Abstract
Bullous pemphigoid (BP) is an autoimmune disease of the skin characterized by subepidermal blistering accompanied by severe itch, causing a profound decrease in quality of life and conferring significant mortality risk. Because age is a primary risk factor, this rare condition is becoming more common as our population ages. Consequently, there is a need for enhanced recognition and appropriate management of BP. The objective of this endeavour was to develop a series of practical recommendations to improve the diagnosis and management of BP based on available evidence and expert opinion where evidence was lacking, with a focus on disease management within the Canadian context. A panel of 9 Canadian dermatologists with interest and experience in BP identified key topics in its diagnosis and management. A broad literature review, along with clinical expertise and opinion, supported the development of manuscript sections on each identified topic. Where appropriate, the panel developed clinically relevant recommendations that were adopted by consensus following a modified Delphi process and prespecified agreement cut-off of 80%. Panel members reviewed draft consensus statements and rated their level of agreement with each using an anonymous online survey platform. Statements not achieving consensus were discussed and updated in a live virtual meeting, after which another round of anonymous voting was held. Through this process, 18 recommendations were approved by the panel. These statements can guide healthcare providers in the practical management of BP in Canada and beyond.
Keywords
Introduction
As the number of elderly people in Canada increases, so will the incidence of diseases where age is a primary risk factor. The number of Canadians over the age of 75 is predicted to double in the time period from 2017 to 2037, 1 and this changing demographic will require physicians to become more familiar with the treatment of conditions that were once less common.
One such example is bullous pemphigoid (BP), an autoimmune disease of the skin characterized by subepidermal blistering accompanied by severe itch. It has a considerable impact on quality of life and is associated with a nearly 3-fold increase in 1-year standardized mortality risk. 2 While relatively rare in the general population (2.4-23 cases/million), incidence increases 10-fold among individuals 80 years and older (190-232 cases/million). Overall incidence of BP has increased ~2- to 4-fold over the past 2 decades, and this trend can be expected to continue given our aging population. 3
While formal guidelines for the management of BP have been published for European jurisdictions, 4 there are key considerations that should be emphasized for the Canadian context. The purpose of this document is to guide the assessment and treatment of BP through a series of recommendations derived via a consensus process involving an expert panel of Canadian dermatologists.
Materials and Methods
An expert panel of 9 Canadian dermatologists was assembled, with panel members identified and invited by a steering committee representing the Dermatology Association of Ontario (F.E.L., M.B.S., P.L., and A.M.) based on their expertise and interest in this subject area. This panel met virtually on July 11, 2023, to identify key topics and develop a structure for a manuscript, including a series of evidence-informed recommendations for BP management in Canada. Following this meeting, topics were assigned to faculty pairs who identified key literature, prepared the relevant section of the manuscript, and generated draft recommendations based on available data, guidelines, and clinical experience.
Completed sections and practice points were shared with the entire panel, and individual members voted anonymously on their level of agreement with each recommendation using a 5-point Likert scale ranging from strongly disagree to strongly agree. Voting occurred using a modified Delphi consensus technique with a prespecified cutoff agreement of 80% agree or strongly agree. 5
Proposed recommendations that did not reach the threshold for consensus were discussed in a virtual meeting of 7 panel members that took place on December 13, 2024. The recommendations were amended through open discussion, and an anonymous vote was held via online poll. Revised statements that reached consensus among the panel members in attendance were sent to the remaining 2 panel members for their anonymous vote; statements for which the consensus threshold was not reached within the virtual meetings are noted in the body of the manuscript. A measure of the consensus supporting each statement is recorded in Table 1 as both the degree of consensus (pooled number of panel members who responded “agree” and “strongly agree”) and as a weighted average (scores of 1-5 assigned to responses of “strongly disagree” to “strongly agree” divided by the number of votes received).
All Recommendations. Consensus Strength is an Average Score Based on Panel Votes, With 1 Point Being Conferred for “Strongly Disagree” and 5 Points for “Strongly Agree.”

Abbreviations: AEs, adverse events; BP, bullous pemphigoid; C3, complement component 3; DEJ, dermal-epidermal junction; DIF, direct immunofluorescence; IgG, immunoglobulin G; IVIg, intravenous immunoglobulins; Q4W, every 4 weeks.
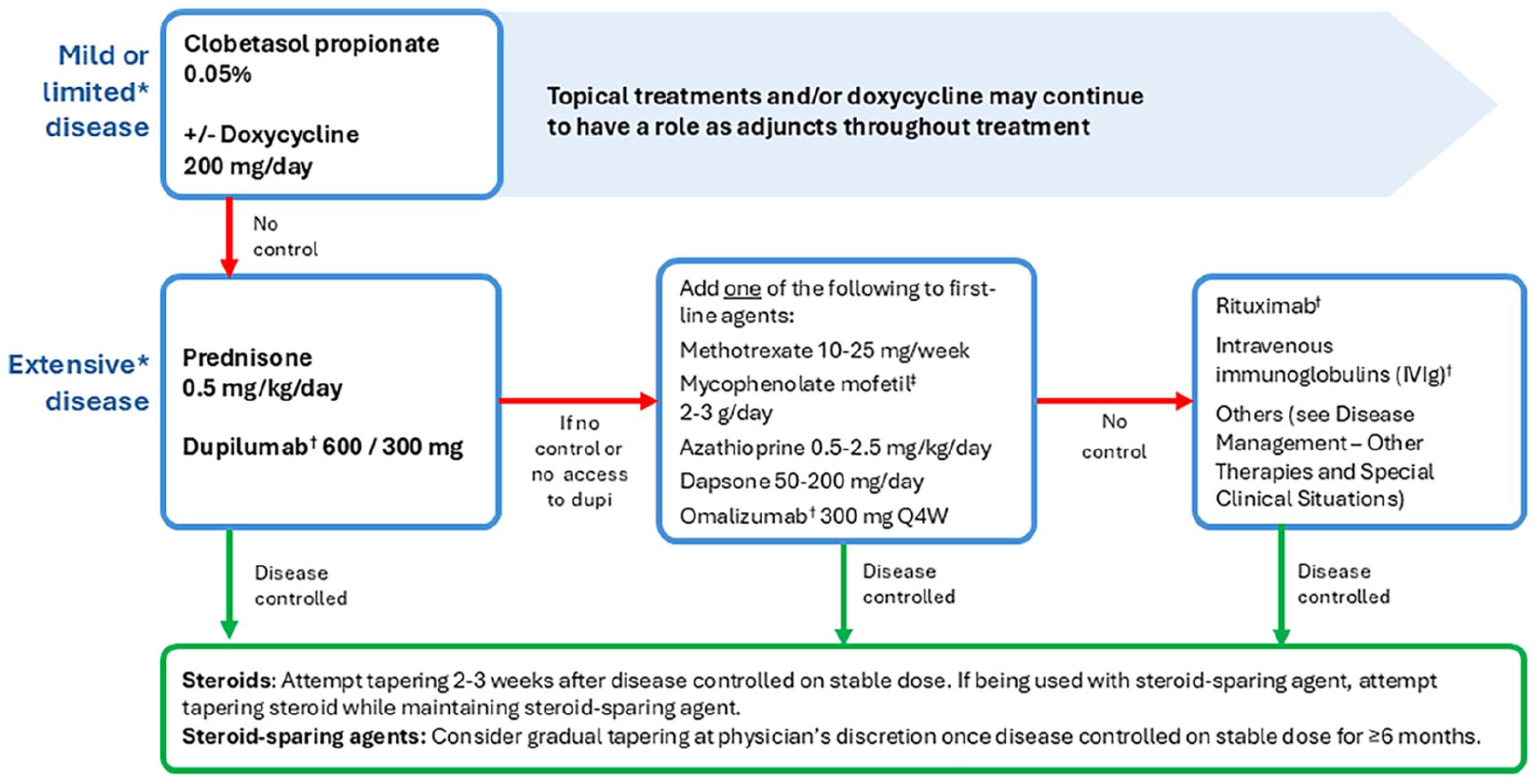
Treatment algorithm. Disease control is defined as a cessation in the development of new symptoms and the amelioration of existing symptoms. Bolded text represents preferred options; lack of bolding indicates no strong recommendation for first-choice therapies at that stage. Note that this algorithm is constructed with consideration of medication access limitations at the time of publication. Future approval of any medication for the treatment of BP and reimbursement criteria may influence treatment options. BP, bullous pemphigoid; Q4W, every 4 weeks.
Pathophysiology
BP is an autoimmune disease characterized by the disruption of the basement membrane zone (BMZ), leading to the formation of subepidermal bullae. 6 The primary mechanisms driving BP autoimmunity are poorly understood and likely include hereditary and external influences. 7
Patients with BP have tissue-bound and circulating autoantibodies against BP180 (Collagen XVII) and/or BP230, both components of the dermal-epidermal junctional (DEJ) adhesion complex. Binding of autoantibodies to these self-antigens triggers complement activation primarily through immunoglobulin G (IgG), although IgE is also often present. IgG1 can recruit complement component 3 (C3) and begin a complement-dependent cascade leading to the formation of terminal membrane attack complexes, which exert direct cytotoxic effects in the epidermal basal cells, disrupt the DEJ, and promote blister formation. 8
In parallel with complement activation, Th2 pathways are also key drivers of BP pathophysiology. Activated Th2 cells release cytokines such as IL-4, IL-13, and IL-31 to activate B cells and to attract and activate eosinophils, promote cytokine and chemokine production by mast cells, and may contribute directly to pruritic symptoms. 8 This process promotes the infiltration of the DEJ by eosinophils, accompanied by neutrophils and macrophages. 8 These cells secrete proteases which contribute to dermo-epidermal separation by degrading the extracellular matrix and splitting DEJs.9-11 IL-4 and IL-13, partially through STAT6 activation, promote B-cell class switching, leading to the production of IgG1 and IgE.12,13 Activation of the IL-17/IL-23 axis also seems to occur in at least a subset of patients with BP, but a clear picture of its involvement in BP pathophysiology has yet to emerge from ongoing research.8,14
Treatments for BP target steps along these cascades. Corticosteroids have broad anti-inflammatory properties and continue to be the mainstay of acute BP treatment. Similarly, azathioprine, methotrexate, and mycophenolate mofetil have broad immunomodulatory effects and have been widely used as steroid-sparing agents. Dapsone downregulates eosinophil and neutrophil migration, whereas doxycycline is thought to also inhibit neutrophil migration. Other effective strategies include blocking IL-4 and IL-13 with dupilumab, targeting IgE with omalizumab and depleting B cells using rituximab. 4 Lastly, Janus kinase (JAK) inhibitors reduce cytokine-induced cellular signalling, and some case reports describe their efficacy to improve disease activity in BP.15-19 Future therapies may target components of the complement system or molecules involved in eosinophil recruitment, as detailed in the future of therapy section.
Diagnosis and Workup
BP diagnosis is based on a combination of clinical features and immunofluorescence histopathology findings.
The “classic” presentation of BP is intensely pruritic, tense bullae, often with symmetrical distribution on inflamed and non-inflamed skin. “Non-classical” presentations may include pauci-bullous or localized eczema, localized disease (eg, pre-tibial BP), dyshidrosiform-like or pompholyx-like, and prurigo nodularis-like. In early stages, the presentation may be of urticarial plaques accompanied by itch out of proportion with visible disease, sometimes referred to as pre-bullous BP. In all presentations, mucosal involvement remains rare. 4
Given the potential polymorphous presentation of BP, physicians should consider a wide range of differential diagnoses, including pemphigus herpetiformis, dermatitis herpetiformis, linear IgA bullous dermatosis, bullous lichen planus, epidermolysis bullosa acquisita, bullous lupus erythematosus, pemphigus foliaceus, and in non-classic presentations, eczema, urticaria, prurigo nodularis, impetigo, erythema multiforme, Sweet’s syndrome, and Stevens-Johnson syndrome/toxic epidermal necrolysis. 6
The “classical” clinical and histopathological criteria for BP diagnosis are summarized in Table 2. A positive DIF result is essential for a correct diagnosis of BP with very few exceptions. Biopsy specimens for DIF should be taken from perilesional skin (this can be erythematous skin but not from a bulla) and should be stored in Michel’s medium or saline prior to analysis. DIF sample exposure to formalin or aldehydes should be avoided altogether as this can produce false-negative results. Specimens typically show linear deposits of IgG and/or C3 along the DEJ. The location and serration pattern of these deposits can help differentiate BP from other autoimmune blistering disorders. 4 Although DIF is reliable for the diagnosis of BP, false-negative or indeterminate results occur in a small number of patients (Tables 3 and 4). When BP is clinically suspected but the initial DIF is negative, a repeat biopsy for DIF should be considered, taking care to follow the practical recommendations listed above.20,21
Clinical and Histopathological Criteria for BP Diagnosis. 4

Abbreviations: BMZ, basement membrane zone; BP, bullous pemphigoid; C3, complement component 3; DEJ, dermal-epidermal junction; DIF, direct immunofluorescence; ELISA, enzyme-linked immunosorbent assay; IgG, immunoglobulin G; IIF, indirect immunofluorescence; OR, odds ratio.
Diagnostic Performance of Different Tests Supporting BP Diagnosis Based on Analysis of 1125 Patients in a Single-Centre Study. 22
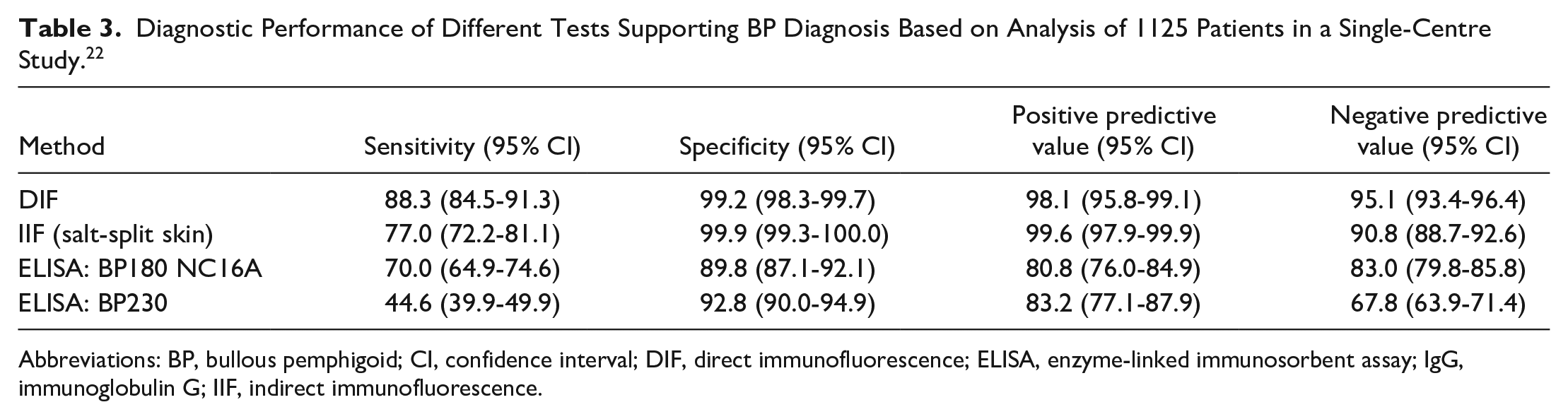
Abbreviations: BP, bullous pemphigoid; CI, confidence interval; DIF, direct immunofluorescence; ELISA, enzyme-linked immunosorbent assay; IgG, immunoglobulin G; IIF, indirect immunofluorescence.
Medications Associated With Increased Incidence of BP. Reporting From Swiderski et al 23 is Limited to Findings Where OR >2.0 for ≥1000 Patients.
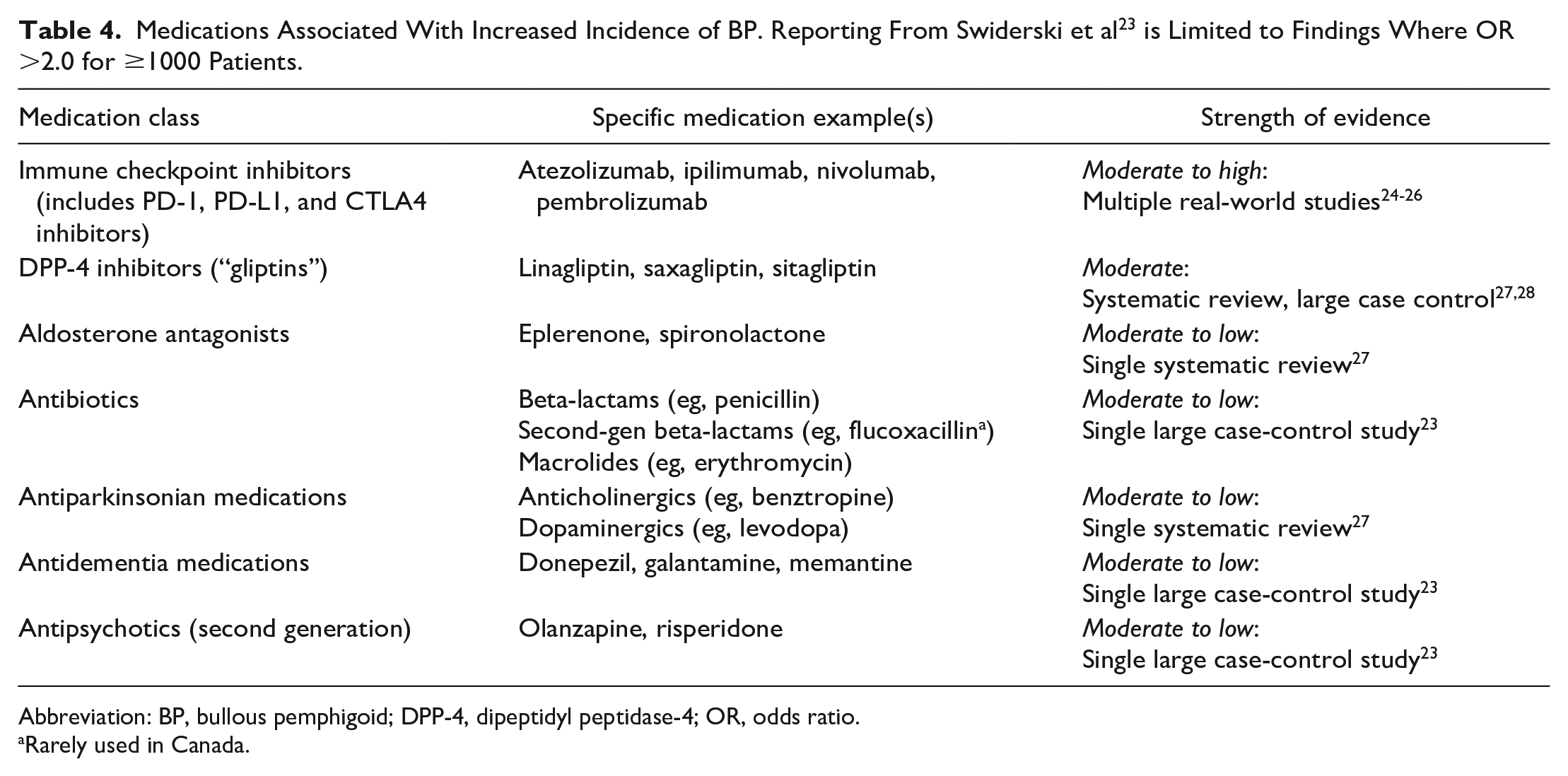
Abbreviation: BP, bullous pemphigoid; DPP-4, dipeptidyl peptidase-4; OR, odds ratio.
Rarely used in Canada.
Circulating IgG autoantibodies may be identified in serum using indirect immunofluorescence for IgG anti-BMZ antibodies in NaCl-separated normal human skin (“salt-split skin”), or enzyme-linked immunosorbent assay for anti-BP180 or anti-BP230 IgG. 4 Other immunoassays, such as addressable laser bead immunoassays or multiplex assays, may also be used to detect relevant antigens.
BP has been linked with a variety of comorbidities. Several epidemiological studies have provided evidence of an association between BP and neurologic disorders, including multiple sclerosis, dementia, Parkinson’s disease, epilepsy, and stroke. Active BP is also pro-inflammatory and prothrombotic, increasing the risk of thromboembolic events compared to age-matched controls. The development of BP has been associated with a number of medications (see section on Medication-associated BP) and therefore a thorough medication review is important in these patients. The association between BP and malignancy remains unclear, so further work-up is generally warranted only if prompted by symptoms or routine age-appropriate cancer screening.6,28
Treatment Goals
The primary goals of treatment are to clear and/or prevent skin lesions, reduce itch, and prevent or reduce the risk of recurrence, all while minimizing side effects. 4 Control of disease activity (CDA) is defined as the point at which new lesions or pruritic symptoms cease to form and established lesions begin to heal. 29
Clinically practical measures of BP disease activity are not well established. The panel considered a recommendation around the use of validated scales for monitoring treatment efficacy and relapse. Consensus on such a recommendation was not achieved due to practical concerns associated with administering validated scales in real-world scenarios. Additionally, pruritus scores in BP have not been demonstrated to correlate with blister activity or number. 30 As such, the panel did see a value in using the Itch NRS for some patients, but considered scales such as the BP Disease Area Index (BPDAI) and the Autoimmune Bullous Skin Disorder Intensity Score as appropriate for research settings. A daily blister count has been suggested by other sources as a measure of disease activity. 4
There is no generally accepted classification of BP disease severity. Some sources recommend classification using the BPDAI score, or into mild, moderate, and severe disease based on body surface area (BSA) involvement.31,32 These classifications may not be practical or even feasible in clinical practice, and may not adequately represent the impact of BP on the life of a given patient. Practically, it may be more useful to classify BP into “limited” and “extensive” disease, based on whether patients are willing and able to self-administer topical treatment to the entirety of the affected area and on patient-reported symptom severity. Given that BP generally affects elderly or medically complex patients, both disease severity and the overall patient condition should be taken into consideration when selecting treatment for BP. 33
Disease Management
Topical and Systemic Steroids
The 2022 European Academy of Dermatology and Venereology (EADV) guidelines recommend either super-potent topical corticosteroids (TCS) or oral corticosteroids as first-line treatments for BP regardless of severity or affected BSA. 4 However, the administration of topical regimens, especially in patients with significant BSA involvement, may require as much as 30 to 45 min of dedicated nursing care per application, 4 which makes them impractical in many Canadian healthcare settings. In Canada, patients with BP are rarely admitted for inpatient care, and with limited community nursing resources, administration of treatment usually falls to caregivers or the patients themselves. While topical treatments have a crucial role in BP management, practical considerations for the Canadian context necessitate less emphasis on topical treatments relative to the European recommendations.
A systematic review of 14 studies concluded that the high-potency TCS clobetasol propionate is an effective and safe treatment for BP. 34 In a randomized comparison of clobetasol propionate cream (40 g/day) versus oral prednisone (0.5 or 1 mg/kg/day, depending on extent of disease), the topical regimen resulted in more patients achieving CDA at 3 weeks, a lower incidence of severe adverse events (AEs), and lower mortality. When adjusted for patient age and Karnofsky score (a validated measurement of how well a person can perform daily activities), this safety benefit was most apparent in patients with extensive disease. 35
Moderate-certainty evidence (GRADE criteria) showed that treatment with a lower cumulative dose of clobetasol propionate (10-30 g/day, tapered over 4 months) is equally effective with similar AEs as standard dosing (40 g/day, tapered over 12 months).34,36
The panel recommends initiating topical management with clobetasol propionate cream once or twice a day to a maximum of 20 g/day for areas excluding the face, which is frequently spared in BP, and folds, which are atrophy-prone. 4 Panel members use alternative super-potent class 1 TCS in clinical practice and may step down to moderate-potency TCS for adjunctive use, but clobetasol is best supported by evidence. Lack of disease control after 2 weeks with topical clobetasol (or equivalent) should be taken as an indicator for systemic therapy. 36
Non-adherence to topical treatments is reported to be higher than other methods of administration 37 and is associated with poorer clinical outcomes, increased disease burden, and higher healthcare costs. 38 A combination of patient, treatment, and disease factors contributes to rates of non-adherence, which are often higher in elderly patients who have physical difficulty applying medication. The chronic nature of BP is also likely to impose additional practical and economic barriers to adherence. 38
The panel considered a recommendation on the use of alternative classes of topical treatment, such as calcineurin inhibitors and JAK inhibitors, but consensus could not be achieved due to a lack of evidence supporting the efficacy of these medications for the treatment of BP. At the time of writing, there is no evidence supporting the use of topical JAK inhibitors for the treatment of BP, and only anecdotal case reports to support the use of topical calcineurin inhibitors as an alternative or adjunct to topical steroids in generalized and localized disease.39-42 Importantly, the panel emphasized that failure of super-potent TCS indicates an escalation to systemic therapy rather than a switch to any other class of topical medication.
Shared decision-making with patients should determine the feasibility of a topical-only regimen (see treatment goals section). If topical treatment is impractical, prednisone is an effective therapy for BP. Oral corticosteroids at 0.5 mg/kg/day have shown good efficacy in patients with milder forms of BP, with between 69% and 75% of patients achieving CDA after 3 weeks of treatment, and have a more favourable safety profile than higher-dose therapy.34,43 However, prednisone 0.5 mg/kg/day appears less efficacious in patients with more severe disease. 43 Some panel members recommended escalation to 1.0 mg/kg/day based on clinical experience if disease control is not achieved in 1 to 3 weeks of therapy, although an increase to 0.75 mg/kg/day may be sufficient based on an older study showing no benefit of 0.75 mg/kg compared to 1.25 mg/kg.4,44
The panel agrees with the EADV guidelines recommendation to initiate progressive steroid tapering after 15 days of CDA at a stable dose according to the following schedules 4 :
TCS therapy. Taper application frequency over 4 to 12 months in phases of at least 1 month each: daily, every 2 days, 2×/week, and 1×/week.
Oral corticosteroid therapy. Gradual reduction to a dose of 0.1 mg/kg/day within 4 to 6 months of initiation; therapy may be discontinued after 3 to 6 months disease-free at this minimal therapeutic dose.
Patients should be screened for infection risks as per normal practice before initiating systemic corticosteroids and/or certain immunosuppressive agents. Ideally, patients should be up to date on age-appropriate vaccinations before initiating therapy with immunosuppressants; however, treatment of acute BP should not be delayed based on vaccination status. Skin tests for tuberculosis in appropriate patients should be done as early as possible (ie, within 1 month of therapy with an equivalent of ≥15 mg/day of prednisone). 45
Many patients with BP are at high risk for AEs associated with long-term corticosteroid use, such as osteoporotic fracture and serious infection: events that can occur at doses of 5 mg or less/day.46,47 Patients beginning long-term corticosteroids should consider vitamin D supplements, adequate calcium (ideally from dietary sources), and bisphosphonate therapy, and undergo usual monitoring including blood pressure, blood sugar, and bone density.4,48 Relatively common AEs associated with corticosteroids can be managed per routine clinical practice, such as the use of proton pump inhibitors for gastroesophageal reflux disease. The panel emphasizes the importance of collaboration with the patient’s primary care team to coordinate monitoring and care on issues that fall outside the dermatologic scope.
Steroid-Sparing Agents
Several immunomodulatory therapies, such as dupilumab, methotrexate, azathioprine, and mycophenolic acid, have demonstrated efficacy in the treatment of BP; however, robust, high-level evidence is limited. 34 Panel members frequently initiate a steroid-sparing immunosuppressant concomitantly with systemic corticosteroids to facilitate steroid tapering upon resolution of symptoms. Conventional immunosuppressants should also be considered when BP is not adequately controlled by combined topical and systemic corticosteroids, and for continued disease control after systemic corticosteroid tapering. Non-steroidal immunosuppressants without systemic steroids may also have a role in BP management where patient characteristics contraindicate the use of corticosteroids (eg, uncontrolled diabetes, congestive heart failure, or severe osteoporosis). 4
Dupilumab at an initial dose of 600 mg and subsequent doses of 300 mg Q2W thereafter should be considered as the first choice among steroid-sparing therapies due to its anticipated efficacy and safety. Its efficacy has been reported in numerous case series, including some long-term cases, and includes evidence of efficacy after failure of systemic corticosteroids.49-51 It is generally safe in patients with multiple comorbidities and those taking multiple medications. 49 In the LIBERTY-BP ADEPT Phase 2/3 clinical trial, dupilumab 300 mg SC Q2W was initiated with oral prednisone or prednisolone according to local standard of care, and steroids tapering started no later than 6 weeks after initiation. Compared to placebo, dupilumab treatment was associated with significantly higher rates of remission at 16 and 36 weeks, a lower rate of relapse, and a reduced need for rescue therapy. Overall safety was consistent with the known dupilumab safety profile. 52
Low-dose methotrexate (10-25 mg weekly, and on occasion, doses as low as 5 mg weekly) in combination with topical clobetasol propionate has the potential to shorten the required duration of high-potency topical treatment. 53 In a randomized noninferiority trial, a 4- to 6-week course of topical steroids in combination with continued methotrexate resulted in a lower rate of relapse after 9 months (25%) compared to topical steroids alone (42.5%). 54 A retrospective study recently confirmed that low-dose methotrexate following 3 months of clobetasol propionate is safe and leads to satisfactory control of BP over a median follow-up of 11 months. 55
Patients prescribed low-dose methotrexate should be monitored regularly for toxicities. With appropriate dose modifications (doses as low as 5-10 mg/week), methotrexate appears safe among patients with BP who have moderately impaired renal clearance at baseline (estimated glomerular filtration rate (eGFR) ≥45 mL/min/1.73 m2). 56 However, current Canadian labelling for methotrexate indicates that it should be avoided in patients with eGFR <50 mL/min/1.73 m2.57,58
The recommendation that azathioprine and mycophenolate mofetil may be considered in certain instances aligns with EADV guidelines. Limited data are available to support the efficacy of either intervention.4,34
A randomized, prospective study comparing the safety and efficacy of methylprednisolone combined with either azathioprine or mycophenolate mofetil found that the 2 immunosuppressants had comparable efficacy; however, mycophenolate mofetil was better tolerated than azathioprine with significantly lower liver toxicity (low-certainty evidence according to GRADE criteria).34,59
Use of these immunosuppressants should follow usual monitoring and screening procedures, including dose adjustment of azathioprine according to thiopurine methyltransferase activity. 4
Omalizumab at a dose of 300 mg Q4W may also be considered for some patients when first-line therapies have failed. Evidence supporting omalizumab is limited to case series and retrospective studies, as reviewed elsewhere. 60 The largest series so far reported included 100 patients with prior treatment failure, 77% of whom achieved complete remission after a median of 3 months. 61 However, the clinical experience of some panel members has not reflected rates seen in published literature. The mechanism of omalizumab suggests that it may be more effective when IgE levels are higher, though this relationship is not clearly identified in retrospective studies.62-64
The panel considered a recommendation for doxycycline as an alternative to systemic corticosteroids in first-line therapy, but did not achieve consensus. Some saw more value for doxycycline as an adjunctive therapy throughout treatment, and some took issue with the design of a study meant to show non-inferiority of first-line treatment with doxycycline 200 mg/day compared to 0.5 mg/kg/day prednisolone (including the breadth of the 25% non-inferiority margin). 65 In this study, doxycycline was also found to be associated with fewer severe, life-threatening, and fatal events compared to prednisolone, but some efficacy outcomes favoured prednisolone, such as the proportion of patients who were completely blister-free after 6 weeks. TCS use for BP symptom relief was also higher for patients initiated on doxycycline compared to prednisolone. Doxycycline is thus recommended as adjunctive therapy throughout treatment. There are no trials confirming the effectiveness of nicotinamide as an adjunct to treatment with doxycycline. Nicotinamide 500 mg up to 3× daily is considered highly safe and may be used in combination with doxycycline at the discretion of the health care professional (HCP).
Discontinuation of steroid-sparing agents may be a treatment goal for some patients. There is no evidence supporting a specific timeline or process for withdrawal of steroid-sparing agents. The panel suggests considering a gradual taper of these medications in appropriate patients only if patients have been completely disease-free with no new symptoms or flares for at least 6 months.
Other Therapies
Dapsone is another possible choice for steroid-sparing, non-biologic treatment of BP. The panel considered a recommendation around its use as monotherapy and part of a combination regimen, but it did not achieve consensus. The evidence supporting its use is taken from multiple case series, and randomized trial data are lacking. 66 We support a role for dapsone in neutrophil-rich disease and/or in patients with heavy immunosuppression or who are ineligible for other treatments. Appropriate screening should be performed before initiating dapsone, with hematological monitoring throughout treatment. Dapsone can be initiated at 50 mg, with gradual escalation of dose up to a maximum of 200 mg/day based on tolerability. 4
Rituximab and IVIg are both highly effective therapies used as described in recent guidelines. 4 However, due to their association with potentially serious AEs, particularly in older populations, these therapies are typically reserved for when multiple treatments have failed.
The panel considered a recommendation supporting the use of systemic JAK inhibitors when other lines of therapy have failed, but the statement did not reach consensus due to a lack of quality evidence to support their use. While there are case reports supporting the use of various JAK inhibitors to treat BP, no clinical trials are registered with the National Institutes of Health at the time of writing. JAK inhibitors may be considered in later lines of therapy when other treatment options have been exhausted. One report describes the use of baricitinib 4 mg/day as a first-line therapy in an elderly male with uncontrolled hypertension and concomitant psoriasis. 15 Tofacitinib has been used successfully at a dose of 5 to 10 mg BID in several case reports, both as monotherapy and in combination with systemic steroids; often after failure of more commonly used therapies, including prednisone, minocycline, and dupilumab.16,17 Upadacitinib 15 mg/day has also been effective in the treatment of BP after failure of prednisone 50 mg/day, and in the treatment of concomitant BP and psoriasis vulgaris.18,19
Special Clinical Situations
Because BP is most prevalent among older populations, adjustments to the therapeutic approach may often be required based on comorbidities and concomitant medications.
Patients who are elderly, especially with multiple comorbidities, need special consideration. Immunosuppression associated with systemic corticosteroids is often undesirable, and dupilumab would be our first-line treatment of choice in these patients, if available, due to the lack of drug-drug interactions and low rates of AEs. Omalizumab may also be considered if dupilumab is not effective or accessible. The panel suggests considering methotrexate if not contraindicated. Azathioprine should be avoided in these patients given the potential for liver toxicity, but mycophenolate may be considered in some scenarios. Rituximab or IVIg remains a later-line option, though caution should be exercised due to the potential for volume overload and other AEs.
Malignancy
The management of BP in any patient with an active malignancy should be done in close collaboration with the treating oncologist; however, dermatologists will likely have more experience managing BP and with biologic therapy, and as such should lead these discussions. Again, since the use of systemic corticosteroids is often undesirable in those with an active malignancy being treated with immunotherapy, dupilumab therapy should be considered as a first-line therapy. While panel members prioritize biologics in patients with active malignancy, rituximab should be used with extreme caution in these patients, though it does not appear to increase the risk of a second primary malignancy. 67 Consider medication-induced BP in patients with active malignancy taking an immunotherapy (eg, anti-PD-1, anti-PD-L1, or anti-CTLA4; see section on medication-associated BP). In cases where immunotherapy is withheld due to the occurrence of BP, re-challenging with immunotherapy can be considered if control of BP can be achieved with a steroid-sparing agent such as dupilumab.
Pregnancy
Patients who are pregnant should undergo a thorough evaluation to examine the possibility of other pregnancy-associated blistering diseases, such as pemphigoid gestationis. Many therapies commonly used for BP may be used in pregnancy, although treatments with known risk to the fetus should be avoided (eg, methotrexate, mycophenolate). 68 The risk-benefit profiles of treatment options should be discussed with the patient and their obstetrician as necessary.
Children and Adolescents
Childhood BP remains relatively rare. It occurs most often in the first year of life, with another peak in incidence at a median age of 8 years. 69 Infantile BP commonly affects acral sites. Among older children, BP is less likely to involve hands and feet, but more likely to involve external genitalia. 69 Mucosal involvement may be more likely in BP of early adolescence (10-13 years old), although BP is very rare overall in the adolescent population. 70 The histopathology of childhood BP is similar to that seen in adult disease.69,71 Evidence supporting treatment of BP in juvenile populations is restricted to case studies, which suggest that systemic corticosteroids are effective in most cases and may be supplemented with methotrexate, dapsone, doxycycline, or mycophenolate, with dosing appropriate for the patient’s weight and/or age.69,70 There are single case studies supporting the use of dupilumab in adolescents and cyclosporin in infants.72,73 Successful treatment has also been reported with azathioprine, IVIg, and rituximab, but we suggest reserving these therapies for resistant cases.69,70,74 TCS should be used with the usual precautions in pediatric populations.
Medication-Associated BP
BP incidence is associated with several medication classes according to a variety of case reports and retrospective analyses as described below. The mechanism of medication-associated BP is unclear and must be multifactorial, given that only a small proportion of people taking risk-conferring medications develop BP. However, some reports are conflicting, and not all are substantiated by the single systematic review and meta-analysis that has been conducted at the time of writing.27,75,76 This meta-analysis of case-control studies, cohort studies, and a randomized clinical trial did not find any association between BP incidence and medications including thiazide or loop diuretics, antibiotics, or ACE inhibitors. A significant association was seen for aldosterone antagonists [odds ratio (OR) 1.75, 95% confidence interval (CI) 1.28-2.40], dipeptidyl peptidase-4 (DPP-4) inhibitors (OR 1.92, 95% CI 1.55-2.38), anticholinergic medications for Parkinson’s disease (OR 3.12, 95% CI 1.54-6.33), and dopaminergic medications for Parkinson’s disease (OR 2.03, 95% CI 1.34-3.05). 27 The association with DPP-4 inhibitors was further supported by a large case-control study from a UK database (OR 2.77, 95% CI 2.37-3.23). 23 This study found several other significant associations with BP incidence, with ORs >2 for classes including antibiotics (OR 2.08, 95% CI 1.99-2.17 after adjustment) and second-generation antipsychotics (OR 2.58, 95% CI 2.20-3.03). 23 It is important to emphasize that, even with 2- or 3-fold increases in ORs, BP associated with these medications remains very rare. There is no recommendation to avoid them in patients without a history of BP linked to this drug class.
Notable omissions from the above studies were immune checkpoint inhibitors (ICIs), which are used for the treatment of a variety of malignancies. Approximately 30% of patients taking ICIs experience cutaneous immune-related AEs, which include BP.24,26 An analysis of a US database estimates the overall incidence of BP to be about 3.5% among patients taking ICIs. 25 BP has been reported with all classes of ICI (PD-1, PD-L1, and CTLA4 inhibitors), with PD1 inhibitors associated with the highest risk in the class.24,25 Risk factors of ICI-induced BP are male gender, older age, and presence of skin cancer.25,26 The median time to onset was 22 weeks after initiation according to a systematic review of literature and 29 weeks in an analysis of registry data, but BP may occur even after discontinuation of the ICI.24,25 Pruritus is a common prodromal symptom, and lesions on the face, neck, and mucosal surfaces are relatively common (~1 in 5 patients) compared to classic BP. 24
Management of medication-associated BP should consist of discontinuing the suspected medication and treating the patient the same as non-medication-induced BP. In many cases, alternatives to the suspect medication can be found in collaboration with the patient’s usual care team.
For ICIs, depending on their indication and whether the medication is the optimal choice for ongoing malignancy treatment, an attempt could be made to resume ICI therapy once CDA is achieved and pruritus is managed. Ongoing therapy to control BP is recommended if ICIs will be used to re-challenge the individual. Unfortunately, BP results in the discontinuation of the ICI in over 50% of cases. 24
Future of Therapy
There are currently a limited number of trials underway investigating the potential of established and novel medications for the treatment of BP. There is an ongoing trial to compare the effectiveness of methotrexate 15 mg/week versus dapsone 100 mg/day as adjunctive medications to prednisolone 0.75 mg/kg/day. This should help, which will provide insight into selecting combination therapies with systemic steroids. 77
The phase 2/3 LIBERTY-BP ADEPT trial evaluated the efficacy and safety of the IL-4/IL-13 receptor blocker dupilumab as a treatment for BP. 78 Given their similar mechanisms of action, lebrikizumab, tralokinumab, and nemolizumab may have a future role in BP management; however, no publications detailing their use in this context were identified at the time of publication. The BALLAD phase 2/3 trial will test the efficacy of efgartigimod for the treatment of BP, a therapeutic strategy that will block neonatal Fc receptors to prevent recycling of IgG back into the bloodstream.79,80
Other potential therapeutic targets have been identified for the treatment of BP, including eosinophil signalling pathways and complement system components. Unfortunately, several clinical trials exploring new avenues have recently been terminated due to resource concerns or futility. A summary is shown in Table 5.
A Selection of Candidate Medications for Treatment of BP and Their Current Investigation Status.
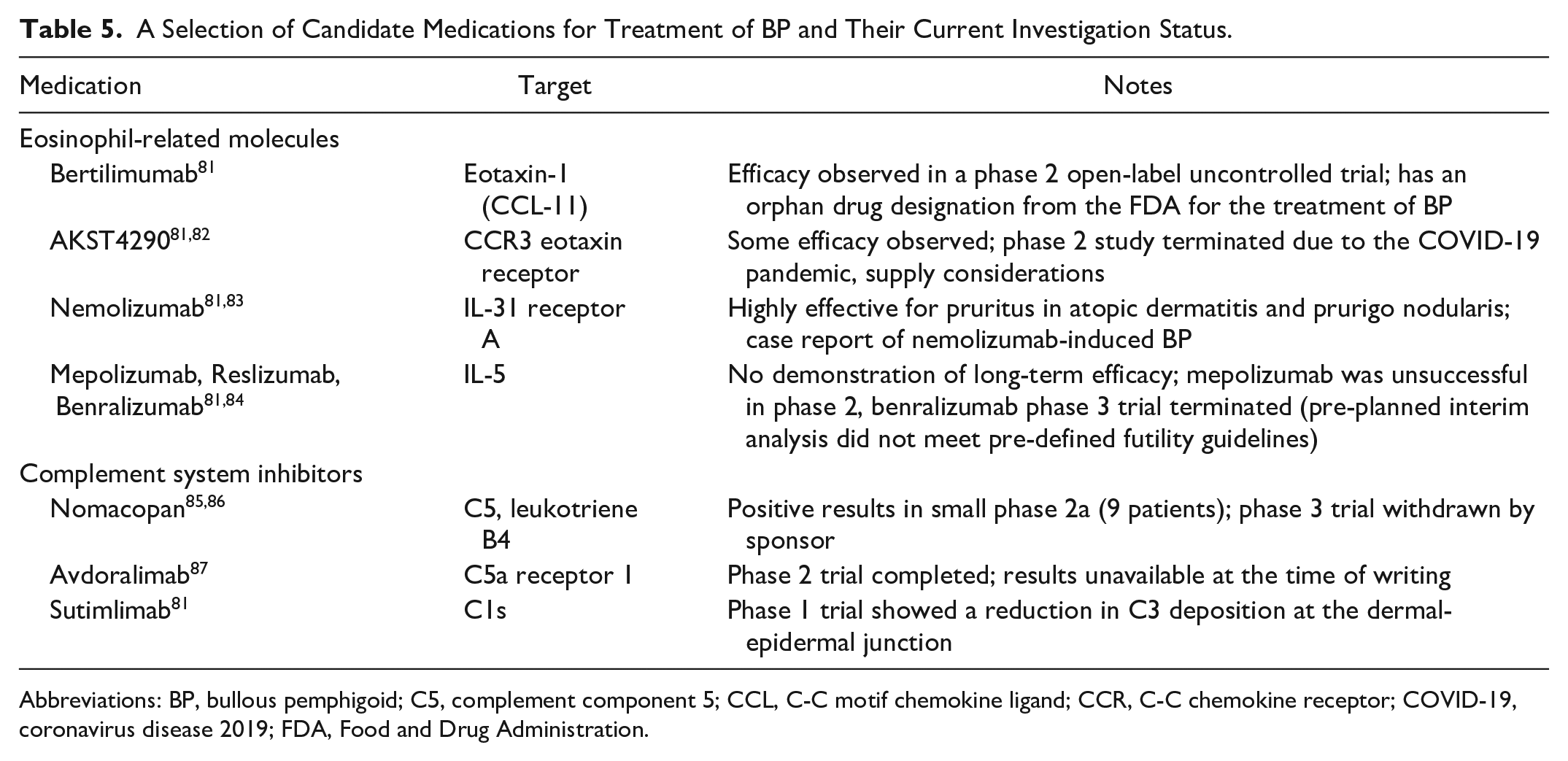
Abbreviations: BP, bullous pemphigoid; C5, complement component 5; CCL, C-C motif chemokine ligand; CCR, C-C chemokine receptor; COVID-19, coronavirus disease 2019; FDA, Food and Drug Administration.
As research into the pathophysiology and treatment of BP progresses, the therapeutic approach will continue to evolve. For example, the involvement of IL-17 and the Th17 axis in BP is currently being investigated in a clinical trial context. 88 While there are some case reports of IL-17 and IL-23 inhibitors being used in BP, no formal trials are underway of any of these medications at the time of writing.81,88 Further research into the role of type 2 inflammation and IgE signalling in BP may provide new avenues for the treatment of the disease and improve outcomes for patients. 89
Conclusions
Given the growth of this segment of the Canadian population, it is reasonable to expect increased incidence and prevalence of diseases primarily associated with elderly people, including BP. This manuscript provides insight into the identification and management of BP within the Canadian context. While the disease remains relatively rare, research continues in hopes of expanding treatment options and improving patient outcomes.
Footnotes
Author Contributions
Authors worked in pairs to develop initial drafts of each section and the associated recommendations, where applicable. All authors reviewed the document and recommendations in their entirety, with F.E.L. acting as the Chair in the implementation of any revisions. All authors approved the final draft.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.E.L. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for AbbVie, Amgen, Arcutis, Bausch Health, Bio-JAMP, Boehringer-Ingelheim, Bristol Meyers Squibb, Celgene, Celltrion, Eli Lilly, Galderma, GSK, Incyte, Janssen, LEO Pharma, Novartis, Pfizer, Regeneron, Sandoz, Sanofi-Genzyme, Sun Pharma, and UCB. A.M. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for Abbvie. M.B.S. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for Sanofi, Novartis, and Galderma. J.B. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for AbbVie, Amgen, Arcutis, Beiersdorf, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Concert, Galderma, Eli Lilly, Evelo, Incyte, Janssen, Johnson and Johnson, Leo Pharma, L’Oréal Group, Novartis, Pfizer, Sanofi Genzyme, Sun Pharma, Reistone, UCB, and Vyne. J.P.D. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for Eli Lilly, Janssen, Solius, Astra Zeneca, AbbVie, Amgen, Bausch, Bristol Myers Squibb, Celltrion, Galderma, Leo, Janssen, Novartis, Pfizer, and Sanofi. M.G.K. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for National Capital Skin Disease Foundation, AbbVie, Amgen, Arcutis, Bausch, BioJamp, BMS, Boehringer Ingelheim, Eli Lilly, Incyte Janssen, Leo, Novartis, Pfizer, Recordati, Sanofi-Genzyme, Therakos, and UCB. P.R.M. declares no conflicts of interest. K.S.P. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for AbbVie, Amgen, Arcutis, Boerhinger-Ingleheim, BMS, Bausch Health, Eli Lilly, Incyte, Janssen, Leo, Novartis, Pfizer, Recordati, Sanofi, Sun Pharma, and UCB. P.L. has served as an advisor/consultant for, has received grants/honoraria from, and/or has served as a speaker for AbbVie, Acelyrin, Amgen, Arcutis, Apogee Therapeutics, Bio-JAMP, Bausch Health, Boehringer Ingelheim, Bristol-Myers-Squibb, Celgene (Amgen), Demira, Eli Lilly Canada, Fresenius Kabi Canada, Fusion MD, Galderma, Incyte Pharma, Johnson and Johnson, LEO Pharmaceuticals, Novartis, Pfizer, Sandoz, Sanofi-Genzyme, Sun Pharmaceuticals, and UCB.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No authors received compensation for their work on this manuscript. We would like to thank Incyte Biosciences Canada, LEO Pharma, Inc. Canada, Novartis Pharmaceuticals Canada, Pfizer Canada ULC, and Sanofi-Aventis Canada, Inc. for supporting the development of this manuscript through unrestricted grants to the Dermatology Association of Ontario. We thank LiV Agency, Inc. for logistical, organizational, and medical writing support (Ian Hellstrom and Lisa Kartusch).
