Abstract
Background:
The use of shared decision-making (SDM) in atopic dermatitis (AD) management guidelines is limited.
Objective:
To develop a tool for implementing the Aiming High in Eczema and Atopic Dermatitis (AHEAD) SDM approach in AD clinical practice.
Methods:
The international group of 7 expert dermatologists responsible for leading the development of the AHEAD approach initially drafted the tool. A global patient research study and an advisory board that included the leaders of 10 patient advocacy groups captured global patient and nonclinician considerations.
Results:
The AD SDM tool provides a simple, practical tool for implementing SDM using the AHEAD approach in clinical practice. The tool enables patients to identify and rank their most troublesome AD symptoms, and clinicians to tailor treatment targets to address them. The tool will enable patients to better communicate their disease burden, increase treatment adherence, and improve continuity of care.
Conclusions:
The AD SDM tool provides a simple, practical tool for implementing SDM using the AHEAD approach. The consolidation of routine outcome measures should streamline the collection and interpretation of clinically relevant patient information. Feedback from patients and clinicians who use the tool will allow for the assessment of its value in clinical practice.
Keywords
Introduction
Atopic dermatitis (AD) is a common, chronic, systemic, relapsing inflammatory skin disease, which can lead to significant impacts on physical, mental, and social functioning.1 -3
The complex and variable nature of AD requires identification of individualized, patient-relevant treatment targets to ensure that patients’ treatment is tailored to their individual needs. 4 The 2021 treat-to-target initiative in AD recognized this need and developed a multidimensional approach to AD management that included both patient-reported outcome (PRO) and clinician-reported outcome (CRO) measures to assess both physical and nonphysical AD symptoms. 5 In this approach, clinicians decide which outcome measures are used to assess disease severity and assign treatment targets. Therefore, clinicians may underestimate disease severity by selecting an outcome measure that focuses primarily on cutaneous signs of AD (eg, Eczema Area and Severity Index [EASI]) and thus fails to capture a “complete picture” of their patient’s disease burden. 5 Furthermore, evidence suggests that the assigned treatment targets may be insufficient to achieve optimal patient outcomes. 6 These limitations, in conjunction with a global patient perception of feeling unheard during consultations and having their disease burden underestimated,7,8 highlight the need for a decision-making approach that incorporates higher treatment targets, helps patients to more effectively communicate their disease burden, and actively involves patients in decisions regarding their AD management.
In 2021, an international group of expert dermatologists recognized these gaps and began developing the Aiming High in Eczema and Atopic Dermatitis (AHEAD) recommendations. 9 These international, expert, consensus-based recommendations introduce a novel concept for minimal disease activity (MDA) criteria and emphasize the importance of a new, shared decision-making (SDM) approach to AD management. 9
In the “AHEAD approach,” patients are asked to identify which 1 to 3 AD symptoms are most important for them to resolve, and corresponding PRO measures are selected to tailor disease-severity assessments and treatment targets to their individual needs. 9 The clinician also chooses at least 1 CRO measure to provide an objective assessment of the patient’s overall disease. Finally, the clinician and patient discuss the chosen CROs and PROs and engage in SDM to select “moderate” or “optimal” treatment targets. Clinicians and patients/caregivers are encouraged to aim for optimal treatment targets, with the achievement of optimal targets defined as MDA.
SDM approaches to disease management are widely accepted as a core feature of providing good healthcare and improving patient outcomes.10,11 Indeed, in other therapy areas, such as asthma and Crohn’s disease, SDM has improved patients’ knowledge of treatment options, treatment adherence, patient–clinician communication, and patient satisfaction.10 -14 It has also been associated with reduced healthcare resource use and costs, and may reduce clinician burnout.10 -14 However, while some AD management guidelines explicitly recommend SDM,15 -18 others do not explicitly mention SDM in their decision-making algorithms.19 -21
While the incorporation of SDM into routine clinical practice offers clear benefits for patients, clinicians’ satisfaction with decision aids that support SDM remains a crucial factor in successful implementation.10,11,22 Resistance to adopting decision aids often arises from time constraints, limited familiarity, and uncertainty about their practical value or relevance to specific clinical situations.11,23 -25 The authors therefore recognized the need for a tool centered on feasibility to support the implementation of the AHEAD approach in clinical practice.
Here, we present the AD SDM tool, which aims to provide clinicians and patients with a simple and practical tool for implementing the AHEAD approach in clinical practice. This tool is for use with adult patients (≥18 years) with AD of all severities.
Materials and Methods
Development of the Initial Tool
The AD SDM tool was developed following a global, ethnically diverse, qualitative patient research study on the disease burden of adults with AD, and the AHEAD recommendations, which were published separately.8,9 An overview of the development of the AD SDM tool can be found in Figure 1.8,9
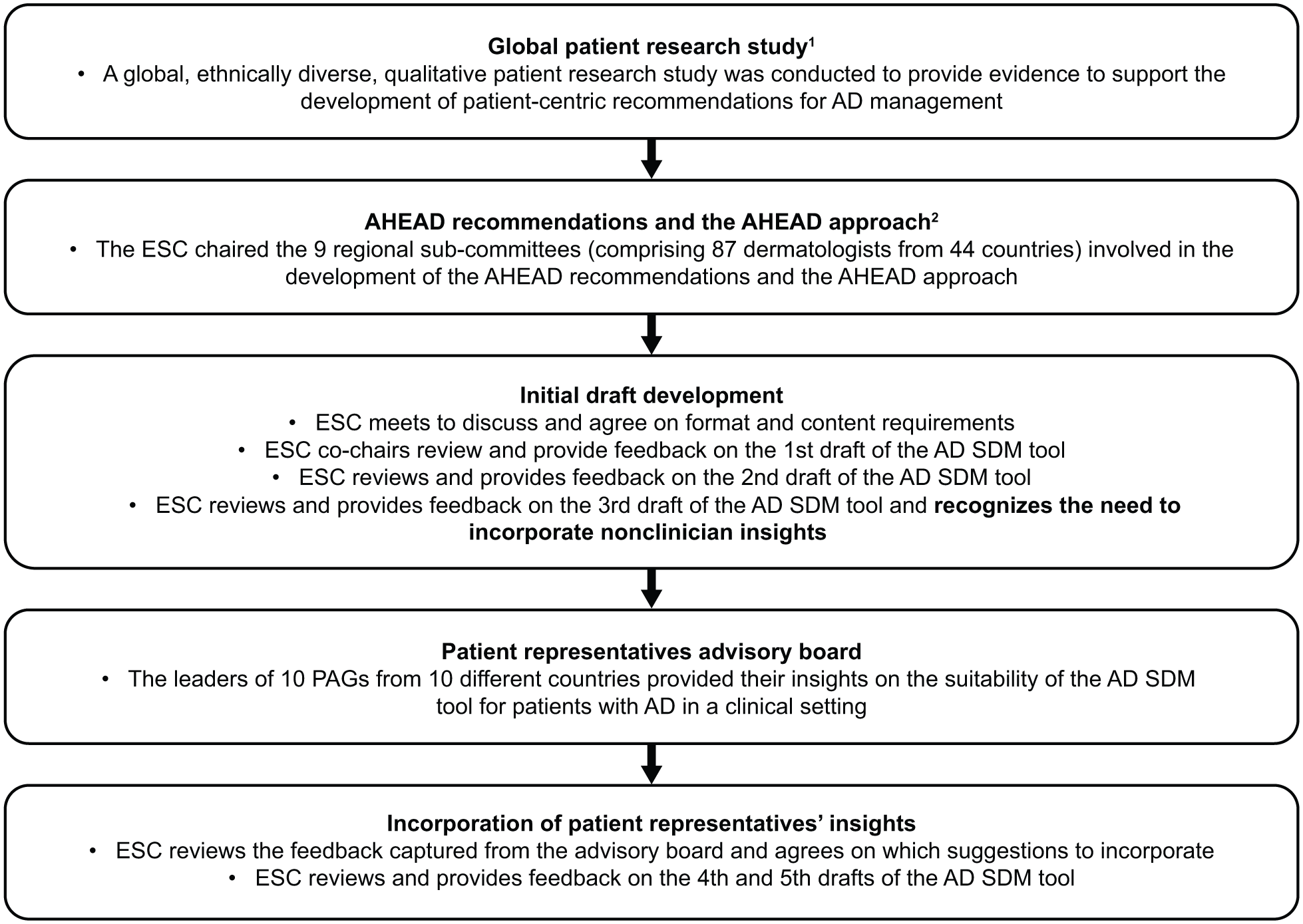
Overview of the development of the AD SDM tool. AD, atopic dermatitis; AHEAD, Aiming High in Eczema and Atopic Dermatitis; ESC, Executive Steering Committee; PAG, patient advocacy group; SDM, shared decision-making.
An international group of 7 expert dermatologists, comprising the Executive Steering Committee (ESC) responsible for leading the 9 regional subcommittees (including 87 dermatologists from 44 countries) involved in the development of the AHEAD recommendations and the AHEAD approach, contributed to the development of the AD SDM tool (Supplemental Table S1). 9
The ESC first met to agree on the format and content requirements of the AD SDM tool, and an initial draft was developed based on these requirements. The ESC co-chairs reviewed the initial draft, provided their feedback, and worked to develop an updated version of the tool. This updated version was sent to all members of the ESC for review, and the experts’ comments were incorporated to produce the initial AD SDM tool.
To ensure the tool was valuable and feasible for use by patients, the ESC recognized the need to capture nonclinician insights. Therefore, the ESC agreed to conduct an advisory board to capture a diverse range of nonclinician perspectives from the leaders of patient advocacy groups, herein referred to as “patient representatives.”
Refinement of the Tool According to Patient Representatives’ Feedback
In total, an international group of 10 patient representatives representing 10 countries (Austria, Brazil, Canada, France, Germany, Netherlands, South Korea, Spain, Switzerland, and Turkey) participated in the advisory board. This included 3 workshops where patient representatives were able to provide their insights on the suitability of the AD SDM tool for patients with AD in a clinical setting, and the potential benefits and challenges to its value and implementation in clinical practice (Supplemental Table S2).
Following the advisory board, the ESC reviewed the patient representatives’ feedback, and their recommendations were incorporated if the ESC deemed them feasible in clinical practice. An overview of the patient representatives’ feedback can be found in Supplemental Table S3.
Results
A copy of the AD SDM tool can be found in the Supplemental Appendix. In total, 7 international dermatology experts from 7 countries combined their local clinical insights with nonclinician perspectives (generated from feedback provided by the 10 patient representatives from 10 countries who participated in the patient representatives’ advisory board, and the perspectives captured from 88 patients from 15 countries who participated in the global patient research study) to develop the AD SDM tool (Supplemental Tables S1 and S2). 8
An overview of how the AD SDM tool should be used to implement the AHEAD approach in clinical practice is provided in Figure 2. 9 The AD SDM tool was designed to be completed within 5 to 10 minutes and aims to provide clinicians and patients with a simple, practical, and standardized approach for implementing the AHEAD approach in clinical practice. This tool will therefore enable future studies to assess the feasibility and value of the AHEAD approach in clinical practice.
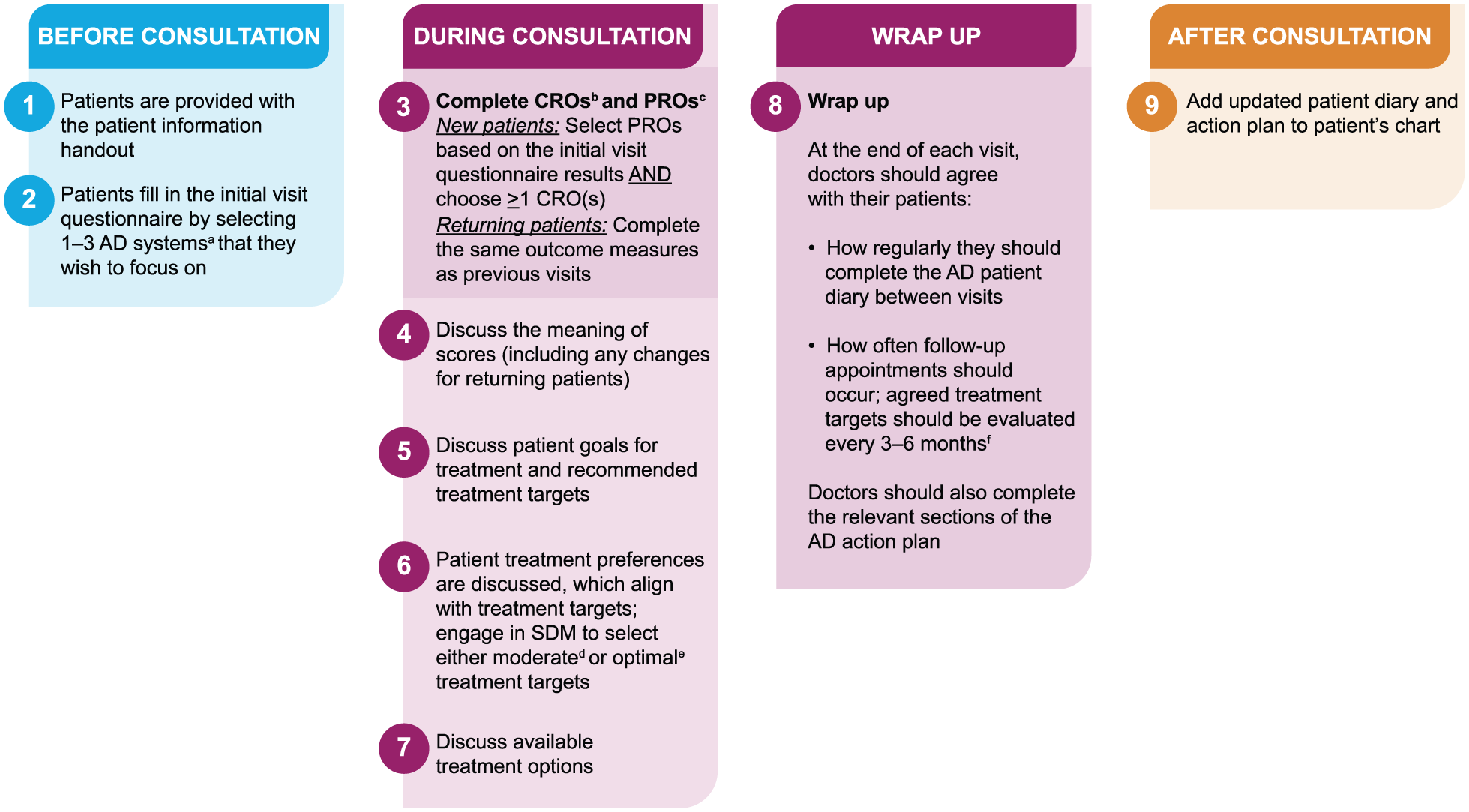
Overview of how the AD SDM tool should be used to implement the AHEAD approach in clinical practice. AD, atopic dermatitis; AHEAD, Aiming High in Eczema and Atopic Dermatitis; BSA, body surface area; CRO, clinician-reported outcome; DLQI, Dermatology Life Quality Index; EASI, Eczema Area and Severity Index; EASI-75/90, 75%/90% improvement in EASI; HADS, Hospital Anxiety and Depression Scale; HADS-A, HADS-anxiety; HADS-D, HADS-depression; IGA, Investigator’s Global Assessment; MDA, minimal disease activity; NRS, Numeric Rating Scale; POEM, patient-oriented eczema measure; PRO, patient-reported outcome; SCORAD, SCORing Atopic Dermatitis; SCORAD-50/75, 50%/75% improvement in SCORAD; SDM, shared decision-making.
Patient Materials
The patient materials comprise 4 sections: the patient information handout, the initial visit questionnaire, the patient appointment resources, and an optional AD patient diary.
The patient information handout consists of 2 pages and informs the patient what to expect during their consultation. Page 1 includes a flowchart explaining the 4-step process that the patient will experience. Page 2 provides the patient with a checklist detailing what they should bring to appointments and some questions they may wish to consider asking their clinician. On review of the initial draft, the patient representatives perceived the language used in the flowchart to be too complex and clinician-centric, stating that it did not serve as a useful guide for patients in its current form. The experts therefore revised the language used throughout to ensure that it was patient-friendly. For example, “Based on the results from questionnaires and discussions, your doctor will work with you to set a treatment goal” was revised to “You and your doctor will work together to set a treatment goal” to simplify the language, reduce word count, and emphasize that the AHEAD approach is a patient-centric, collaborative approach to AD management.
The initial patient visit questionnaire asks patients to select their 1 to 3 most troublesome AD symptoms that they would like their treatment plan to focus on. There are 7 domains for the patient to choose from, with 6 of these domains corresponding to a different PRO used to assess the patient’s disease severity and assign treatment targets. 8 The seventh domain, “other,” does not correspond to a PRO used in the AHEAD approach. The authors agreed to include this domain following feedback from the patient representatives, who highlighted that patients should be able to communicate issues beyond the preexisting 6 domains (eg, difficulties with their treatments or side effects). The patient representatives agreed with asking patients to select their most troublesome AD symptoms, but noted that ranking them may be challenging for individuals who perceive all symptoms as equally important. They also advised that the tool should not restrict patients to selecting only 3 AD symptoms because all 6 may be considered equally important. While the ESC acknowledged that all AD symptoms may hold equal importance, they expressed concern that requiring clinicians to complete 6 outcome measures within the limited time available during consultations would be unfeasible. The final version therefore requires patients to select up to 3 symptoms rather than rank them.
Furthermore, the patient representatives perceived the initial visit questionnaire to be clinician-centric, stating that it focused primarily on treatment goals and not the patient’s disease burden. Therefore, further revisions included modifying the language to ensure that patients feel that clinicians understand and appreciate their disease burden. For example, the term “AD features” was revised to “AD symptoms” to remove the implication that the 6 selectable domains are a benefit of AD, and an introductory statement was added to convey that clinicians understand that limiting patients to choosing 3 AD symptoms may be difficult if all symptoms are equally troublesome. Some patient representatives expressed concern that patients may be confused about why they were being asked to select 1 to 3 AD symptoms, noting that some patients may perceive this as a “test.” To address these concerns, the experts revised the questionnaire to include a simplified explanation of how patients’ responses will be used by clinicians to assess disease severity and develop a treatment plan.
The patient appointment resources span 2 pages and were designed to help patients monitor their treatment progress. Patients are asked to record their treatment targets and upcoming appointment dates and are provided with space to record the scores of PROs between appointments. Patients are also instructed to record the development of new symptoms and advised to take photographs to document changes to their skin. Finally, patients are reminded to bring completed questionnaires, photographs, and an optional AD patient diary to their next appointment.
The optional AD patient diary is designed to help patients monitor their treatment progress by enabling them to record the presence and severity of their symptoms weekly, between appointments. Scores from 0 (no impact on daily life) to 10 (severe impact on daily life) can be recorded for each of the 6 AD symptoms assessed in the AHEAD approach. These scores can serve as reference points, helping patients to communicate their disease burden. On review of the AD patient diary, patient representatives stated that the tool should clarify whether its completion is a prerequisite for the clinician to develop a treatment plan, citing the low usage of disease-tracking methods among patients. 26 The experts agreed and thus revised the name to “optional AD patient diary” to ensure that patients did not feel forced to complete it.
Patient Representatives’ Insights on the Benefits and Challenges to Implementing the AD SDM Tool in Clinical Practice
A summary of the patient representatives’ feedback on the patient-facing materials in the AD SDM tool can be found in Supplemental Table S3.
In summary, patient representatives reported that they believe the AD SDM tool will lead to improved clinician–patient communication, patient knowledge of AD, treatment options, and outcomes. In addition, patient representatives stated that the tool’s consideration of patient preferences may lead to improved treatment adherence. Some patient representatives believed that the incorporation of an AD action plan and optional AD patient diary will improve the continuity of care for patients who see different clinicians at each appointment. However, patient representatives recognized that the limited time available during appointments, and difficulties implementing a culture of SDM among clinicians, were major challenges to implementing the AD SDM tool in clinical practice.
Clinician Materials
The clinician materials comprise 3 sections: the implementation guide, the AD action plan, and supplementary materials.
Clinicians are provided with an implementation guide, which contains an outline of the process to follow before, during, and after a consultation. The AD action plan (designed to be used in conjunction with the implementation guide) consists of 2 pages and is intended to help clinicians implement the AHEAD approach, monitor their patient’s treatment progress, and outline how treatment targets should be selected. The first page of the AD action plan contains 2 tables: the first table details which PROs should be used to evaluate patients’ disease severity (each PRO corresponds to an AD feature selected by the patient in their initial visit questionnaire), and the second table details cutoff scores for the “moderate” and “optimal” treatment targets for each CRO and PRO used in the AHEAD approach. The second page contains a treatment target tracker, enabling clinicians to record patients’ agreed treatment targets, scores generated from PROs and CROs, and the dates they were completed.
To ensure familiarization with the AHEAD approach, clinicians are provided with the AD SDM tool supplementary materials. These materials contain descriptions of each CRO and PRO, how each score is calculated, and what each score means. Furthermore, they contain background information on the development of the AD SDM tool, the AHEAD approach, and the global patient research study.8,9
Discussion
Insights from an international group of dermatology experts, combined with global nonclinician and patient perspectives, led to the development of the AD SDM tool, which aims to provide clinicians and patients with a simple, practical tool for implementing the AHEAD approach in clinical practice.
Providing clinicians and patients with a simple tool to implement the AHEAD approach should help increase the uptake of this novel approach to AD management, and therefore help improve the current standard of care through higher treatment targets and increased patient involvement. 9 Tailoring PROs to patients’ most troublesome symptoms, and enabling them to choose between “moderate” and “optimal” treatment targets (some patients may have limited access to, or reject the advanced treatment options that may help them meet “optimal” targets), engages patients in SDM and will help to improve patient knowledge of treatment options, treatment adherence, patient–clinician communication, and patient satisfaction, and reduce healthcare resource use, healthcare costs, and clinician burnout.10 -14 Furthermore, encouraging patients to aim for “optimal” targets wherever possible, and recommending treatment modification or escalation if treatment targets are not met within the agreed timeframe, will improve patient outcomes by ensuring that patients are receiving the treatments they need to achieve “optimal targets” (and thus achieve MDA). 9
A key strength of the AD SDM tool is its inclusion of international clinician perspectives, its consideration of the universal challenges that exist in AD management, and its focus on feasibility of use across a diverse range of clinical practice settings. The tool was therefore not designed to serve as a diagnostic instrument or to dictate specific treatments because different countries follow varying AD management guidelines to inform diagnosis and treatment decisions. Rather, it aims to empower patients by facilitating a patient-centered SDM approach, supporting collaboration between clinicians and patients in setting treatment goals and discussing management options. During the initial ESC meetings, it was agreed that time constraints during appointments, combined with the perception among clinicians that the AHEAD approach would require “more work,” would pose key barriers to implementation of the AHEAD recommendations in clinical practice. Therefore, to minimize the time required to implement the AHEAD approach, the tool was made to be as concise as possible. Furthermore, the 6 domains that patients choose from when selecting their 1 to 3 most troublesome AD symptoms each correspond to outcome measures that should be routinely assessed in clinical practice. Thus, the consolidation of these outcome measures into a single tool should streamline the collection of clinically relevant information. Further efforts to minimize the AD SDM tool’s impact on appointment times include the patient information handout and the initial patient questionnaire. These preappointment materials should ensure that patients are prepared for their initial appointment by knowing what to expect, what to bring, and which 1 to 3 AD symptoms they wish to focus on. The tool was designed for completion within 5 to 10 minutes; however, completion of the preappointment materials prior to the patient’s initial appointment should reduce this time by minimizing the time clinicians spend introducing the AHEAD approach, leaving more time available to discuss treatment options, answer questions, and develop a treatment plan. The ESC’s focus on maximizing the feasibility of the tool should increase the adoption of the AHEAD approach and therefore increase the number of patients who will benefit from higher, individualized treatment targets and increased patient involvement compared with the current standard of care. 9
While feasibility of the AD SDM tool with clinicians was important, the ESC recognized the need to capture nonclinician insights to ensure the tool’s suitability for patients. The incorporation of insights from an international group of patient representatives, and perspectives generated from a global, ethnically diverse, qualitative patient research study, ensures that the AD SDM tool considers a diverse range of patient needs, further helping to improve its uptake.
Although SDM has improved treatment adherence and clinical outcomes in other therapy areas, its use within AD management is limited.15 -27 While the limited use of SDM in AD management provided the experts with the opportunity to develop a new tool, there are limitations. The benefits of using the tool have yet to be formally evaluated, providing an opportunity for future research. Furthermore, the tool is currently only available in English and is limited to use with adult patients (≥18 years). However, there are plans to translate and localize the tool for use in additional languages and cultural contexts, with international uptake offering opportunities to gather feedback and make further refinements. In addition, there may be barriers to its adoption in clinical practice, particularly among clinicians who are less familiar with the outcome measures used in the tool, or who have limited time in which to complete them during routine consultations. Indeed, limited appointment times have been shown to represent a major barrier to the incorporation of SDM into routine clinical practice, and may therefore limit the feasibility of the tool, particularly in patients with mild AD, where clinicians may not see the value of the perceived extra work. 27 However, evidence suggests that integrating SDM does not necessarily prolong consultations when supported by a multifaceted implementation strategy, adapted workflows, and targeted clinician training. 28 Moreover, patient decision aids, such as the preappointment materials and AD diary, may further streamline consultations because patients may arrive better informed and better prepared for their consulations. 28
While the AD SDM tool enables patients to communicate a broad range of AD symptoms they may be experiencing and provides clinicians with corresponding PROs to assess disease severity and guide individualized treatment, significant gaps remain. For example, standard CRO measures, such as EASI and SCORing Atopic Dermatitis, have demonstrated limited reliability and validity in patients with darker skin tones. 29 Newer instruments, including the PDCA-Derm, are being developed to better assess postinflammatory hyper- and hypopigmentation; however, additional studies are needed to establish their validity. 30 To ensure the tool remains relevant and up to date with emerging scientific findings, future translations and localization efforts should look to incorporate recently validated outcome measures where appropriate.
Due to the need to capture in-depth feedback from patients who are highly educated regarding AD management in their respective countries, the ESC decided that patient representatives, not patients, should review the tool. Furthermore, despite the patient representatives’ in-depth knowledge of AD management, some of their recommendations were omitted from the final version of the tool. For example, patient representatives stated that patients should be able to select all 6 AD symptoms in the initial visit questionnaire as symptoms that they wish to focus on, adding that all 6 AD symptoms may be of equal importance to patients with severe AD. Although the ESC agreed with this suggestion in principle, they were concerned that requiring clinicians to assess and discuss more than 3 PROs during an appointment may limit the feasibility of the tool and thus reduce its uptake in clinical practice. Therefore, the experts agreed to limit patients to choosing 1 to 3 AD symptoms, as per their initial consensus-based recommendations. 9
Despite efforts to capture global expert, patient, and patient-representative perspectives, we, the authors, believe that there would be great value in assessing the feasibility and value of the tool in a real-world, clinical setting. Previous SDM tools have often overlooked the importance of generating awareness and interest, leading to limited uptake or adoption failure despite otherwise robust design. 31 We believe that future efforts to translate and localize the tool across additional languages and healthcare contexts will enhance its accessibility and encourage broader use. This, in turn, will create opportunities for patients, clinicians, and other allied healthcare professionals, such as nurses and pharmacists, to provide valuable feedback, supporting further refinement and improvement.
Supplemental Material
sj-docx-1-cms-10.1177_12034754261427403 – Supplemental material for The Atopic Dermatitis Shared Decision-Making Tool: Implementing the AHEAD Approach in Clinical Practice
Supplemental material, sj-docx-1-cms-10.1177_12034754261427403 for The Atopic Dermatitis Shared Decision-Making Tool: Implementing the AHEAD Approach in Clinical Practice by Andreas Wollenberg, Melinda Gooderham, Norito Katoh, Valeria Aoki, Andrew E. Pink, Yousef Binamer and Jonathan I. Silverberg in Journal of Cutaneous Medicine and Surgery
Footnotes
Acknowledgements
This manuscript was developed following an AbbVie-sponsored program that included a literature review on AD, a global patient research study, expert discussion meetings, and eDelphi voting. However, the program adhered to complete objectivity with no active involvement from AbbVie, and AbbVie was not involved in the development of the publication. AbbVie selected the patient representatives and reviewed the publication for scientific accuracy; however, decisions regarding the final publication content were made solely by the authors. All authors made substantial contributions to the publication or critically revised it for important intellectual content and approved the final version. AbbVie provided funding to invited participants, including honoraria for their attendance at the meetings. No payments were made to the authors for the development of this publication. Samuel Devereux, MPharm, of 2 the Nth (Cheshire, UK), provided medical writing and editorial support to the authors in the development of this publication, and financial support for these services was provided by AbbVie.
Author Contributions
A.W. and J.I.S. conceived of and developed the initial AD SDM tool. All authors contributed to, critically revised, and approved the final AD SDM tool.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.W. is a consultant for AbbVie, Aileens, Almirall, Beiersdorf, Eli Lilly, Galderma, GlaxoSmithKline, Janssen, LEO Pharma, Loreal, MedImmune, MSD, Novartis, Pfizer, Pierre Fabre, Regeneron Pharmaceuticals, Inc, and Sanofi; an investigator for AbbVie, Almirall, Anacor Pharmaceuticals, Eli Lilly, Galapagos, Galderma, Glenmark, LEO Pharma, MedImmune, Novartis, Pfizer, Regeneron Pharmaceuticals, Inc, Sanofi, and UCB; and received research grants from Pierre Fabre. M.G. is an investigator, speaker, and/or advisor for AbbVie, Akros, Amgen, AnaptysBio, Arcutis, Aristea, Aslan, Bausch Health, BMS, Boehringer Ingelheim, Celgene, Dermavant, Dermira, Eli Lilly, Galderma, GlaxoSmithKline, Incyte, Janssen, Kyowa Kirin, LEO Pharma, MedImmune, Meiji, Merck, Moonlake, Nimbus, Novartis, Pfizer, Regeneron Pharmaceuticals, Inc, Roche, Sanofi-Genzyme, Sun Pharma, UCB, and Ventyx. N.K. has received honoraria as a speaker/consultant for AbbVie, Celgene Japan, Eli Lilly Japan, Janssen Pharma, Kyowa Kirin, LEO Pharma, Maruho, Mitsubishi Tanabe Pharma, Otsuka Pharmaceutical, Pfizer, Sanofi, Taiho Pharmaceutical, and Torii Pharmaceutical; and received grants as an investigator from A2 Healthcare, AbbVie, Boehringer Ingelheim Japan, Eisai, Eli Lilly Japan, Janssen Pharma, Kyowa Kirin, LEO Pharma, Maruho, Sun Pharma, Taiho Pharmaceutical, and Torii Pharmaceutical. V.A. is an investigator for Eli Lilly and Sanofi (clinical trials); and a consultant and/or speaker for AbbVie and Pfizer. A.E.P. is an advisory board member of, investigator for, recipient of grants from, or recipient of educational support from AbbVie, Almirall, Amgen, BMS, Boehringer Ingelheim, Celgene, Eli Lilly, Galderma, Janssen, LEO Pharma, Novartis, Pfizer, Sanofi-Genzyme, and UCB. Y.B. has received speaker honoraria for serving as a consultant for and travel support from AbbVie, Eli Lilly, Janssen, Kyowa Kirin, NewBridge, Novartis, and Sanofi; and received research grants from Novartis and Sanofi. J.I.S. is an investigator for AbbVie, BMS, Eli Lilly, GlaxoSmithKline, Incyte, Kiniksa Pharmaceuticals, LEO Pharma, Realm Therapeutics, and Regeneron Pharmaceuticals, Inc; a consultant for AbbVie, Alamar, Amgen, AObiome, Arcutis, Arena, Asana, Aslan, BioMX, Biosion, Bodewell, Boehringer Ingelheim, Cara, Castle Biosciences, Celgene, Connect Biopharma, Dermavant, Dermira, Dermtech, Eli Lilly, Galderma, GlaxoSmithKline, Incyte, Kiniksa Pharmaceuticals, LEO Pharma, Menlo, Novartis, Optum, Pfizer, RAPT, Regeneron Pharmaceuticals, Inc, Sanofi-Genzyme, Shaperon, and Union; and a speaker for AbbVie, Eli Lilly, LEO Pharma, Pfizer, Regeneron Pharmaceuticals, Inc, and Sanofi-Genzyme.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AbbVie funded the study and reviewed the publication for accuracy prior to submission. However, decisions regarding the publication content were made solely by the authors. No payments were made to the authors for the development of this publication.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
