Abstract
Trihexyltetradecylphosphonium bis(2-ethylhexyl)phosphate (phosphonium phosphate) ionic liquid is soluble in non-polar lubricants. It has been proposed as an effective anti-wear additive comparable to zinc dialkyldithiophosphate. Previously, phosphonium phosphate has shown a better anti-wear performance under some conditions such as high temperature. In this work, the tribological performance and the lubrication mechanism of phosphonium phosphate are compared with that of zinc dialkyldithiophosphate when lubricating silicon under various tribological conditions. This can lead to an understanding of the reasons behind the superior anti-wear performance of phosphonium phosphate under some conditions. A micro-scale study is conducted using a nanotribometer. The results show that both additives lead to a considerable reduction in both friction and wear coefficients. The reduction in the wear coefficient is mainly controlled by the formation of the tribofilm on the rubbing surfaces. Zinc dialkyldithiophosphate can create a thicker tribofilm, which results in a better anti-wear performance. However, the formation of a thicker film will lead to a faster depletion and thus phosphonium phosphate can provide better anti-wear performance when the depletion of zinc dialkyldithiophosphate starts.
Keywords
Introduction
A large number of recent studies in tribology have focused on the mitigation of wear and friction. This mitigation will lead to an elongated lifetime of the mechanical systems and enhance the overall efficiency. The increase in the components’ lifetime can decrease maintenance cost, the consumption of material and the energy required to manufacture replacements. Furthermore, reduction in friction increases system efficiency due to the reduction of the energy consumption associated with running the component. For instance, in passenger automobiles and heavy-duty machines, the reduction in the friction coefficient can reduce fuel consumption by about 18% and 14%, respectively. Consequently, the reduction in fuel consumption decreases the emission of CO2, which makes the mechanical system more environmentally friendly.1,2
A number of approaches have been suggested and investigated in the literature to decrease wear and friction that include the use of advanced lubricants such as ionic liquids (ILs),3–6 effective anti-wear additives such as zinc dialkyldithiophosphate (ZDDP), 7 advanced coatings such as diamond-like coatings (DLCs) 8 and surface texturing. 9
In terms of the use of anti-wear additives, ZDDP showed a significant reduction in wear due to its ability to react with the interacting surfaces to form a protective layer called tribofilm. The mechanism of the formation of ZDDP tribofilms was studied in detail in the literature.10–15
These studies covered the formation of the tribofilm and its impact on the tribological behaviour of bearing steel surfaces. The results confirmed the tribofilm formation and its substantial role in the reduction of wear. However, adding the ZDDP additive to engine oils produces ash that poisons the catalyst of the exhaust system, thus reducing its efficiency to reduce harmful emissions. This motivated researchers to find different alternatives to ZDDP.16–18 The ashless dialkyldithiophosphates (DDPs) have been suggested as good candidates to replace ZDDP.16–18 The results showed that DDPs can provide comparable anti-wear performance to ZDDP.
ILs have also been suggested as effective alternatives to ZDDP. 19 ILs have been proposed as either potential lubricants or additives due to their desirable physical properties such as high thermal stability, low vapour pressure and high thermal conductivity. In addition, ILs showed the ability to react with interacting surfaces and to form a tribofilm. The tribofilm formation can provide a desirable tribological performance to potentially protect the surface from wear or reduce the friction coefficient. However, the use of ILs as additives is not applicable in non-polar lubricants due to their poor solubility.3–6 A novel phosphonium phosphate (PP) IL that is soluble in non-polar lubricants was proposed to have good solubility. 19 The wear protection of PP IL was comparable with ZDDP when lubricating different contacts such as steel–DLC, 20 Al–Si–cast iron, 7 silica, 21 aluminium 22 and steel–steel. 23 The results showed that both ZDDP and PP additives react with the rubbing surfaces and exhibited a considerable reduction in wear. In addition, PP showed a higher wear reduction at high temperature and a higher anti-corrosion when lubricating steel–steel contacts.
The tribofilm formation mechanism of PP was studied by Zhou et al. 24 The results showed that a very thin film, <50 nm, is generated, which exhibited a reduction in wear. The reactivity of ZDDP and PP with silicon surfaces at elevated temperature and the formation mechanism of thermal films were studied in Al-Sallami et al. 25 The results showed that ZDDP can form a thicker thermal film than PP.
However, there is a question that remains to be answered in relation to the anti-wear mechanism of PP compared to ZDDP: what is the exact reason behind the superior anti-wear performance of PP under a particular condition (at high temperature)? This can be answered by studying the tribological performance and the formation of the tribofilms of both additives under various tribological conditions including varying contact pressures, sliding distances and temperatures.
In addition, the understanding of the lubrication of Al–Si alloy is crucial due to its widespread use in various mechanical systems such as engines, plain bearings and compressors of refrigeration systems. 26 Burkinshaw et al. 27 studied the tribological performance and the tribochemistry of both Al–Si alloy and silicon crystal. They found that both showed a comparable tribological performance and the silicon grains controlled the tribochemical behaviour.
This study is an extension of previous studies25,27 that were aimed at understanding the tribological behaviour and the lubrication mechanism of silicon surfaces, relevant to the powertrain in the internal combustion engine. It is also an extension of the very recently published work that aims to understand the thermal film formation of PP and compares it with that of the ZDDP thermal film to understand their lubrication mechanisms.
Materials and experimental methods
Materials
The tribopair is a silicon wafer, n-type, as a substrate and a steel ball (AISI 52100) as a counterface and their average surface roughness values are 0.2 and 125 nm, respectively. The surface roughness was measured using an atomic force microscope and white light interferometry for the silicon substrate and the steel ball, respectively. The silicon wafer was cut into small samples, 7 × 7 mm2. The diameter of the steel ball is 2 mm. The mechanical properties of both materials are presented in the Supplemental Material.
Three lubricants were used, i.e. polyalphaolefin (PAO), PAO + 1.08 wt% PP and PAO + 0.74 wt% ZDDP. These concentrations of the additives were selected to obtain the same phosphorus content at both mixtures. The blending process has been presented in the previous work in Al-Sallami et al. 25 Table 1 shows the chemistry, vendors and purity of the utilized lubricants.
The utilized lubricants.
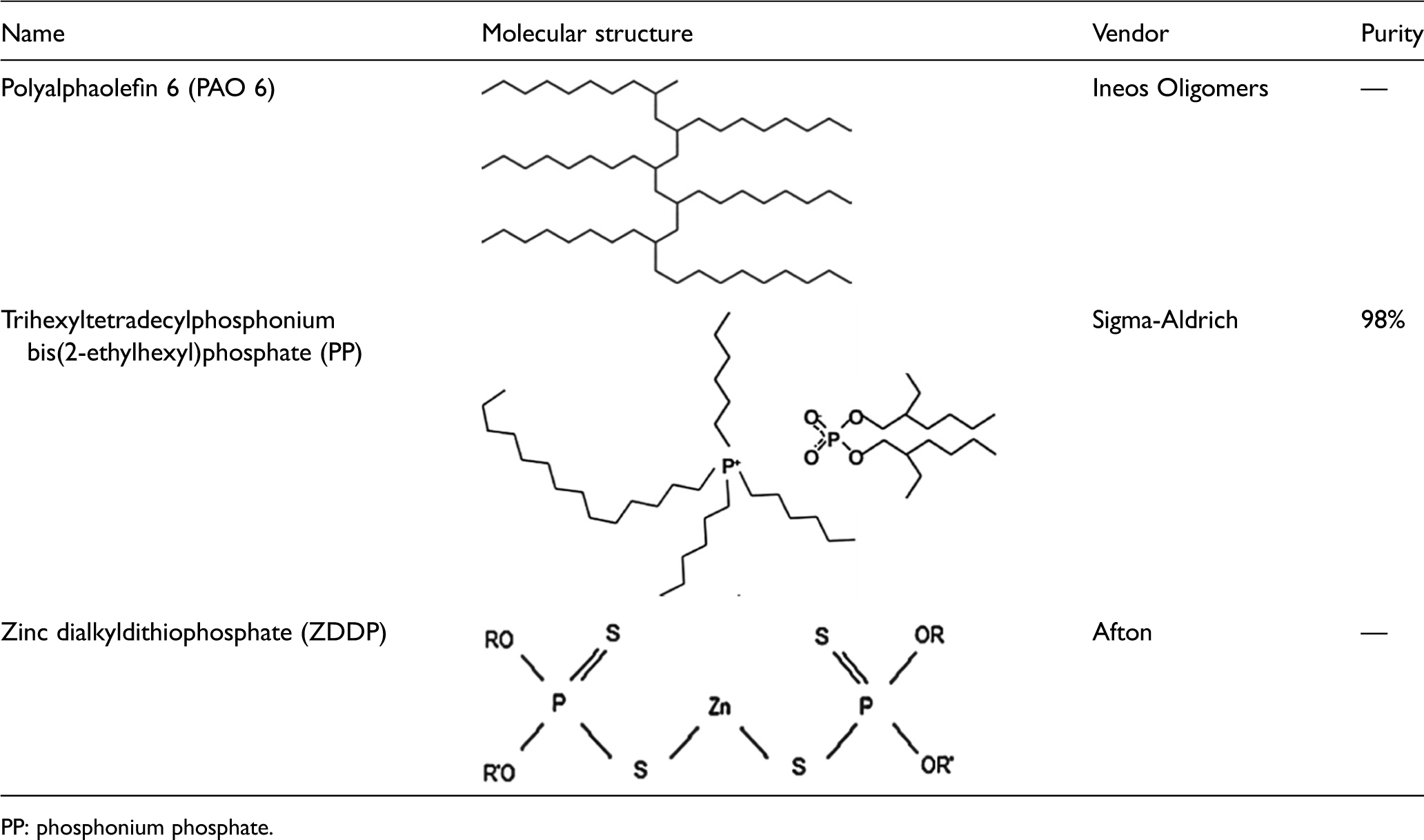
PP: phosphonium phosphate.
Tribological tests
The tribological experiments were performed by applying a reciprocating motion using a nanotribometer with a ball-on-plate configuration. The high load cell was used when the maximum and minimum applicable loads were 1 N and 0.5 mN, respectively, with a 3 µN load resolution.
A bespoke component was designed to conduct high-temperature experiments, as shown in Figure 1. A cartridge heater is inserted inside the component. A thermocouple is also inserted inside the component to sense the temperature. A digital controller is used to supply the electrical power to the cartridge heater and stop it once the desired temperature is reached (the signal comes from the utilized thermocouple).

The configuration of high-temperature experiments using the bespoke design. NTR: nanotribometer.
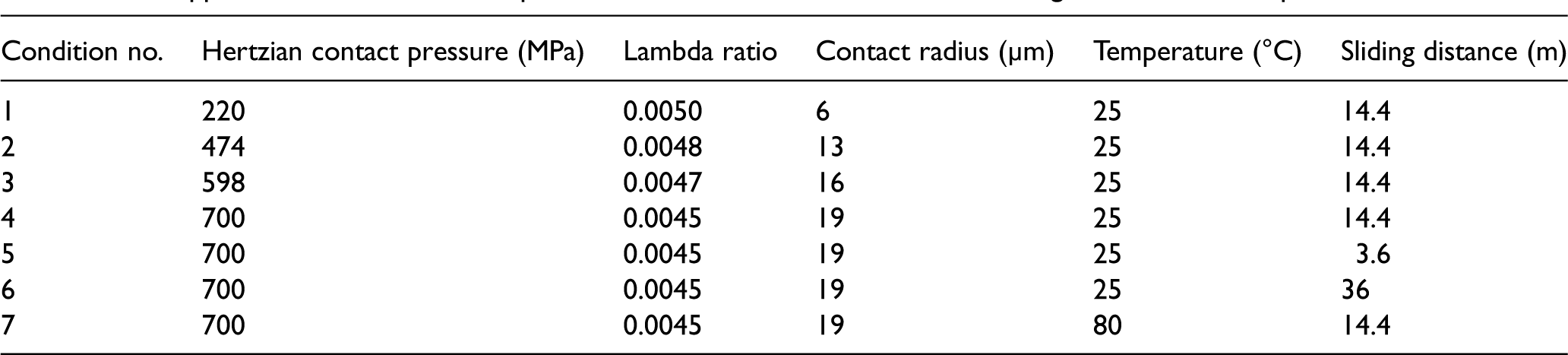
Hamrock and Dowson 28 formulae were used to calculate the contact pressure, contact radius and lambda ratio. The details of the calculations of the contact pressure and lambda ratio are provided in the Supplemental Material. The velocity and sliding amplitude were kept constant, which were 2 mm/s and 2 mm, respectively. Table 2 shows the operating conditions in this study. The first four conditions were applied to investigate the effect of contact pressure. Conditions 4–6 were applied to investigate the effect of the sliding distance, whereas conditions 4 and 7 were applied to investigate the effect of temperature.
The applied conditions for the experiments to assess the effects of load, sliding distance, and temperature.
The friction coefficient was measured in situ by calculating the ratio of the friction force to the normal force at each cycle (every 2 s). Each experiment is repeated at least two times and in most cases three times. The presented values are the average values with error bars showing the difference between the results of the conducted experiments under each condition.
The presentation of the friction coefficient behaviour in Figure 2 has resulted from the average values of at least two tests. Surface morphology was characterized using three-dimensional (3D) optical microscopy including the quantification of the wear volume. The following equation was used to calculate the wear coefficient: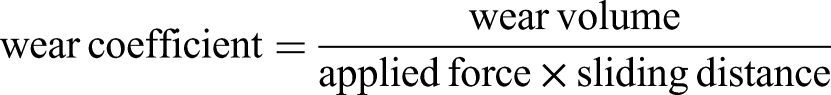
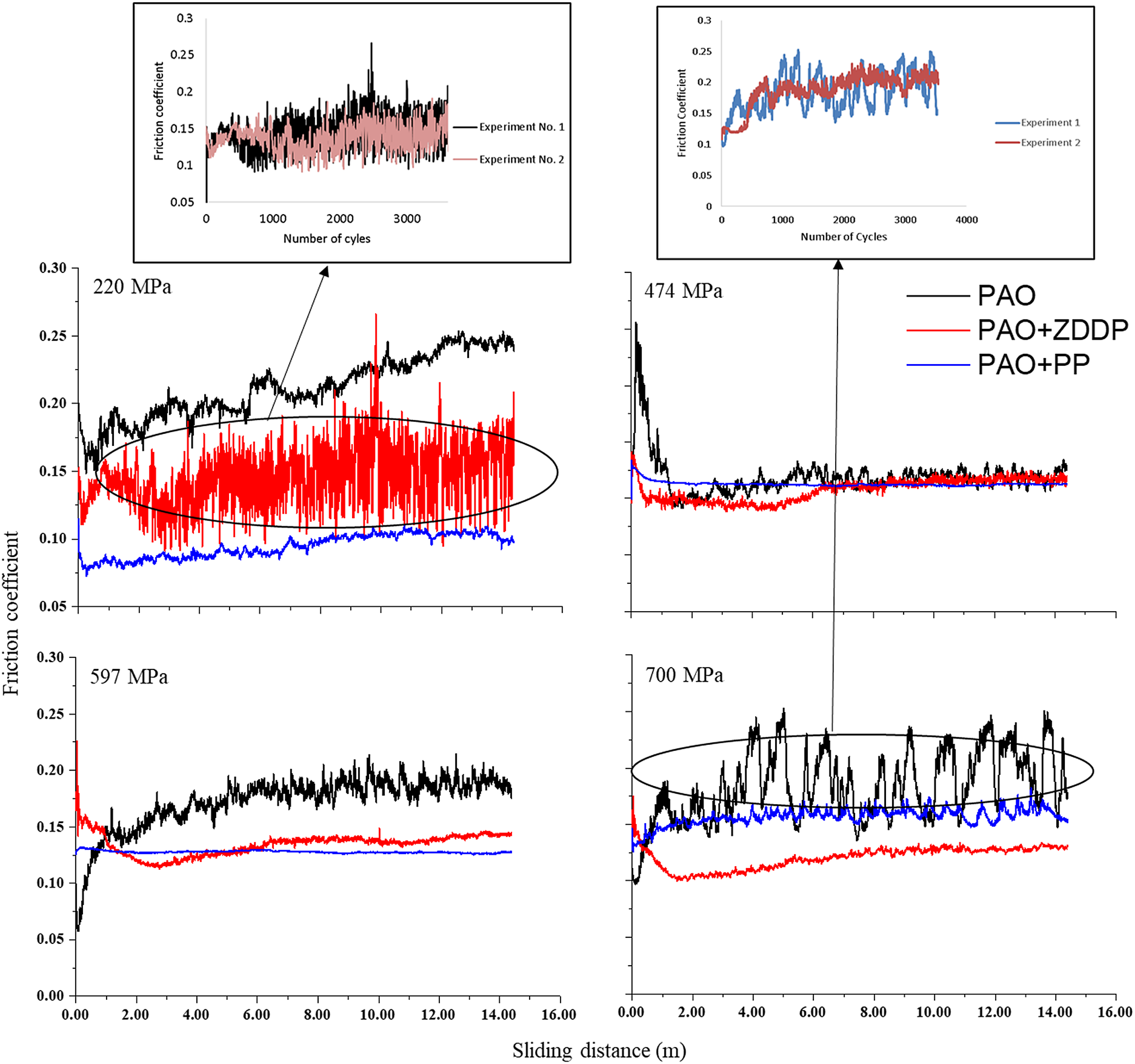
Evolution of friction coefficient as a function of the sliding distance at a range of contact pressures. PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate.
Surface morphology
The surface morphology of the samples was characterized using non-destructive and non-contact white light interferometry (NPFLEX Bruker). A vertical optical profiler with a 10× objective was used to obtain a 3D map of the surface topography and surface roughness and to quantify the wear volume. To quantify the wear volume, a stitching scan was performed to cover a 3 × 0.82 mm2 area with a lateral resolution of 0.38 µm, which was then analysed using Vision64 software (Bruker).
Surface chemistry
Scanning electron microscope–energy dispersive X-ray spectroscopy (SEM–EDS)
A Hitachi SEM–EDS was used to assess the surface chemistry inside and outside the wear tracks. Silicon samples require a preparation before measurement including carbon coating to avoid charging. 29 The applied voltages were 2 and 15 kV to collect electron images and scan surface chemistry, respectively. At least two points were scanned inside and outside the wear track. The chemical analysis and calculation of the chemical composition were performed using AZtec software.
X-ray photoelectron spectroscopy (XPS)
A Thermo Escalab 250 XPS was used to quantify the surface chemistry and identify the tribofilm species. A monochromatic Al Kα (1486.6 eV) beam with a 500 µm spot size was used. The position of the spot was specified at the centre of the wear track. Three sweeps were applied to acquire the survey and 10 sweeps for the high-resolution regions. The pass energies were 160 and 20 eV for the survey and high-resolution regions, respectively. To avoid the interference between the silicon electrons, which might result from the plasmon effect, both phosphorus and sulphur peaks of P 2s and S 2s were acquired instead of P 2p and S 2p since the recognition with silicon electrons is easier in the former.30,31 CasaXPS was used for curve fitting using Gaussian–Lorentzian peak shapes. The charge correction was performed based on the aliphatic carbon peak in the C 1s spectrum by shifting its binding energy to 285 eV.
Results
Friction coefficient
The behavior of the friction coefficients during the test and their values are shown in Figures 2 and 3. It can be clearly observed that the relationship between the friction coefficient and contact pressure of PAO is inconsistent, i.e. the increase in contact pressure causes either an increase or decrease in the friction coefficient. This can be related to the variation of surface roughness due to the wear and smearing of contacting asperities. 32
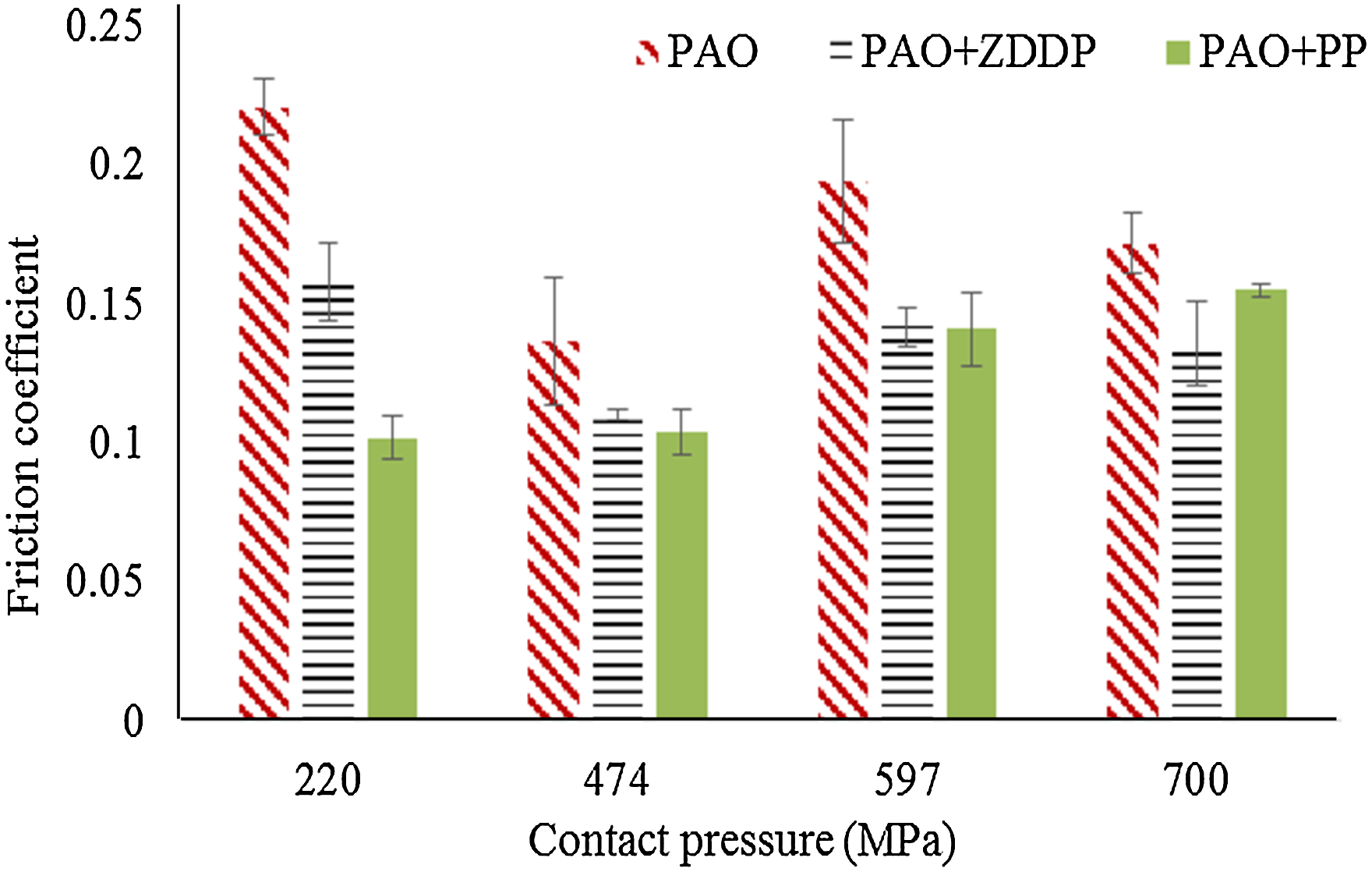
Friction coefficients at various contact pressures (sliding distance 14.4 m, velocity 2 mm/s and stroke length 2 mm). PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate.
Adding PP or ZDDP leads to a decrease in the friction coefficient. Within the same contact pressure, adding either PP or ZDDP appears to have the same reduction effect on the friction coefficient, except for the case of 220 MPa at which the PP additive reduces the friction coefficient by 50% more than the ZDDP. The reduction in the friction coefficient at high contact pressures (474 MPa and higher) as a result of using anti-wear additives (ZDDP and PP) can be related to the decrease in the production of wear debris. 33
The evolution of the friction coefficient is also investigated over time during the tribological experiments, as shown in Figure 2. The PP additive shows the highest stability, i.e. the lowest fluctuations, at all contact pressures. The ZDDP additive also shows the high stability except at the lowest contact pressure. In contrast, PAO shows the highest fluctuation, especially at the highest contact pressure. The fluctuations in the friction coefficient can possibly be related to the production of wear debris, which increase with wear. 33 This suggests that the lower the wear debris in the tribo contact the more stable the friction coefficient. The high fluctuations in the case of the ZDDP additive at the lowest contact pressure (same behaviour is observed for three tests) seem to result from the balance in the formation and removal of the protective tribofilm.
In addition, the friction coefficient can also be affected by the viscosity of the lubricant and the combined roughness of the rubbing surfaces.32,34 In this study, the effect of viscosity is negligible since all the tested lubricants exhibit the same viscosity. 7 On the other hand, the average surface roughness shows a considerable effect on the final (stable) friction coefficient of PAO (see Table 3 and Figure 2). For a neat PAO, the reduction in surface roughness leads to a decrease in the final friction coefficient and vice versa (at least in the cases where wear was measurable). This confirms that in the absence of any additive, the friction coefficient is mainly controlled by the average surface roughness.
Average surface roughness inside wear track at various contact pressures.

PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate.
Adding either PP or ZDDP leads to a considerable reduction in the average surface roughness (see Table 3), apart from the highest contact pressure. This suggests that the reduction in the average surface roughness contributes to the reduction in the friction coefficient. However, at the highest contact pressure (700 MPa), PP shows a lower average surface roughness while it exhibits a higher final friction coefficient. This can reveal that in the presence of an anti-wear additive (PP or ZDDP), the surface roughness is not the main factor that controls the friction coefficient.
Figures 4 and 5 show the effect of increasing temperature on the friction coefficients and the behaviour of friction coefficients during the tribological experiments, respectively. In the case of ZDDP, the friction coefficient increases when the temperature increases from 25 °C to 80 °C while in the case of PP it decreases.
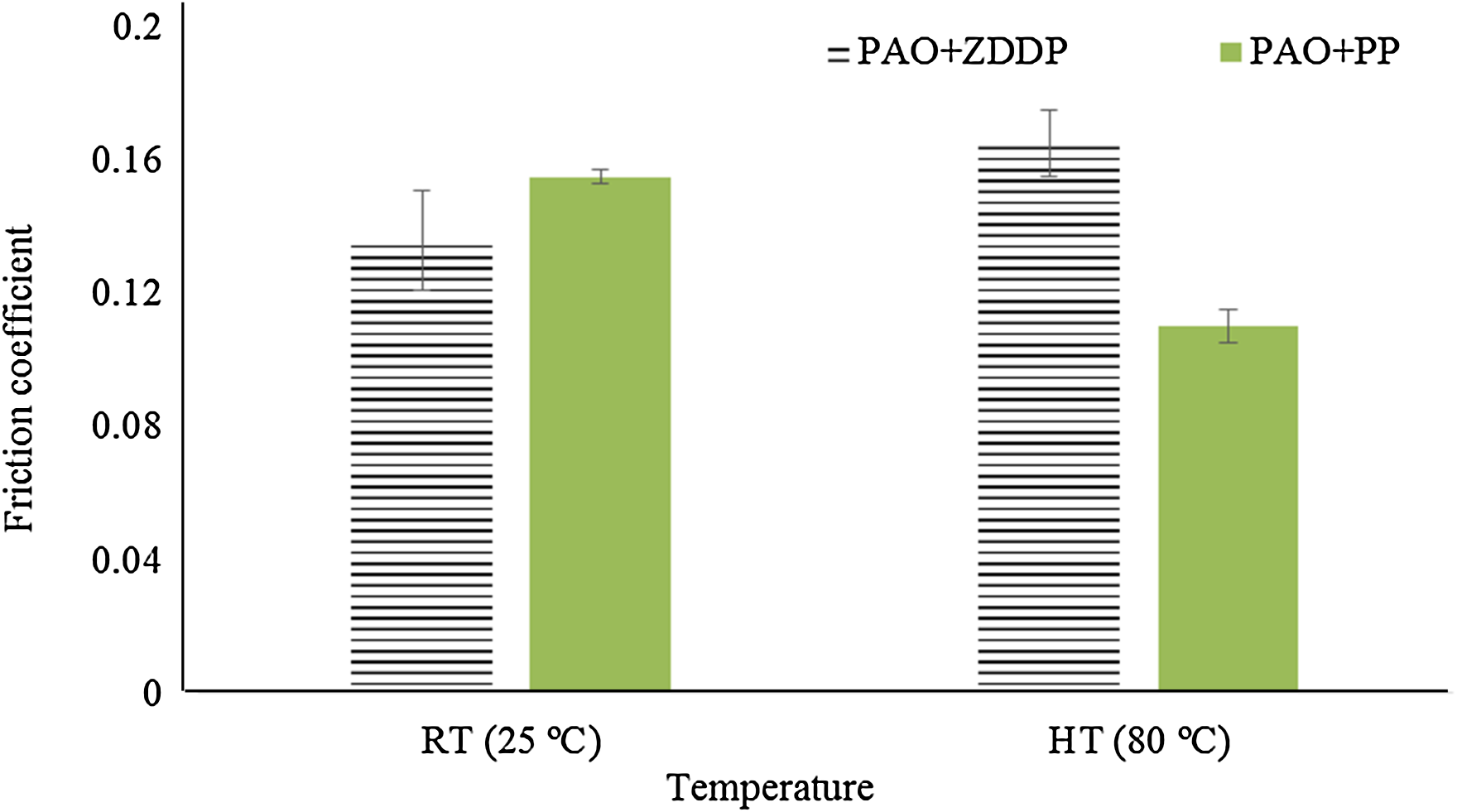
The friction coefficients for PAO + ZDDP and PAO + PP at room temperature and high temperature (at contact pressure 700 MPa, sliding distance 14.4 m, velocity 2 mm/s and stroke length 2 mm). PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate; RT: room temperature; HT: high temperature.
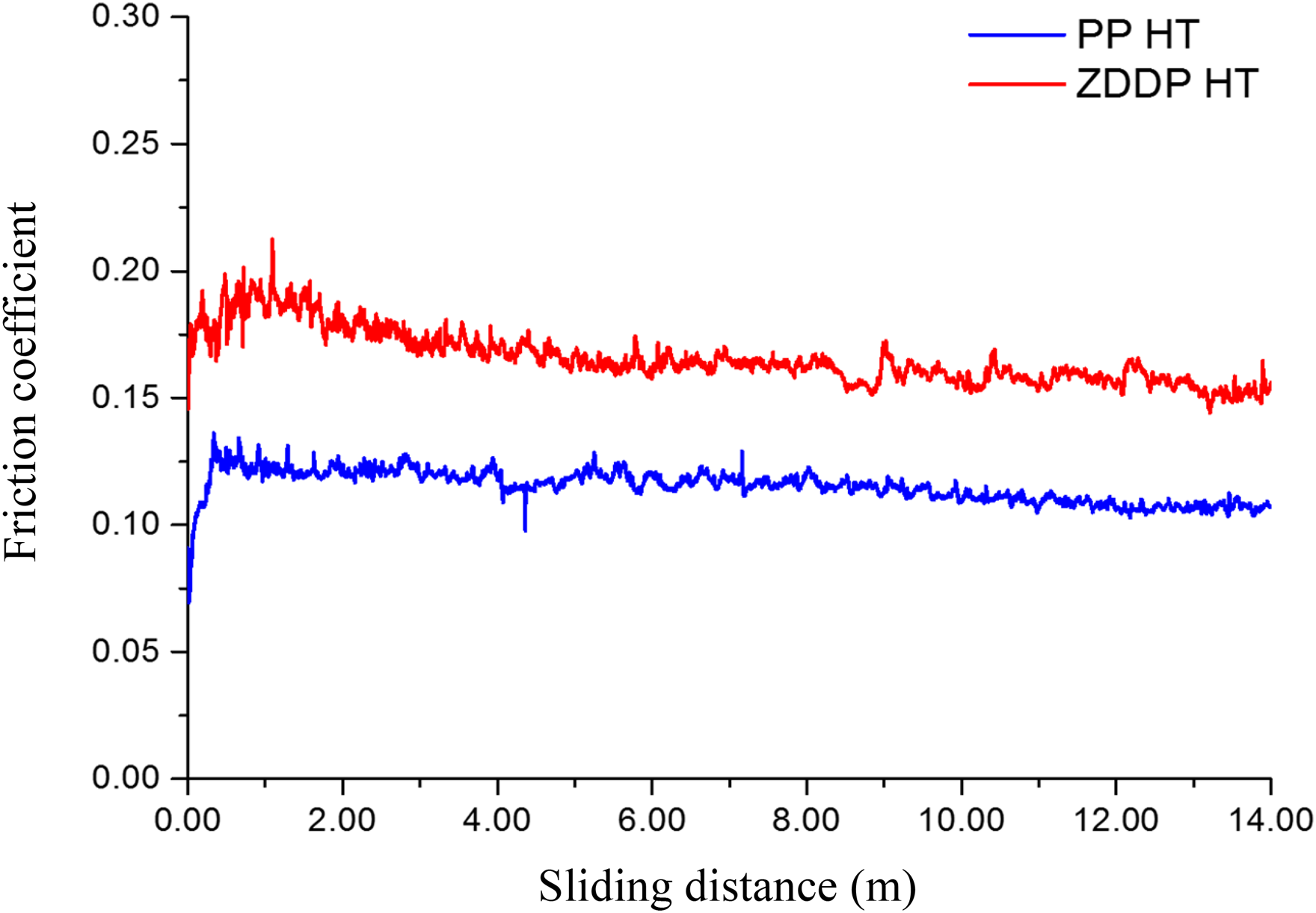
The behaviour of friction coefficients for both PAO + ZDDP and PAO + PP at high temperature (80 °C). PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate; HT: high temperature.
The increase in temperature shows a slightly more stable friction coefficient for both additives over the sliding distance (see Figures 2 and 5). This can be related to the faster formation of the tribofilm, which leads to a reduction in the production of wear debris. To confirm this finding, again, the results of both wear coefficients and surface chemistry must be considered together.
The effect of the sliding distance is also investigated by applying three various sliding distances, which are 3.6, 14.4 and 36 m. Figure 6 shows the friction coefficients for both additives at the three sliding distances. The results show that the friction coefficient of ZDDP is lower than the one of PP (as an average for the whole test of 36 m). By increasing the sliding distance from 3.6 to 14.4 m, the friction coefficient increases. The friction coefficients after 36 m of sliding are comparable with those observed after 14 m of sliding.
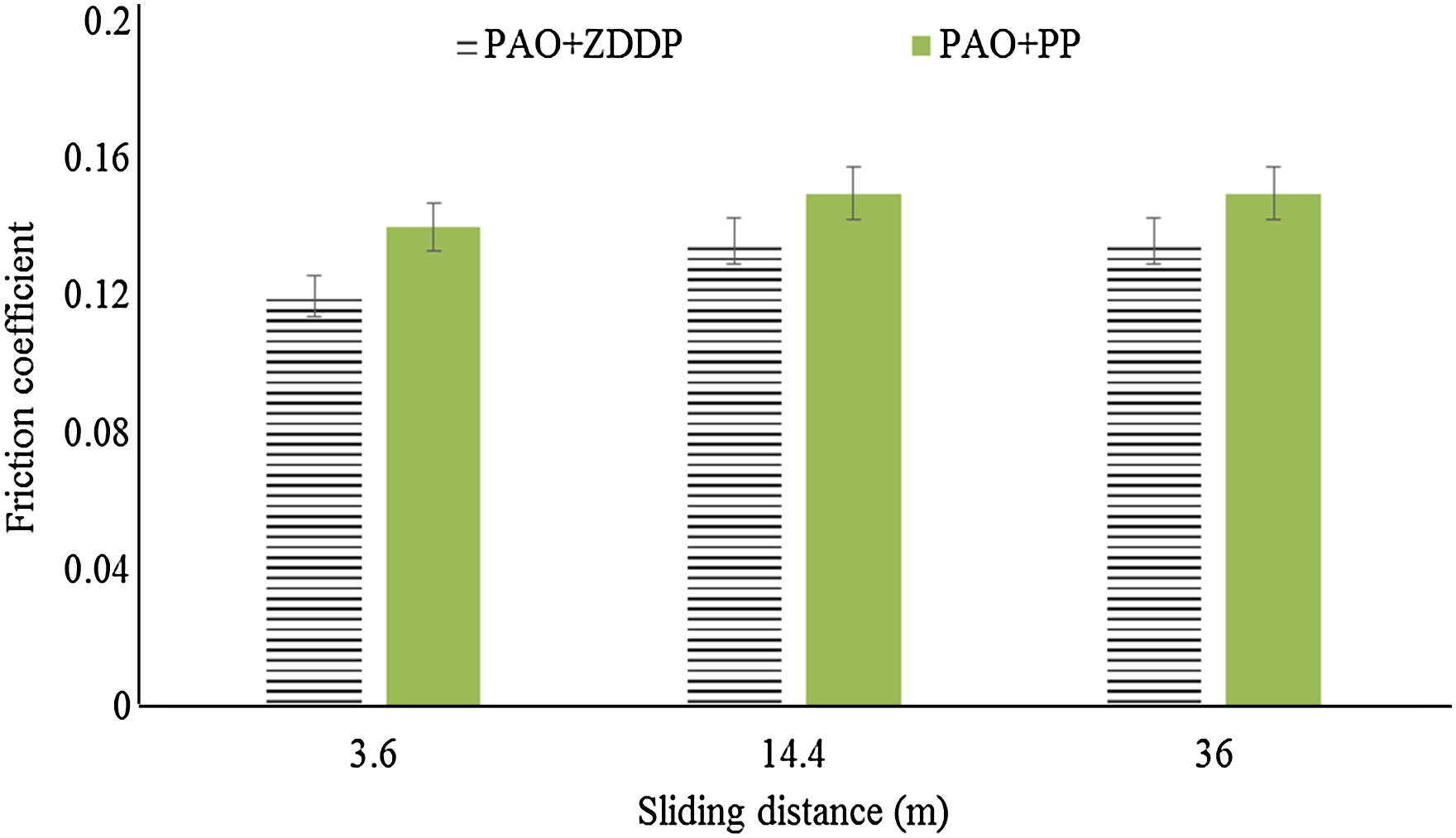
The friction coefficients for both additives at various sliding distances (at contact pressure 700 MPa, velocity 2 mm/s and stroke length 2 mm). PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate.
The evolution of friction coefficients of both additives over 36 m of the sliding distance is shown in Figure 7. The friction coefficient of ZDDP starts with a lower value and then increases until it reaches a steady state of 0.145. The friction coefficient of PP is higher than ZDDP and increases slightly until reaching about 0.165 and then decreases and becomes 0.125 at the end of the test (at 36 m of sliding).
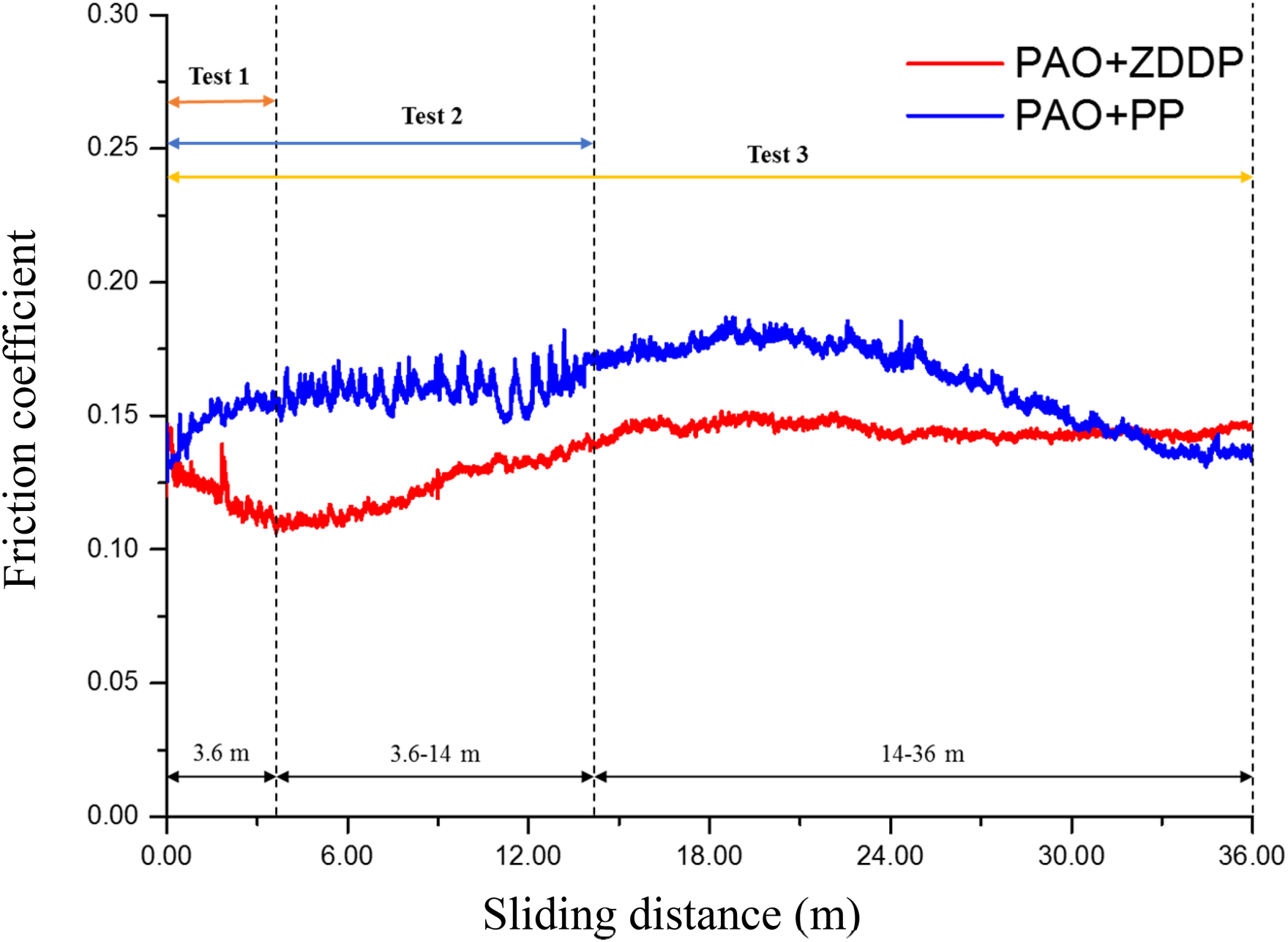
Friction coefficient behaviour of both additives at three various sliding distances. PAO: polyalphaolefin; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate.
Wear coefficient
Figure 8 shows the wear coefficients for all lubricants at various contact pressures. No measurable wear can be detected at the lowest contact pressure of 220 MPa. For the rest of the contact pressures, the PP and ZDDP additives provide a comparable reduction in the wear coefficient. The reduction has resulted from the reduction in wear depth and width (see Figure 9). The ZDDP provides a higher reduction, which suggests that a thicker or more durable protective tribofilm is formed by ZDDP.
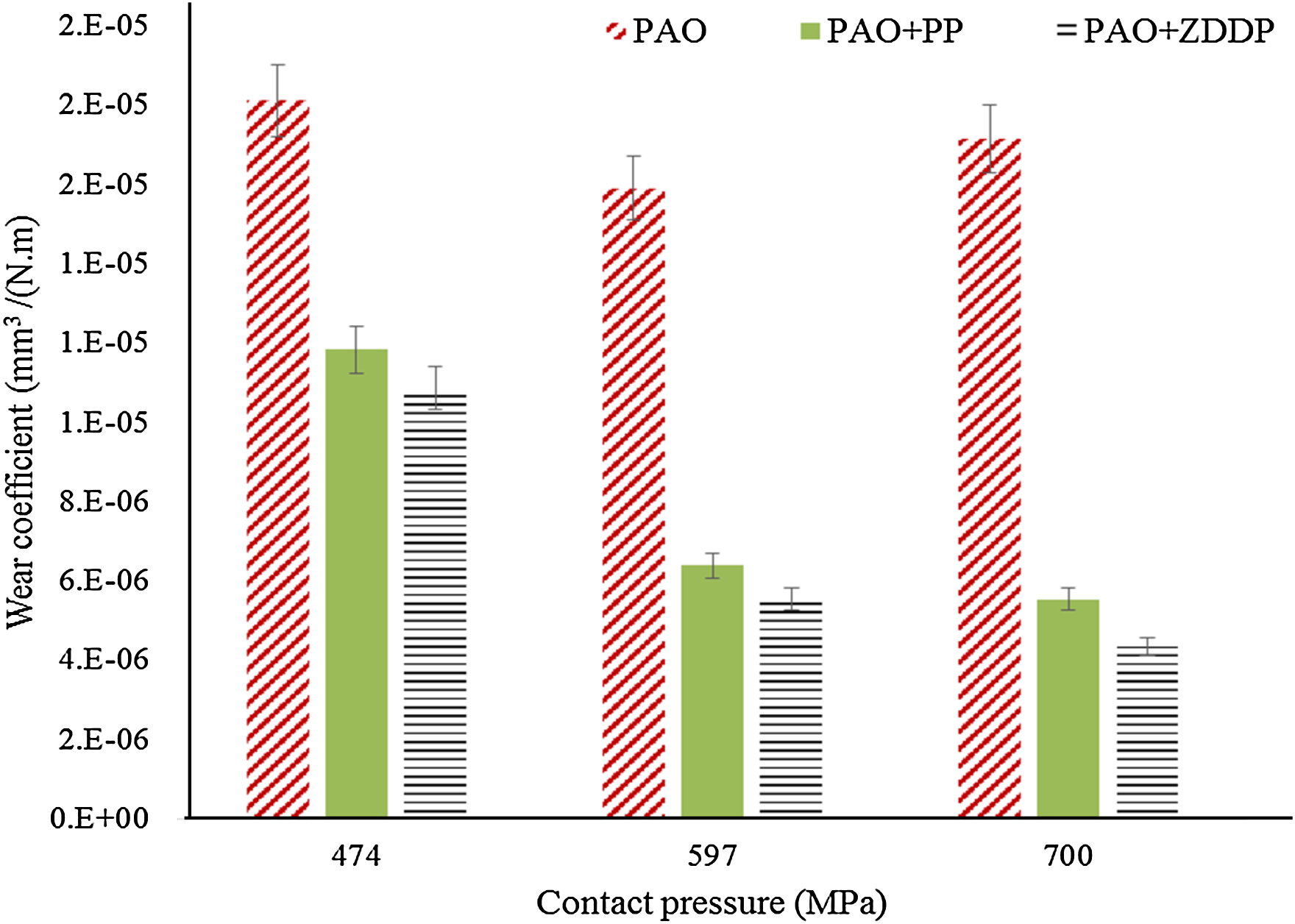
Wear coefficient at various contact pressures (sliding distance 14.4 m, velocity 2 mm/s and stroke length 2 mm). PAO: polyalphaolefin; PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate.
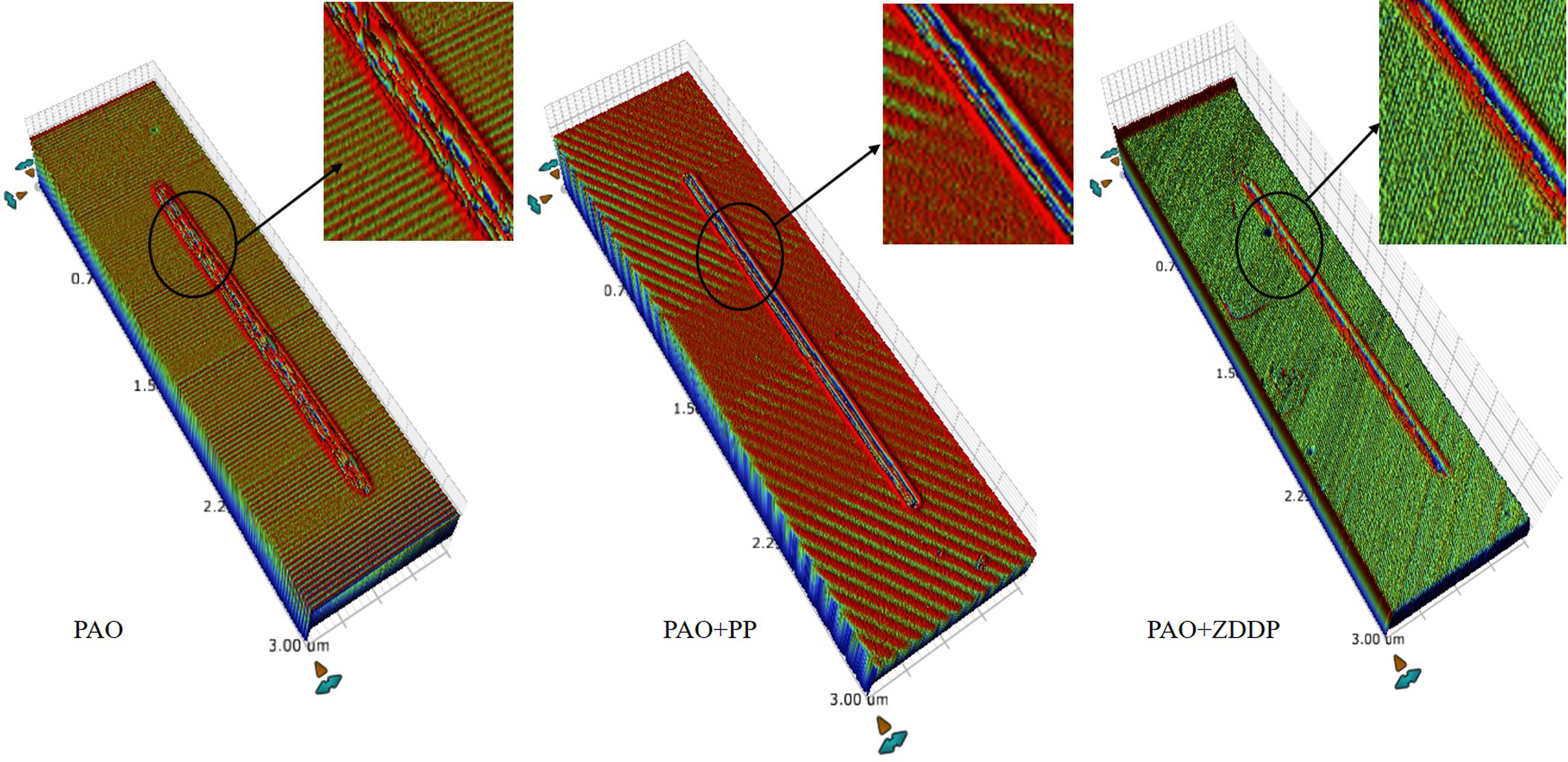
A 3D view of the wear tracks that resulted at a contact pressure of 700 MPa. 3D: three-dimensional; PAO: polyalphaolefin; PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate.
The increase in temperature causes a significant reduction in the wear coefficients of both additives, as shown in Figure 10. This reduction can be due to the formation of tribofilms and thermal films by the additives on silicon, as presented in Al-Sallami et al. 25 A higher reduction is achieved by ZDDP at high temperature, which is expected since it is able to generate thicker tribofilms and thermal films at high temperature10–15 while the reactivity of PP with a silicon surface is self-limited. 25
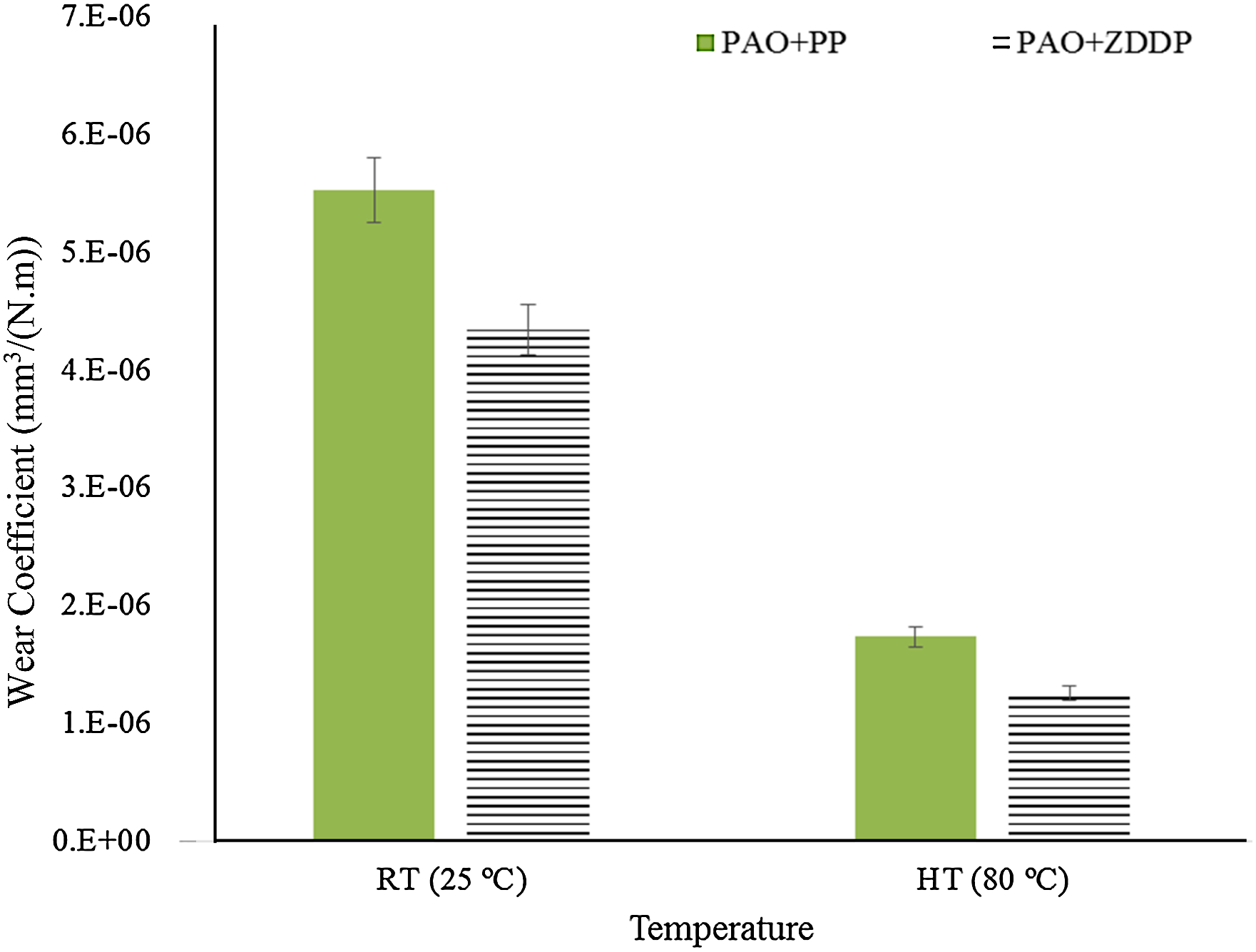
Wear coefficients for PAO + ZDDP and PAO + PP at room temperature and high temperature (contact pressure 700 MPa, sliding distance 14.4 m, velocity 2 mm/s and stroke length 2 mm). PAO: polyalphaolefin; PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate; RT: room temperature; HT: high temperature.
Figure 11 shows the wear coefficients of ZDDP and PP additives at two sliding distances (14.4 and 36 m). No wear could be detected at the lowest sliding distance (3.6 m). A considerable reduction in the wear coefficient is achieved as the sliding distance increases to 36 m (not wear volume). Referring to equation (1), the increase in the sliding distance will decrease the wear coefficient when the increase in the wear volume is not significant. This reduction also can be related to the formation of a thicker tribofilm that leads to higher protection against wear. The PP shows higher wear protection than ZDDP at the highest sliding distance (36 m). This can be related to its thicker tribofilm at this condition (longest sliding distance).
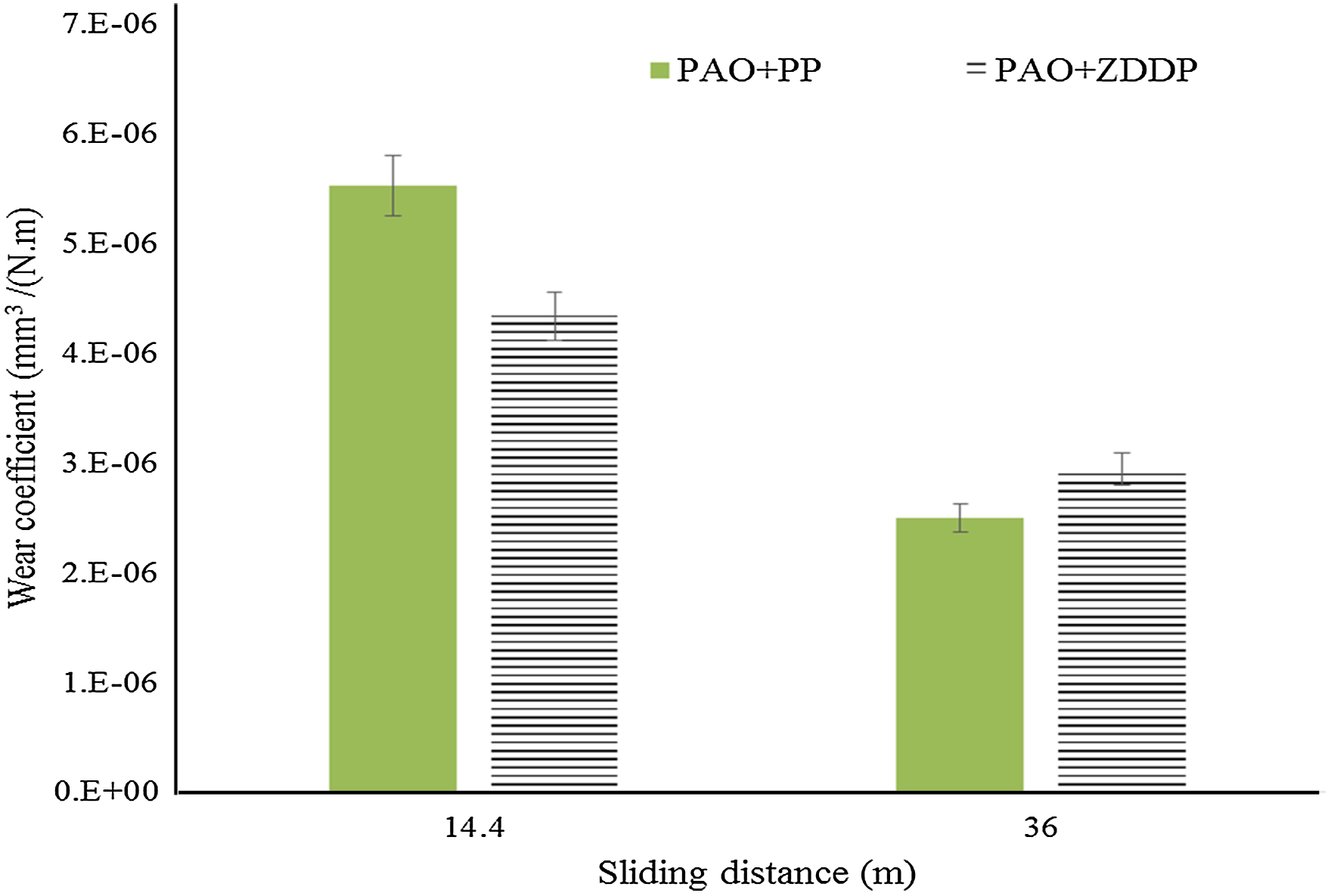
Wear coefficient behaviour of both additives at two various sliding distances (contact pressure 700 MPa, velocity 2 mm/s and stroke length 2 mm). PAO: polyalphaolefin; PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate.
Surface chemistry
SEM–EDS was used to evaluate the surface chemistry inside and outside the wear track. Figure 12 shows the scanned points for three samples at the maximum contact pressure (700 MPa). The results from the inside and outside of the wear tracks show that only silicon, carbon and oxygen exist in all samples (see Table 4). This suggests that the tribofilm is either not formed or formed but not thick enough to be detected by EDS.

Scanned spots by SEM–EDS for wear tracks that resulted at a contact pressure of 700 MPa. SEM–EDS: scanning electron microscope–energy dispersive X-ray spectroscopy; PAO: polyalphaolefin; PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate.
EDS results at a contact pressure of 700 MPa.

EDS: energy dispersive X-ray spectroscopy; PAO: polyalphaolefin; PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate.
To check the formation of any tribofilm, XPS was used and only the samples lubricated with PAO + ZDDP and PAO + PP were examined since the formation of any tribofilm is expected in these cases only.
For the case of the substrates lubricated with ZDDP, the presence of P, S or Zn can confirm the formation of a protective tribofilm. Figures 13 and 14 show the XPS results for Zn and P. As the two elements exist inside the wear track, this suggests that the tribofilm forms on the contacting surfaces. In contrast, no sulphur can be detected inside the wear track under various conditions, see Figure 15. This suggests that either sulphur is not included in the formation of the tribofilm or it might have been formed but then removed by rubbing. This is supported by the previous results of Al-Sallami et al., 25 which showed that the formation of the ZDDP thermal film on the silicon surface does not always include sulphur.
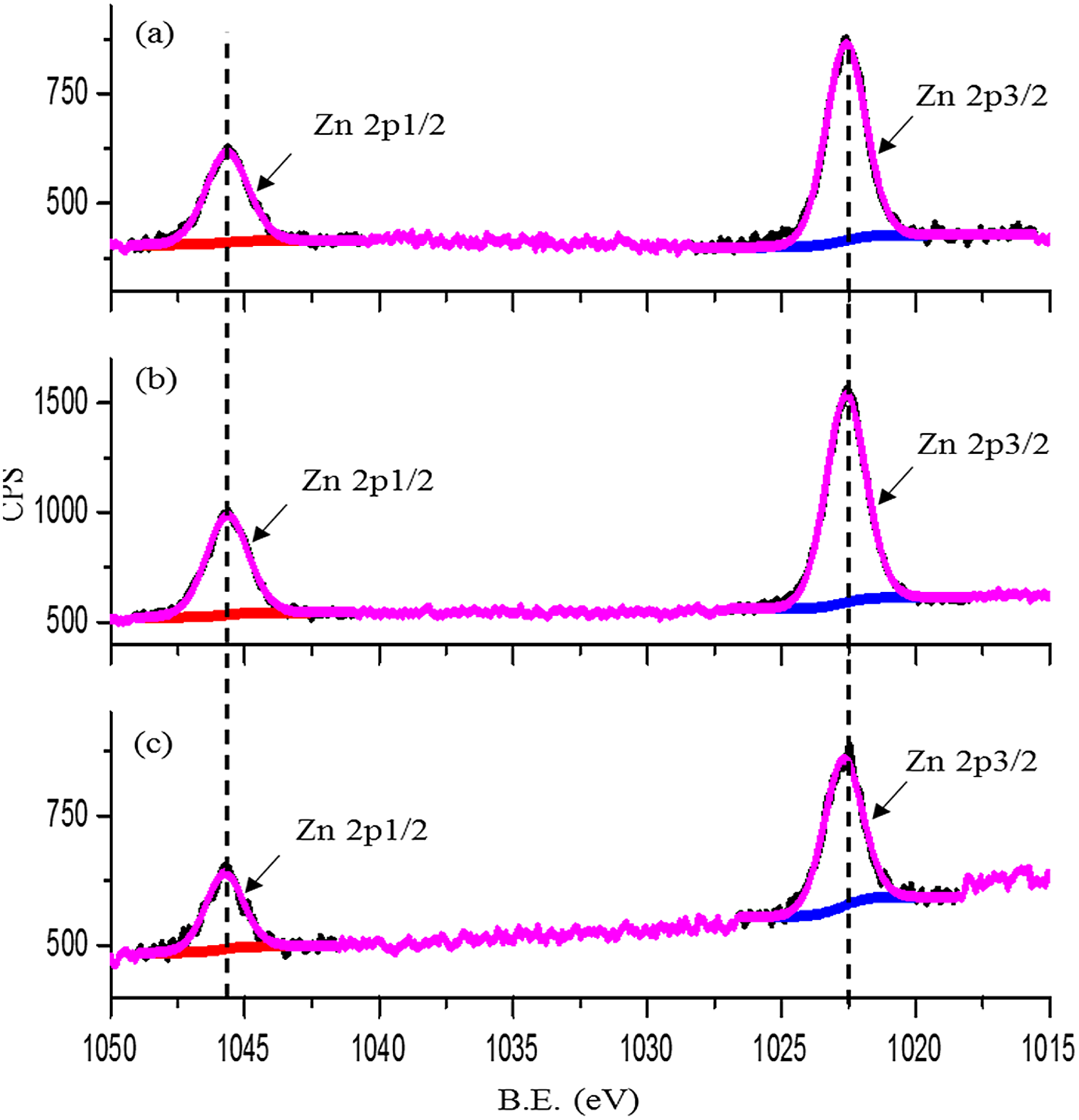
XPS results for Zn 2p from inside wear track for samples lubricated by ZDDP at a contact pressure, temperature and sliding distance of: (a) 700 MPa, 25 °C and 14.4 m; (b) 700 MPa, 80 °C and 14.4 m; and (c) 700 MPa, 25 °C and 36 m. XPS: X-ray photoelectron spectroscopy; ZDDP: zinc dialkyldithiophosphate; CPS: counts per second.
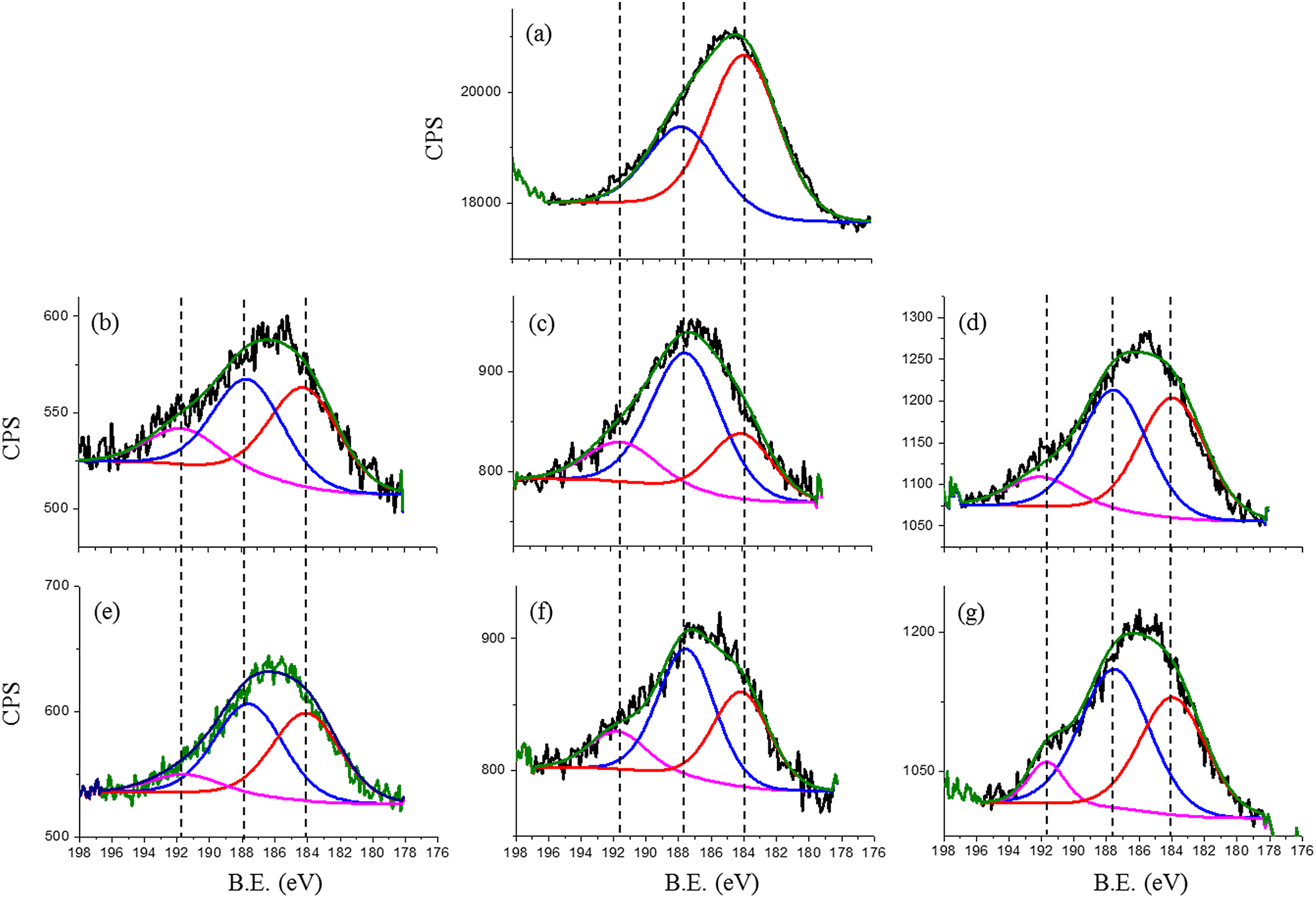
XPS results of P 2s peak for (a) silicon before experiments, the rest are inside wear track that resulted from (b) ZDDP tribofilm at a contact pressure of 700 MPa, 25 °C and a sliding distance of 14.4 m, (c) ZDDP tribofilm at a contact pressure of 700 MPa, 80 °C and a sliding distance of 14.4 m and (d) ZDDP tribofilm at a contact pressure of 700 MPa, 25 °C and a sliding distance of 36 m, (e) PP tribofilm at a contact pressure of 700 MPa, 25 °C and a sliding distance of 14.4 m, (f) PP tribofilm at a contact pressure of 700 MPa, 80 °C and a sliding distance of 14.4 m and (g) PP tribofilm at a contact pressure of 700 MPa, 25 °C and a sliding distance of 36 m. XPS: X-ray photoelectron spectroscopy; ZDDP: zinc dialkyldithiophosphate; PP: phosphonium phosphate; CPS: counts per second.
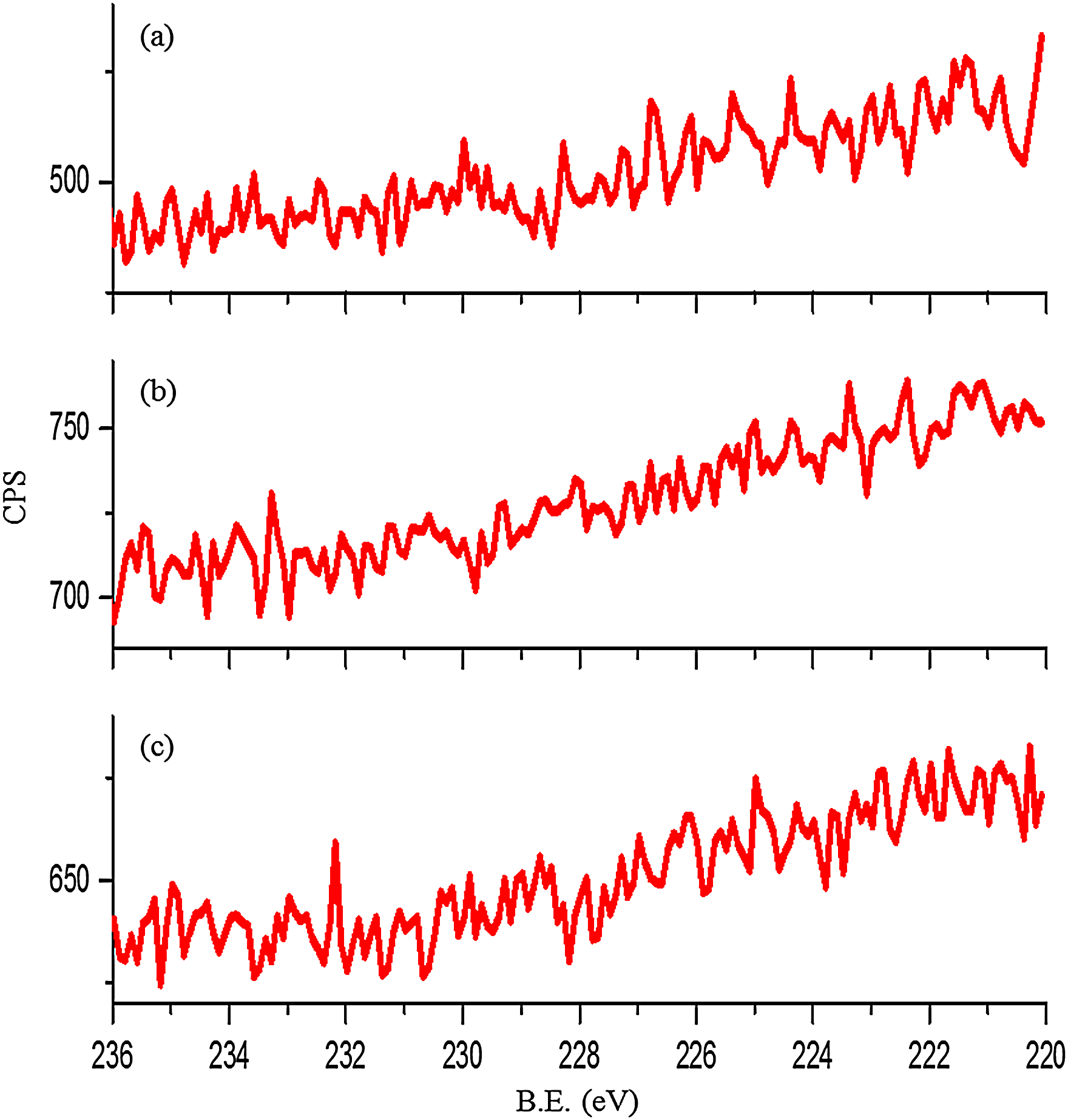
XPS results for S 2s from inside wear track for samples lubricated by ZDDP at: (a) contact pressure 700 MPa, 25 °C and sliding distance 14.4 m; (b) contact pressure 700 MPa, 80 °C and sliding distance 14.4 m; and (c) contact pressure 700 MPa, 25 °C and sliding distance 36 m. XPS: X-ray photoelectron spectroscopy; ZDDP: zinc dialkyldithiophosphate; CPS: counts per second.
The obtained peak of Zn 2p3/2 at 1022.6 eV can be assigned to ZnO. 35 This would suggest that either the formed tribofilm is thin (<8 nm) or phosphorus prefers to react with the silicon surface rather than zinc, which results in a ZnS (or ZnO) layer on the top of a thick SiPO4 layer. The presence of ZnO was also observed in the ZDDP thermal film on the silicon surface. 25 The increase of either the temperature or the sliding distance results in a slight change at the ratio of P 2s/Zn 2p to Si 2s, whereas their binding energies are kept the same. The increase in temperature leads to an increase in the ratio of Zn 2p to Si 2s. In contrast, the increase in the sliding distance shows a decrease in the intensity of the ratio of Zn 2p to Si 2s.
To detect phosphorus, it is recommended to scan P 2s instead of P 2p when the substrate is silicon. The differentiation between the silicon-related peak (comes from the plasmon effect) and the phosphorus peak is easier when P 2s is scanned.30,31
In addition, the presence of phosphorus in n-type silicon needs to be considered. The increase in the ratio of the P 2s peak to the Si 2s peak suggests an increase in the phosphorus content that comes from the formation of the tribofilm. Figure 14 shows the XPS results for P 2s before the experiment and inside the wear tracks. Phosphorus is scanned and the ratio of P 2s to Si 2s is calculated. The ratio of P 2s to Si 2s is about 0.5 before the experiment. The significant increase in this ratio is correlated with the increase in the phosphorus content inside the wear track that comes from the formation of a tribofilm.
The formation of a tribofilm not only increases the presence of phosphorus but also results in a new peak at 191.5 eV, which is assigned to P–O–Si. 30 This indicates the formation of a tribofilm by both additives. In terms of the effect of increasing temperature, Figures 13(b) and 14(c) show that the increase in temperature leads to an increase in the thickness of the tribofilm since the ratio of both Zn 2p and P-related peaks to Si 2s increased (see Table 5). However, the obtained results by XPS suggest that the film chemistry remains the same at both temperatures since the peaks of both Zn and P were obtained at the same binding energies (see Figures 13(a), (b) and 14(b), (c)). Again, the increase of the sliding distance causes a decrease in the tribofilm thickness, as presented in Table 5.
The ratio of P 2s related peaks to Si 2s peak after the increase of either the temperature or the sliding distance.

PP: phosphonium phosphate; ZDDP: zinc dialkyldithiophosphate.
For PP, the results also suggest that the thickness of the formed tribofilm increases with temperature since the ratio of P-related peaks to Si 2s significantly increased (see Table 5). The ratio of P–O–Si to phosphorus also increases, which suggests that the formation of Si–O–P increases with temperature (see Figure 14(e) and (f)). It is also clear that the thickness of the PP tribofilm increases with increasing the sliding distance since the ratio of P 2s to Si 2s peaks increases (see Table 5). In contrast, a slight reduction in the ZDDP tribofilm thickness is suggested by XPS results since the ratio of P 2s related peaks to Si 2s decreases (see Table 5). In addition, the binding energy of the detected peaks is still the same.
Discussion
Friction coefficient
The overall results suggest that the friction coefficient of PP and ZDDP additives is mainly controlled by the formation of a protective tribofilm on the rubbing surfaces. The formation of a tribofilm appears to decrease the friction coefficient (see Figures 3, 4 and 6) and increase its stability (see Figures 2, 5 and 7) in addition to the reduction in the wear coefficient (see Figures 8 to 10). This suggests that the reduction in the friction coefficient results from the reduction in wear debris in the contact. The wear debris can cause an increase in the friction coefficient and a decrease in its stability. 32
Despite the reduction in the friction coefficient after the formation of either PP or ZDDP tribofilm in comparison with a neat PAO, the ZDDP tribofilm shows a higher friction coefficient than PP. The increase in the ZDDP tribofilm thickness (after increasing temperature) increases the ZDDP friction coefficient (see Figure 4 and Table 5). This finding is confirmed when the reduction in the ZDDP tribofilm thickness (after increasing the sliding distance) decreased the ZDDP friction coefficient (see Figure 6 and Table 5). In contrast, the increase in the PP tribofilm thickness decreased the friction coefficient after increasing either the temperature or the sliding distance (see Figures 4 and 6 and Table 5). This is mostly obtained due to the difference in the chemistry between the PP and ZDDP tribofilms. The presence of ZnO in the ZDDP tribofilm might contribute to its increasing friction coefficient.
Wear coefficient and the lubrication mechanism
Wear was controlled by the formation of the tribofilm. The highest wear coefficients at all contact pressures were obtained in PAO due to the absence of a protective layer (tribofilm). The formation of a tribofilm by both additives reduced the wear coefficient (see Figures 8 and 14). This finding is confirmed when the increase in temperature increased the tribofilm thickness of both additives, which in turn decreased their wear coefficients (see Figure 10 and Table 5). Further confirmation is observed after the increase of the sliding distance where the ZDDP tribofilm thickness decreased and thus increased the wear coefficient (see Figure 11 and Table 5).
The higher reduction in the wear coefficient of ZDDP compared to PP additive (except at the longest sliding distance) originates from its thicker tribofilm and chemistry, as stated in the literature.19,24,25 Zhou et al. 24 and Al-Sallami et al. 25 proposed that the formation of a tribofilm or thermal film by PP is limited by the presence of either a direct contact with a cationic surface or wear debris. However, the formation of a thicker film by ZDDP is due to the presence of Zn cation. This makes ZDDP able to form a thicker tribofilm than PP. The chemistry of ZDDP and PP tribofilms is different since Zn just exists in the ZDDP tribofilm. This finding was pointed out earlier by Sharma et al. 36 This could contribute to the superior anti-wear performance of ZDDP under most conditions in this study.
The most probable scenario that results a better anti-wear performance by PP in this study and in Qu et al. 19 is that the higher formation rate of the ZDDP tribofilm causes a faster depletion of ZDDP from the bulk lubricant, i.e. PAO + ZDDP. The superior anti-wear performance of PP after about 40 m of sliding (at 100 °C) of PAO + ZDDP in Qu et al. 19 cannot be correlated with the effect of viscosity since both oils had very comparable viscosities. In addition, ZDDP shows a superior anti-wear performance after the increase of temperature to 80 °C at a lower sliding distance (14.4 m).
Conclusions
An experimental investigation has been conducted to compare the tribological performance and the lubrication mechanism of PP and ZDDP when added to PAO to lubricate silicon substrate under various conditions including various contact pressures, temperatures and sliding distances. The main conclusions are as follows:
The friction coefficient of a neat PAO is mainly controlled by surface roughness, whereas the friction coefficient of PAO + PP and PAO + ZDDP is mainly controlled by the formation of a tribofilm on the rubbing surfaces. Both additives provide a considerable reduction in the friction coefficient and exhibit a comparable friction coefficient. Both additives provide a substantial reduction in the wear coefficient and ZDDP exhibits a lower wear coefficient than PP under all conditions except at the highest sliding distance. The formation of a tribofilm controls the wear coefficient of both additives and the increase in its thickness decreases the wear coefficient. The better anti-wear performance of ZDDP results from its thicker tribofilm since the formation of the PP tribofilm is self-limited. The depletion of ZDDP from the bulk oil is faster due to not only its higher reactivity but also due to the self-limited tribofilm and thermal film formation of PP. This results in better anti-wear performance by PP at the highest sliding distance.
Footnotes
Acknowledgements
The authors would like to thank Dr Filippo Mangolini for his help in this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This project was funded by the University of Leeds, LARS fellowship.
