Abstract
L-Carnitine, an amino acid derivative critical for fatty acid metabolism, exhibits anti-inflammatory, antioxidant, and cytoprotective properties, with emerging applications in dermatology. This review explores its therapeutic potential, both topically and systemically, and its role as a biomarker in various skin disorders. A systematic search of Medline, EMBASE, and PubMed (1946-January 2025) was conducted, focusing on L-carnitine and dermatologic conditions, excluding acetyltransferases, acyltransferases, breast, and genetic metabolic diseases. From 420 articles, 59 were included after screening in Covidence. Data on study design, sample size, carnitine type, and outcomes were extracted and validated by two authors. Topically, L-carnitine (1%-5%) reduces acne lesions, sebum production, post-inflammatory hyperpigmentation, and cellulite, particularly when combined with agents like licochalcone-A or salicylic acid. Systemic L-carnitine mitigates inflammation, oxidative stress, and treatment-related side effects (eg, from corticosteroids, isotretinoin, vismodegib) while improving skin elasticity, microcirculation, wound healing, and fibrosis in conditions like Raynaud’s disease, venous leg ulcers, and sclerotic disorders. As a biomarker, altered carnitine levels and metabolites reflect disease mechanisms in genodermatoses (eg, restrictive dermopathy, recessive dystrophic epidermolysis bullosa), inflammatory dermatoses, and melanoma, aiding diagnosis and monitoring. L-Carnitine shows promise as a therapeutic and diagnostic tool in dermatology, with benefits in acne, sebum control, post-inflammatory hyperpigmentation, cellulite, inflammation, and tissue repair. Its biomarker potential enhances disease insights and personalized care. Further dermatology-specific trials are needed to optimize dosing, formulations, and long-term safety, particularly regarding trimethylamine-N-oxide-related cardiovascular risks.
Keywords
Introduction
Carnitine, an amino acid derivative synthesized from L-methionine and L-lysine, exists primarily in its active form, L-carnitine, along with several other bioactive derivatives including propionyl-L-carnitine (PLC) and acetyl-L-carnitine (ALC). 1 L-carnitine is crucial for transporting long-chain fatty acids into mitochondria for β-oxidation and energy production. 2 Acylcarnitines, formed through conjugation of L-carnitine and fatty acids, contribute to the cellular carnitine pool and support mitochondrial function. 1
Beyond its metabolic function, L-carnitine exhibits anti-inflammatory, antioxidant, and cytoprotective effects, with emerging relevance in cardiovascular diseases, cancer and neuroprotection. 3 ALC, in particular, enhances mitochondrial efficiency, supports cholinergic transmission and protects neural tissue. 4
This review examines carnitine's growing relevance in dermatology, both as a therapeutic agent, used topically and systemically, and as a potential biomarker in various skin disorders and conditions (Table 1).
Clinical Applications and Biomarker Relevance of Carnitine in Dermatology.
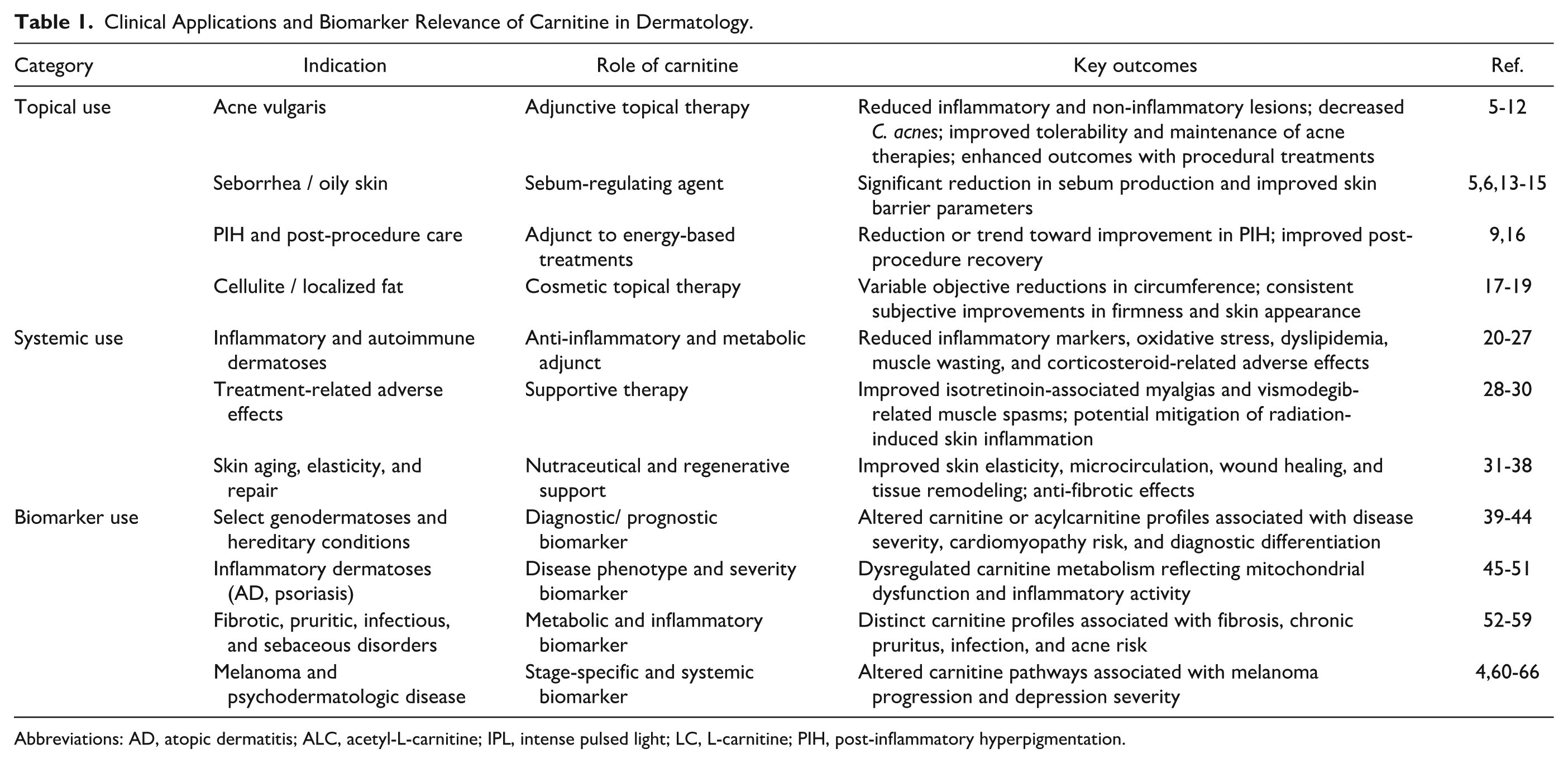
Abbreviations: AD, atopic dermatitis; ALC, acetyl-L-carnitine; IPL, intense pulsed light; LC, L-carnitine; PIH, post-inflammatory hyperpigmentation.
Methods
The following bibliographic databases were searched individually on January 11, 2025: Medline (1946 to January 10, 2025), and EMBASE (1974 to 2025 January 09) via Ovid; PubMed (1946-2025) directly through the US National Library of Medicine. Systematic searches were completed using a combination of subject headings, such as Medical Subject Headings (MeSH), wherever they were available in the database and keywords. The search strategy consists of two main concepts: (1) carnitine and (2) dermatology, including skin diseases. Articles focused on the acetyltransferases or acyltransferases were excluded, and so were articles discussing breast diseases and metabolic diseases. No limits for publication date were used, but the search was restricted to the English language, those pertaining to human studies, and those with abstracts. The titles and abstracts were first screened for relevance in Covidence, and then the full-text was screened to ensure eligibility. Data was extracted from the papers at the full-text assessment. A total of 420 articles were found and after screening and reviewing the articles, 59 were included for data extraction. Two authors (EC, MS) independently evaluated the validity of the qualifying studies by evaluating their titles, abstracts and findings. This review excluded animal studies, books, conference abstracts, non-systematic reviews, and grey literature. The following information was extracted and checked by the two authors: first author’s name, nation, year of publication, type of study, study sample size, specific carnitine molecule, and other data were retrieved for further analysis.
Topical Use of L-Carnitine in Dermatology
Numerous studies evaluated the topical use of L-carnitine in dermatology, focusing on acne, sebum reduction, post-inflammatory hyperpigmentation (PIH) and cellulite management. The researchers employed various L-carnitine concentrations (1-5%), often in combination with other active ingredients. Most were randomized controlled trials (RCT), except for one multicenter observational study. 5 Table 2 summarizes the studies reviewed.
Summary of Studies with Carnitine as Topical Therapy.
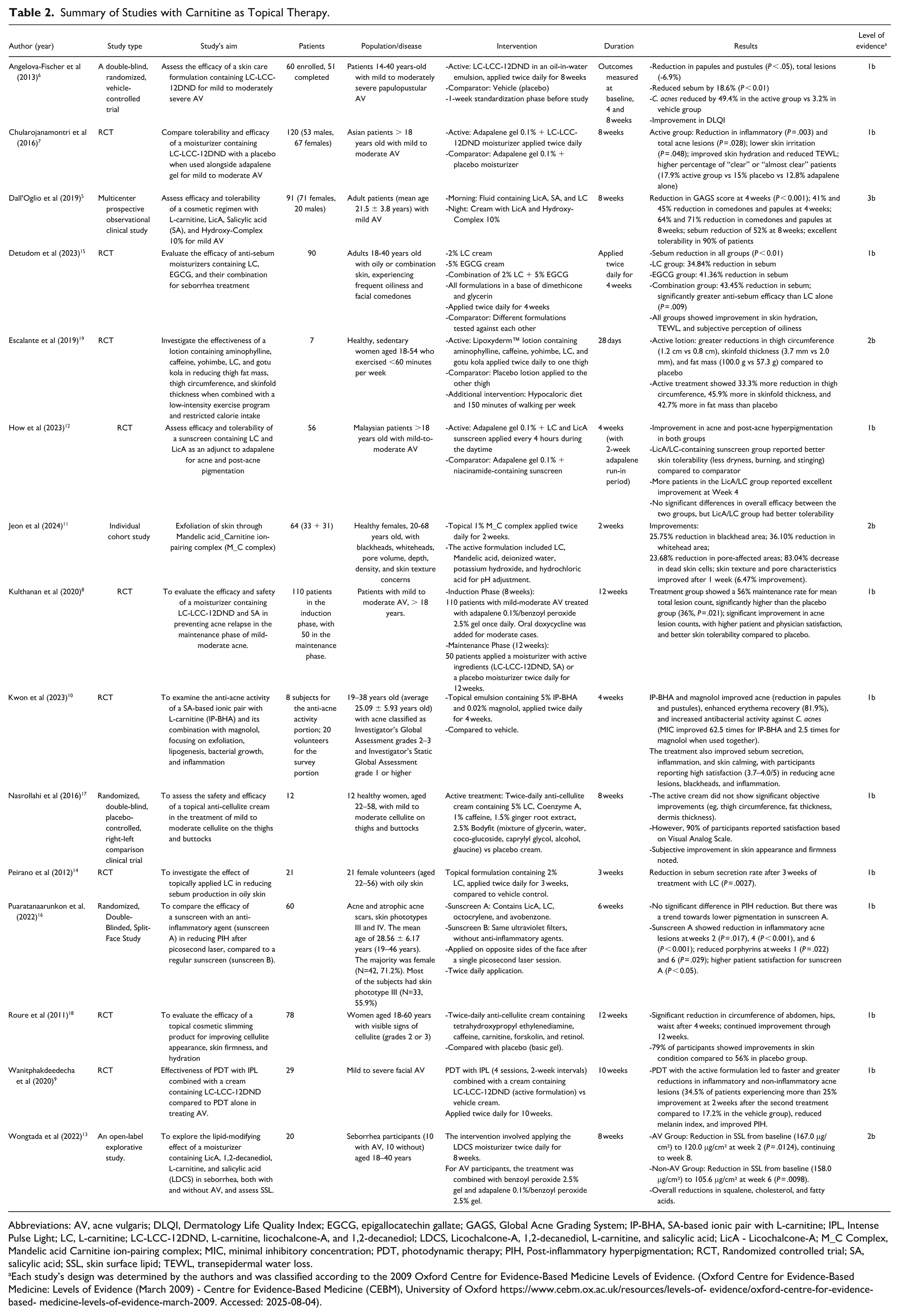
Abbreviations: AV, acne vulgaris; DLQI, Dermatology Life Quality Index; EGCG, epigallocatechin gallate; GAGS, Global Acne Grading System; IP-BHA, SA-based ionic pair with L-carnitine; IPL, Intense Pulse Light; LC, L-carnitine; LC-LCC-12DND, L-carnitine, licochalcone-A, and 1,2-decanediol; LDCS, Licochalcone-A, 1,2-decanediol, L-carnitine, and salicylic acid; LicA - Licochalcone-A; M_C Complex, Mandelic acid Carnitine ion-pairing complex; MIC, minimal inhibitory concentration; PDT, photodynamic therapy; PIH, Post-inflammatory hyperpigmentation; RCT, Randomized controlled trial; SA, salicylic acid; SSL, skin surface lipid; TEWL, transepidermal water loss.
Each study’s design was determined by the authors and was classified according to the 2009 Oxford Centre for Evidence-Based Medicine Levels of Evidence. (Oxford Centre for Evidence-Based Medicine: Levels of Evidence (March 2009) - Centre for Evidence-Based Medicine (CEBM), University of Oxford https://www.cebm.ox.ac.uk/resources/levels-of-evidence/oxford-centre-for-evidence-based-medicine-levels-of-evidence-march-2009. Accessed: 2025-08-04).
Acne Treatment
Topical L-carnitine reduced acne lesion counts, particularly when combined with other active ingredients. A 2013 trial of 51 patients demonstrated that a formulation with L-carnitine, licochalcone-A, and 1,2-decanediol (LC-LCC-12DND) reduced inflammatory acne lesions compared to a vehicle control. 6 A 2016 study found that combining LC-LCC-12DND with adapalene enhanced lesion reduction, with greater decreases in both inflammatory and total acne lesions. 7 In a multicenter study, a regimen incorporating LC-LCC-12DND and salicylic acid (SA) led to over 60% reduction in comedones and papules by week 8, suggesting efficacy against both non-inflammatory and inflammatory lesions. 5 The use of LC-LCC-12DND containing moisturizer was also shown to maintain the benefits from active acne treatment better than placebo in an RCT of 50 patients. 8 Finally, Wanitphakdeedecha et al 9 evaluated the combination of Intense Pulsed Light (IPL) therapy with an LC-LCC-12DND containing cream, reporting more rapid and substantial lesion reductions, indicating enhanced benefit when paired with procedural treatments.
A small RCT testing a SA-L-carnitine ionic pair with magnolol showed significant reductions in inflammatory lesions, along with antibacterial, anti-inflammatory, and exfoliating properties, demonstrating greater potency against Cutibacterium acnes than clindamycin. 10 Jeon et al 11 studied a mandelic acid-L-carnitine complex and found improvements in comedones, skin texture and pore appearance. However, an RCT by How et al 12 compared a sunscreen with L-carnitine and licochalcone-A to a niacinamide-based formulation as adjuncts to adapalene and found no significant difference in lesion reduction, though there was better tolerability of the adapalene in former group. This suggests that the efficacy of L-carnitine may vary depending on the comparator used.
In summary, the findings support topical L-carnitine’s potential role in acne management, particularly when combined with other active ingredients. While some findings vary, the overall evidence supports its role in reducing both inflammatory and non-inflammatory lesions and enhancing standard treatment regimens.
Sebum Reduction
Topical L-carnitine has been shown to reduce sebum production, a key factor in acne and seborrhea. A 2013 double-blind, randomized, vehicle-controlled trial reported decreased sebum levels after 8 weeks of treatment with a formulation containing LC-LCC-12DND in individuals with mild to moderate acne. 6 Similarly, a 2022 open-label study observed reduced sebum following 8 weeks of treatment with a moisturizer containing LC-LCC-12DND and salicylic acid. 13 Dall'Oglio et al 5 also noted reduced sebum in users of a combination of L-carnitine and licochalcone A. In another vehicle-controlled trial, a 2% L-carnitine formulation significantly lowered sebum secretion within 2 weeks. 14 Detudom et al 15 further demonstrated a 34.84% sebum reduction after 4 weeks using L-carnitine with epigallocatechin gallate.
These findings support topical L-carnitine as an effective agent for sebum control, with potential applications in managing acne, seborrhea, and related disorders.
Post-Inflammatory Hyperpigmentation
Topical L-carnitine shows potential in managing PIH, though findings are mixed. Wanitphakdeedecha et al 9 reported a significant PIH reduction at 1 month (P = .015) using IPL photodynamic therapy (endogenous photosensitizers) combined with a topical L-carnitine formulation. In contrast, Puaratanaarunkon et al 16 observed only a trend towards reduced PIH with a L-carnitine-based sunscreen following picosecond laser treatment. These results suggest topical L-carnitine may aid PIH, particularly when used alongside IPL therapy.
Cellulite and Fat Reduction
Several studies have explored topical L-carnitine for cellulite and fat reduction. Nasrollahi et al 17 reported subjective improvements in skin firmness with 5% L-carnitine cream, though no objective changes were noted. Roure et al 18 found that a formulation with L-carnitine, caffeine, and forskolin led to significant reductions in body circumference and improved skin tone after 12 weeks. Escalante et al 19 observed reductions in thigh circumference, skinfold thickness, and fat mass after 28 days of topical L-carnitine use, particularly when combined with diet and exercise. These findings suggest that topical L-carnitine may improve skin appearance and support fat reduction, though results vary with treatment protocols.
Section Summary
Topical L-carnitine is beneficial in dermatology, especially in acne treatment, sebum reduction, PIH management, and cellulite/fat reduction. Many studies have demonstrated its efficacy, often in combination with other active ingredients such as licochalcone-A. Since many studies were funded by Beiersdorf, the manufacturer of L-carnitine-based products, there could be a degree of bias. Only one study by Peirano et al 14 evaluated L-carnitine as a single ingredient, emphasizing the need for further research to fully ascertain its effects.
Future studies should focus on determining the optimal concentrations and vehicle formulations for L-carnitine, as well as investigating its mechanisms of action. The current evidence supports its role in enhancing acne treatment, reducing PIH, controlling sebum production, and improving skin appearance in cellulite and fat reduction.
Oral or Systemic Carnitine in Dermatology
Several studies assessed oral L-carnitine in dermatology. Although most were RCTs, some were systematic reviews and case reports. L-carnitine, along with derivatives like ALC and PLC, was primarily used alone but occasionally combined with other supplements. The studies focused on L-carnitine’s effects on inflammatory and autoimmune skin diseases including acne, skin aging, wound healing, and metabolic dysregulation. The therapeutic potential of L-carnitine may stem from its ability to reduce inflammation, oxidative stress, and metabolic dysfunction in chronic inflammatory skin diseases20,21, 22 Table 3 summarizes the studies reviewed.
Summary of Studies with Carnitine as Systemic Therapy.
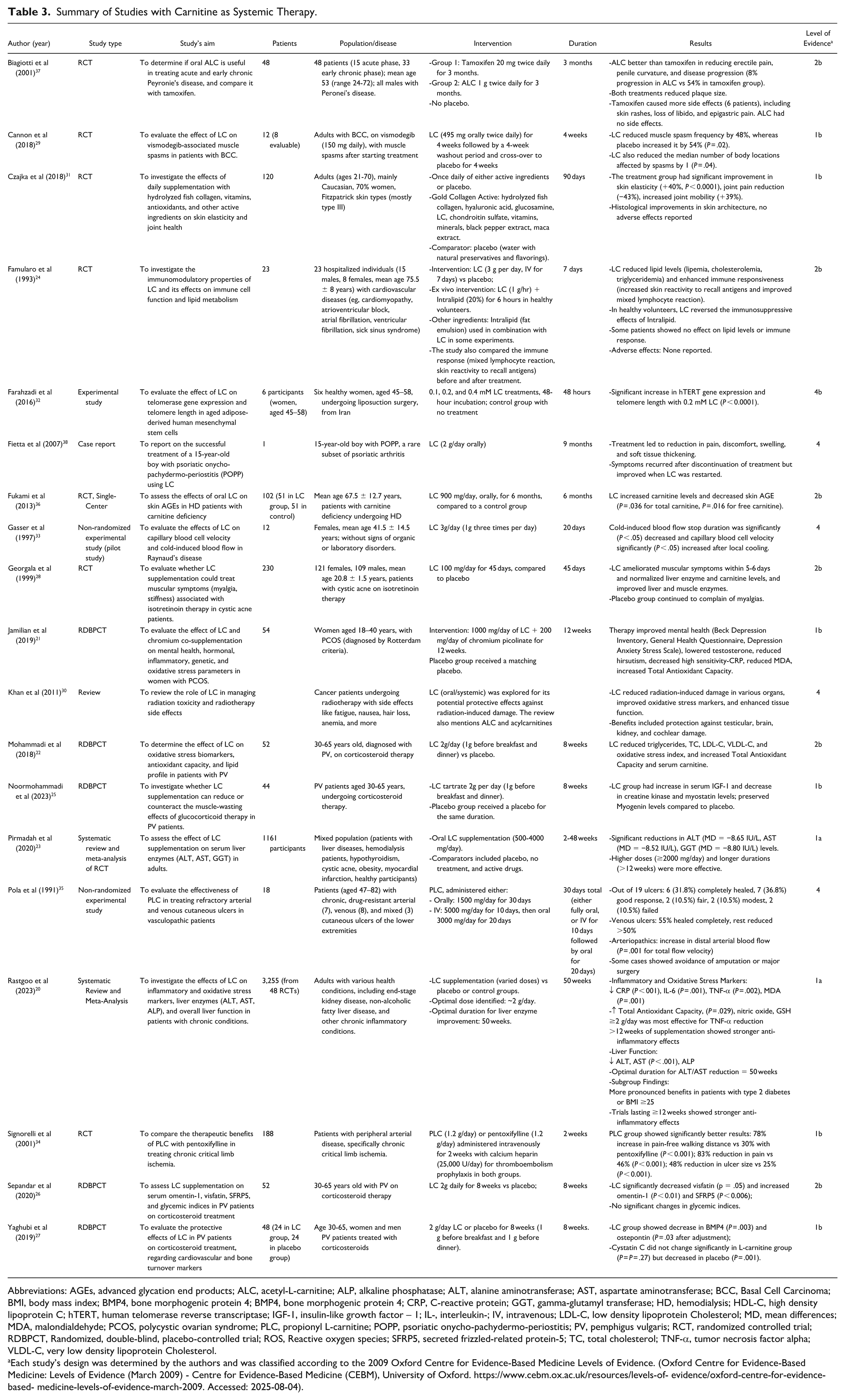
Abbreviations: AGEs, advanced glycation end products; ALC, acetyl-L-carnitine; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCC, Basal Cell Carcinoma; BMI, body mass index; BMP4, bone morphogenic protein 4; BMP4, bone morphogenic protein 4; CRP, C-reactive protein; GGT, gamma-glutamyl transferase; HD, hemodialysis; HDL-C, high density lipoprotein C; hTERT, human telomerase reverse transcriptase; IGF-1, insulin-like growth factor – 1; IL-, interleukin-; IV, intravenous; LDL-C, low density lipoprotein Cholesterol; MD, mean differences; MDA, malondialdehyde; PCOS, polycystic ovarian syndrome; PLC, propionyl L-carnitine; POPP, psoriatic onycho-pachydermo-periostitis; PV, pemphigus vulgaris; RCT, randomized controlled trial; RDBPCT, Randomized, double-blind, placebo-controlled trial; ROS, Reactive oxygen species; SFRP5, secreted frizzled-related protein-5; TC, total cholesterol; TNF-α, tumor necrosis factor alpha; VLDL-C, very low density lipoprotein Cholesterol.
Each study’s design was determined by the authors and was classified according to the 2009 Oxford Centre for Evidence-Based Medicine Levels of Evidence. (Oxford Centre for Evidence-Based Medicine: Levels of Evidence (March 2009) - Centre for Evidence-Based Medicine (CEBM), University of Oxford. https://www.cebm.ox.ac.uk/resources/levels-of-evidence/oxford-centre-for-evidence-based-medicine-levels-of-evidence-march-2009. Accessed: 2025-08-04).
Anti-inflammatory and Immunomodulatory Effects
Although not originally studied within dermatology, several investigations into oral L-carnitine have yielded findings with clear relevance to dermatologic care. A systematic review and meta-analysis (SR-MA) of 48 RCTs, demonstrated that L-carnitine supplementation significantly reduced inflammatory markers such as C-reactive protein (CRP), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α). 20 Importantly, these studies also reported improvements in liver enzymes, alanine aminotransferase (ALT) and aspartate aminotransferase (AST), particularly in trials of ≥ 12 weeks duration. Similarly, another SR-MA found notable reductions in ALT, AST and gamma-glutamyl transferase (GGT) with higher L-carnitine doses (≥2 g/day), reinforcing its hepatoprotective properties. 23 These findings are especially pertinent in dermatology, where systemic therapies such as isotretinoin and methotrexate pose risks of hepatotoxicity, suggesting L-carnitine may offer a supportive role in liver function monitoring or adjunctive care.
Further, Famularo et al 24 demonstrated immunomodulatory effects of L-carnitine in cardiovascular patients, showing improved lymphocyte responsiveness and increased skin reactivity to recall antigens, findings potentially applicable to dermatologic conditions with altered immune reactivity such as allergic contact dermatitis. In another study, Jamilian et al 21 found that L-carnitine combined with chromium improved inflammatory and hormonal parameters in women with polycystic ovary syndrome (PCOS), including reductions in high-sensitivity CRP and testosterone levels. This is particularly relevant given the high prevalence of acne and hidradenitis suppurativa in PCOS patients, conditions influenced by both inflammation and androgen levels.
Together, these studies support L-carnitine’s systemic anti-inflammatory, hormonal, and hepatic benefits, all of which are highly relevant in dermatology. Whether used to support patients on hepatotoxic medications, modulate immune reactivity in eczematous conditions, or aid in managing hormonally driven dermatoses, L-carnitine represents a promising adjunctive therapeutic avenue deserving further exploration in dermatologic populations.
Alleviating Treatment-Related Side Effects
L-carnitine can alleviate treatment-related adverse effects in patients undergoing dermatology therapies including corticosteroids, isotretinoin, vismodegib, and radiation. In patients with pemphigus vulgaris (PV) treated with oral corticosteroids, a randomized double-blind, placebo-controlled trial (RDBPCT) demonstrated that 2g of L-carnitine daily for 8 weeks improved muscle biomarkers [increased serum insulin-like growth factor 1 (IGF-1)] and reduced myostatin and creatine kinase levels, key indicators of reduced muscle atrophy. 25 This indicates the potential role of L-carnitine in preventing muscle wasting, a common side effect of long-term corticosteroid use. Another RDBPCT of 52 patients showed that oral L-carnitine improved insulin sensitivity and reduced inflammation in PV patients treated with corticosteroids. 26 Mohammadi et al 22 demonstrated reductions in lipid markers [triglycerides, low-density lipoprotein cholesterol (LDL-C), very-low-density lipoprotein cholesterol (VLDL-C)] and oxidative stress, in a similar population of PV patients, suggesting L-carnitine's potential in managing dyslipidemia and oxidative damage, common concerns in PV patients receiving corticosteroid therapy. Beyond metabolic effects, Yaghubi et al 27 found that L-carnitine reduced bone turnover biomarkers [bone morphogenic protein 4 (BMP4) and osteopontin] in PV patients treated with corticosteroids, indicating its potential to ameliorate bone and cardiovascular risks associated with corticosteroid use. These studies suggest L-carnitine may serve as a useful addition to reduce muscle wasting, metabolic disturbances, bone loss and oxidative stress in patients undergoing corticosteroid therapy.
In patients treated with isotretinoin for severe acne, Georgala et al 28 administered 100mg of L-carnitine daily for 45 days and found relief from muscle pain and stiffness, enabling continued isotretinoin treatment without dose reduction. In addition, Cannon et al 29 demonstrated that L-carnitine reduced muscle spasms by 48% in patients with basal cell carcinoma undergoing vismodegib therapy. Khan et al 30 suggested that L-carnitine could help mitigate radiation-induced skin damage (erythema and inflammation), although more studies are needed for dermatologic confirmation.
These findings highlight L-carnitine as an adjunct in alleviating various side effects associated with dermatologic treatments including corticosteroids, isotretinoin, vismodegib, and radiation therapy. Its ability to address muscle wasting, metabolic disturbances, and oxidative stress suggests it as a useful therapy for improving patient outcomes in dermatologic care.
Elasticity and Aging
Systemic L-carnitine has been shown to improve skin elasticity and combat aging. Czajka et al 31 found that a nutraceutical containing L-carnitine, hydrolyzed fish collagen, vitamins, and antioxidants improved skin elasticity by 40% over 90 days (P < 0.0001), suggesting a significant role in collagen synthesis and skin rejuvenation, although the specific attribution of the effect to L-carnitine can not be proven. Additionally, Farahzadi et al 32 showed L-carnitine’s ability to increase telomerase gene expression and telomere length in human mesenchymal stem cells, suggesting a potential role in skin rejuvenation at the cellular and stem cell level. These findings indicate that L-carnitine could be an important ingredient in anti-aging formulations for collagen synthesis and promotion of cellular regeneration.
Microcirculation, Wound Healing and Anti-Fibrotic Effects
L-carnitine’s systemic administration has demonstrated potential benefits in dermatology-related contexts by improving microcirculation, enhancing wound healing, and exerting anti-fibrotic effects across various clinical settings. Several studies have highlighted L-carnitine’s role in improving vascular function and microcirculation, which may be relevant in dermatologic conditions associated with impaired blood flow. For example, Gasser et al 33 evaluated patients with primary Raynaud’s disease and found that a 20-day course of oral L-carnitine (3 g/day) significantly increased capillary blood cell velocity and reduced the cold-induced blood flow stop duration, indicating enhanced microcirculatory function. Similarly, Signorelli et al 34 demonstrated that PLC improved pain-free walking distance and reduced ulcer size in patients with chronic critical limb ischemia, suggesting benefits in circulation and wound healing. In a study of 18 patients with chronic, drug-resistant arterial and venous lower leg ulcers, Pola et al 35 used oral or intravenous PLC for 30 days resulting in complete healing in 32% of the ulcers and significant improvement in another 37%, with a marked increase in distal arterial blood flow among arteriopathic patients. These findings are particularly notable given that patients had previously failed to respond to standard therapies.
Beyond its effects on circulation, L-carnitine has shown potential in tissue regeneration and anti-fibrotic activity. Fukami et al 36 reported that L-carnitine supplementation reduced advanced glycation end products (AGEs) in hemodialysis patients, improving skin elasticity and supporting tissue regeneration. Biagiotti et al 37 found ALC to be more effective than tamoxifen in improving tissue remodeling in patients with Peyronie’s disease, showcasing its potential in addressing fibrosis, which may have implications in sclerotic skin disorders. Additionally, Fietta et al 38 reported reduced pain, swelling, and tissue thickening in a patient with psoriatic onycho-pachydermo-periostitis following L-carnitine treatment, further supporting its role in tissue repair.
Collectively, these findings suggest that oral L-carnitine may offer supportive therapeutic roles in dermatology by improving microcirculation, promoting wound healing, and modulating fibrotic processes, warranting further exploration in targeted dermatologic conditions.
Section Summary
Systemic L-carnitine shows promise as an adjunctive therapy in dermatology, with demonstrated benefits including anti-inflammatory and immunomodulatory effects, mitigation of treatment-related side effects, improved skin elasticity, enhanced microcirculation, and wound healing, as well as anti-fibrotic activity. While existing studies suggest meaningful clinical potential, many are limited by small sample sizes and variation in dosage and formulations. Future well-designed RCTs are needed to define optimal use in dermatologic care. Nonetheless, L-carnitine represents a versatile systemic agent that aligns with dermatology’s broader goals of reducing inflammation, minimizing treatment toxicity and supporting overall skin health.
Use of Carnitine as a Biomarker for Dermatologic Disease or Disease-State
Given its central role in fatty acid metabolism and mitochondrial function, carnitine has emerged as a promising biomarker across a range of dermatologic and systemic conditions. Alterations in carnitine levels and derivatives reflect underlying metabolic, inflammatory, or fibrotic processes, offering diagnostic and prognostic insights. This section examines how shifts in carnitine metabolism, whether through depletion, accumulation, or altered ratios, can inform on disease presence, subtype, or progression in both hereditary and acquired dermatologic disorders. Table 4 summarizes the studies reviewed.
Summary of Studies with Carnitine as Biomarker.
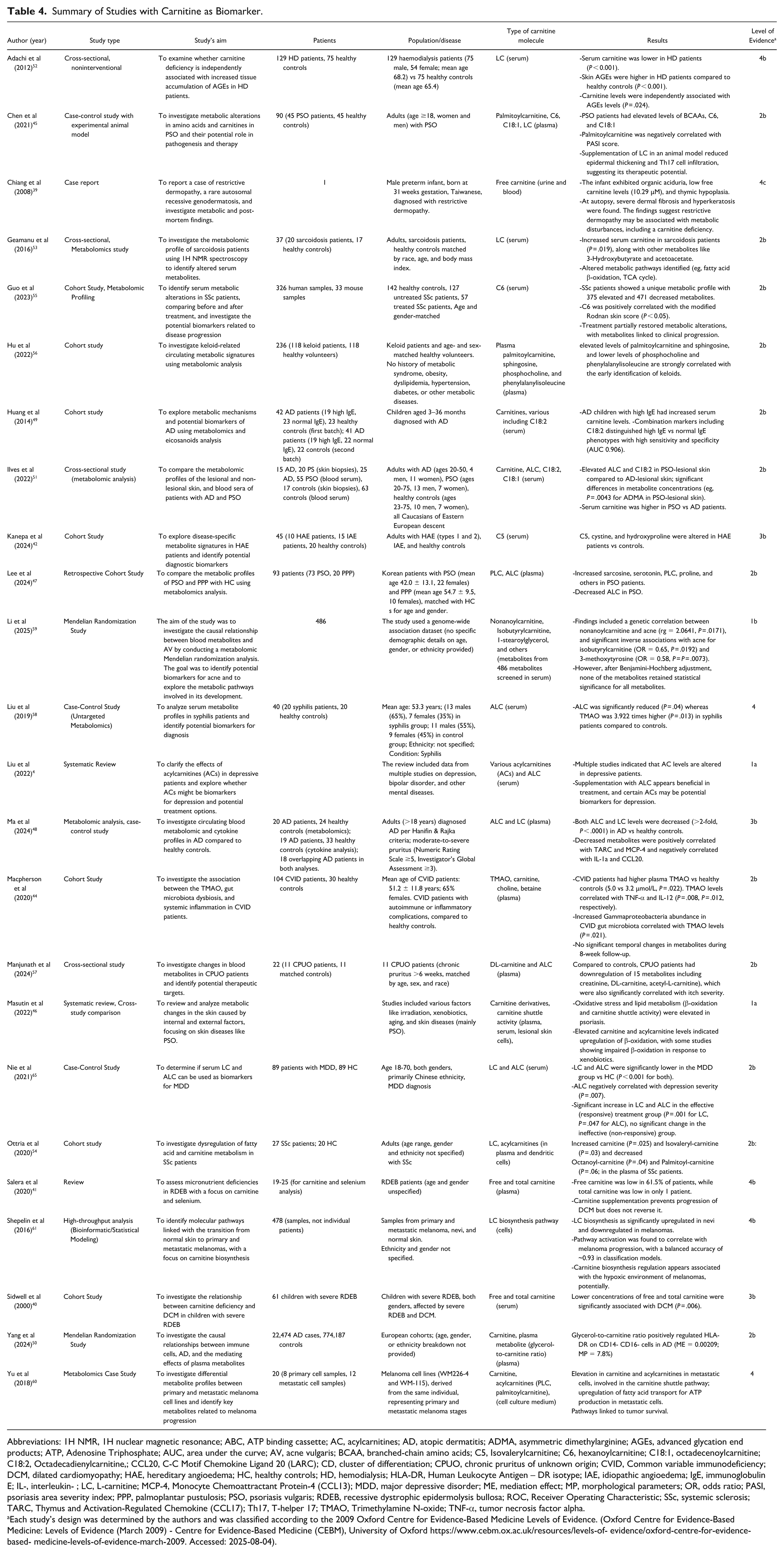
Abbreviations: 1H NMR, 1H nuclear magnetic resonance; ABC, ATP binding cassette; AC, acylcarnitines; AD, atopic dermatitis; ADMA, asymmetric dimethylarginine; AGEs, advanced glycation end products; ATP, Adenosine Triphosphate; AUC, area under the curve; AV, acne vulgaris; BCAA, branched-chain amino acids; C5, Isovalerylcarnitine; C6, hexanoylcarnitine; C18:1, octadecenoylcarnitine; C18:2, Octadecadienylcarnitine,; CCL20, C-C Motif Chemokine Ligand 20 (LARC); CD, cluster of differentiation; CPUO, chronic pruritus of unknown origin; CVID, Common variable immunodeficiency; DCM, dilated cardiomyopathy; HAE, hereditary angioedema; HC, healthy controls; HD, hemodialysis; HLA-DR, Human Leukocyte Antigen – DR isotype; IAE, idiopathic angioedema; IgE, immunoglobulin E; IL-, interleukin- ; LC, L-carnitine; MCP-4, Monocyte Chemoattractant Protein-4 (CCL13); MDD, major depressive disorder; ME, mediation effect; MP, morphological parameters; OR, odds ratio; PASI, psoriasis area severity index; PPP, palmoplantar pustulosis; PSO, psoriasis vulgaris; RDEB, recessive dystrophic epidermolysis bullosa; ROC, Receiver Operating Characteristic; SSc, systemic sclerosis; TARC, Thymus and Activation-Regulated Chemokine (CCL17); Th17, T-helper 17; TMAO, Trimethylamine N-oxide; TNF-α, tumor necrosis factor alpha.
Each study’s design was determined by the authors and was classified according to the 2009 Oxford Centre for Evidence-Based Medicine Levels of Evidence. (Oxford Centre for Evidence-Based Medicine: Levels of Evidence (March 2009) - Centre for Evidence-Based Medicine (CEBM), University of Oxford https://www.cebm.ox.ac.uk/resources/levels-of-evidence/oxford-centre-for-evidence-based-medicine-levels-of-evidence-march-2009. Accessed: 2025-08-04).
Carnitine as a Biomarker in Genodermatoses and Hereditary Conditions
Carnitine and its metabolites show promise as biomarkers in genetic and hereditary dermatoses, offering insights into disease mechanisms, progression, and therapeutic strategies.
Chiang et al 39 reported low free carnitine levels in a case of restrictive dermopathy, a rare autosomal recessive condition. Although this was based on a single case, this study points to the potential of carnitine as a potential biomarker in this rare disease. Early detection of carnitine deficiency could enable interventions like supplementation to improve outcomes.
In recessive dystrophic epidermolysis bullosa (RDEB), Sidwell et al 40 and Salera et al 41 found significantly reduced carnitine levels, linking deficiency to dilated cardiomyopathy (DCM). While supplementation did not reverse established DCM, it prevented further deterioration, highlighting the importance of monitoring carnitine levels to mitigate cardiovascular risks in RDEB.
In hereditary angioedema (HAE), Kanepa et al 42 and Wang et al 43 showed decreased isovalerylcarnitine (C5) levels, with a diagnostic ratio distinguishing HAE from idiopathic angioedema with high sensitivity and specificity. This suggests that carnitine metabolites, like C5, could be used as a biomarker for diagnosing rare and often misdiagnosed conditions like HAE.
Macpherson et al 44 found elevated trimethylamine-N-oxide (TMAO), a carnitine metabolite, in patients with Common Variable Immunodeficiency (CVID). TMAO levels correlated with inflammatory markers and gut microbiota, indicating its potential as biomarker for chronic inflammation in immune-mediated skin diseases.
These findings highlight carnitine and its metabolite as valuable biomarkers in genodermatoses and hereditary conditions, from restrictive dermopathy to RDEB, and HAE. Monitoring carnitine levels could enhance the management of these conditions, through further research is needed to confirm clinical utility.
Carnitine as Biomarker in Atopic Dermatitis and Psoriasis
Carnitine studies in atopic dermatitis (AD) and psoriasis (PSO) illustrate its potential as a biomarker in these chronic diseases. While altered carnitine metabolism is consistently shown, discrepancies exist in the direction and implications of these changes.
Both PSO and AD show altered carnitine metabolism. In PSO, systemic carnitine depletion correlates with disease severity, 45 , 46 while increased lesional carnitine suggests increased β-oxidation in inflamed skin. 46 Lee et al 47 further refined the biomarker potential of carnitine-related metabolites by identifying elevated PLC and decreased ALC in PSO patients compared to healthy controls. In AD, systemic carnitine changes appear to vary by disease phenotype, indicating different metabolic adaptations. Ma et al 48 reported significant reductions in plasma L-carnitine and ALC levels in AD patients. Conversely, Huang et al 49 observed elevated serum carnitine levels in AD patients with high immunoglobulin E levels, suggesting possible mitochondrial dysfunction and impaired fatty acid β-oxidation. Additionally, Yang et al 50 found that the glycerol-to-carnitine ratio significantly influenced inflammatory responses. A metabolomic study by Ilves et al 51 further highlighted differences in carnitine metabolism between PSO and AD. They found that serum carnitine levels were higher in PSO than in AD, and certain carnitine metabolites were more elevated in PSO-lesional skin compared to AD-lesional skin.
The evidence supports carnitine as a potential biomarker in PSO and AD, though its utility may depend on disease phenotype, systemic versus localized changes, and possibly environmental factors. Future research should integrate metabolomic and immunologic data to optimize its role in inflammatory skin diseases and guide personalized treatment strategies.
Carnitine as Biomarkers in in Other Chronic Dermatologic and Systemic Disorders
Carnitine’s role in skin pathology may extend beyond metabolic support to include potential utility as a biomarker of fibrosis and tissue remodeling. While a cross-sectional study of 129 hemodialysis patients by Adachi et al 52 found that carnitine deficiency was associated with increased accumulation of AGEs - known contributors to skin damage, aging, and fibrosis - other studies suggest that elevated carnitine levels may also reflect fibrotic processes. For example, a metabolomics study of patients with sarcoidosis 53 and a separate cohort study in systemic sclerosis (SSc) 54 both reported increased carnitine and acylcarnitine species, suggesting impaired fatty acid oxidation and mitochondrial dysfunction. Moreover, hexanoyl-carnitine was positively correlated with skin fibrosis in SSc 55 while elevated palmitoylcarnitine levels were linked to inflammation and fibroblast proliferation in keloid patients. 56 Taken together, these findings indicate that both deficiency and dysregulation of carnitine metabolism may serve as markers of skin pathology, whether through accumulation of toxic metabolites like AGEs or through altered mitochondrial function associated with fibrosis and inflammation.
Interestingly, while elevated carnitine levels have been associated with fibrotic and inflammatory skin conditions, a contrasting pattern emerges in disorders characterized by chronic irritation or microbial etiology. In chronic pruritus of unknown origin (CPUO), reduced levels of L-carnitine and ALC have been correlated with more severe pruritus, implicating mitochondrial dysfunction and impaired epidermal barrier repair. 57 Similarly, metabolomic profiling in syphilis patients has revealed diminished ALC, likely reflecting oxidative stress and compromised energy metabolism during systemic infection and cutaneous involvement. 58 In acne, a condition marked by sebaceous gland hyperactivity and immune dysregulation, a recent Mendelian randomization study identified elevated nonanoylcarnitine as a potential risk marker, while isobutyrylcarnitine may exert protective effects further supporting the role of lipid metabolism perturbations in disease pathogenesis. 59 These findings suggest that distinct carnitine profiles, whether elevated or depleted, may signify divergent pathological processes, with deficiencies aligning more closely with chronic itch, infectious dermatoses, and microbial inflammation.
Carnitine as Biomarker in Melanoma
Carnitine metabolism plays a significant role in melanoma progression, with two key studies, one by Yu et al 60 and another by Shepelin et al 61 offering complementary insights into its potential as a biomarker. Yu et al 60 used metabolomics to compare primary and metastatic melanoma cell lines and found that carnitine and acylcarnitines were significantly upregulated in metastatic melanoma. These metabolites are key intermediates in the carnitine shuttle pathway, which transports fatty acids into mitochondria for β-oxidation and adenosine triphosphate (ATP) production, a process that becomes crucial when metastatic cells face glucose deprivation. Their findings suggest that enhanced carnitine shuttle activity supports metastatic survival and could serve as biomarker of advanced disease, as well as therapeutic target.
In contrast, Shepelin et al. 61 performed a bioinformatics analysis and found that carnitine biosynthesis (distinct from shuttle activity), was upregulated in nevi but progressively downregulated in primary and metastatic melanoma. This suggests that endogenous carnitine production may be more active in early stages (nevi), but becomes suppressed as melanoma progresses, potentially reflecting a shift in how melanoma cells acquire and utilize metabolic substrates.
Rather than being contradictory, these studies highlight different aspects of carnitine metabolism across melanoma stages. Yu et al. 60 emphasized the importance of carnitine shuttle function in metastasis, while Shepelin et al. 61 highlight the loss of carnitine biosynthesis early in tumor development. Together, they imply that carnitine-related pathways may have stage-specific diagnostic of therapeutic relevance – biosynthesis-related pathways for early detection, and shuttle-related metabolites for monitoring metastasis. Clinically, carnitine metabolism warrants further investigation as a potential biomarker system and therapeutic target in melanoma.
Carnitine as Biomarker of Depression
Although depression is not specifically a dermatologic condition, there is an intricate and well-documented relationship between psychiatric disorders and the skin. Many dermatologic diseases significantly impact patients’ quality of life, with an estimated one third of patients in dermatology clinics needing assessment for mental health symptoms. 62 In turn, psychiatric medications may produce cutaneous side effects, and some dermatologic treatments can influence mental health status.63,64 In this context, carnitine has drawn attention as a shared biomarker in both domains. A case-control study of 89 patients reported significantly lower serum levels of L-carnitine and ALC in individuals with Major Depressive Disorder (MDD), with ALC levels inversely correlating with depression severity. 65 Similarly, a 2022 systematic review found that depressed patients had reduced free carnitine and short-chain acylcarnitines, suggesting that impaired mitochondrial function and altered neurotransmitter regulation may contribute to mood instability. 4 Moreover, a 2018 SR-MA concluded that ALC supplementation significantly decreases depressive symptoms compared with placebo. 66 These findings propose that carnitine metabolism may not only serve as a biomarker for depression but may also help explain overlapping metabolic dysfunction in dermatologic conditions with psychiatric comorbidities, such as psoriasis, acne, and chronic pruritus.
Section Summary
Current evidence supports the potential of carnitine and its derivatives as emerging biomarkers in dermatology. Altered carnitine metabolism provides insight into disease mechanisms, severity, and progression across genodermatoses, inflammatory, fibrotic, pruritic, neoplastic, and psychodermatologic conditions. While promising, variability in study design and metabolite profiling highlights the need for standardized, dermatology-focused research. Future studies should aim to validate these biomarkers in larger cohorts and explore their integration into personalized care.
Discussion
L-carnitine is emerging as a multifaceted agent in dermatology, with growing evidence supporting its therapeutic, adjunctive, and diagnostic potential across a wide range of skin diseases. Systemically, its has demonstrated anti-inflammatory, metabolic, and immunomodulatory effects. It also has a potential role in mitigating treatment-related adverse effects from agents like isotretinoin, and corticosteroids. Its roles in tissue repair, microcirculation, and fibrosis modulation further highlight its utility in wound healing and sclerotic skin disorders. Topically, L-carnitine demonstrates promise in acne management, sebum regulation, post-inflammatory hyperpigmentation, and cellulite, with favorable results across multiple studies, particularly when combined with other active agents.
Beyond its therapeutic applications, carnitine and its metabolites are increasingly recognized as potential biomarkers, offering insight into disease pathophysiology, severity, and subtype differentiation. Altered carnitine profiles have been observed in genodermatoses, inflammatory dermatoses, fibrotic conditions, and neoplastic disease such as melanoma, as well as in psychodermatologic overlaps, suggesting that carnitine metabolism may reflect systemic immune or metabolic dysfunction relevant to skin disease.
Importantly, most studies have not shown significant adverse effects associated with carnitine supplementation at standard doses. While some reviews list multiple side effects,20,67 closer examination suggests these may not be directly attributable to carnitine itself. For instance, one study reported gastrointestinal symptoms in patients taking orlistat plus carnitine, 68 though orlistat alone is known to cause such effects. Other studies reported adverse event rates similar to placebo.69,70 Taken together, the evidence supports a favorable safety profile for systemic carnitine.
While carnitine supplementation is generally safe, concerns remain regarding its association with cardiovascular disease (CVD) through increased production of trimethylamine-N-oxide (TMAO), a gut-derived metabolite of carnitine, choline, and phosphatidylcholine. 71 , 72 Although TMAO has been linked to atherosclerosis via macrophage activation, 71 its direct role in CVD remains unclear. Several studies have found that elevated TMAO levels following carnitine supplementation do not consistently correlate with traditional cardiovascular risk markers, 73 , 74 suggesting that TMAO may serve more as a biomarker rather than a causative agent in CVD. 71 Emerging evidence also indicates that probiotics, such as Lactobacillus rhamnosus GG, can lower TMAO levels by modulating gut microbiota, 72 offering a potential strategy to mitigate risks. Given carnitine’s therapeutic benefits for skin conditions, further research into microbiome-targeted interventions may help optimize its safe and effective use in dermatology.
Conclusion
L-carnitine represents a multifaceted agent in dermatology, offering therapeutic, adjunctive, and diagnostic benefits across a wide range of skin diseases. Its systemic and topical effects, ranging from anti-inflammatory action to metabolic regulation and barrier repair, are supported by a growing body of evidence. Furthermore, its emerging role as a biomarker opens the door to personalized dermatologic care, with potential applications in diagnosis, disease monitoring, and stratified treatment approaches. Although questions remain regarding long-term safety and disease-specific dosing strategies, L-carnitine’s favorable tolerability profile and broad physiologic relevance position it as a valuable tool in the evolving landscape of integrative dermatology. Future research should focus on dermatology-specific clinical trials, biomarker validation, and mechanistic studies to fully define its place in routine dermatologic practice.
Supplemental Material
sj-docx-1-cms-10.1177_12034754261427402 – Supplemental material for L-Carnitine in Dermatology: A Systematic Review of Therapeutic Potential and Biomarker Applications
Supplemental material, sj-docx-1-cms-10.1177_12034754261427402 for L-Carnitine in Dermatology: A Systematic Review of Therapeutic Potential and Biomarker Applications by Eunice Y. Chow and Mariusz Sapijaszko in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-2-cms-10.1177_12034754261427402 – Supplemental material for L-Carnitine in Dermatology: A Systematic Review of Therapeutic Potential and Biomarker Applications
Supplemental material, sj-docx-2-cms-10.1177_12034754261427402 for L-Carnitine in Dermatology: A Systematic Review of Therapeutic Potential and Biomarker Applications by Eunice Y. Chow and Mariusz Sapijaszko in Journal of Cutaneous Medicine and Surgery
Footnotes
Author Contributions
Dr. Chow and Dr. Sapijaszko contributed equally to the conception of the review. Dr. Chow conducted the literature search, performed data extraction, prepared the tables, and drafted the manuscript. Dr. Sapijaszko contributed to data verification and critical revision of the manuscript. Both authors reviewed and approved the final version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
There are no human participants in this article
Informed Consent Statement
Informed consent is not required.
Supplemental Material
Supplemental material for this article is available online.
