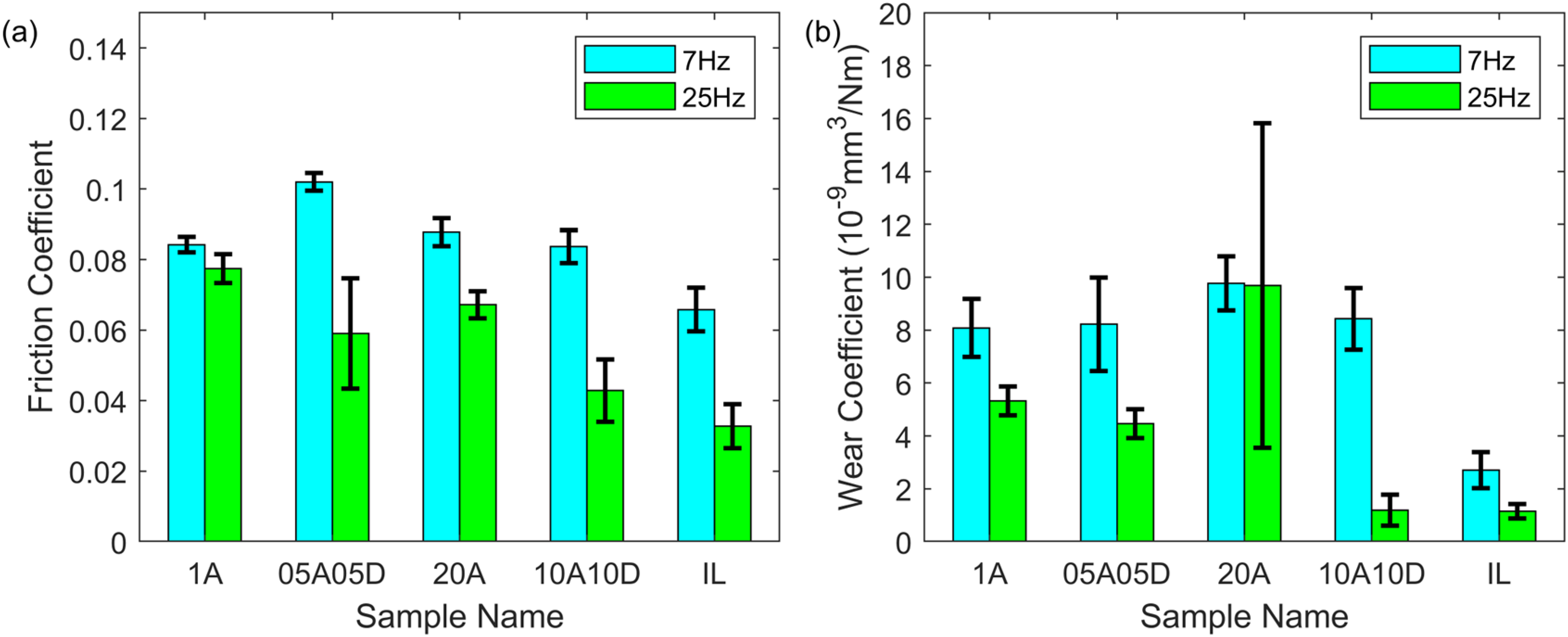
Friction (a) and wear coefficient (b) of oleic acid, additive mixture and ionic liquid under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 25 Hz frequency and 7 mm stroke length.
Figure 7 shows the tribological results to study the instant friction response to the frequency. The running frequency was switched between 7 and 25 Hz, with the friction monitored simultaneously. During the first period under 7 Hz, the IL gave the lowest friction, the same as the steady state. When the frequency was first shifted to 25 Hz, an instant drop in friction was seen for all samples, down to the level of their stable state for the 25 Hz condition. The reduction for Hexa probably originates from the increase of higher entrainment speed, which is the same case for 20A as sliding speed had limited influence on the chemical interaction between oleic acid and contact surfaces. Considering the dramatic change in friction for 10A10D, one plausible explanation could be that high frequency promoted in-situ interaction between acid and amine, which formed an extra separating tribo-film, consequently reducing friction. As the frequency was changed back to 7 Hz between 2400 and 3000 s, the friction for 20A, Hexa and IL returned to the first 7 Hz period immediately, which is an indication that the cause of the friction change did not originate from the change of a separating tribo-film. However, for 10A10D, friction did not return to the original level since the in-situ interaction from higher frequency kept a certain level of ability to separate the contacts better than the first 7 Hz period. During the last testing period under 25 Hz, friction was close to the first 25 Hz period.
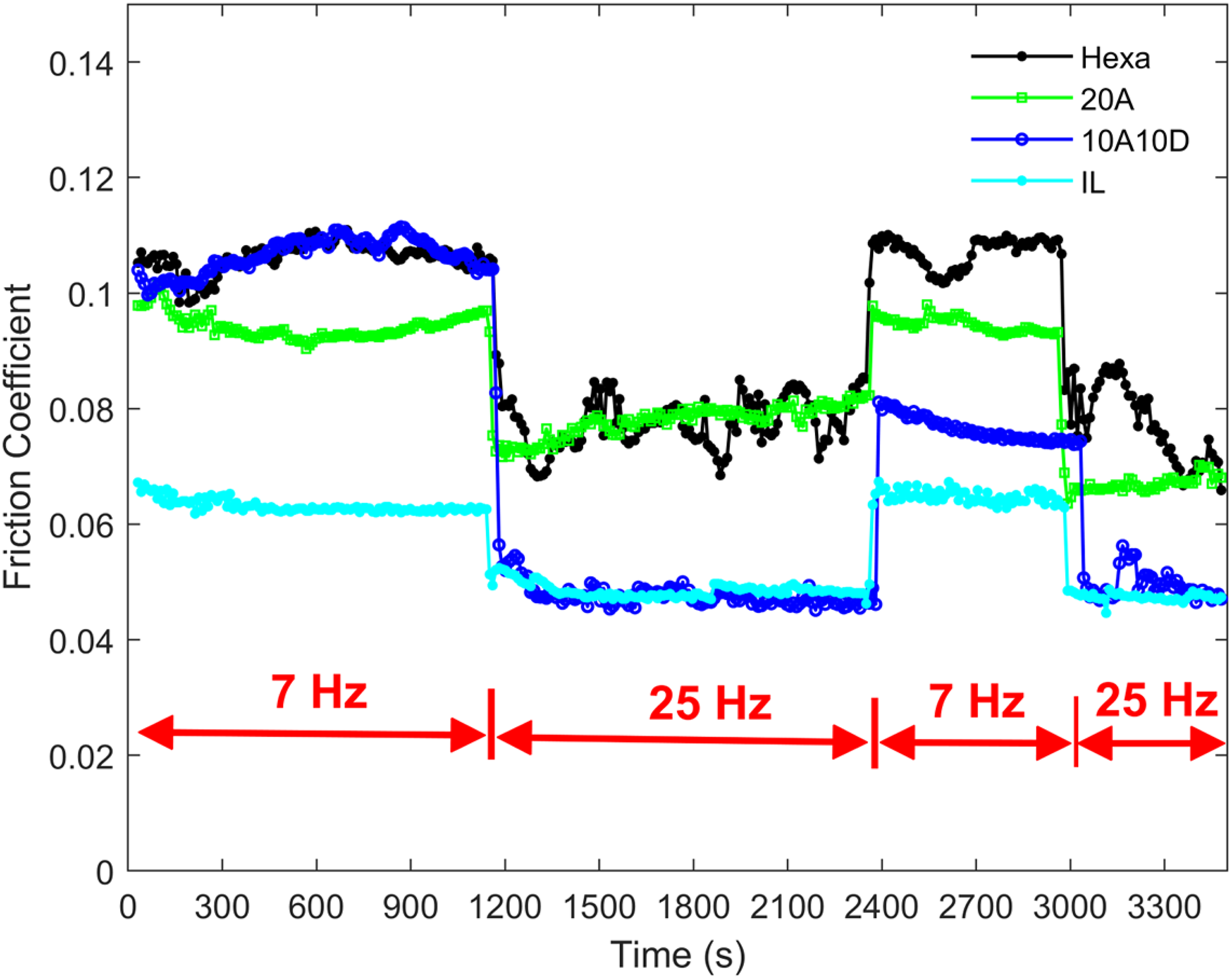
Friction curve of additives hexa, 10A10D, 20A and ionic liquid under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 mm stroke length and switching frequency between 7 and 25 Hz.
Surface analysis
SEM analysis
To compare the wear mechanisms, scanning electron microscopy was applied to obtain the images for the wear scars with 2000 magnification. For the surface lubricated with Hexa without additives, the abrasive traces can be seen in Figure 8. Hence, the main wear mechanism for the pure hexadecane without any additives is abrasion, showing the highest wear coefficient compared to all other samples. For samples, 05A05D, 1A, 10A10D and 20A, the surface morphology became more homogeneous with less character of abrasion; however, there were very slight signs of adhesive wear. The addition of additives mitigated the strong ploughing effects by forming an organic boundary layer on the metal surface, therefore shifting the abrasive wear for the pure hexadecane to very slight adhesive wear for samples with additives. Moreover, among the lubricant samples with additives, the effects of different reciprocating frequencies, additive types, and additive concentrations on the wear mechanism were negligible. For the IL as a neat lubricant, the surface appears ‘rougher’ with more ‘abrasive traces’; however, the ‘traces’ did not come from the ploughing between the counter surfaces but from the original surface. This phenomenon also proves that the IL has a better ability to protect original surface features, leaving the worn scar surface almost as in its original condition.

Scanning electron microscopy (SEM) images (2000x, 10 kV). for wear scars on the pins under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 and 25 Hz frequency and 7 mm stroke length.
EDX was utilised to analyse the content for chemical elements on the wear scar, and results are given in Table 3. Caution should be taken when reviewing the results, as the interaction depth between the electron beam and the detected surface is in the micron range which is much larger than the actual tribo-film thickness. The carbon content stabilised at about 3 wt% for all samples and the significant variation was between oxygen and iron. Wear scars lubricated with the low total concentration (1 mM) additives showed a higher oxygen content than that for 20 mM total concentration under both 7 and 25 Hz conditions. This suggests that a larger amount of additive may reduce the oxidation of the steel surface. There is no clear evidence that oscillating frequency influences the chemical element concentration.
Representative element composition for wear scar obtained with EDX.
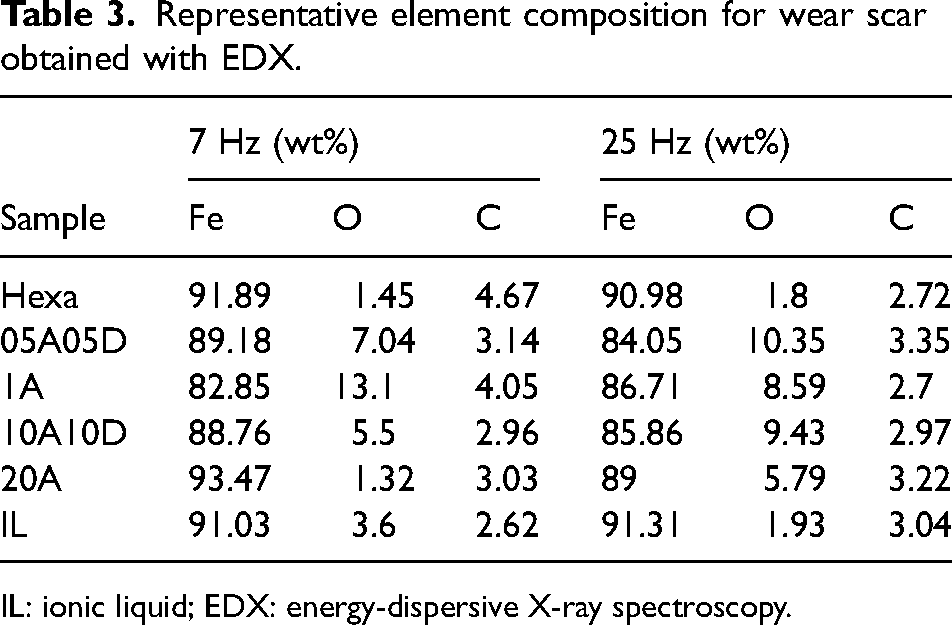
IL: ionic liquid; EDX: energy-dispersive X-ray spectroscopy.
ToF-SIMS analysis
To understand the lubricating mechanism, ToF-SIMS analysis was conducted on the wear tracks from plates to detect representative molecular fragments such as oleic acid and dodecyl amine. Figure 9 gives the information of the negative ions for the oleic acid. From part (a), there are strong peaks at the position 281.26 amu for oleate anion from the IL, 10A10D and 20A, suggesting the existence of oleic acid deposited on the wear tracks. The intensity was normalised according to the method described in the experimental part to compare the amount of oleate anion on the worn surfaces. From Figure 9(b), the largest amount of oleate anion was found for neat IL, followed by the additive mixture 10A10D. The normalised intensity for 20A is only about 10% of that for IL and 20% for 10A10D. Moreover, oscillating frequency has little effect on the amount of oleate anion as the normalised intensity is almost identical for both 7 and 25 Hz for each sample.
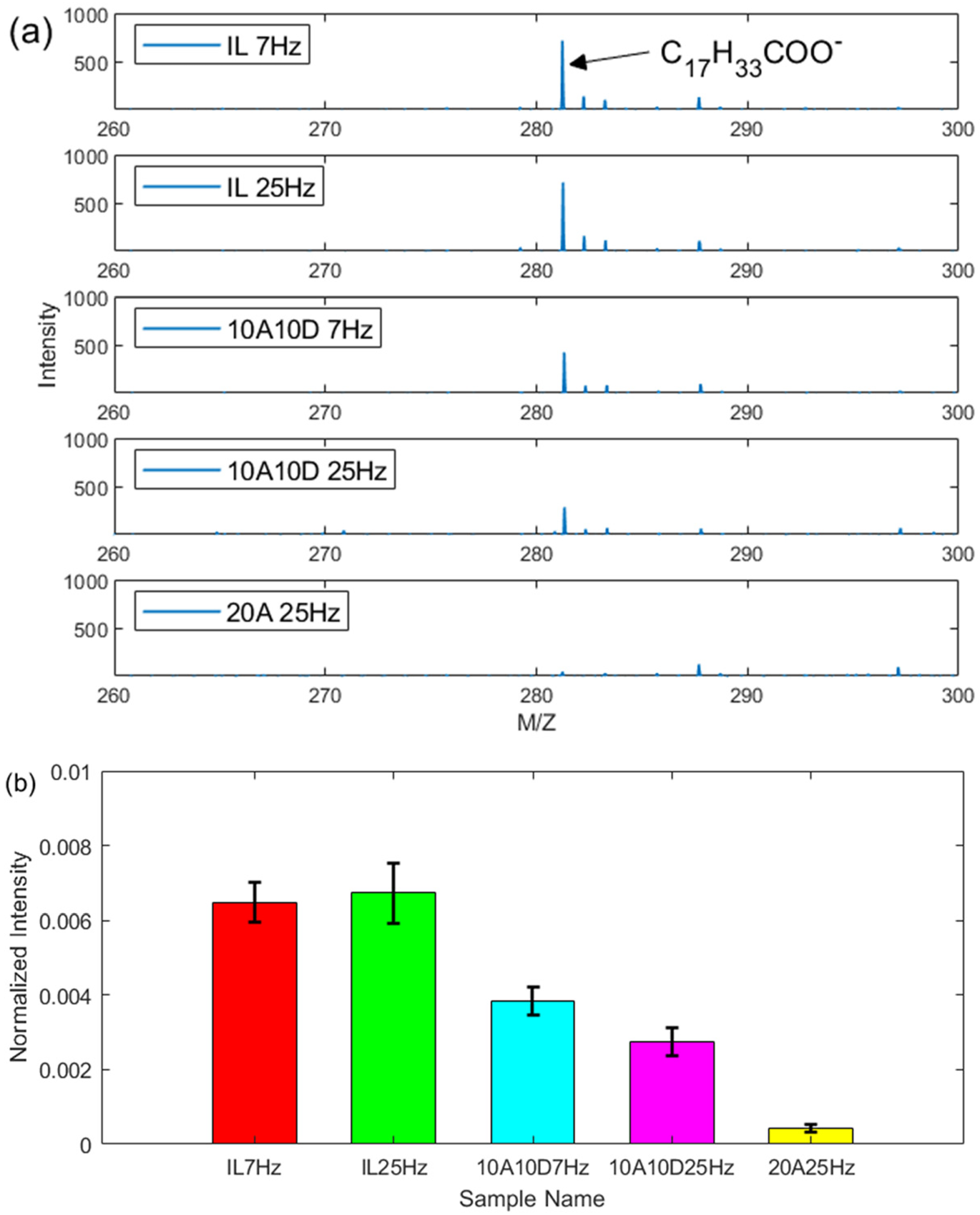
Negative ions spectra for wear tracks (a) oleate anion peak intensity, (b) normalised intensity for oleate anion, under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 and 25 Hz frequency and 7 mm stroke length.
The spectra of dodecyl amine are given in Figure 10. The protic dodecyl amine at the position 186.24 u was found for both IL and 10A10D from part (a), indicating a product with an ionic character was formed during the tribological process. Under 25 Hz, the normalised intensity for protic amine is almost double that for 7 Hz. This also suggests that higher frequency can facilitate the protonation of dodecyl amine.
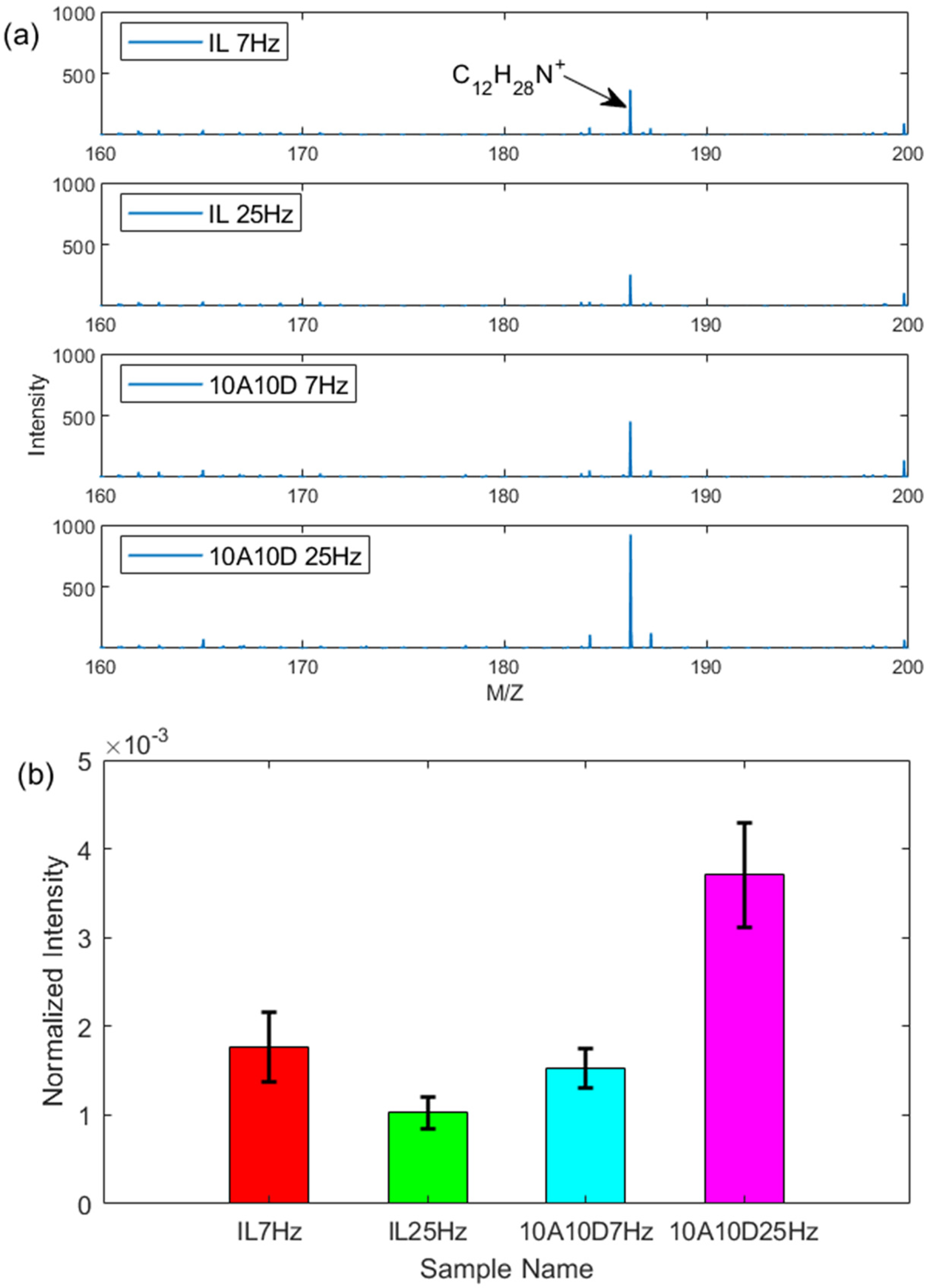
Positive ions spectra for wear tracks (a) peak intensity for dodecyl amine, (b) normalised intensity for dodecyl amine, under 1 h reciprocating sliding, 50 °C, 1 GPa maximum contact pressure, 7 and 25 Hz frequency and 7 mm stroke length.
Image analysis of the distribution for both negative and positive ions is given in Figure 11. In the first row for negative ions, the oleate anion was seen scattered homogeneously across the whole image for all samples under both 7 and 25 Hz. The height of the image is 500 µm, which is larger than the wear track; however, there was no clear boundary inside and outside the wear track. This suggests that both tribological and thermal effects can promote the adsorption of negative ions on metal surfaces at a similar level of influence. For the second row in Figure 11, similarly to negative ions for the IL, ions were distributed evenly on the whole surface with no distinguishable boundaries. For the additive mixture, 10A10D, the protic amine stayed mainly in the middle of the wear track especially for the 25 Hz testing condition, indicating the rubbing between surfaces shares an essential role in forming amine salt on the wear tracks.
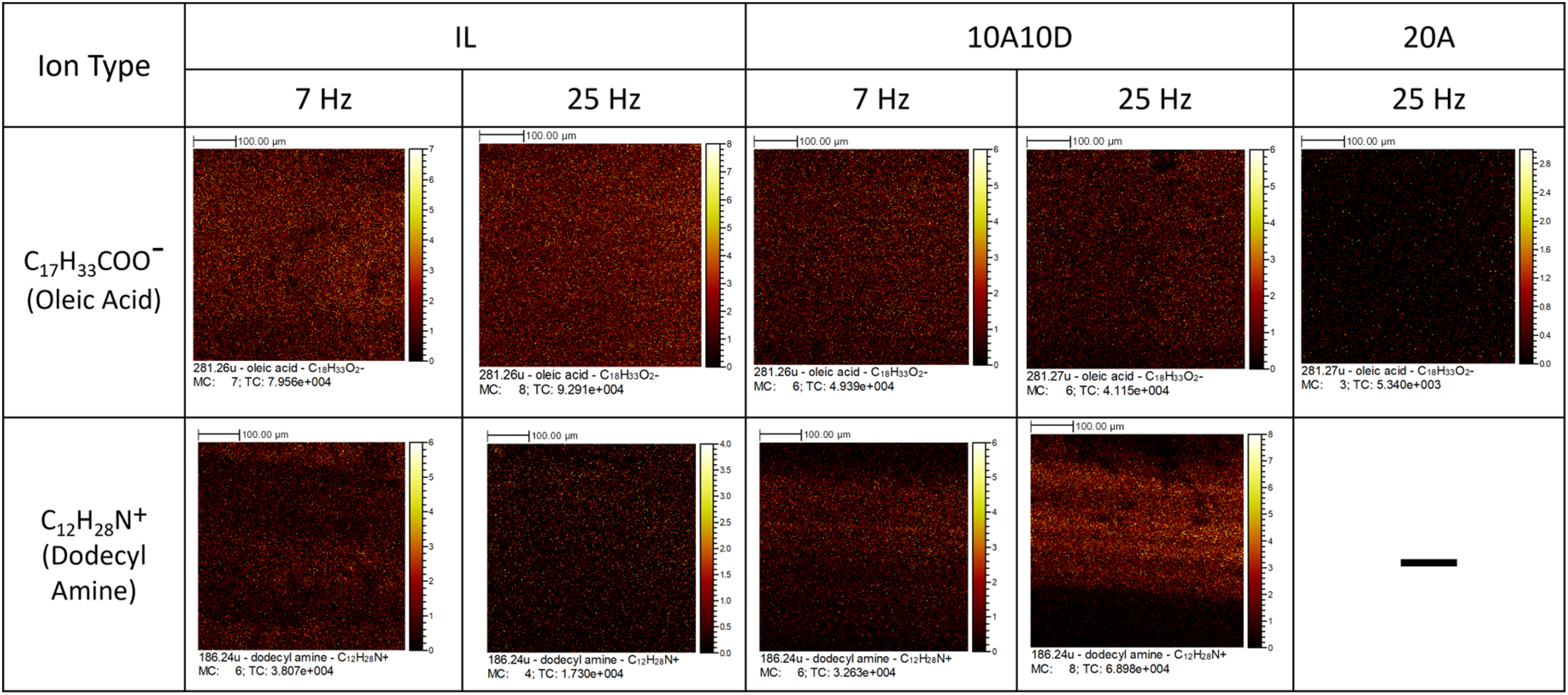
Time-of-flight secondary ion mass spectrometry (ToF-SIMS) images analysis for positive ions and negative ion distribution from wear tracks.
Discussion
Single-additive system
Compared to amines, esters and amides used as a single additive in non-polar base oil, oleic acid has shown a good tribological performance.16,17,53 The reaction between carboxylic groups and metal surfaces facilitates the adsorption of the acids on the surface to reduce friction and wear, by forming a complex ester-like product.54–57 When the adsorption reaches a saturation state, the tribological performance of additives reaches a plateau. Within the testing conditions in the current study, a very low concentration of oleic acid, 1 mM, was sufficient to cover the adequate contact surface to reach the optimal tribological condition, in agreement with what was found in. 25 A higher concentration, 20 mM oleic acid provided negligible benefit in improving the tribological performance. The above results suggest that a self-assembly layer composed of oleic acid has its performance limitation, which may constrain the application of bio-based fatty acid as an additive in single-additive systems.
Bi-component additive system
The IL made of bio-based fatty acid and amine has been studied as a neat lubricant due to its good tribological response, non-corrosive and bio-degradable properties. From the ToF-SIMS analysis in Figure 9, the existence of oleate anion was confirmed on the wear track, and the oleate anion was combined with the metal surface producing an ester-like soap. These complex compounds are similar to those formed in the contacts lubricated with only oleic acid as an additive. The positive ions from dodecyl amine in Figure 10 proved that IL also adsorbed on the wear track, forming a dense separating layer, which also accounted for the good lubricity of IL.
ToF-SIMS detected only the top few nanometres of the worn surface. For both 7 and 25 Hz conditions, the estimated film thickness was over 30 nm. Even though the cleaning process removed some IL, the remaining IL layers that can be analysed by ToF-SIMS were almost identical for both cases. Hence, the spectra and normalised intensities for oleic acid and dodecyl amine ions for both 7 and 25 Hz conditions were almost identical. A higher dragging speed from the 25 Hz condition led to a thicker lubricant film. Although this thicker film did not shift the lubrication regime from the boundary regime to a full film condition based on lambda estimation, it adsorbed strongly on the surfaces and transferred the shear plane from an alkyl chain plane to an ionic plane, decreasing the shear force dramatically for 25 Hz, compared to 7 Hz. This friction reduction mechanism has been proposed for IL nanofilms 58 and explains the further friction reduction in the macroscale as the running frequency increases.
In terms of the simple additive mixture containing oleic acid and dodecyl amine, the difference between low (7 Hz) and high (25 Hz) frequency-formed boundary films is illustrated schematically in Figure 12. Under 7 Hz, there is little improvement in tribological performance if the total concentration is the same as oleic acid, and no interaction between acid and amine was witnessed. The adsorbed film contained mainly oleic acid, as shown on the left side of Figure 12. The competition between acid and amine might suppress the effectiveness of acid when the total concentration was as low as 1 mM. On the other hand, the equal molar ratio acid/amine additive mixture showed its advantage over the tribological performance of oleic acid at high reciprocating frequency for both low (1 mM) and high (20 mM) total additive concentrations. As schematically shown in Figure 12, the amine was involved in the film with oleic acid, forming a product with ionic character. The physical properties of acid and amine mixture have been investigated in previous studies. Evidence can be found that equal molar ratio of acid and amine gives the highest ionic characteristics and viscosity, which can provide benefits for lubricating properties. In one early study, 59 equimolar fatty acid and dodecyl amine formed a 1:1 product, which was ammonium salt and showed good solubility in non-polar organic solvents. The product could also form a micelle when the concentration was above the critical micelle concentration. In another study by Saeten et al., 60 it was then confirmed that a fatty acid/fatty amine mixture in heptane with a 1:1 ratio showed the highest relaxation time and static permittivity, which was evidence of the strongest ionic character among different ratio values. In a more recent work by Karlsson et al., 61 the conductivity and bulk viscosity of complexation containing heptanoic acid and heptylamine were measured, showing that an equal molar mixture of acid and amine provided the highest conductivity and viscosity compared to different ratio mixtures. All the above studies prove that equimolar fatty acid and amine possess a stronger ionic character than other ratios of combination. However, the pre-interaction between acid and amine in hexadecane cannot meet all essential conditions to reduce friction and wear as the neat IL lubricant under a low running frequency of 7 Hz. As seen in Figure 6, a high oscillating frequency of 25 Hz in the tribological process is another critical factor in initiating the synergistic effect. This is also supported by the real-time frequency shift test in Figure .7, as the friction dropped almost instantly with an increase of frequency for 10A10D, then increased again as frequency decreased. A potential explanation for this phenomenon is that higher frequency oscillation inputs more tribological energy, enhancing the entanglement between molecules and the ionic character of the ‘pre-formed’ compound. Afterwards, this in-situ product can lubricate the surface with the same mechanism as the neat IL. Moreover, a higher total additive concentration and entrainment speed lead to a more ionic product within contacts so that the tribological performance approaches that for the neat IL, as shown in Figure 6. The above explanation is also supported by the ToF-SIMS analysis in Figure 10, which shows more protic dodecyl amine on the wear track under 25 than 7 Hz. Moreover, the lateral distribution of ions shown in Figure 11, also confirms the formation of protonated dodecyl amine within the wear track.
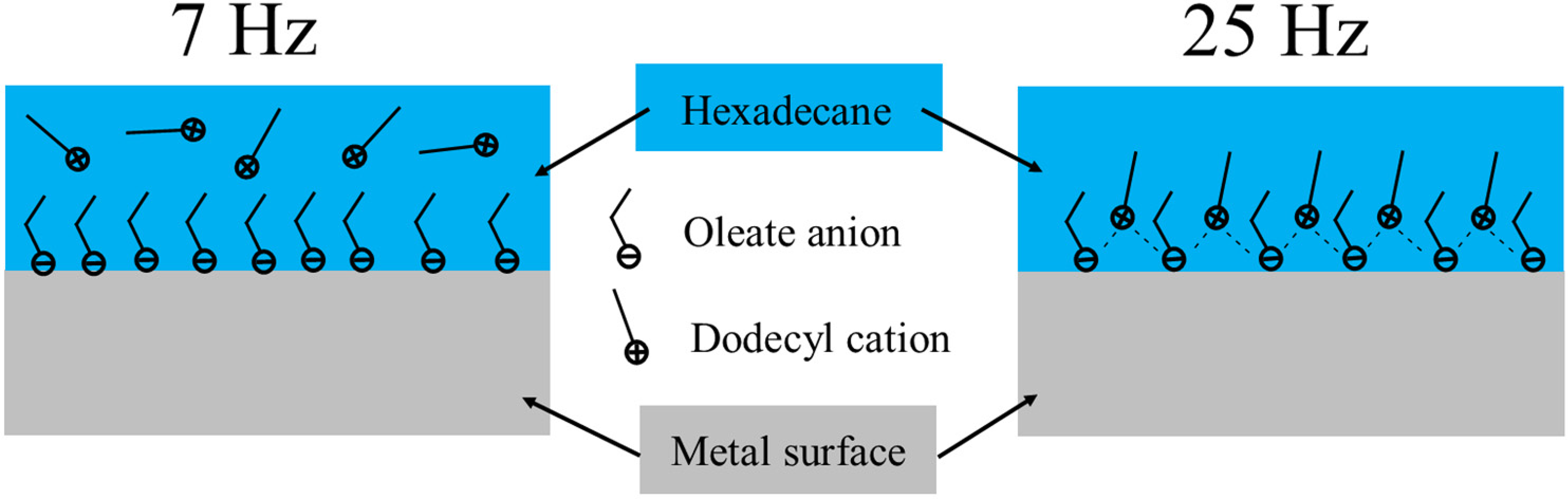
Schematic images of boundary films formed at 7 and 25 Hz test conditions from a fatty acid and amine lubricant mixture.
In general, through the tribological study and surface analysis for the additive mixture, it is manifested that under certain testing conditions, an in situ tribological process can boost the proton transfer from oleic acid to dodecyl amine, enhancing the ionic character of the additive mixture. The ionic product shares a similar lubricating mechanism as the neat IL. Additionally, it can be deduced that this tribologically induced proton transfer is partially reversible as the friction shifts back to a higher level than that for the neat lubricant under 7 Hz in Figure 7. This study highlights the potential of substituting the neat IL with an acid/amine additive mixture while still achieving similar tribological performance under specific testing scenarios. Another advantage of applying additive mixtures instead of neat IL is that it requires much fewer raw materials for the additive mixture than neat IL, so fewer resources are needed. Therefore, the detailed discussion of the synergistic effect of fatty acid and amine highlights the possibility of utilising bio-based materials as lubricant additives with less energy consumption and resources.
Conclusion
This study investigated the potential of using bio-based materials as lubricant additives amid increasing requirements for environmental protection. A systematic comparison of the tribological performance of oleic acid, its mixture with an amine, and neat IL was conducted. The worn surfaces were examined with SEM and ToF-SIMS to understand the lubricating mechanism.
Under 7 Hz reciprocating frequency, oleic acid is key in reducing friction and wear in the additive mixture, while oleic acid and dodecyl amine function separately. Neat IL shows the best performance among oleic acid and additive mixture, regardless of additive concentration value, showing about 25% lower friction and 75% lower wear coefficient, compared to the sample with the additive mixture. This is due to both a chemical reaction with metal surfaces to form carboxylic soap and physical adsorption on the surface as a viscous separating layer.
Under a reciprocating frequency of 25 Hz, this higher frequency provides little benefit to the performance of a lubricant with only oleic acid as an additive. On the other hand, around 50% decrease in friction and 75% decrease in wear coefficient was observed when a lubricant mixture of 20 mM oleic acid and dodecyl amine additive was tested at 25 Hz, compared to 7 Hz tests. This synergistic effect is mainly seen when the molar ratio between acid and amine is 1:1. The low friction and wear performance of an equimolar mixture is close to that of the neat IL, indicating that the higher frequency tribological process enhances the ionic character of the additive mixture shifting the shear plane from an alkyl chain to an ionic plane. The mechanistic understanding developed from this study opens the opportunity to develop advanced lubricants using bio-based chemical additives, minimising the environmental impact of current lubricants.
Supplemental Material
sj-docx-1-pij-10.1177_13506501241251524 - Supplemental material for Tribological performance of fatty acid, acid/amine additive mixture and ionic liquid
Supplemental material, sj-docx-1-pij-10.1177_13506501241251524 for Tribological performance of fatty acid, acid/amine additive mixture and ionic liquid by Ju Shu, Cayetano Espejo, Mitjan Kalin and Ardian Morina in Proceedings of the Institution of Mechanical Engineers, Part J: Journal of Engineering Tribology
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union's Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No. 860246, Slovenian Research and Innovation Agency (ARIS), research core funding No. P2-0231 and the Engineering and Physical Sciences Research Council Grant No. EP/S030476/1.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
