Abstract
Silicone elastomer medical implants are ubiquitous in medicine, particularly for breast augmentation. However, when these devices are placed within the body, disruption of the natural biological interfaces occurs, which significantly changes the native energy-dissipation mechanisms of living systems. These new interfaces can introduce non-physiological contact pressures and tribological conditions that provoke inflammation and soft tissue damage. Despite their significance, the biotribological properties of implant-tissue and implant-extracellular matrix (ECM) interfaces remain poorly understood. Here, we developed an in vitro model of soft tissue damage using a custom-built in situ biotribometer mounted onto a confocal microscope. Sections of commercially-available silicone breast implants with distinct and clinically relevant surface roughness (
Keywords
Introduction
Silicone elastomers are used in a panoply of implantable medical devices (e.g., shunts, ports, drains, biosensors, and prostheses) to adjust form and restore function. Their widespread use is partly due to their material and mechanical properties (e.g., low surface tension, thermal and chemical stability, and tunable elastic modulus) but also due to their perceived “biocompatibility.”
1
These implantable materials, widely considered “inert,” may provoke the foreign body response (FBR) and lead to clinical complications; certain silicone devices have been linked with chronic inflammation,
2
capsular contracture,
3
and even cancers.4–7 Unsurprisingly, biocompatibility is a system property; there is no such thing as a universally “biocompatible material.”
8
Intrinsically biocompatible systems enable materials to perform with appropriate host responses in specific situations.9,10 It is difficult, if not impossible, to predict in vivo device performance without fundamental knowledge of tribological challenges across device-tissue interfaces. A wide gap in the knowledge remains regarding the delicate balance between normal and adverse cellular remodeling near implanted devices and the possible role of mechanical stresses in mediating healthy wound healing or peri-implant fibrosis. This knowledge gap is partly responsible for the lack of regulations involving surface texturing of soft breast implants. One notable case involved aggressively roughened or “macrotextured” designs (average surface roughness
The foreign body response (FBR) initiated post-implantation begins similarly to physiological wound healing.15,16 The FBR is characterized by a series of overlapping stages, including initial adsorption of proteins and formation of a provisional extracellular matrix (ECM), acute inflammation, and immune cell invasion associated with chronic inflammation (Figure 1). 17 The ECM provides structural support to surrounding cells as a three-dimensional network of proteins, primarily composed of collagens. Under inflamed conditions, this collagen-rich scaffold becomes a contributor to advancing fibrosis. Additionally, implant fibrosis and chronic macrophage activation and foreign body giant cell (FBGC) formation are abnormal wound-healing stages of the FBR observed post-implantation. 18 While contact with implant surfaces has been shown to directly cause the FBR, mechanical overloading and wear complications can result in implant failures and further damage, including loss of anchorage, 19 biofilm development,20,21 and a variety of rare complications. 22 Recent advances in tribological instrumentation have enabled friction and rheological measurements between soft implant materials and living cells, 23 cell layers,24,25 and tissues. 26 Our study investigates the relationship between silicone elastomer breast implants of varying surface roughness and damage to soft tissue models in vitro using collagen-coated hydrogels and cell monolayers. We hypothesize that increasing surface roughness will increase frictional shear stresses and damage soft tissues in vitro.
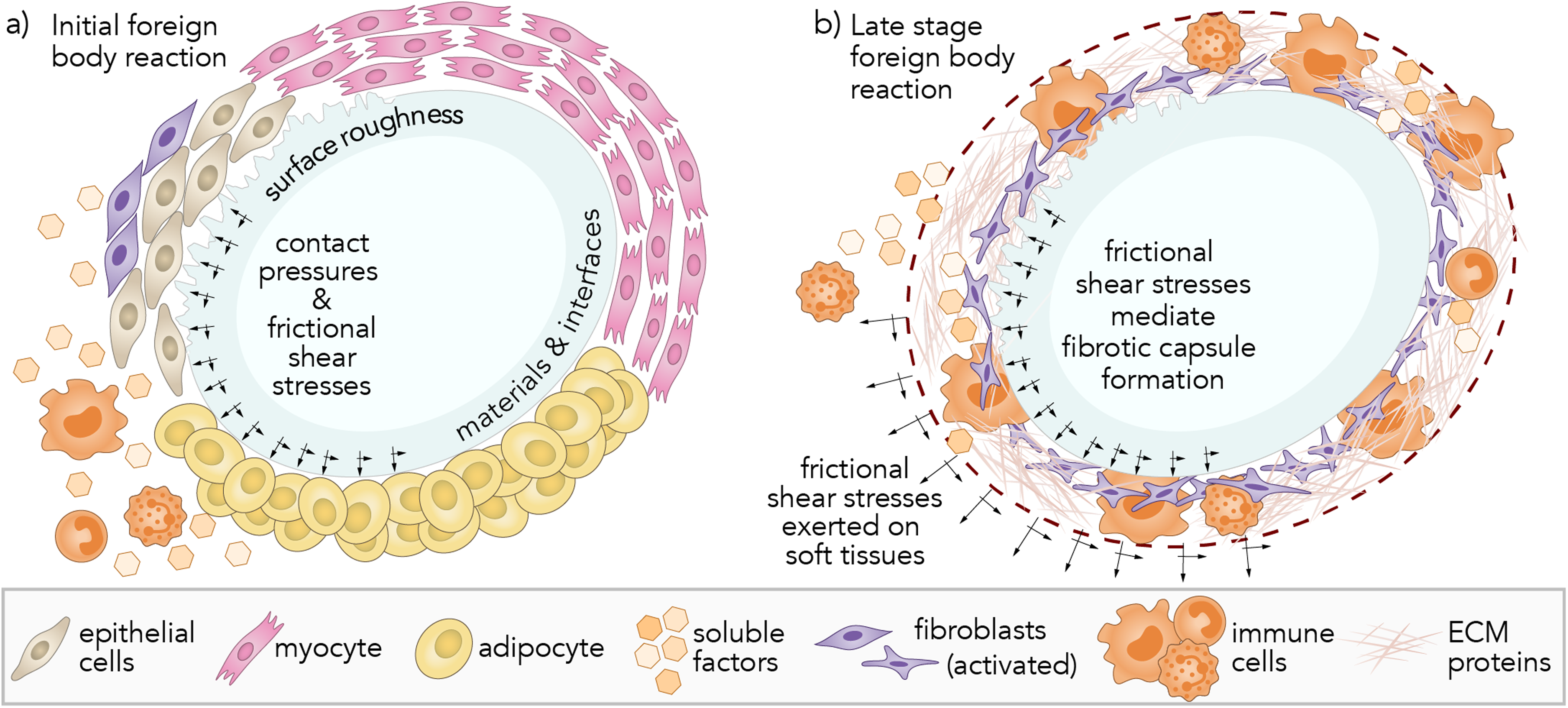
(a) Immediately post-implantation (0–2 weeks), silicone prostheses directly contact and slide against soft tissues, including epithelial cells, myocytes, adipocytes, and extracellular matrix (ECM) proteins, including collagen. The resulting contact pressures and shear stresses across mechanosensitive surfaces are hypothesized to provoke the immune response and initiate the wound healing process. (b) About 2 weeks post-implantation, frictional shear stresses likely mediate the formation of a fibrotic capsule composed of activated fibroblasts, immune cells, and ECM proteins.
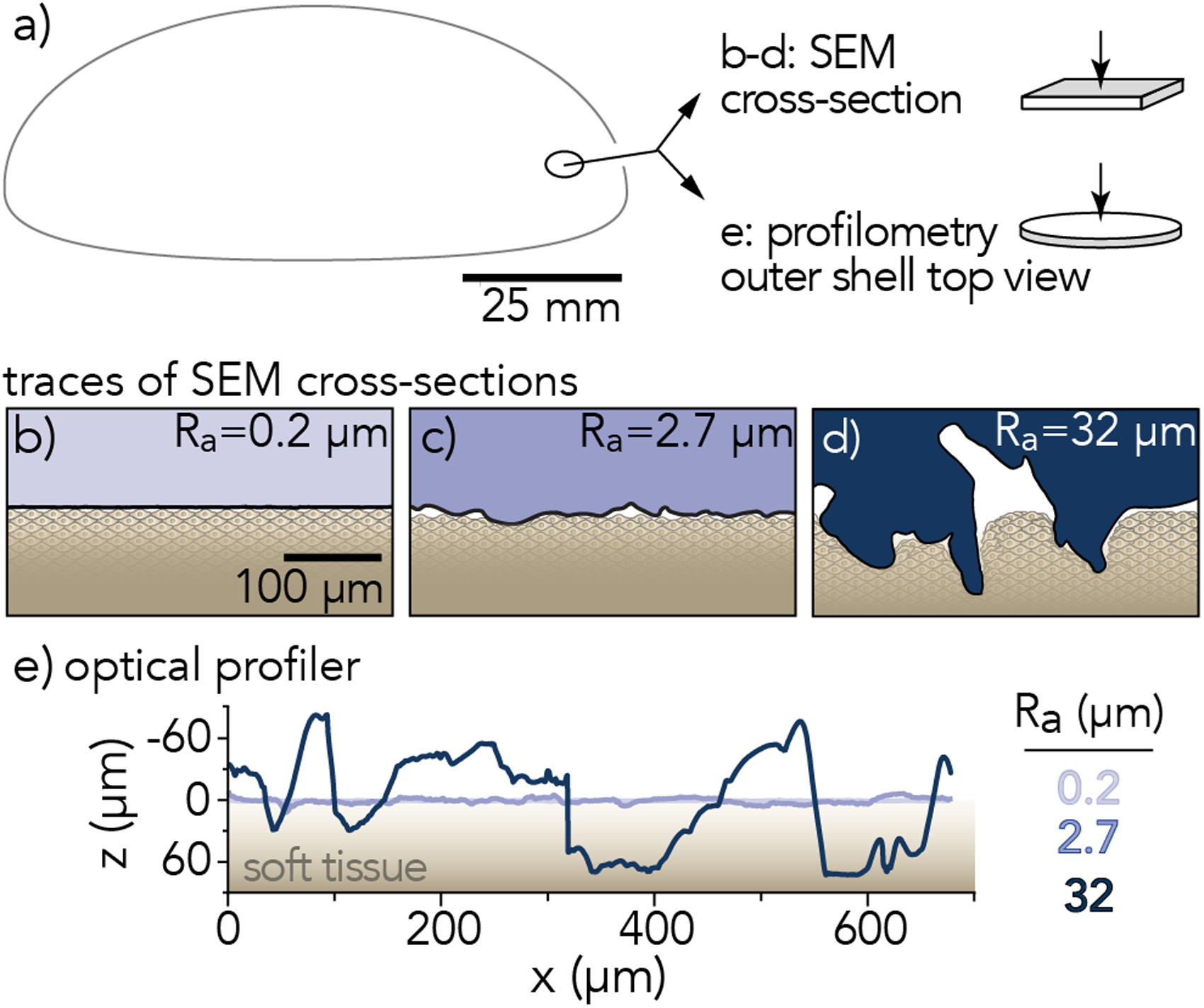
(a) Sections of outer shell removed from breast implant base and prepared for scanning electron microscopy (SEM) and optical surface profilometry. Representative traces of implant surface profiles measured by (b-d) scanning electron microscopy and (e) 3D laser scanning microscopy. Average surface roughness,
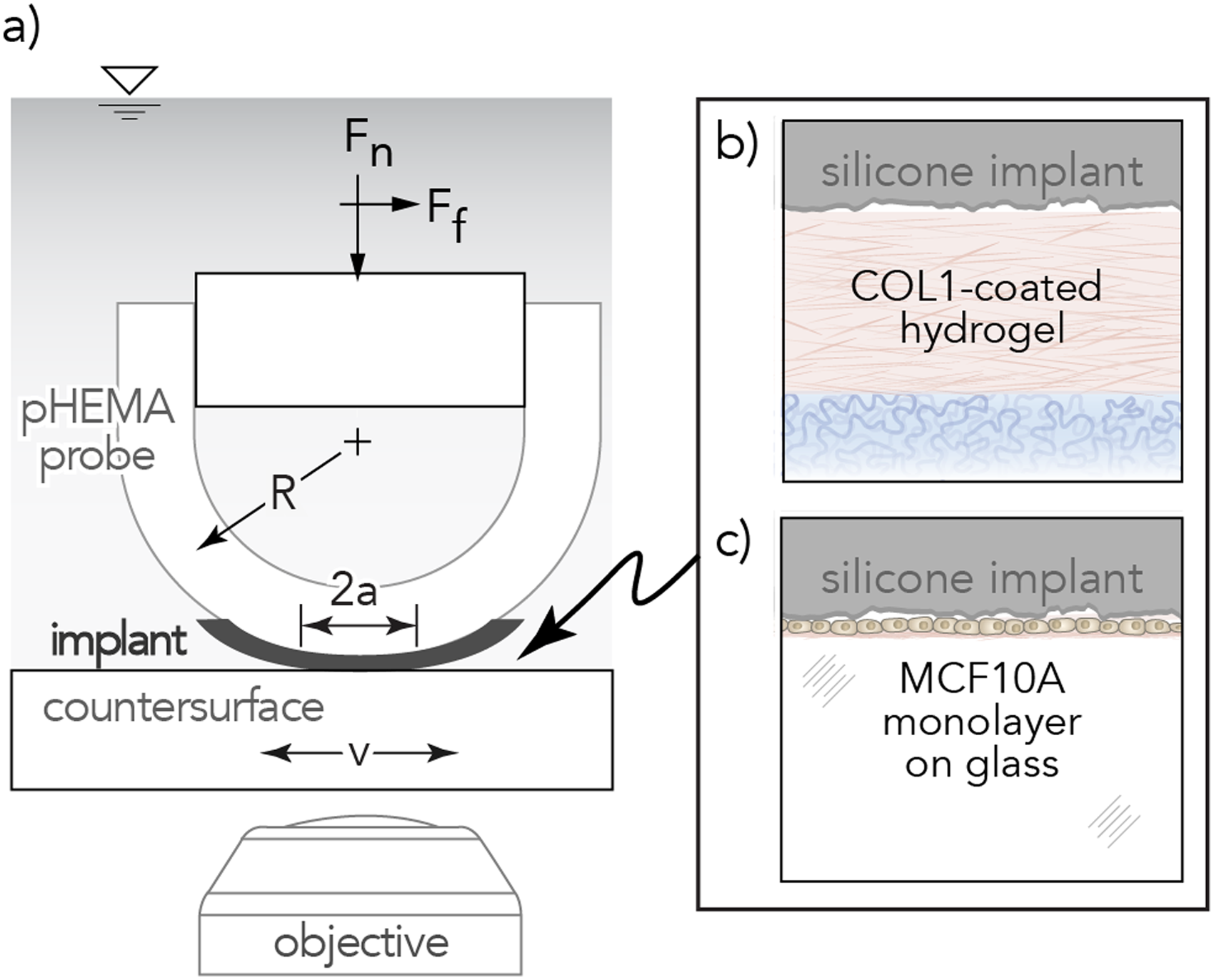
(a) Illustration of biotribometer configuration. Insets of contact between silicone implant and experimental countersurfaces (b) Collagen Type 1, COL1-coated hydrogel and (c) healthy breast epithelia, MCF10A monolayer.
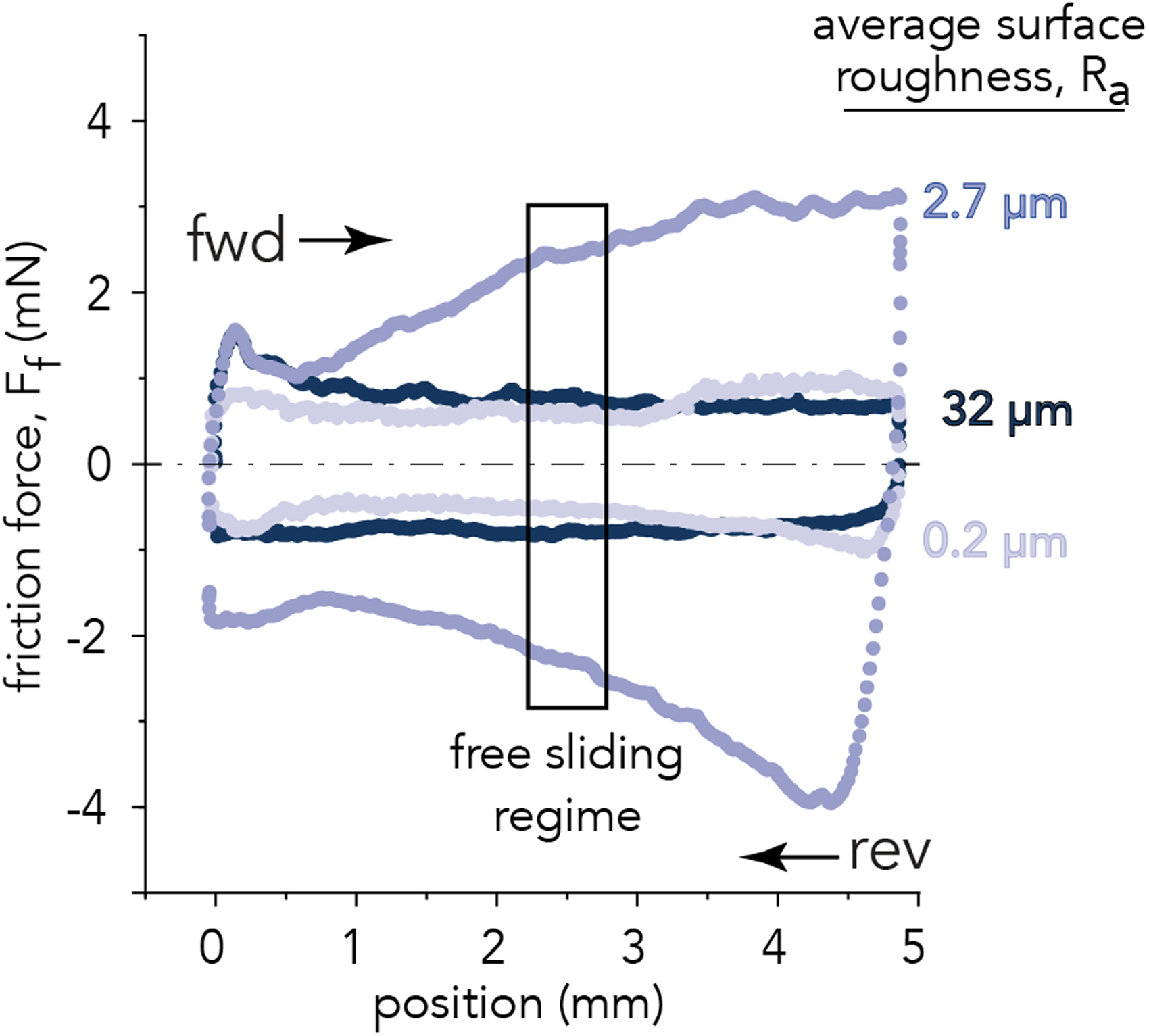
Friction force trace of first reciprocating cycle for three breast implant samples of

(a,c,e) Collagen removed from polyacrylamide hydrogel substrates and (b,d,f) transferred to the breast implant probe after 1,000 mm total sliding distance. (a,b)
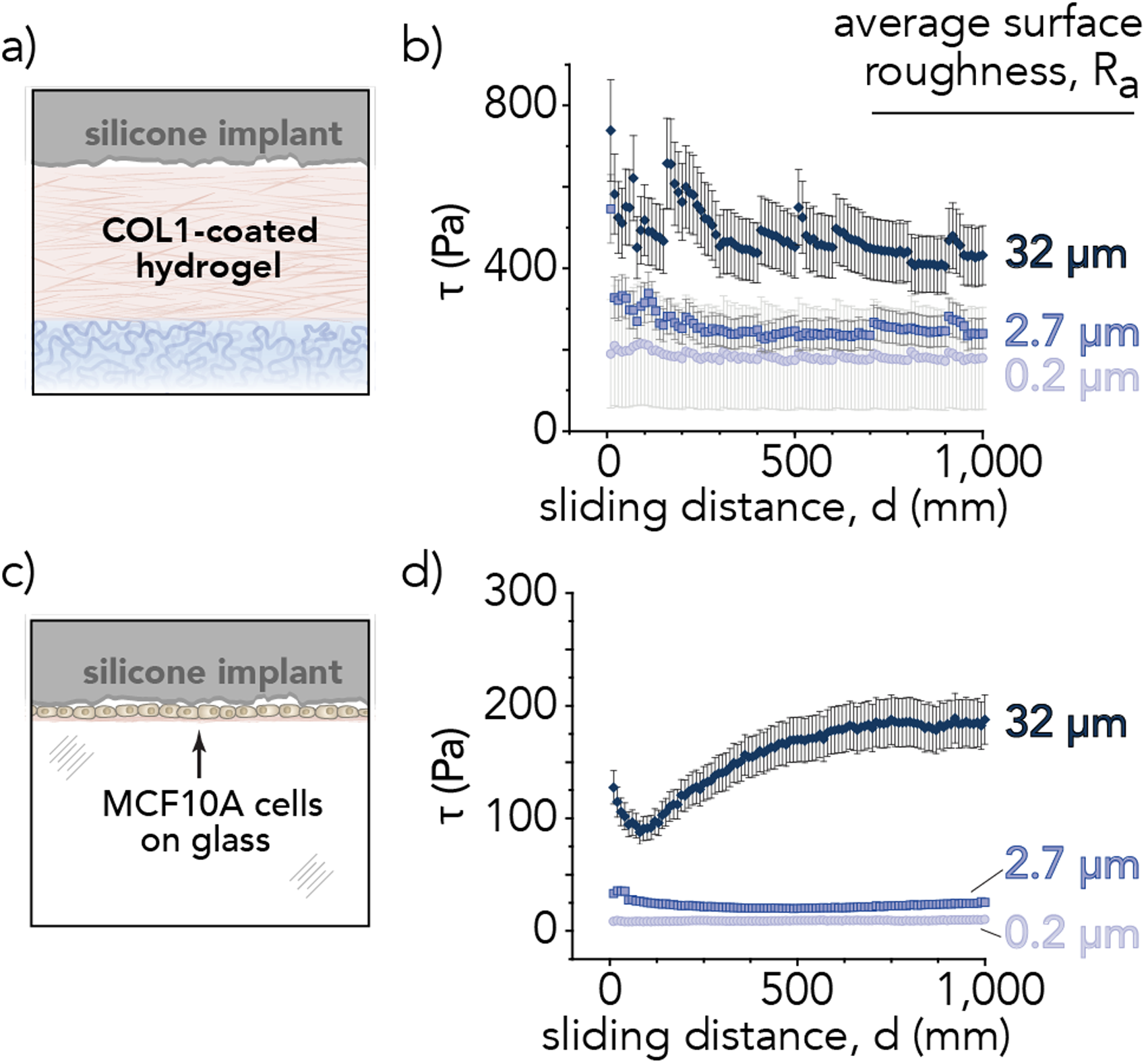
Frictional shear stress as a function of sliding distance for the silicone elastomer implant surfaces (
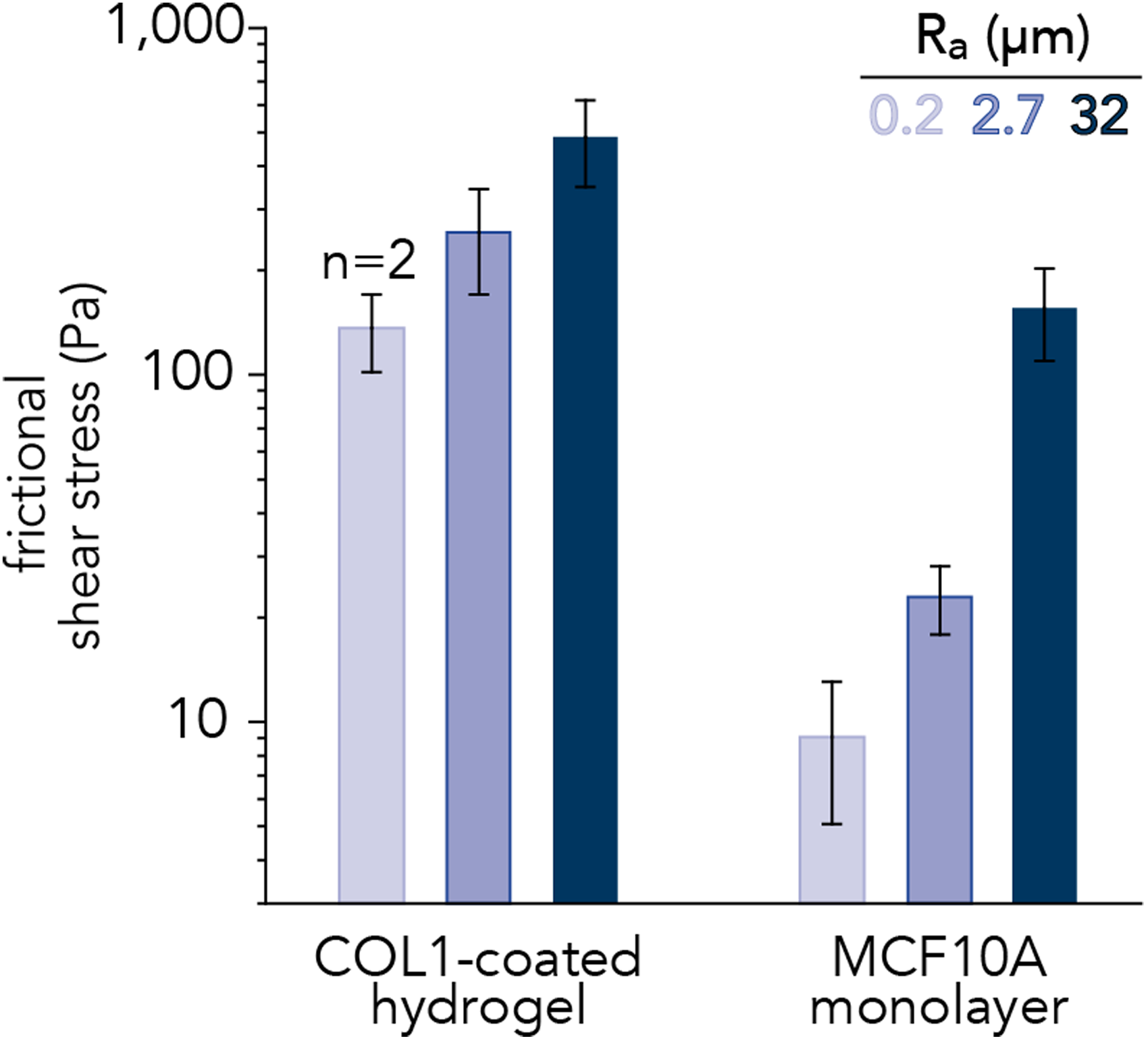
Frictional shear stresses across the sliding interface between silicone implants and model countersurfaces: collagen-coated (COL1) polyacrylamide hydrogels and MCF10A breast epithelial cell monolayers. Shear stress increases with the average surface roughness of breast implant materials (
Materials and methods
Implant surface characterization
The surface topography of soft silicone breast implants was evaluated to investigate the role of surface roughness on soft tissue damage in vitro. Sections of unused and de-gelled commercially-available silicone breast implant shells were removed near the base (Figure 2(a)) for all analyses, where the contact pressures and frictional shear stresses are greatest against the body.
Thin strips (2 mm wide, 6 mm length) of implant shells were cut with a sharp blade and imaged in cross-section using scanning electron microscopy (SEM) to acquire high-resolution micrographs of the implant surface topography that would be in direct sliding contact with soft tissues (Figure 2(b) to (d) and
Soft tissue model surface preparation
To investigate initial damage to the breast pocket following surgical implantation of silicone elastomer implants, we developed simplified in vitro models of the implant-tissue sliding interface that incorporated non-cellular (e.g., ECM) and cellular components.30,31 We conducted tribological measurements (Figure 3(a)) of implants sliding against collagen-coated hydrogels (Figure 3(b)) and healthy breast epithelial cell monolayers (Figure 3(c)).
Collagen-coated hydrogels were composed of polyacrylamide (PAAm) which is a common system used for biological experimentation and protein functionalization.
30
Hydrogel disks (2 mm thickness, 18 mm diameter) were prepared by combining 17.5 wt.% acrylamide, 0.7 wt.% N,N’-methylenebisacrylamide (MBAm), 0.15 wt.% tetramethylenediamine (TEMED), and 0.15 wt.% ammonium persulfate (APS) in ultrapure water (18.2 M
Healthy breast epithelia (MCF10A, ATCC CRL-10317TM) (Figure 3(c)) were used to model the cellular component of breast pockets. Cell monolayers were cultured in basal mammary growth media (MEBMTM Basal Medium, Lonza CC-3150), plated on fibronectin-coated glass-bottomed culture dishes (20 mm well diameter, #0 cover glass, Cellvis D35200N), and grown to 80% confluence prior to tribological measurements. Quasi-static indentation experiments (normal load,
Implant probe preparation and assembly
Sections of silicone breast implant shells (3 mm radius, 0.5 mm thickness) were mounted with cyanoacrylate adhesive on hydrogel probes with spherical shell geometries such that the exterior implant surface would be exposed.
33
Poly(hydroxyethyl)methacrylate (PHEMA) probes (6 mm radius of curvature,
In situ Biotribometer
Tribological testing was conducted using a custom linear reciprocating biotribometer mounted to the condenser turret of an inverted laser scanning confocal microscope (Nikon A1R HD). The probe was fastened to a titanium double-leaf cantilever assembly with normal and tangential stiffness of
For each sliding cycle, friction coefficients (
Results and discussion
Tribological measurements
Surface roughness has long been known to influence the tribological properties of elastic bodies in sliding contact and the relationship is strongly dependent on material,
36
environmental,
37
and testing conditions.38–40 For breast implant materials in sliding contact with COL1-coated hydrogels, the outer shell’s surface roughness did not correlate with friction force (Figure 4). Image analyses of the sliding path revealed that increased surface roughness led to greater removal of collagen from the hydrogel substrate (Figure 5(a,c,e)). Analysis of the wear track over time and sliding distance demonstrated increasing collagen removal at constant normal force
To investigate soft tissue damage in vitro with a simple cellular model of the breast pocket, human breast epithelial (MCF10A) cell monolayers were subjected to identical tribological challenges (with the exception of growth media in place of PBS). Cells were stained using a fluorescent propidium iodide (PI) live/dead assay to clearly identify dead cells (
Frictional shear stress measurements demonstrate the utility of these in vitro soft tissue models and enabled our investigation into COL1- and MCF10A-implant interfaces. These biological interfaces present unmet tribological design challenges for implant materials. These studies are limited by the fact that cell monolayers were plated on fibronectin-coated glass and not on COL1-coated hydrogel substrates. Another limitation is that the sliding experiments were conducted over the span of a few hours, which may be insufficient to observe substantive cellular remodeling or morphological changes. Future investigations will extend this work to provide deeper analysis of cell responses to repeated mechanical microtraumas.
Conclusions
In this investigation, we developed an in vitro method to systematically characterize silicone breast implant surface roughness induced soft tissue damage on COL1-coated polyacrylamide hydrogel surfaces and MCF10A cell monolayers. Our findings suggest that “smooth” silicone elastomer breast implants (
Supplemental Material
sj-docx-1-pij-10.1177_13506501221132897 - Supplemental material for In vitro models of soft tissue damage by implant-associated frictional shear stresses
Supplemental material, sj-docx-1-pij-10.1177_13506501221132897 for In vitro models of soft tissue damage by implant-associated frictional shear stresses by Jonah M. Rosas, Dixon J. Atkins, Allison L. Chau, Yen-Tsung Chen, Rachel Bae, Megan K. Cavanaugh, Ricardo I. Espinosa Lima, Andrew Bordeos, Michael G. Bryant and Angela A. Pitenis in Proceedings of the Institution of Mechanical Engineers, Part J: Journal of Engineering Tribology
Footnotes
Acknowledgements
This manuscript is dedicated to the memory of Duncan and Mabel Dowson, whose generosity of spirit is a continual source of inspiration. We are grateful to Kylie E. Van Meter for helpful discussions on early designs of the probe assembly. We are grateful for helpful discussions with Dr. Juan Manuel Urueña.
Data accessibility
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was partially supported by the National Science Foundation (NSF) Materials Research Science and Engineering Center (MRSEC) at UC Santa Barbara through DMR-1720256 (IRG-3). The use of the shared facilities of the MRSEC is gratefully acknowledged; the UCSB MRSEC is a member of the Materials Research Facilities Network (www.mrfn.org). This work was partially supported by the BioPACIFIC Materials Innovation Platform of the National Science Foundation under Award No. DMR-1933487. This work was partially supported by Establishment Labs, Costa Rica. Establishment Labs did not take part in the study design, collection or analysis of the data; in writing the journal article, or the decision to submit for publication. J.M.R. acknowledges support from the Gates Millennium Scholarship through the Bill and Melinda Gates Foundation and Hispanic Scholarship Fund. A.L.C. acknowledges support of the National Science Foundation Graduate Research Fellowship Program under Grant No. 1650114. R.B. acknowledges support from the Future Leaders in Advanced Materials (FLAM) program (DMR-1720256). R.I.E.L. acknowledges support from the University of California Leadership Excellence through Advanced Degrees (UC LEADS) program. A.A.P. acknowledges funding support from the NSF CAREER award (CMMI-CAREER-2048043).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


