Abstract
Post-inflammatory hyperpigmentation (PIH) and hypopigmentation are heterogenous conditions that often occur secondary to dermatoses (eg, psoriasis, acne, or atopic dermatitis), cutaneous injury (eg, burns or radiation therapy), infection (viral, bacterial, or fungal), or allergic/immunologic dysregulation (eg, contact dermatitis or insect bite reactions). The pathophysiology of PIH is complex with multiple pathways implicated in hypermelanosis including ultraviolet radiation exposure, local skin inflammation, and cytokine and growth factor signalling between keratinocytes, melanocytes, and dermal fibroblasts. PIH is a clinical diagnosis based on patient history and clinical examination and may persist for months to years following the resolution of underlying pathophysiological mechanisms, emphasizing the need for prompt diagnosis and management. PIH can be disfiguring and associated with substantial impairments in quality of life and psychosocial well-being. Patients with skin of colour (ie, Fitzpatrick skin types III-VI) have an increased risk of PIH and often present with more pronounced dyspigmentation, highlighting a high unmet need for improved recognition, awareness, and management in this population. Photoprotection is the cornerstone of PIH management given the photosensitive nature of the condition and is recommended in tandem with other treatment options including topical agents (eg, hydroquinone, retinoids, tranexamic acid), light/laser-based therapies, or chemical peels (glycolic acid and salicylic acid). Despite their use, high-level clinical evidence for these treatment options in PIH remains limited. This review provides an up-to-date overview of PIH pathophysiology, diagnosis and differential diagnosis, and management, and offers expert clinical commentary on practical strategies for identifying, preventing, and managing PIH in routine practice.
Introduction
Cutaneous pigmentation changes encompass both hyperpigmentation and hypopigmentation, reflecting alterations in constitutive skin colour.1,2 Pigmentation changes are more often acquired secondary to conditions that result in skin inflammation or injury; however, there are also congenital forms associated with varying patterns of inheritance. 2 Post-inflammatory hyperpigmentation (PIH) tends to be more prominent and impactful for individuals with pigmented skin (ie, Fitzpatrick skin types III-VI). 1
The etiology of PIH is heterogeneous and can include dermatologic conditions, immunologic dysregulation, cutaneous injury, and infectious triggers (Supplemental Table S1).1-3
PIH is a common complaint accounting for up to 8.5% of dermatology consultations. 2 The condition can be associated with substantial personal burden and impairments in quality of life, self-esteem, and psychosocial well-being.4-6 The burden of PIH may be particularly high when it occurs on highly visible areas. 6
With the substantial prevalence and burden of pigmentary skin changes, it is imperative for dermatologists to recognize and appropriately manage PIH to limit its negative impact on patients’ quality of life and psychosocial functioning. The goals of this review were to provide an up-to-date review of the underlying pathophysiology of PIH, describe its diagnosis and differential diagnosis, and, given the dearth of clinical guidelines on post-inflammatory pigmentation changes, offer an expert clinical commentary on practical strategies for preventing and managing PIH in routine practice.
Pathophysiology
Physiology of Melanin Deposition
Melanin is the major pigment that imparts human skin and hair its colour. Its main biological function is protection against ultraviolet radiation (UVR).7-9 Melanin is produced by melanocytes, which reside in the basal layer of the epidermis of the skin and in the outer root sheath of hair follicles. 7 The term “melanogenesis” refers to melanin synthesis, melanosome formation and transport, and melanin metabolism. 7 Melanin synthesis is sequestered in specialized membrane-bound organelles called melanosomes.7,9 The transfer of melanosomes from melanocytes to surrounding keratinocytes results in deposition of melanin in the epidermis, giving skin its colour.7,9 Keratinocytes in the basal epidermal layer contain 60% to 80% of melanosomes, which are localized over cell nuclei and thus produce a shield-like protection against UVR-induced deoxyribonucleic acid damage. 10
The precursor of melanin is tyrosine, which is oxidized by tyrosinase in a multistep process that ultimately produces 2 main types of melanin in humans: eumelanin, an insoluble brown-black pigment, and pheomelanin, a slightly more soluble, sulphur-containing pigment with a reddish-yellow hue.7,10 Varying ratios of eumelanin and pheomelanin account for the observed differences in skin colour, which is also influenced by the size, quantity, and distribution of melanosomes in the epidermis. 10
Melanosome size and distribution vary across Fitzpatrick skin types, with darker skin having larger and more dispersed melanosomes versus smaller and clustered melanosomes in lighter skin.9,10 The central role of melanin in photoprotection also accounts for observed geographic patterns in skin pigmentation. 10 Humans originating from equatorial regions where UVR is more intense have darker skin tones than those living closer to the poles, where UVR is less intense and the skin thus requires less photoprotection. 10
Pathophysiological Processes in Hyperpigmentation
Hyperpigmentation can result from various endogenous and exogenous stimuli including cutaneous inflammation, injury, and/or UVR exposure. 9 These processes can all lead to increased deposition of melanin in the epidermis or deeper into the dermis.1,9,10 Several factors influence the severity of hyperpigmentation including baseline skin colour, degree and depth of inflammation or injury, extent of dermoepidermal junction disruption, presence of predisposing local and systemic inflammatory conditions, hormonal factors, and melanocyte stability. 11
Multiple signalling pathways have been implicated in PIH, and there is evidence of considerable cross-talk between keratinocytes, melanocytes, and fibroblasts (Figure 1).12,13 Keratinocytes have been shown to release soluble factors that promote melanogenesis: alpha-melanocyte-stimulating hormone (α-MSH), endothelin 1 (ET-1), stem cell factor, hepatocyte growth factor, and prostaglandins. 12 α-MSH production is induced by inflammatory cytokines and stimulates pigmentation signalling pathways including the melanocortin-1 receptor (MC1R), which drives excess melanin synthesis and results in PIH. 14 α-MSH thus acts as a molecular bridge between skin inflammation and pigmentation. Keratinocytes also modulate melanocytes by releasing inflammatory cytokines, which upregulate melanogenesis. 12 Moreover, keratinocytes can indirectly stimulate melanogenesis via the release of inflammatory cytokines through a paracrine feedback loop. 12 Fibroblasts secrete growth factors that can contribute to a hormonal milieu that promotes melanocyte proliferation.15-17
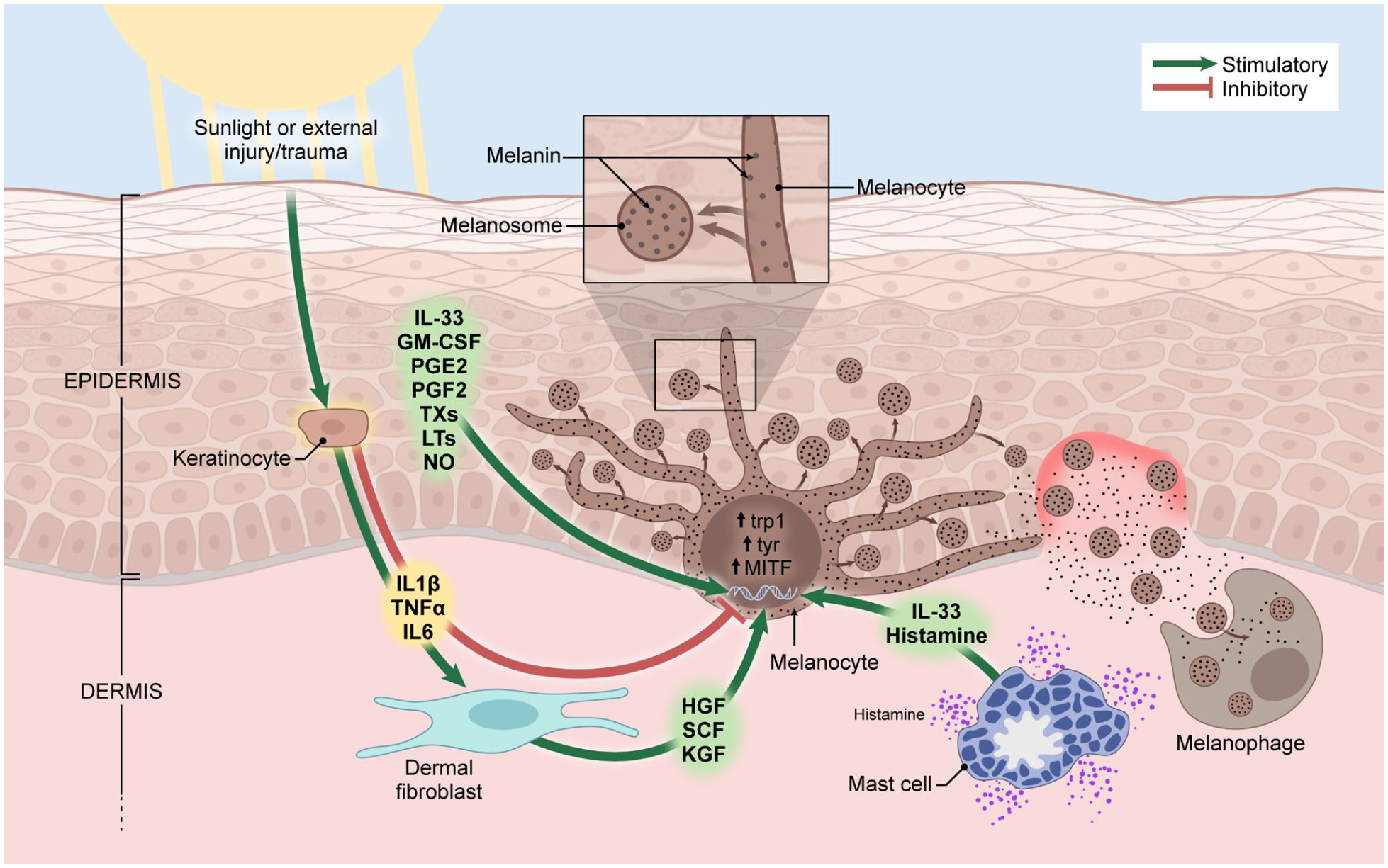
Pathophysiologic mechanisms implicated in post-inflammatory hyperpigmentation.
Exposure to UVR is one of the main exogenous stimuli associated with hypermelanosis and is recognized as a major contributor to skin photoaging, solar lentigos, and melasma.8-10,18 There are 3 distinct phases to UVR-induced hypermelanosis: immediate pigment darkening (IPD), which tends to be transient in nature; persistent pigmentation, which lasts longer than IPD and occurs in response to more intense UVR exposure; and delayed tanning (DT), which involves activation of melanogenesis several days after UVR exposure. 10 Chronic UVR exposure has been associated with increased melanocyte density and melanin deposition in the epidermis, processes that contribute to skin darkening. 9 Thus, adequate photoprotection can help mitigate the effects of chronic UVR exposure on hyperpigmentation in all skin phototypes. Although lighter phototypes exhibit reduced IPD and DT responses than darker phototypes, 10 the higher prevalence of hyperpigmentation disorders in individuals with skin of colour may be related in part to the less frequent use of photoprotection in this population, which could expose them to a higher risk of UVR-induced hypermelanosis. 18
Local skin inflammation is one of the key endogenous factors associated with PIH and can be a consequence of inflammatory skin conditions such as psoriasis or atopic dermatitis. 10 Indeed, post-inflammatory pigmentation changes are relatively common in individuals with psoriasis. In a study of 459 patients with psoriasis, hypopigmentation and hyperpigmentation were present in 10% and 13.7% of patients, respectively. 19 PIH was nearly 4-fold more common in patients with skin of colour whereas hypopigmentation was not associated with any particular skin type. PIH can be triggered by the release by keratinocytes of various inflammatory mediators in the epidermis including cytokines [eg, interleukin (IL)-1β, IL-6, tumour necrosis factor (TNF), interferon γ], reactive oxygen species, prostaglandins, and other pro-inflammatory molecules (eg, neuropeptides, chemokines, leukotrienes, and growth promoters such as epidermal growth factor), which stimulates melanogenesis (Figure 1).3,10,18 Skin inflammation can also induce melanocyte hyperplasia and damage to the epidermal basement membrane, which can facilitate leakage of melanosomes from keratinocytes into the dermis.3,10,18 PIH in the upper dermis is associated with the release of large quantities of melanin, which is phagocytosed by macrophages and imparts a darkened colour to the skin, which can be prolonged or even permanent.3,18 This type of PIH can affect any skin type, but it is more common in Fitzpatrick type III to IV skin types. 10
The observation that PIH occurs in areas previously affected by psoriatic plaques indicates a pathogenic link between psoriasis and PIH. In support of that hypothesis, Wang et al demonstrated that TNF and IL-17 have synergistic effects on melanogenesis in an in vivo cell model of psoriasis. 13 Specifically, IL-17 and TNF synergistically inhibited pigmentation signalling and melanogenesis by suppressing pigmentation-related genes and lowering cellular tyrosinase levels in melanocytes, thereby reducing cellular melanin content. 13 Moreover, IL-17 and TNF jointly induced the expression of melanocyte mitogens, including CXCL1 and IL-8, thus enhancing melanocyte proliferation. Epidermal melanocytes in psoriatic lesions are exposed to elevated levels of IL-17 and TNF. 13 Accordingly, the combined effect of these 2 cytokines causes hypopigmentation and, paradoxically, increases the number of melanocytes by stimulating melanocyte proliferation.13,20 During recovery after inflammation, the effect of IL-17 and TNF leaves a hypopigmented zone on the edge of regressing lesions (ie, Woronoff ring), where melanogenesis is still suppressed despite the increased density of melanocytes along the basement membrane in lesional psoriatic skin.13,20 Progressive recovery of pigmentation gene expression, along with the increased number of melanocytes, can then lead to an abundant melanin production and PIH. 13 Selective therapeutic blockade of TNF and/or IL-17 was shown to result in progressive recovery of PIH in healing psoriatic lesions among treatment responders, 13 further supporting the pathogenic link between skin inflammation and PIH.
The pathophysiology of post-acne PIH appears to be multifactorial. 21 Pigmentation changes can be secondary to acne-associated inflammation as well as external trauma (eg, excoriation). 21 As a result, the term “post-acne hyperpigmentation” or PAH has been suggested to be more inclusive of the broad pathophysiologic mechanisms involved. 21 In addition to the inflammatory pathways described above, trauma-related pathways involving paracrine signalling between keratinocytes, fibroblasts, and melanocytes have been implicated in PAH. 21 For example, Kurita et al found that mechanical stimulus upregulates ET-1 by keratinocytes. 17 ET-1 is a paracrine soluble factor that has both melanogenic and mitogen effects on melanocytes. 22
Hyperpigmentation and hypopigmentation changes are also common after skin injury and can be long-lasting with scarring, particularly after burn wounds.1,23,24 Injuries to the skin resulting from cosmetic procedures (eg, laser/light therapy, chemical peels, dermabrasion), radiation therapy, UVR (eg, melasma), and trauma (eg, surgery, burns) can significantly alter melanogenesis, with variations over time as the skin heals.1,18 For example, in the early post-injury phase before scarring occurs, there is often increased melanin synthesis around the periphery of the wound. 24 After the resolution of inflammation and re-epithelialization, melanin production gradually declines and natural skin colour is partially to fully restored. 24 Disruptions in inflammatory cytokine pathways have also been reported during wound healing. 25 Overall, the pathophysiologic mechanisms underlying PIH during wound healing are not as well elucidated as other types of PIH, and there remains a need for further research in this area.
Diagnosis of PIH
PIH is a clinical diagnosis based on history and clinical examination.11,26 Determining the underlying cause of hyperpigmentation and presence of reactive processes can be helpful in the diagnostic pathway.11,26 A detailed patient history should include background dermatologic disease and the time of disease onset. Specific hyperpigmentation conditions are more commonly associated with congenital (eg, café-au-lait macules, naevus of Ota), childhood (eg, Becker naevus, idiopathic eruptive macular hyperpigmentation), pregnancy (eg, melasma), or middle-age (eg, lentigines, lichens planus pigmentosus) onsets. 26 History should also address prior medication use including topical, oral, and systemic agents, especially if drug-induced pigmentation changes are suspected.11,26 Any previous signs or symptoms of inflammation and systemic symptoms that may reflect the presence of other systemic disorders should be addressed. 26
Physical examination is important to determine the pattern of pigmentation, and clinical examination is used to determine the nature of the pigmentation as epidermal (tan, brown, or dark brown) or dermal (blue/grey).1,11,26 Both dermal hyperpigmentation and epidermal hyperpigmentation may coexist and erythematous papules, nodules, or plaques may be present if the underlying inflammatory process is still active.1,11 A Woods lamp evaluation may be used to reveal fluorescence and distinguish between dermal PIH and epidermal PIH.1,11,26 If the underlying inflammatory etiology or diagnosis is uncertain, a biopsy may be helpful.1,12 Finally, laboratory examinations may be required to rule out conditions like Addison’s disease or systemic lupus erythematosus if the patient presents with concurrent signs or symptoms. 1
Differential Diagnosis
Possible differential diagnoses include, but are not limited to, the following: melasma, ochronosis, cutaneous lupus, hyperpigmented mycosis fungoides, pityriasis lichenoides chronica, amyloidosis cutis dyschromatosis, idiopathic guttate hypomelanosis, trichrome or confetti like vitiligo, phytophotomermatoses, flagellate dermatosis, erythema dyschromicum perstans, acanthosis nigricans, primary localized cutaneous amyloidosis, confluent and reticulated papillomatosis of Gougerot and Carteaud, and medication-induced pigmentary changes.2,11,26,27 Supplemental Table S2 provides a summary of distinguishing features between PIH and common mimickers.
Management
General
Early identification of underlying inflammatory etiology is important for PIH treatment and management to prevent further irritation and pigmentation changes.28,29 A key principle of PIH treatment selection is consideration of the benefit relative to the risk of skin irritation, which may itself contribute to dyspigmentation. 30 There are a variety of treatment options available for PIH, including sun protection, topical agents, oral agents, chemical peeling (chemexfoliation), and laser treatment.27,30-39 The respective mechanisms of action, recommended uses in PIH, advantages, and disadvantages of the most common treatment options are summarized in Supplemental Tables S3 to S5. In addition, novel therapies have emerged for the treatment of several dyspigmentation disorders, such as oral or topical Janus kinase inhibitors for vitiligo and atopic dermatitis.40-43 Although not discussed herein, it is worth monitoring whether evidence is generated for the use of such novel agents in the treatment of PIH.
Photoprotection
PIH is a photosensitive condition that may be exacerbated by exposure to visible light and UV rays.39,44-46 Photoprotection, including broad spectrum sunscreen, reduces induction of reactive oxygen species and inflammatory responses by UV and visible light, and is recommended as an adjunct therapy/protective measure for PIH.28,29,36-39,44-46 All patients should be educated on the importance of sun protection factor (SPF) sunscreen, sun-protective clothing, and use of shade structures, which may be particularly important for individuals with skin of colour since they are at increased risk of PIH, and the importance of sun protection is often under-recognized in these groups.28,44 Broad spectrum sunscreen with SPF 30 to 60 is widely accessible, does not require a prescription, and has minimal side effects other than a low risk of skin reactions and the potential to decrease vitamin D absorption in darker-skinned individuals (daily intake of 1000 IU vitamin D is recommended in high-risk patients).28,29,36-39,44-46 Although direct clinical evidence for the efficacy of sunscreen alone compared with other treatment options for PIH is limited, there is extensive evidence for the benefit of sunscreen and sun avoidance for the prevention of PIH or as an adjunct to other treatment modalities.28,29,36-39,44-46
Topical Agents
Topical agents including hydroquinone, retinoids, azelaic acid, and kojic acid are often used in the first-line treatment of PIH and may be effective alone, in combination regimens, or in tandem with laser therapy or chemical peeling (Supplemental Table S3).27,30,32,33,47 A systematic review of treatment outcomes in patients with PIH found that topical monotherapies resulted in 5.4% complete response, 72.4% partial response, and 22.2% poor/no response, whereas combination therapies resulted in 2.4% complete response, 84.9% partial response, and 12.7% poor/no response. 35 Although topical therapies target the epidermal component of PIH by reducing melanin production, other treatment modalities including lasers and chemical peels may better target deeper layers of the dermis. 35
Hydroquinone
Hydroquinone is a versatile organic compound widely used for the topical treatment of hyperpigmentation disorders, and is often considered a mainstay of PIH treatment.28,47 Hydroquinone may be recommended as a first-line treatment for PIH caused by trauma, acne, and melasma and is typically used on the face, back of hands, and neck/chest area.27,28,35,39,47-50 The effectiveness of hydroquinone for PIH has been demonstrated with monotherapy use as well as in combination with retinoids, glycolic acid, kojic acid, ascorbic acid, vitamin E, sunscreens, topical corticosteroids, lasers, and chemical peels.27,28,35,39,47-50
Retinoids
Topical retinoids have important comedolytic and anti-inflammatory effects, which may be particularly helpful in reducing both acne and associated hyperpigmentation.30,51 They have also been shown to directly affect melanogenesis by inhibiting the transfer of melanosomes to keratinocytes and accelerating epidermal turnover. 52 Retinoids are widely approved for the treatment of acne vulgaris and are often considered a first-line therapy for individuals with acne-related PIH.30,53,54 Topical retinoids including tretinoin, adapalene, tazarotene, and trifarotene have demonstrated effectiveness in lightening PIH lesions and may be particularly useful in reducing hyperpigmentation in patients with skin of colour.28,30,34,55,56 A systematic review found that topical retinoid monotherapies resulted in 4.1% complete response, 70.1% partial response, and 25.8% poor-to-no response in individuals with PIH. 35
Many patients with acne and PIH require a multimodal approach of topical retinoids with other management strategies, including combination with other topicals (eg, hydroquinone, azelaic acid, antibiotics with benzoyl peroxide) or procedures.30,39 Studies exploring topical combinations including clindamycin (1.2%)/tretinoin (0.025%) or microencapsulated hydroquinone (4.0%)/retinol (0.15%) with an antioxidant for acne and hyperpigmentation have shown improved overall hyperpigmentation and lesion intensity, quantity, and size.48,57 Dual combination hydroquinone (4.0%)/retinol (0.15%) delivered in microsponge reservoirs has been shown to effectively treat both melasma and PIH while minimizing skin irritation side effects. 50 Additionally, multimodal therapies including a salicylic acid peel (20%-30%)/topical tretinoin (0.1%) combination may be associated with greater improvement of PIH than either monotherapy alone with good tolerability, low recurrence rate, and minimal side effects.35,58
Based on available evidence for the treatment of melasma, triple topical combination therapies containing hydroquinone, retinoids, and corticosteroids may be a useful treatment for PIH. 27 For example, the Kligman formula (5.0% hydroquinone/0.1% tretinoin/0.1% dexamethasone) may effectively treat both melasma and PIH,35,59 and TriLuma® (4.0% hydroquinone/0.05% tretinoin/0.01% fluocinolone acetonide) is often considered the gold standard topical treatment for patients with melasma.27,60,61 Although treatment of acne-related PIH with TriLuma may be associated with greater improvement of hyperpigmentation than dual combinations of hydroquinone, tretinoin, or fluocinolone acetonide,3,62 the results of a systematic review showed 50% partial response and 50% poor-to-no response in patients with PIH. 35
Collectively, integration of retinoids with other topicals or multimodal therapies may help improve efficacy and reduce side effects associated with individual agents while targeting different aspects of PIH pathophysiology.
Azelaic Acid
Azelaic acid is a naturally occurring dicarboxylic acid isolate that acts as a reversible tyrosinase inhibitor and thus inhibits melanogenesis.28,32,39,63 There is evidence that azelaic acid may be effective for the treatment of PIH related to acne vulgaris and melasma as both monotherapy and in combination with retinoids or glycolic acid, but the data are limited compared with first-line hydroquinone and retinoid treatments.28,32,35,39,63,64
Kojic Acid
Kojic acid is a fungal metabolite that inhibits the activity of tyrosinase by chelating copper at its active site and thus inhibits melanogenesis.28,29,32,65 Kojic acid has been suggested as a second-line option in patients intolerant to first-line therapies.28,29,32,65 Although the clinical evidence for kojic acid in PIH is limited, it has been shown to exhibit similar effectiveness as other topical agents and may be beneficial when used in combination with hydroquinone.28,32
Tranexamic Acid
Topical tranexamic acid (TXA) inhibits the binding of plasminogen to keratinocytes, thereby reducing the activity of melanocyte tyrosinase and the production of arachidonic acid and prostaglandin (known inflammatory mediators of melanogenesis). 66 TXA may be effective alone or in combination with kojic acid, niacinamide, vitamin C, or laser therapy for the treatment of mild-to-moderate PIH, UVR-induced PIH, and melasma-related hyperpigmentation.27,35,47,65-67 Changes in the appearance of hyperpigmentation, skin texture, and skin tone homogeneity can occur within 2 weeks of TXA treatment initiation. 66 Topical and oral formulations of TXA are both available; topical TXA is often preferred over oral formulations for its more favourable safety profile, as it is mostly associated with mild and transient adverse reactions including erythema, pruritus, and stinging. Oral TXA has also been used for the treatment of hyperpigmentation disorders and could be an alternative for individuals not responding well to topical formulations, although the evidence for its use is limited.66,68
Thiamidol
Thiamidol (4-n-butylresorcinol) is a highly potent and selective inhibitor of human tyrosinase and melanin production. 69 Several studies over the last decade have demonstrated the efficacy and safety of thiamidol for the treatment of dyspigmentation disorders, including melasma, acne-induced PIH, UVR-induced PIH, and laser-induced PIH.70-76 Notably, randomized controlled trials comparing thiamidol to hydroquinone have consistently shown thiamidol is generally well tolerated, with a low frequency of side effects that are mostly mild in nature and a low risk of allergic contact dermatitis.74-76 The role of thiamidol in the treatment of hyperpigmentation is comprehensively discussed in a recently published review by Frey et al. 74
Niacinamide
Niacinamide is a vitamin B3 derivative that inhibits the transfer of melanosomes from melanocytes to neighboring keratinocytes, and has been shown to be an effective skin-lightening agent.77-80 Topical niacinamide has been shown to dose-dependently and reversibly reduce facial hyperpigmentation, particularly when combined with N-undecylenoyl phenylalanine, N-acetyl glucosamine, TXA, vitamin C, or ultrasound enhancement.77-82 However, niacinamide has predominantly been tested in small studies of Asian women with hyperpigmented facial spots, with very limited evidence from well-controlled studies in other populations or in PIH specifically.77-80
Methimazole
Topical methimazole is a noncytotoxic and non-mutagenic compound that inhibits melanocyte peroxidase and metabolization of melanin intermediates, thereby interfering with melanin production and biosynthesis of eumelanin and pheomelanin.83,84 Although there is evidence supporting the use of methimazole in patients with melasma and hydroxyquinone-resistance, data on the efficacy in PIH are limited.65,83,84
Cysteamine
Cysteamine is a naturally occurring aminothiol with strong antioxidant activity that allows for lightening of the stratum corneum and inhibition of melanogenic enzymes including peroxidase and tyrosinase.85,86 Topical cysteamine may be an effective treatment for acne-induced and chronic severe PIH, but evidence is limited.46,65,86,87 In one small randomized, controlled trial, cysteamine improved quality of life and had similar efficacy and tolerability as a topical hydroquinone/ascorbic acid combination. 87 Topical cysteamine is generally well tolerated, with mild side effects including erythema, dryness, irritation, burning, and scaling.46,65,86,87
Glutathione
Glutathione is a naturally occurring thiol-tripeptide that helps maintain intracellular redox balance with antioxidant and anti-melanogenic properties and may also inhibit tyrosinase activity and switch melanogenesis from darker eumelanin to lighter pheomelanin synthesis.65,88 Glutathione is available in multiple formulations. Topical and oral glutathione are generally well tolerated and may increase skin lightening and reduce hyperpigmentation, although these effects appear to be temporary.65,88,89 However, evidence for the effect of glutathione on depigmentation (especially in PIH) is scarce. 89 In particular, oral glutathione may have poor bioavailability, and there are minimal and controversial data available for skin-lightening effects of intravenous glutathione.88,89
Chemical Peels
Deeper or dermal PIH may not respond sufficiently to topical agents and may require procedural interventions such as chemical peel or laser therapy, which can be considered as second-line or later options and are often used in combination with topicals.27,28,32,65,90 PIH is a common indication for chemical peels in individuals with skin of colour, although caution must be exercised for darker-skinned individuals since there is a risk of worsening hyperpigmentation (Supplemental Table S4).28,32,90 Chemical peels allow for controlled chemical injury to a specific skin depth, which stimulates new skin growth with potential improvements in skin surface, texture, and appearance.28,90 The exfoliation caused by chemical peels regulates epidermal thickness through uniform dispersion of basal layer melanin and deposition of new collagen with ground substance in the dermis. 90 A systematic review of treatment outcomes in PIH found that chemical peels alone resulted in 33.3% partial response and 66.7% poor-to-no response, while chemical peels combined with topical agents resulted in 85.7% partial response and 14.3% poor-to-no response. 35
A detailed patient history must be taken to minimize irritation and/or PIH exacerbation, with specific consideration of other dermatological conditions, herpes simplex virus (HSV) infection history, skin type, concomitant medications, and previous reactions to cosmetic interventions.28,91,92 Further, the specific chemical peel agent used should be carefully selected to avoid skin reactions and irritation, which can lead to worsening PIH or new lesions.28,92 Superficial chemical peels are typically well tolerated and effective in patients with skin of colour.28,91,92 Currently, the most commonly used chemical peel agents include glycolic acid and salicylic acid.28,90,93 Mandelic acid (usually in combination with salicylic acid) is another useful peeling agent that offers slower penetration and potentially less irritation than glycolic acid given its larger molecular size.94-96
Laser/Light-Based Therapies
Although topical treatments remain the standard of care for PIH, melanin’s wide absorption spectrum as a chromophore and laser therapy’s more recent emergence as a dermatological treatment have led to the exploration of laser therapy as a treatment for PIH.31,37 Laser/light-based therapies may be effective as an alternative or adjunct therapy (often in combination with topicals) and considered as second-line or later options in patients with a suboptimal response to topicals and/or with dermal PIH.27,37,39 However, the device and settings must be carefully selected for each individual to avoid excessive heat production and adverse effects such as worsening hyperpigmentation (Supplemental Table S5).27,37,39 This is of particular importance for patients with skin of colour and/or with dermal PIH with blue-grey or black pigmentation.27,37,39 Shorter wavelength lasers may be used in patients with lighter skin and epidermal PIH, whereas longer wavelength lasers may be used in patients with darker skin and dermal PIH. 37
Many studies have explored the use of lasers in PIH; however, most have methodological limitations and some findings suggest no improvement or worsening of PIH following laser treatment.31,37,39 In a systematic review of treatment outcomes in PIH, laser therapy resulted in 18.1% complete response, 61.2% partial response, and 18.1% poor-to-no response, whereas laser therapy combined with topical agents resulted in 3.6 % complete response, 84.5% partial response, and 11.8% poor-to-no response. 35 There are several different types of lasers and devices, including quality-switched (QS) devices (nanosecond or picosecond lasers) and fractional non-ablative laser devices.27,37 Currently, the QS Nd:YAG laser is the most frequently used laser for the treatment of PIH and has demonstrated promising effectiveness in several studies.31,37,97
Clinical Commentary
The pathophysiology, diagnosis, and optimal management of PIH remain incompletely defined, which is reflected in the lack of evidence-based guidelines for the clinical management of PIH. Given the gaps and limitations in available treatment options, it is imperative that clinicians educate patients with PIH both to minimize further exacerbation of pigmentation changes and to set realistic expectations of the trajectory of PIH and restoration of normal skin colour and tone. Notably, reversal of PIH can take several months or even years; in some cases, changes can be permanent. This can be further distressing to patients and amplify the substantial psychosocial burden associated with PIH. It can be reassuring to patients when their dermatologist acknowledges the psychosocial distress imposed by PIH, and provision of educational resources about PIH and its management is an important part of effective patient counselling. 6
Educating patients about sun protection is also an important part of PIH management. Patients may have misconceptions about the impact of sun tanning to even out pigmentation changes; this practice should be discouraged because it may exacerbate and perpetuate PIH and contribute to long-lasting dyspigmentation, perhaps especially in those with skin of colour, since UVR is a trigger for and can exacerbate hyperpigmentation. Notably, previous reports have indicated that sunscreen use is lower in African American and Asian American individuals than in non-Hispanic White individuals and can be influenced by cultural perceptions of sun protection, extent of knowledge regarding the risks associated with poor sun protection practices, and availability and cost of products. 98 Further, individuals with skin of colour may be less likely to learn about the importance of sunscreen from a dermatologist, more likely to prefer brands owned by others with skin of colour (which may not be broadly accessible), and have unique concerns associated with sunscreen use than non-Hispanic White people. 98 Therefore, an emphasis on culturally competent education and discussions regarding individual barriers to sunscreen use may improve adherence to appropriate sun protection practices.
Tools for objectively assessing and monitoring pigmentation changes in clinical practice are also lacking. The post-acne hyperpigmentation index (PAHPI) and melanin index have been developed and validated as outcome measures to assess pigmentation severity and to compare treatment efficacy in clinical trials. 99 For example, the PAHPI evaluates size, intensity, and quantity of lesions, and has good interindividual and intraindividual reliability and validity. 99 The melanin index is a sensitive measure that uses spectrophotometric comparison of pigmentation from involved and adjacent uninvolved skin of PIH lesions. 99 Although these tools may provide objective and efficient means of assessing treatment responses among individuals with PIH in the clinical trial setting, their applicability in routine practice is likely limited. The Woods lamp can be useful in distinguishing epidermal from dermal or mixed patterns of PIH, and this evaluation can be efficiently incorporated into practice. However, its utility in monitoring changes in pigmentation over time and response to treatment may be limited. From a practical perspective, simple measures such as serial photography, clinician rating of severity (eg, mild/moderate/severe or worsened/unchanged/improved posttreatment), and patient-reported measures (eg, patient global impression of change) could be implemented to monitor outcomes and document progress in routine care.
The pathophysiological mechanisms that underlie pigmentation changes are incompletely understood, particularly how they differ or overlap with endogenous stimuli such as inflammatory dermatoses. Whether the erythema component of PIH is associated with a specific pathophysiologic process or part of the active inflammation itself remains a controversial question without a clear answer. A better understanding of the pathophysiological processes involved in hyperpigmentation and hypopigmentation could ultimately uncover new targets for treatment.
In terms of treatment sequencing, escalation from topical to systemic and/or procedural therapy does not necessarily need to be considered incrementally. For example, dermal dyspigmentation may be less likely to respond to topical therapies than epidermal PIH and may warrant initiation of other modalities including laser and chemical peels. Such an approach could save the patient time and reduce exposure to agents that may have a lower chance of success. However, evidence to inform the optimal sequencing and timing for escalation of PIH treatment modality is highly limited, requiring clinicians to use their best judgment in recommending an initial approach for patients.
Finally, the impact of earlier control of inflammatory dermatoses on the emergence and/or duration of dyspigmentation and what factors define this “window of opportunity” for treatment remain questions of research interest that could have important clinical consequences.
Conclusions
PIH is a common dermatologic condition associated with cosmetic disfigurement, impaired psychosocial well-being, and reduced quality of life. PIH may persist for months to years following resolution of underlying pathophysiological mechanisms, highlighting the need for prompt diagnosis and management. It is also important for clinicians to consider PIH as a sequela of a preceding initial dermatologic condition or insult and a potential consequence of treatment during management planning. Patients with skin of colour have an increased risk of developing PIH, and there is a particularly high unmet need for improved recognition, education, and safe and effective treatments in this patient population.
In addition to prophylactic photoprotection, several treatment options are available for PIH, including topical agents, chemical peels, oral agents, chemexfoliation, and laser treatment; however, high-level clinical evidence for their use remains limited. Although dermatologists and other clinicians should consider treatment benefit relative to the risk of adverse reactions in patients with different skin tones, the lack of clear evidence for the available treatments presents an ongoing challenge for treatment decision-making. Future research exploring pathophysiological mechanisms of pigmentation changes may reveal new therapeutic targets that improve treatment success. In addition, efforts to standardize and validate tools used to characterize and assess pigmentation changes related to PIH in real-world clinical practice could improve diagnosis and monitoring of treatment effects. Finally, there remains a need for further research including high-quality randomized controlled trials, which would help inform evidence-based guidelines with the ultimate goal of improving care and outcomes for patients with PIH.
Supplemental Material
sj-docx-1-cms-10.1177_12034754261427395 – Supplemental material for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment
Supplemental material, sj-docx-1-cms-10.1177_12034754261427395 for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment by Geeta Yadav, Maxwell Sauder, Sanjay Siddha, Rami Zeinab, Maxime Barakat and Marissa Joseph in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-2-cms-10.1177_12034754261427395 – Supplemental material for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment
Supplemental material, sj-docx-2-cms-10.1177_12034754261427395 for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment by Geeta Yadav, Maxwell Sauder, Sanjay Siddha, Rami Zeinab, Maxime Barakat and Marissa Joseph in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-3-cms-10.1177_12034754261427395 – Supplemental material for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment
Supplemental material, sj-docx-3-cms-10.1177_12034754261427395 for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment by Geeta Yadav, Maxwell Sauder, Sanjay Siddha, Rami Zeinab, Maxime Barakat and Marissa Joseph in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-4-cms-10.1177_12034754261427395 – Supplemental material for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment
Supplemental material, sj-docx-4-cms-10.1177_12034754261427395 for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment by Geeta Yadav, Maxwell Sauder, Sanjay Siddha, Rami Zeinab, Maxime Barakat and Marissa Joseph in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-5-cms-10.1177_12034754261427395 – Supplemental material for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment
Supplemental material, sj-docx-5-cms-10.1177_12034754261427395 for A Review of Post-Inflammatory Pigmentation Changes: Pathophysiology, Diagnosis and Treatment by Geeta Yadav, Maxwell Sauder, Sanjay Siddha, Rami Zeinab, Maxime Barakat and Marissa Joseph in Journal of Cutaneous Medicine and Surgery
Footnotes
Acknowledgements
Medical writing assistance for this manuscript was provided by Christina Clark, MSc, of CC Medical Writing Inc; and Ari Mendell, MSc, PhD; and Kate Nicholson, MSc, PhD, of Compass Leaf Medical Communications Inc. Illustration created by Kryski Biomedia. This support was funded by Bausch Health Canada Inc.
Author Contributions
Geeta Yadav, Marissa Joseph, and Rami Zeinab were involved in study conception and design. All authors were involved in the acquisition, analysis, and interpretation of evidence. All authors were involved in drafting and critically reviewing/revising the manuscript and provided final approval of the manuscript.
Data Availability
No original data were generated in the development of this review article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Geeta Yadav has received honoraria for advisory board and speaker services from Bausch Health and L’Oreal. Maxwell Sauder has received honoraria, consulted and/or served as an investigator for Bausch Health, Janssen, and L’Oreal. Sanjay Siddha serves as an investigator for Bausch Health and has served as a consultant, adviser and/or speaker for Bausch Health and L’Oreal. Rami Zeinab and Maxime Barakat are employees of Bausch Health Canada Inc. Marissa Joseph has received honoraria for advisory board and speaker services from Bausch and L’Oreal.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Bausch Health Canada Inc.
Ethical Considerations
No original data were generated in the development of this review article; therefore, ethics approval was not required.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
