Abstract
Background:
Hidradenitis suppurativa (HS) is a highly comorbid condition understudied in pediatric populations. Furthermore, the underrepresentation of Canadians in HS research limits our evaluation of care in real-world Canadian settings.
Objectives:
To describe the demographics, clinical characteristics, psychosocial burden, and management of pediatric HS at a Canadian hospital.
Methods:
We developed the infrastructure for a Canada-wide HS registry and piloted it at a pediatric HS tertiary care centre. A retrospective review of patients seen was performed using data collection forms (DCFs) developed for the registry.
Results:
The 39 patients included were 53.8% female and 100.0% nonsmokers. Asians were overrepresented compared to other North American studies. Most had Hurley stage II (60.5%) or III (28.9%) disease. Mean delay to diagnosis was 2.1 ± 2.0 years. Obesity (57.9%), acne (30.7%), folliculitis (23.1%), and trisomy 21 (23.1%) were the most common comorbidities. Patients with trisomy 21 had an age of HS onset 3 years younger (P = .0087), and 44.0% had concomitant disseminate and recurrent infundibulofolliculitis. Moderate-to-severe quality of life (QOL) impact was seen in 56.8% of patients, and 17.6% had reported bullying. Of patients prescribed biologics (59.0%), most used 1 (56.6%), while 39.1% switched to a second and 4.3% to a third biologic. Most adalimumab-initiated patients clinically improved (81.0%), but only 2 patients achieved complete remission. Follow-up patients (n = 14) had significant improvements in QOL (P = .038) and pain (P = .026) scores.
Conclusions:
Distinct demographics, severe disease, and low remission rates characterized our complex HS cohort. Prospective use of these DCFs will enhance our understanding of HS in Canada.
Introduction
Hidradenitis suppurativa (HS) is a chronic, inflammatory condition characterized by recurrent, painful nodules, abscesses, sinus tracts and scarring predominantly involving apocrine gland-bearing regions of the skin. It has a prevalence of approximately 1% of individuals worldwide, up to 3.8% in Canada, and has been reported to disproportionally affect women and individuals of African descent.1 -3 There are 2 peaks in the age of onset, the first during teenage years, followed by a second peak in the mid-40s. 4 Compared with adults with HS, children have higher rates of obesity, acne, and polycystic ovary syndrome. 5 The psychosocial impact of HS can also be particularly debilitating for pediatric patients, correlating with increased rates of bullying, absenteeism, and adolescent depression. 6
Despite nearly 30% of patients with HS experiencing an onset of disease before the age of 18 years, pediatric HS remains understudied and undertreated. 6 Knowledge gaps exist around the pathogenesis, phenotypic variability, clinical outcomes, and prognostic indicators of this population. Furthermore, there is inadequate representation of Canadian patients in the literature as the majority of data were derived from US-based studies. 2 There is a need to evaluate these gaps in a real-world setting that includes the range of diverse patient backgrounds and different clinical features of HS seen in Canada. In this pilot study, we provide a preliminary report of the demographic characteristics, clinical phenotypes, management strategies and treatment outcomes for patients seen at Canada’s first multidisciplinary pediatric HS clinic.
Materials and Methods
Study Design
We developed the infrastructure for a Canada-wide HS registry and piloted it at a single pediatric HS clinic at a tertiary care centre in Toronto, Ontario. Patients seen at this clinic required an established clinical diagnosis of HS, with a failure of 3 months of systemic antibiotics or complex comorbidities. Appointments consisted of comprehensive comorbidity screening, completion of validated quality of life (QOL) and mental health questionnaires [Children’s Dermatology Life Quality Index (cDLQI), 7 Patient Health Questionnaire-9, 8 Generalized Anxiety Disorder-7, 9 pain visual analogue scale (VAS) 10 integrated with the pediatric-validated faces scale, 11 and a modified worst itch numeric rating scale (NRS) 12 , and assessment by a multidisciplinary team. This team consisted of pediatric dermatologists, wound care nurses, and a social worker. Follow-ups occurred on an annual basis until a coordinated transition could be made to adult HS care. We performed a Research Ethics Board-approved retrospective review of patients seen to date, using data collection forms (DCFs) developed for the registry. Consent was waived for retrospective DCF piloting. Data were stored on a secure REDCap database.
Statistical Analysis
Descriptive statistics were used for continuous and discrete numerical variables, and proportions were used for categorical variables to outline the demographic and clinical parameters of HS during the initial consultation appointment (“baseline”). Baseline subanalyses between groups of interest were performed using the Mann-Whitney U test. Differences in disease severity and QOL scores were assessed for baseline and follow-up visits using the McNemar test for binary categorical data, and the Wilcoxon signed-rank test for paired ordinal data, where P values <.05 were considered statistically significant. All statistical analyses were performed using Python (version 3.8).
Results
In total, 39 patients were included (Table 1). Mean age at intake was 15.6 ± 1.9 years (range 10-17), and 53.8% (21/39) were female. Mean age of symptom onset and delay to diagnosis were 10.9 ± 3.4 and 2.1 ± 2.0 years, respectively. The most frequently reported comorbidity was obesity (57.9%, 22/38). At baseline, patients had a mean body mass index (BMI) of 31.6 ± 9.0 kg/m2 and a z-score of 2.22 ± 1.38. Other common comorbidities included acne (30.7%, 12/39), folliculitis (23.1%, 9/39), trisomy 21 (23.1%, 9/39), and developmental delay (21.0%, 8/39). The 9 patients with trisomy 21 had a significantly younger mean age of HS symptom onset than those without (8.4 ± 3.2 years vs 11.7 ± 3.1 years, P = .0087), and 4 (44.4%) had comorbid disseminate and recurrent infundibulofolliculitis (DRIF). Psoriasis was reported in 6/39 patients, of which 5 were deemed likely a paradoxical reaction to adalimumab. Four patients had other components of the follicular occlusion tetrad (2 with acne conglobata, 2 with pilonidal disease, 0 with dissecting cellulitis). Among female patients, 95.2% were menarcheal with a mean age of menses onset of 11.4 ± 0.99 years. Family history of HS was reported in 20.5% (8/39) of patients, with 62.5% (5/8) of those being a first-degree relative. No patients reported smoking cigarettes.
Baseline Demographics and Outcome Measures of Pediatric HS Patients.
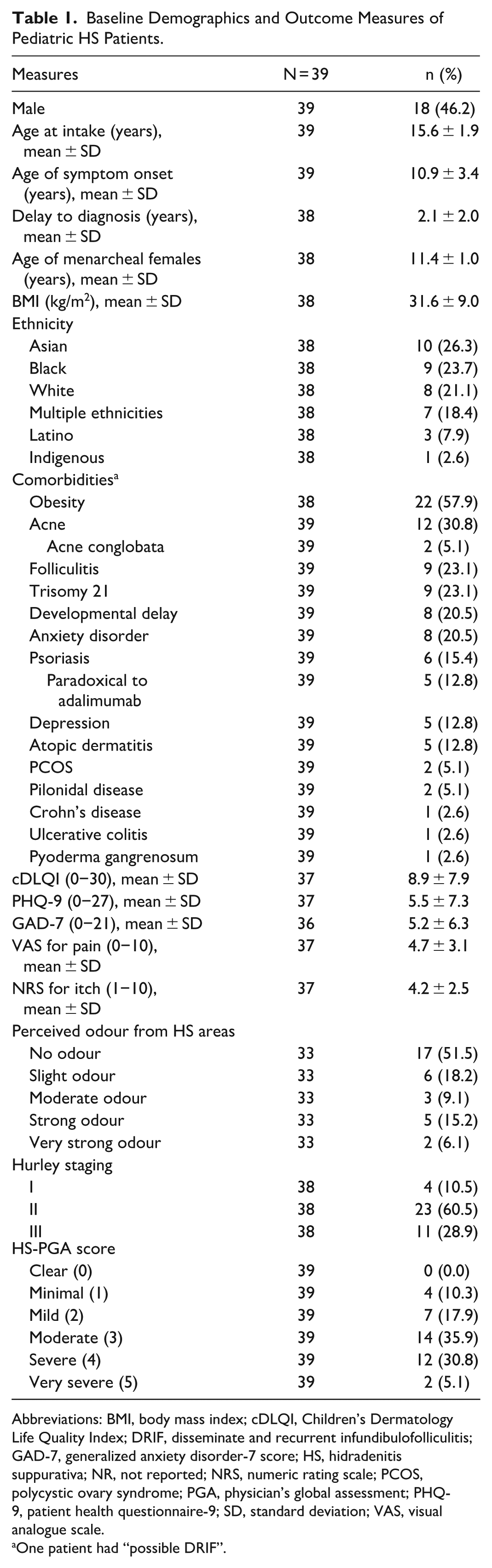
Abbreviations: BMI, body mass index; cDLQI, Children’s Dermatology Life Quality Index; DRIF, disseminate and recurrent infundibulofolliculitis; GAD-7, generalized anxiety disorder-7 score; HS, hidradenitis suppurativa; NR, not reported; NRS, numeric rating scale; PCOS, polycystic ovary syndrome; PGA, physician’s global assessment; PHQ-9, patient health questionnaire-9; SD, standard deviation; VAS, visual analogue scale.
One patient had “possible DRIF”.
With regard to HS symptoms, 69.2% of patients reported the presence of drainage, and 41% reported odour. Mean pain score (VAS) was 4.7 ± 3.1, and mean itch score (NRS) was 4.2 ± 2.5. Depression and anxiety questionnaire scores of moderate or greater severity were reported in 21.6% (8/37) and 22.2% (8/36) of patients, respectively. Moderate-to-severe QOL impact was reported in 56.8% of patients. Current or previous bullying was reported in 17.6% of the cohort.
Disease severity assessment revealed predominance of Hurley stage II disease (60.5%), with 28.9% presenting with Hurley stage III disease. Only 4 patients presented with Hurley stage I disease; these patients were being maintained on systemic antibiotics and all had complex comorbidities. This severity was also demonstrated in the HS-Physician’s Global Assessment (PGA) score, where 35.9% of patients presented with severe (4) or very severe (5) disease. At the time of baseline assessment, 64.1% of patients were considered to be flaring. Lesion distribution, which morphologically consisted of inflammatory nodules, abscesses, draining fistulae, sinus tracts, tombstone comedones, and/or scarring, most commonly involved the groin (87.2%), axillae (84.6%), and buttocks (69.2%; Figure 1). Atypical site involvement was observed in a substantial proportion of patients, including head and neck (23.1%), chest (38.5%), and abdominal (28.2%) involvement. In this cohort, axillary and groin involvement was frequently observed across all Hurley stages. However, Hurley stage I patients had predominantly groin (75.0%) and axillary (50.0%) involvement with some posterior involvement (25.0% back, 25.0% buttocks). In Hurley stage II disease, there was increased involvement of anterior sites including chest (52.2%) and abdomen (34.8%), along with higher rates of axillary (82.6%) and groin (87.0%) involvement. Hurley stage III disease demonstrated the most extensive involvement, with all patients having axillary and groin involvement (100.0%), high rates of buttock involvement (90.9%), but lower prevalence of atypical site involvement than Hurley stage II patients. Patients with atypical site involvement had significantly higher mean BMI than patients without atypical sites (BMI 35.3 ± 9.3 kg/m2 vs BMI 27.8 ± 6.6 kg/m2, P = .0093); age, gender, ethnicity, and presence of trisomy 21 did not differ significantly between groups (P > .05).
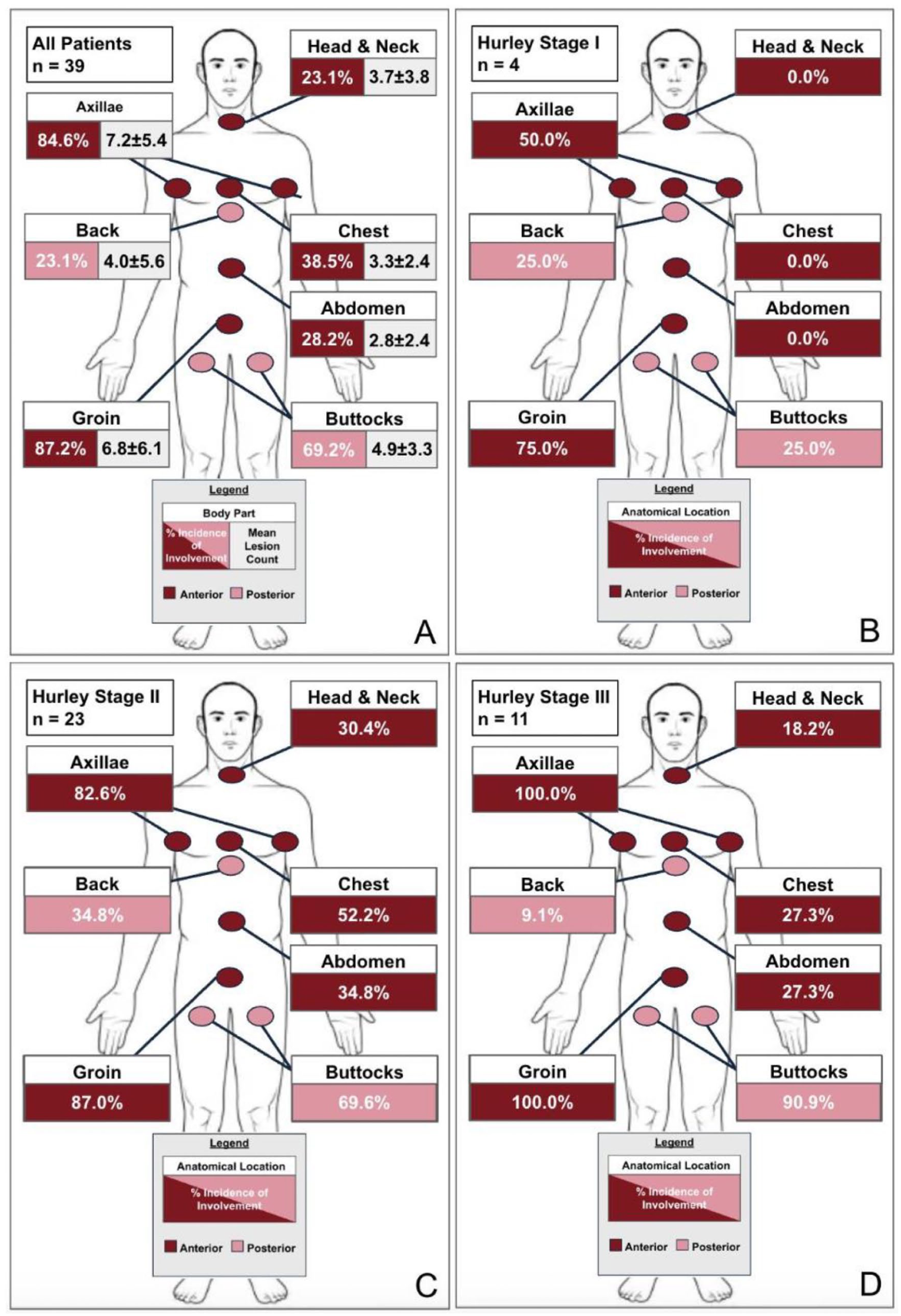
Baseline lesion distribution of pediatric HS patients overall and by Hurley stage. (A) Overall lesion distribution and mean lesion count among pediatric HS cohort; (B) lesion distribution among pediatric HS patients with Hurley stage I disease; (C) lesion distribution among pediatric HS patients with Hurley stage II disease; (D) lesion distribution among pediatric HS patients with Hurley stage III disease. HS, hidradenitis suppurativa.
Baseline laboratory investigations revealed mildly elevated inflammatory markers, with 84.4% of patients having elevated C-reactive protein and 55.2% having elevated erythrocyte sedimentation rate (Table 2). Many also exhibited lipid panel abnormalities, with low high-density lipoprotein cholesterol in 75.8% of patients and elevated triglycerides in 55.9% of patients. Glycemic control remained largely within normal limits. Vitamin D deficiency was present in 74.2% of patients. Zinc deficiency was found in 30.3% of patients. Of the 13 patients for whom fecal calprotectin testing was ordered, 5 were on adalimumab at the time of testing and 7 were not. Elevated fecal calprotectin levels >50 µg/g were seen in 40.0% (2/5) of patients on adalimumab therapy, compared with those in 57.1% (4/7) of patients who were not. However, of these 6 total patients with elevated fecal calprotectin values, most (5/6) were mildly elevated (<200 µg/g). Only 1 patient had clinically significant elevations in fecal calprotectin (1801 µg/g). This patient was symptomatic and had endoscopic findings consistent with inflammatory bowel disease (IBD). They were not on adalimumab at the time of fecal calprotectin testing.
Baseline Laboratory Values for Pediatric HS Patients.
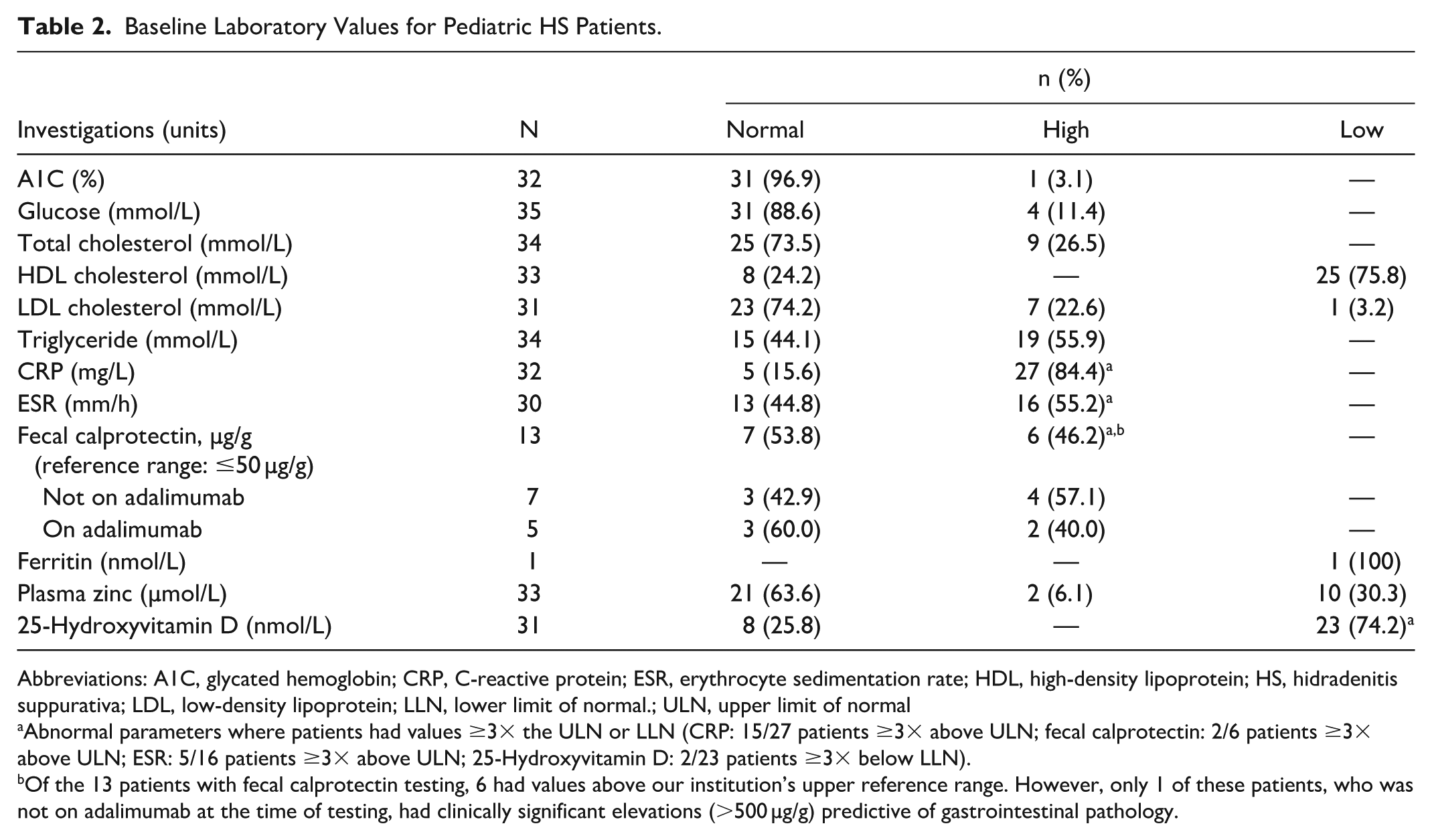
Abbreviations: A1C, glycated hemoglobin; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HDL, high-density lipoprotein; HS, hidradenitis suppurativa; LDL, low-density lipoprotein; LLN, lower limit of normal.; ULN, upper limit of normal
Abnormal parameters where patients had values ≥3× the ULN or LLN (CRP: 15/27 patients ≥3× above ULN; fecal calprotectin: 2/6 patients ≥3× above ULN; ESR: 5/16 patients ≥3× above ULN; 25-Hydroxyvitamin D: 2/23 patients ≥3× below LLN).
Of the 13 patients with fecal calprotectin testing, 6 had values above our institution’s upper reference range. However, only 1 of these patients, who was not on adalimumab at the time of testing, had clinically significant elevations (>500 µg/g) predictive of gastrointestinal pathology.
At baseline, systemic antibiotics (79.5%), biologics (56.4%), and involvement of other specialists (48.7%) were frequently recommended by the multidisciplinary team (Table 3). Notably, surgical intervention was recommended in 30.8% of patients.
Treatment Recommendations for Pediatric HS Patients at Baseline.
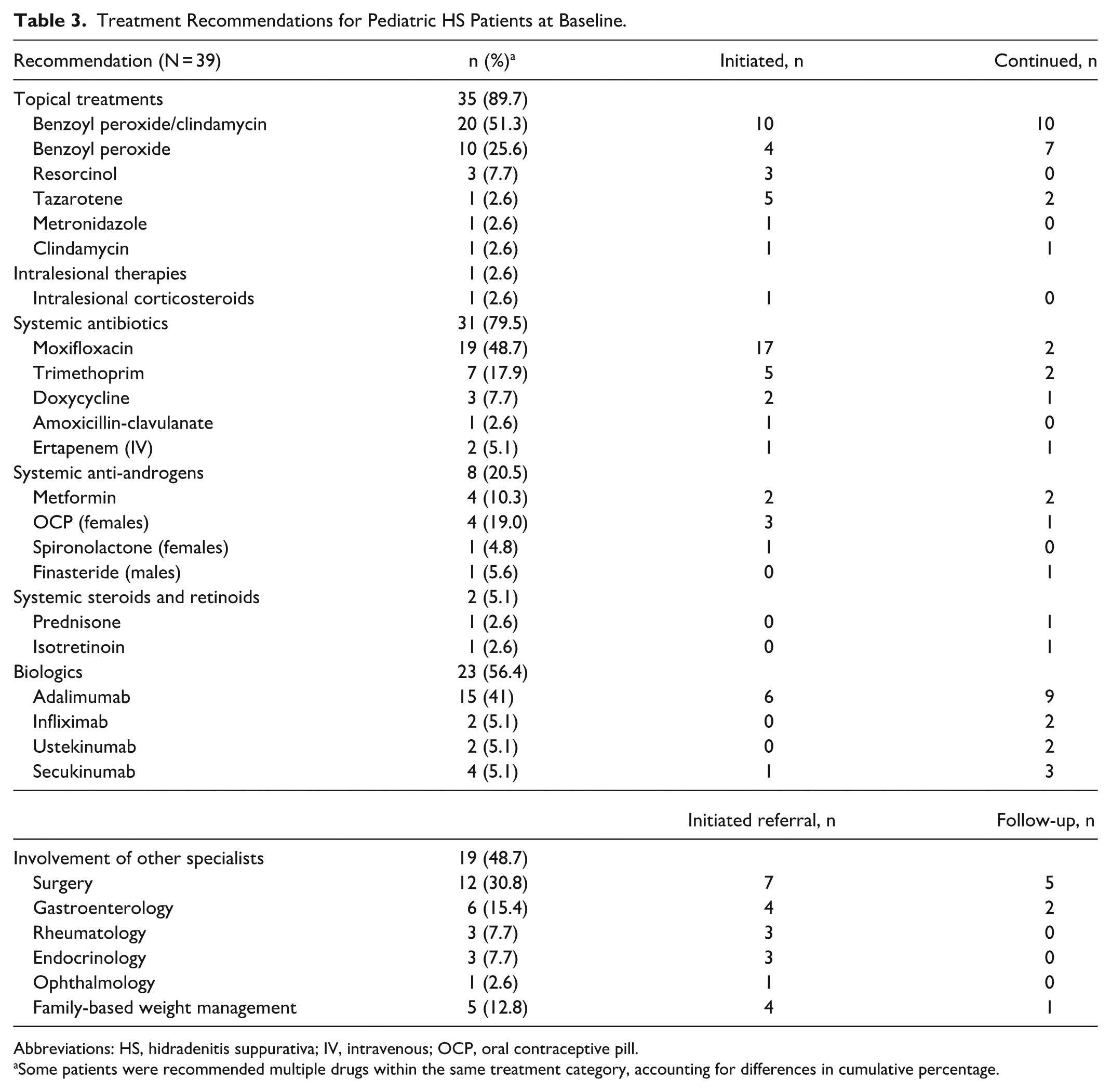
Abbreviations: HS, hidradenitis suppurativa; IV, intravenous; OCP, oral contraceptive pill.
Some patients were recommended multiple drugs within the same treatment category, accounting for differences in cumulative percentage.
Across patients’ entire disease course, the most commonly prescribed systemic treatments were doxycycline (71.8%, 28/39), moxifloxacin (64.1%, 25/39), and adalimumab (53.8%, 21/39). Biologics had been prescribed for 59.0% (23/39) of patients, of which, 39.1% (9/23) reported 2 different biologics, and 4.3% (1/23) reported 3 different biologics (Figure 2). Previous procedural or surgical HS interventions were reported in 20.5% (8/39) of patients. Of these patients, 7/8 had I&Ds, 2/8 had HS deroofings, 1/8 had radiofrequency, and 1/8 had wide local excision surgery.
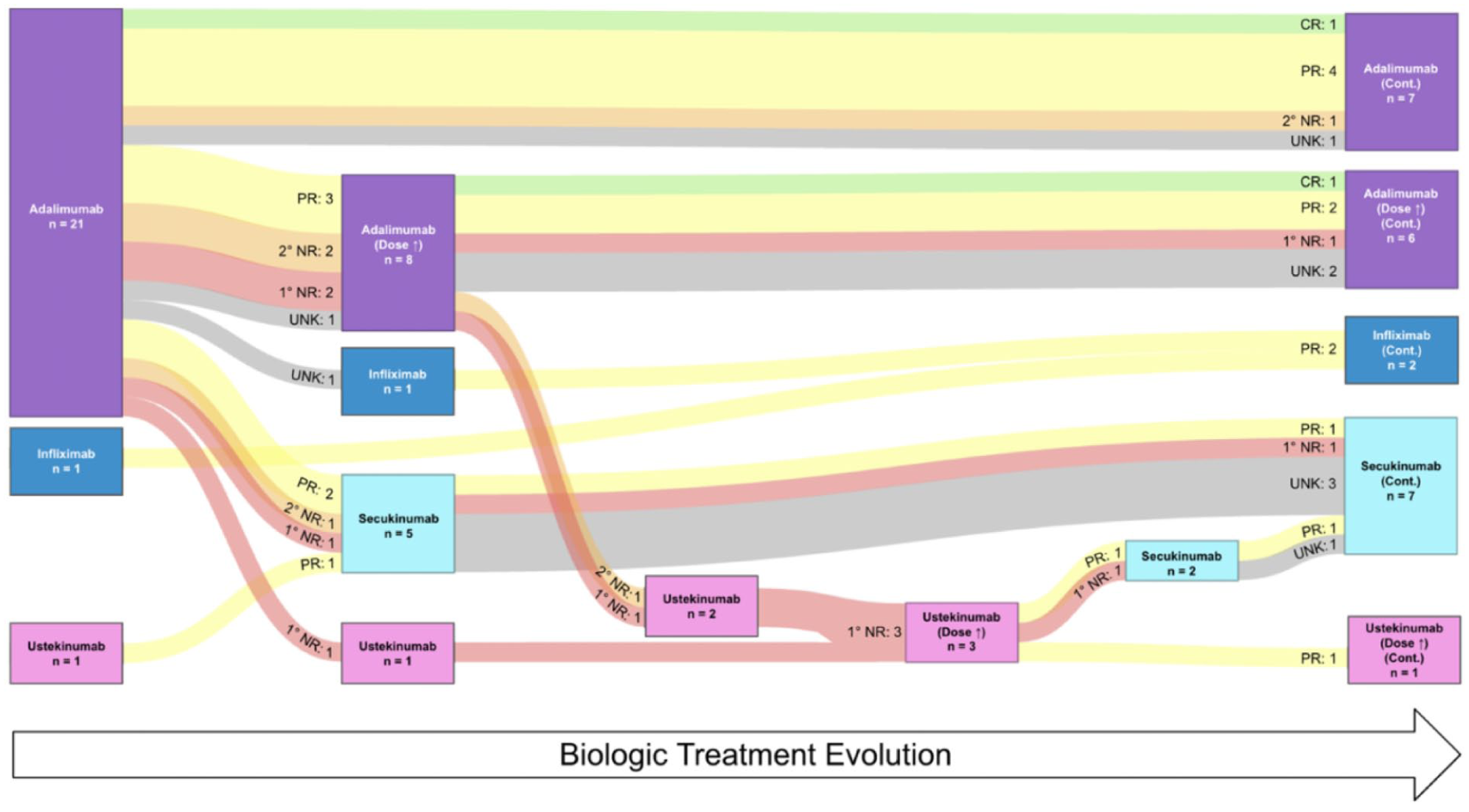
Sankey diagram of biologic treatment evolution and therapeutic response. Patients with UNK response either had since transitioned to adult care, had initiated the biologic just prior to baseline or follow-up with treatment duration that was inadequate to determine response, or had not attended a follow-up visit since initiation of the biologic. 1° NR, primary non-responder; 2° NR, secondary nonresponder; cont., continued; CR, complete remission; PR, partial response; UNK, unknown.
Figure 2 provides a depiction of biologic treatment evolution and associated response to therapy. Overall, 81.0% (17/21) patients started on adalimumab showed some (partial, complete, or initial) clinical response. However, both primary and secondary nonresponse was observed across all biologic classes. Complete remission was observed in only 8.7% (2/23) of biologic-prescribed patients, both of whom were on adalimumab. Among the 21 patients initiated on adalimumab, 7 remained on their prescribed dose without optimization, with 5 demonstrating sustained clinical improvement (1 complete remission, 4 partial response), with a mean time to maximum response of 7 months. Dose optimization occurred in 8 patients on adalimumab, with 3 experiencing sustained clinical improvement (1 complete remission, 2 partial response). Among the 5 other patients on a dose-optimized adalimumab regimen, there were 2 primary nonresponders, 1 secondary nonresponder, and 2 patients with unknown outcomes. All 3 patients who switched to ustekinumab following nonresponse to adalimumab also failed to respond to ustekinumab initially, but 2/3 showed partial response after ustekinumab dose optimization. Of the 7 patients who switched to secukinumab (5 from adalimumab, 2 from ustekinumab), 2 patients achieved partial response (after partial response to adalimumab and dose-optimized ustekinumab, respectively), 1 patient was a primary nonresponder (after primary nonresponse to adalimumab), and 4 patients had no follow-up outcomes at the time of review.
Table 4 provides a comparison of clinical outcome measures during baseline and follow-up appointments among those who attended both visits (n = 14). The median time to follow-up was 364 (IQR 317-371) days. There was a statistically significant improvement (decrease) in mean scores at follow-up for total cDLQI score (9.4-5.4, P = .038) and pain VAS (5.4-3.3, P = .026). A nonsignificant decrease in mean itch NRS (4.9 to 4.4, P = .360), anxiety (5.6-5.0, P = .500) was observed, as was a nonsignificant increase in depression scores (4.7-5.9, P = .939). Improvement in odour scores were marginally nonsignificant (P = .052). At follow-up, more patients with previously severe (4) or very severe (5) HS-PGA scores reported clear (0) to moderate (3) scores, but this difference was not statistically significant (P = .146).
Comparison Between Baseline and Follow-Up Outcomes in Pediatric HS Patients Who Attended Follow-Up.
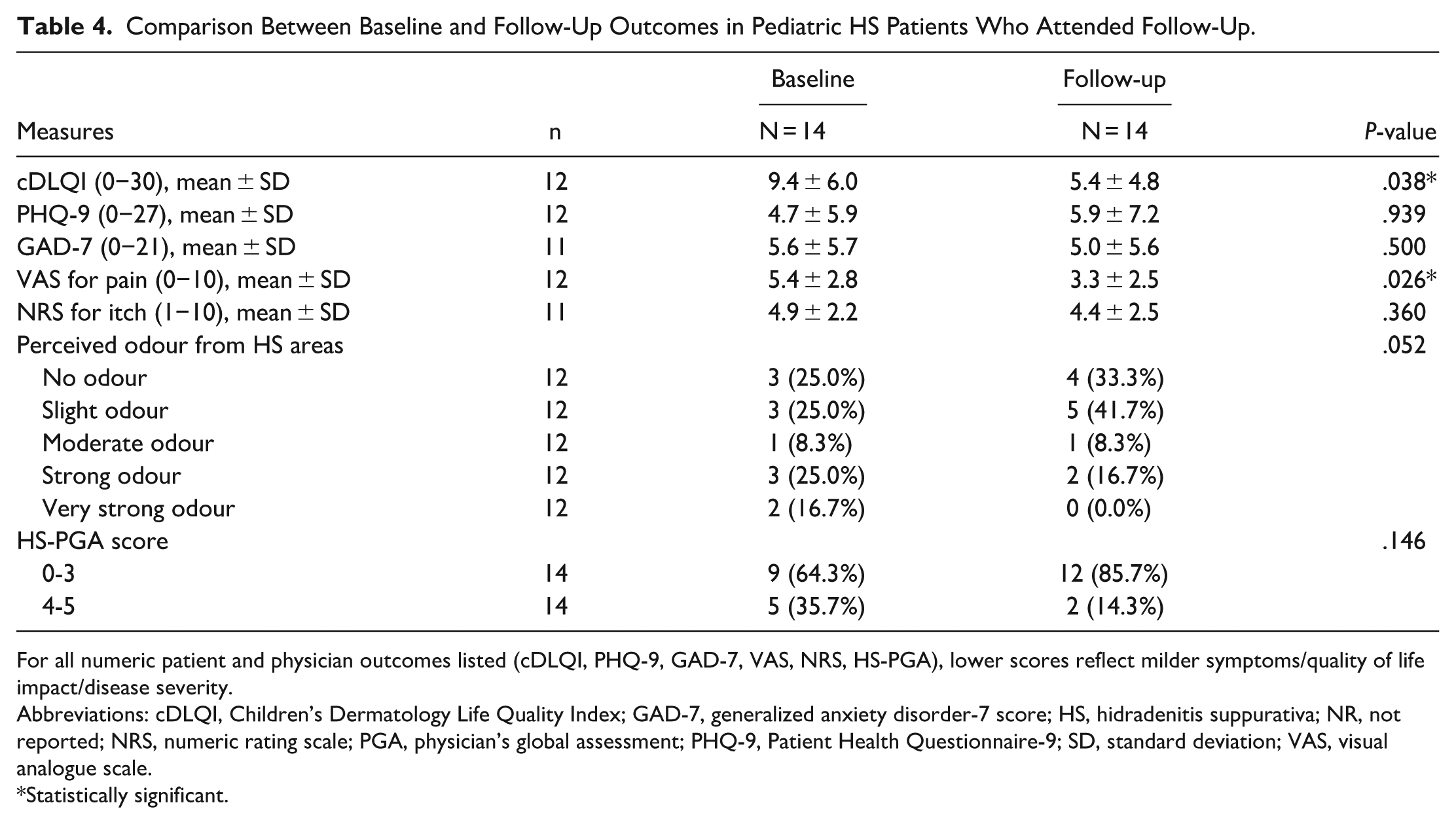
For all numeric patient and physician outcomes listed (cDLQI, PHQ-9, GAD-7, VAS, NRS, HS-PGA), lower scores reflect milder symptoms/quality of life impact/disease severity.
Abbreviations: cDLQI, Children’s Dermatology Life Quality Index; GAD-7, generalized anxiety disorder-7 score; HS, hidradenitis suppurativa; NR, not reported; NRS, numeric rating scale; PGA, physician’s global assessment; PHQ-9, Patient Health Questionnaire-9; SD, standard deviation; VAS, visual analogue scale.
Statistically significant.
Discussion
In this pilot study, we created DCFs for an HS registry that successfully facilitated robust data capture and retrospective analysis of the pediatric patients with HS referred to our tertiary care centre. We observed several demographic and clinical characteristics that both corroborate and diverge from existing literature. An international multicentre analysis by Liy-Wong et al examining 481 pediatric HS patients revealed a 2 year diagnostic delay, with symptom onset at age 12.5 years and formal diagnosis occurring at 14.4 years. 13 Patients were predominantly female (80%), had high rates of HS family history (41%), and frequently had comorbid obesity (65%) and acne vulgaris (29%). 13 In our cohort, the mean diagnostic delay of 2.1 ± 2.0 years aligns with these findings. However, our age of symptom onset of 10.9 ± 3.4 years was younger than most reported cohorts. A meta-analysis by Lam et al demonstrated a significantly younger age of HS symptom onset for patients with trisomy 21 (−6.24; 95% CI, −10.01 to −2.24). 14 This younger age, with a notable prevalence of trisomy 21 as well as Hurley stage III disease in nearly 30% of patients, reflects the complexity of patients referred to our institution.
Furthermore, while epidemiological studies have demonstrated gender variation of HS by geographic origin, the near-equal gender distribution observed contrasts with the established female predominance in both adult and pediatric North American and European cohorts.13,15 -17 The gender distribution observed at our institution more closely mirrors those of Eastern countries, where male predominance or equal gender distributions is found.18,19 Indeed, the ethnic distribution of our cohort was 26.3% Asian, 23.7% Black, 21.1% White, 7.9% Latino, 2.6% Indigenous, and 18.4% with multiple ethnicities. This distribution differs from previous North American epidemiological studies in pediatric HS, which have consistently demonstrated comparatively higher prevalence among Black populations and lower prevalence among Asian populations.20,21 Some adult HS studies have suggested that the higher prevalence of cigarette smoking among male patients from South, Southeast, and East Asian countries likely contributed to these gender differences; however, cigarette smoking was not reported by any patient in our pediatric cohort.17,19,22,23 Alternatively, Lee et al suggested that this difference may reflect higher prevalence of γ-secretase genes NCSTN, PSENEN, and PSEN1 among Asian populations.18,24 The genetic basis of HS typically manifests in 2 distinct patterns when inherited as a monogenic trait. The first involves alterations in the Notch/γ-secretase pathway, where 41 sequence variants have been identified and which produces predominantly comedonal disease, while the second involves mutations affecting inflammasome function, resulting in a systemic inflammatory phenotype often accompanied by arthritis. 25 The relatively high proportion of Asian patients in our cohort may reflect such genetic variations, regional demographics of central Ontario, or may suggest potential under-recognition of HS in Asian patients in previous studies.
Several comorbidities in our cohort were highlighted. The high rate of obesity aligns with established metabolic associations in pediatric HS. However, the 23.1% prevalence of trisomy 21 substantially exceeds previously reported rates of approximately 2.4% in pediatric HS populations. 26 Many (4/9) of our patients with trisomy 21 also had concomitant DRIF, which may be underdiagnosed when assessed as routine folliculitis. There is a significantly increased likelihood of HS in patients with trisomy 21. 14 This elevated risk of HS in trisomy 21 may be attributable to impaired Notch signalling through overexpression of amyloid precursor protein (APP), which is found on chromosome 21.14,27,28 APP functions as a competitive substrate for γ-secretase, and its overexpression promotes keratinocyte hyperproliferation and follicular plugging. 29 While some trisomy 21 cohorts have documented the presence of atypical HS site involvement, 30 it is unclear whether this is at rates greater than patients without trisomy 21, and this was also not a finding in our study. Instead, patients with atypical sites had a significantly higher BMI than those without, suggesting that friction or occlusion of these non-intertriginous sites may play a role. Interestingly, this did not translate to a higher prevalence of Hurley III disease in these patients; rather, Hurley II disease was overrepresented, similar to findings by João et al. 31 While the higher comorbidity prevalence in our cohort could suggest differential rates of underlying genetic susceptibility, it is more likely a reflection of the increased clinical and psychosocial complexity of this cohort.
The high prevalence of paradoxical psoriasis observed in our cohort (5/6 psoriasis cases, 83.3%) illustrates the complex immunological implications of TNF inhibition in this population. This phenomenon is thought to be mediated through dysregulated plasmacytoid dendritic cell activation and overexpression of type I interferons. 32 Among patients with paradoxical psoriasis who discontinued tumor necrosis factor inhibitor (TNFi) therapy (4/5), 3/4 were treated with secukinumab. One patient had a significant improvement in both psoriasis and HS, another had persistence of both conditions, and the third patient did not have follow-up data available. Notably, 1 patient managed their psoriasis with topicals while remaining on TNFi therapy, as their psoriasis was responsive to these topicals unlike their HS. They also had a positive family history of Crohn’s disease, which made class switching to anti-IL-17s a less attractive option. 33
In our cohort, those who underwent fecal calprotectin testing while on adalimumab had lower mean values than those without. Only 1/13 patients tested had elevated fecal calprotectin levels considered to be a positive screen for IBD (>500 µg/g). The fecal calprotectin test is a nonspecific measure of intestinal inflammation; mild elevations <300 µg/g can often result from NSAID use, alcohol, and/or other causes of low-grade intestinal inflammation. 34 The utility of fecal calprotectin screening in patients concurrently receiving adalimumab therapy may be limited, given the potential masking and treatment of unrecognized IBD with adalimumab. In patients with perceived IBD risk, obtaining a baseline fecal calprotectin may be a reasonable screening tool, either prior to suppression by adalimumab or prior to anti-IL-17 initiation. 35
This pediatric cohort demonstrated significant disease burden at baseline, with 56.8% of patients reporting moderate-to-severe impact on the cDLQI. The prevalence of moderate-severe depression (21.6%) and anxiety (22.2%) symptoms, as well as reports of bullying, reaffirms the psychological impact of early-onset disease. The integration of social work was particularly emphasized in our clinic model, which provided ongoing psychosocial assessment, counselling, and community resource connection. Given the overall improvements in QOL metrics over time, this integrated approach appears beneficial for addressing both the physical and psychosocial burden of pediatric HS.
Study limitations include the single-centre design, retrospective methodology, and potential referral bias. Additionally, we were underpowered to perform additional subgroup analyses owing to our limited sample size and availability of follow-up data. Lastly, among Asian patients, we did not differentiate between East, Southeast, and South Asian individuals in our analysis, despite their genetic heterogeneity, limiting the conclusions we can draw from these populations.
Conclusion
Our preliminary analysis revealed distinct demographic characteristics among the pediatric HS patients seen at our centre. Notable findings include comparatively higher Asian prevalence, near-equal gender distribution, and higher prevalence of trisomy 21 and paradoxical psoriasis. Expansion of our database to additional other Canadian sites is currently underway. Future prospective data collection with these DCFs across adult and pediatric HS centres will provide improved long-term characterization of patients in Canada with HS.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Irene Lara-Corrales has received honoraria for consulting and/or speaking for Novartis, Sanofi, and UCB and has received research support from AbbVie. Dr Raed Alhusayen has received honoraria for consulting and/or speaking for AbbVie, Fresenius Kabi, Incyte, Janssen, Novartis, Pfizer, and UCB and received research funding from AbbVie, Incyte, and Jansen. Latoya Palmer has received honoraria or fees for consulting and/or speaking for Novartis and Boehringer Ingelheim Pharmaceuticals. Dr David Croitoru has received consultation fees and educational grants from AbbVie, Amgen, Arcutis, Bausch Health, BioJAMP, Boehringer-Ingelheim, Eli-Lilly, Janssen, Leo, Novartis, Pfizer, Sanofi, Sandoz, Sun Pharmaceuticals, and UCB. Dr Marissa Joseph has received honoraria consulting and/or speaking from, AbbVie, Amgen, Arcutis, Bausch, Boehringer Ingelheim, BMS, Celgene, Eli Lilly, Galderma, Incyte, Janssen, Kenvue, Leo, Loreal, Novartis, Pierre Fabre, Pfizer, Sanofi Genzyme, Sun Pharma, and UCB. Dr Vincent Piguet has received grants from AbbVie, Bausch Health, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Eli Lilly, Incyte, Janssen, LEO Pharma, L’Oréal, Novartis, Organon, Pfizer, Sandoz, and Sanofi; received payment or honoraria for speaking engagement from Sanofi; participated on an advisory board for LEO Pharma, Novartis, Sanofi, Union Therapeutics, Abbvie, and UCB; and received equipment donation from L’Oréal. There are no further disclosures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Skin Investigation Network of Canada (SkIN Canada).
