Abstract
Background:
The prevalence of vulvar diseases has a significant impact on quality of life (QoL). Measurement may not be consistently collected during assessments. Incorporating a QoL measurement tool may help optimize patient-centred care.
Objective:
To determine the impact of the validated Vulvar Quality of Life Index (VQLI) questionnaire during consultation assessments.
Methods:
A randomized controlled trial from a single dermatology centre was completed over 10 months. The intervention group completed the VQLI at the baseline/initial appointment, and both the intervention and control groups completed the questionnaire at follow-up, with these scores compared. Secondary outcomes compared scores within the intervention group; analyzed treatment adherence; and surveyed self-reported symptom improvement, whether patient health-related concerns were addressed, and well-being. Data were analyzed descriptively, with significance between means and proportions assessed using t-tests and Fisher’s exact tests, respectively.
Results:
Forty-two patients participated. Scores within the intervention group, baseline (18) versus follow-up (8.3), were statistically significant. Follow-up VQLI scores in the intervention group (n = 23), 8.3, trended lower than the control (n = 19), 12.8, but were not statistically significant (P = .1529). Although treatment adherence (P = .428), symptom improvement (P = .684), and feeling of whether health-related concerns were addressed (P = .391) were similar, improvement in well-being (P = .017) in the intervention group was statistically significant.
Conclusions:
In addition to the use of VQLI in vulvar dermatology assessments as an aid in identifying the impact of vulvar conditions on QoL, we recommend its use to improve the patients’ sense of well-being.
Keywords
Introduction
Vulvar complaints are among the most frequent causes for a woman to visit a healthcare provider. 1 Vulvar dermatoses are common dermatological conditions affecting the vulva, and can cause considerable pain, irritation, pruritus, and burning. 1 They have a significant impact on quality of life (QoL), 2 affecting every aspect of a woman’s life, including activities of daily living, sexuality, relationships, feelings of self-worth, social, psychosocial, and psychological well-being.1,3 Most women attending a vulvar clinic have reduced QoL related to their disease and a higher baseline anxiety score compared with the general population. 4 A recent study involving 187 new consultations in a single vulvar dermatology clinic found that the impact of the patient’s disease on QoL or sexual function was seldom documented, and those who were asked about the impact on sexual function reported significant impairment, avoidance, or fear of intercourse-related condition. 5 The prevalence and impact of vulval skin conditions are likely to be underestimated, and exact figures are not known as many women delay in seeking medical advice. 3
Health-related QoL (HRQoL) measurement tools are a key component of patient-reported outcomes measures, widely recognized as standards for assessing outcomes in research and clinical management settings, with their measurements serving as a major goal of dermatology. 6 Research on the use of HRQoL as a therapeutic intervention in clinical practice is limited, with mixed results.7,8
HRQoL measurements are important when treating conditions where laboratory tests to determine severity are not available, 9 as is the case with vulvar dermatoses. The importance of patient-reported perception of disease impact is particularly relevant for chronic genital disorders. 9 Current QoL assessment for disease of the vulva involves several separate scales, making their use impractical, and has limitations in their scope, often focusing on sexual function or certain conditions. 2 The Vulvar Quality of Life Index (VQLI) is a validated, simple, and reliable tool that is easy to understand and can be completed in less than 5 minutes2,10 (Figure 1). The VQLI measures the global patient-perceived impact of vulvar disease on QoL.2,10 It can be used to monitor patient symptoms, effectiveness of treatment, and treatment adherence, and can provide consistency to future research studies in vulvar disease.2,10
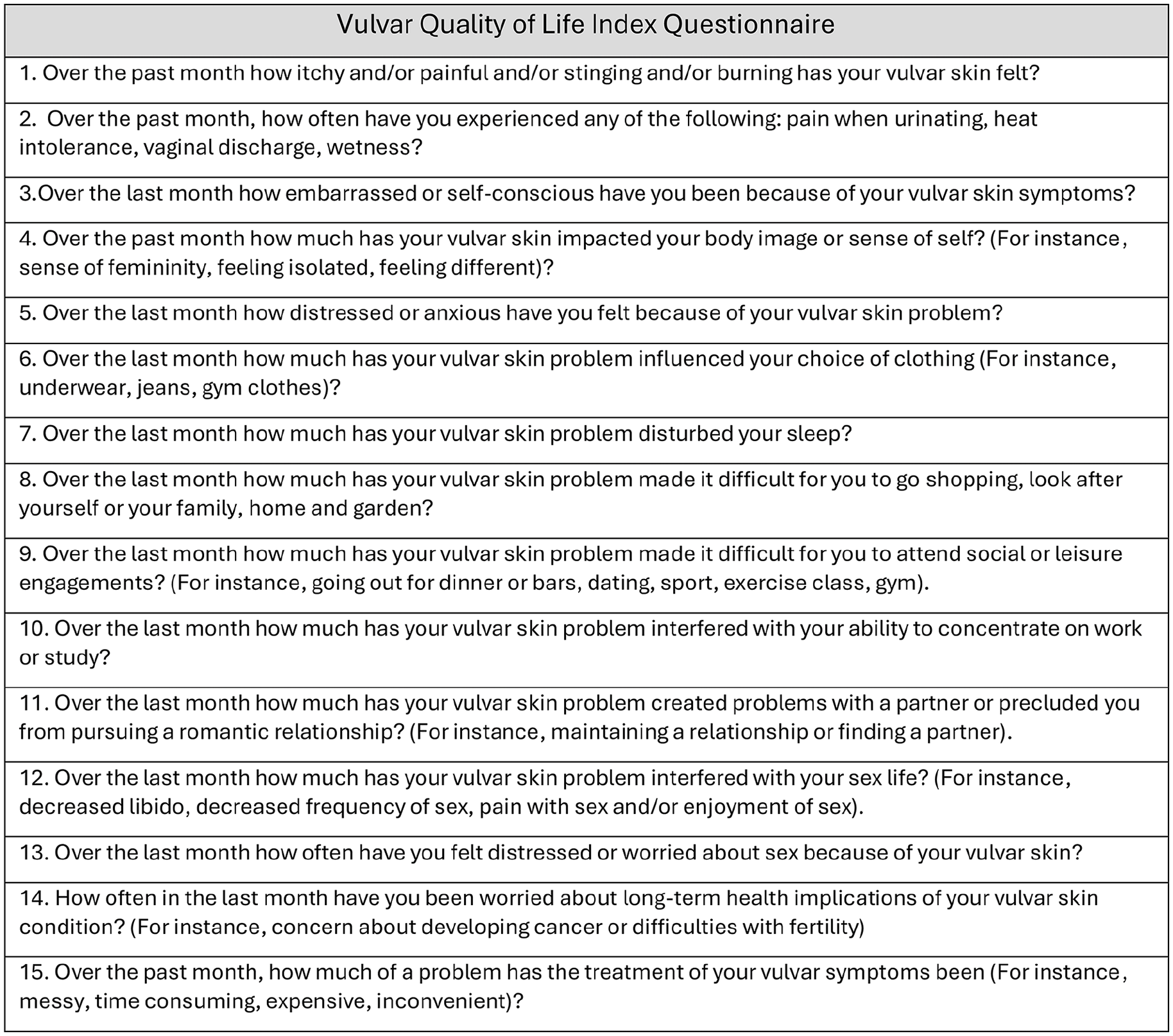
Vulvar Quality of Life Index Questionnaire. Scoring: 0 representing no symptom or complaint (“Not at all” or “Not applicable”), 1 “A little,” 2 “A lot,” and 3 “Very much,” resulting in a maximum score of 45/45 and a minimum score of 0/45. Impact of vulvar dermatoses on quality of life scored as follows: Minimal (0-5); Mild (6-13); Moderate (14-23); Severe (24-37); and Very Severe (38-45).
Methods
Design and Setting
A randomized controlled clinical trial (RCT) 11 was performed to assess whether completion of the VQLI at baseline new patient assessments in a vulvar dermatology clinic results in lower follow-up VQLI scores at subsequent visits, reflecting patient-perceived improved vulvar disease outcomes, treatment adherence, and overall QoL captured in the VQLI.
The study intervention consisted of measuring the global impact of vulvar disease on QoL using the validated VQLI at the baseline new patient assessments in the academic center vulvar dermatology clinic (Figure 1). Outcomes were measured using the VQLI at follow-up visits, along with patients completing a self-reported questionnaire (Figure 1). The intervention group completed a baseline-VQLI, and both the intervention and control groups completed a follow-up-VQLI (Figure 1). Follow-up VQLI scores were compared between the intervention and control groups to gauge the impact of baseline VQLI on patients’ sense of QoL.
New consultations in the academic center vulvar dermatology clinic in Edmonton, Alberta, Canada, were assessed over a period of 10 months from September 2022 to June 2023. The vulvar dermatology clinic is located in an academic center, and chart notes were written by rotating residents, medical students, and the attending staff physician. This study was approved by the University of Alberta Research Ethics Board (Pro00121296) and received operational approval from the Northern Alberta Clinical Trials and Research Center’s Edmonton Zone Research Administration (Protocol Bank ID PB100071).
VQLI Questionnaire
The VQLI2,10 is comprised of 15 questions graded 1-4 on a Likert scale, with 0 representing no symptom or complaint (“Not at all” or “Not applicable”), 1 “A little,” 2 “A lot,” and 3 “Very much,” resulting in a maximum score of 45/45 and a minimum score of 0/45. Overall VQLI score ranges were as follows regarding the impact on QoL: Minimal (0-5); Mild (6-13); Moderate (14-23); Severe (24-37); and Very Severe (38-45).
Participants
The RCT was designed such that all patients with undiagnosed vulvar dermatoses referred to the vulvar dermatology clinic were invited to participate. Inclusion criteria were as follows: new referral for a vulvar condition, age equal to or greater than 18 years of age, female sex, and diagnosis of any vulvar condition. Exclusion criteria were as follows: palliative, unable to provide informed consent, age less than 18 years, and no vulvar condition or lesion (Supplemental File 1). The power of study12,13 was calculated using a power of 80% and a significance level of 5%, with an effect size of 10, and determined to be 36 participants (Supplemental File 2).
Patients were invited to participate upon arriving at their in-person appointment by the study team. Written consent was obtained. Participants were then allocated to the intervention or the control group via a simple randomization (no stratification or blocking) from the central REDCapTM server’s randomization module (ensuring irreversible and concealed allocation) prior to their scheduled new consult visit. Those assigned to the intervention group completed a baseline VQLI before proceeding with their outpatient assessment in the clinic. Those assigned to the control group proceeded with their outpatient assessment in the clinic without completing a baseline VQLI. Both groups then returned for a follow-up assessment, at which point, a follow-up VQLI was completed. Outcome assessors at follow-up were blinded to the patient’s group allocation. The trial concluded when at least 36 participants had experienced the primary outcome event, that is, completion of the follow-up VQLI.
Measurement and Analysis
Outcomes were generated from a chart audit where a follow-up VQLI was completed 1-4 months following the initial in-person outpatient consult assessment and optional participant in-person surveys (Supplemental File 1). The primary outcome was the comparison of follow-up VQLI scores (Supplemental File 1) 1-4 months following the initial in-person consult assessment. Secondary outcomes included treatment adherence between the 2 groups, self-reported improvement, and well-being. Furthermore, the baseline and follow-up VLQI scores of the intervention group were compared. In addition, demographic, referral, and clinic visit data were analyzed.
Data gathered for analyses included: Age; sex; location of residence; distance of residence to vulvar clinic; referring provider specialty; wait time from referral to appointment date; diagnosis at time of referral; prior treatments; type of symptoms; impact on QoL; impact on sexual function; relevant skin pathology results; clinical diagnosis at vulvar clinic, treatment prescribed, and status at follow up visits. Data were analyzed and expressed as mean and confidence interval (continuous variables), as well as counts and proportions (categorical variables). Hypothesis testing was conducted by comparing the means and proportions of study outcomes using Student’s t-tests and Fisher’s exact tests as applicable. All statistical tests were two-sided using P-values ≤ .05 to determine statistical significance.
Results
Study Participants
A total of 51 screened participants were randomly allocated to either the intervention or the control group, with 42 completing the study after 9 were excluded after randomization (Figure S1). Of those that completed the study, 23 were assigned to the intervention group, and 19 to the control group, allowing for case-complete analysis (Figure S1).
The mean time for follow-up was 2.18 months in the control group and 2.14 months in the intervention group. There was no statistical difference in time between assessment and follow-up between the groups (P = .9193).
Use of the VQLI
Within the intervention group, the mean baseline VQLI was 18 (6, 36), and the mean follow-up VQLI was 8.3 (0, 18). The change in the mean VQLI score from baseline to follow-up within the intervention group was 9.3 (1, 27). The mean VQLI scores at follow-up within the intervention group were significantly lower than the mean VQLI at the initial assessment (unequal t-test P-value < .0001; Table 1).
Comparison of Baseline and Follow-Up Mean VQLI Scores in Intervention and Control Groups With Standard Deviation.
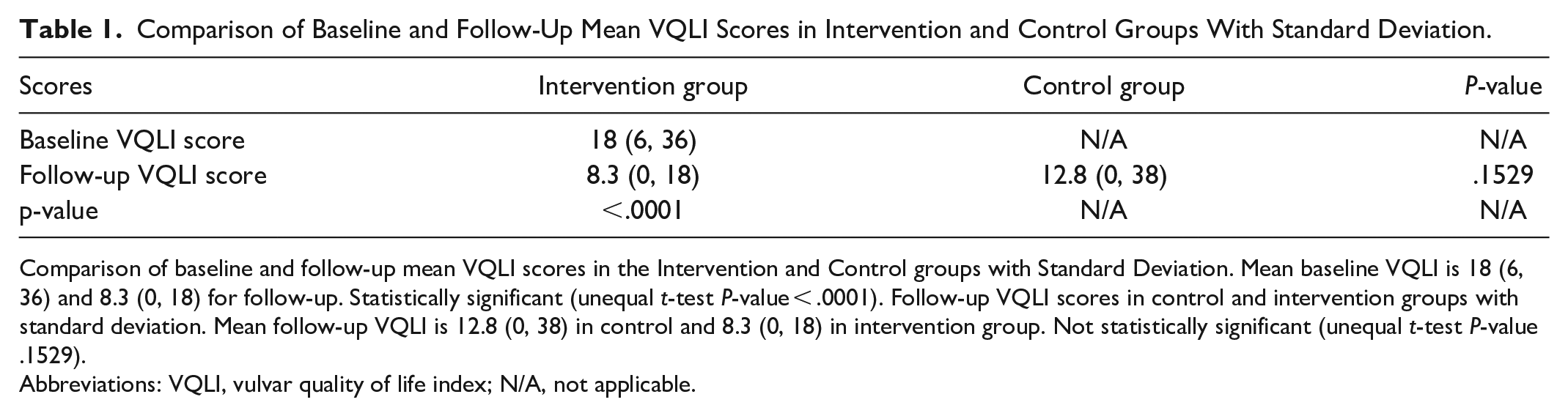
Comparison of baseline and follow-up mean VQLI scores in the Intervention and Control groups with Standard Deviation. Mean baseline VQLI is 18 (6, 36) and 8.3 (0, 18) for follow-up. Statistically significant (unequal t-test P-value < .0001). Follow-up VQLI scores in control and intervention groups with standard deviation. Mean follow-up VQLI is 12.8 (0, 38) in control and 8.3 (0, 18) in intervention group. Not statistically significant (unequal t-test P-value .1529).
Abbreviations: VQLI, vulvar quality of life index; N/A, not applicable.
The mean follow-up VQLI in the intervention group was 8.3 (0, 18) and 12.8 (0, 38) in the control group. While there was a trend toward lower follow-up VLQI scores in the intervention group, this was not significant, with an unequal t-test P-value of .1529 (Table 1).
Treatment adherence was similar (P = .428) between the control group, 89.5% (17), and the intervention group, 78.3% (18; Figure 2). Moreover, the patient’s perception of the cumbersomeness of the prescribed treatment was assessed by the patient’s response to Question 15 of the VQLI, “Over the past month, how much of a problem has the treatment of your vulvar symptoms been (For instance messy, time consuming, expensive, inconvenient)?” Among the intervention group, 60.87% (14) reported difficulties with the prescribed treatment, and 63.16% (12) reported difficulties in the control group. There was no significant difference (Fisher’s exact P-value = 1.000; 1-sided Fisher’s = .567) between the groups (Figure 2). When this same response was compared between both groups who were non-adherent to the prescribed treatment (7), there was no significant difference between the patients’ perception of the cumbersomeness of the prescribed treatment. The Fisher’s exact and 1-sided Fisher’s exact were .397 and .236, respectively.
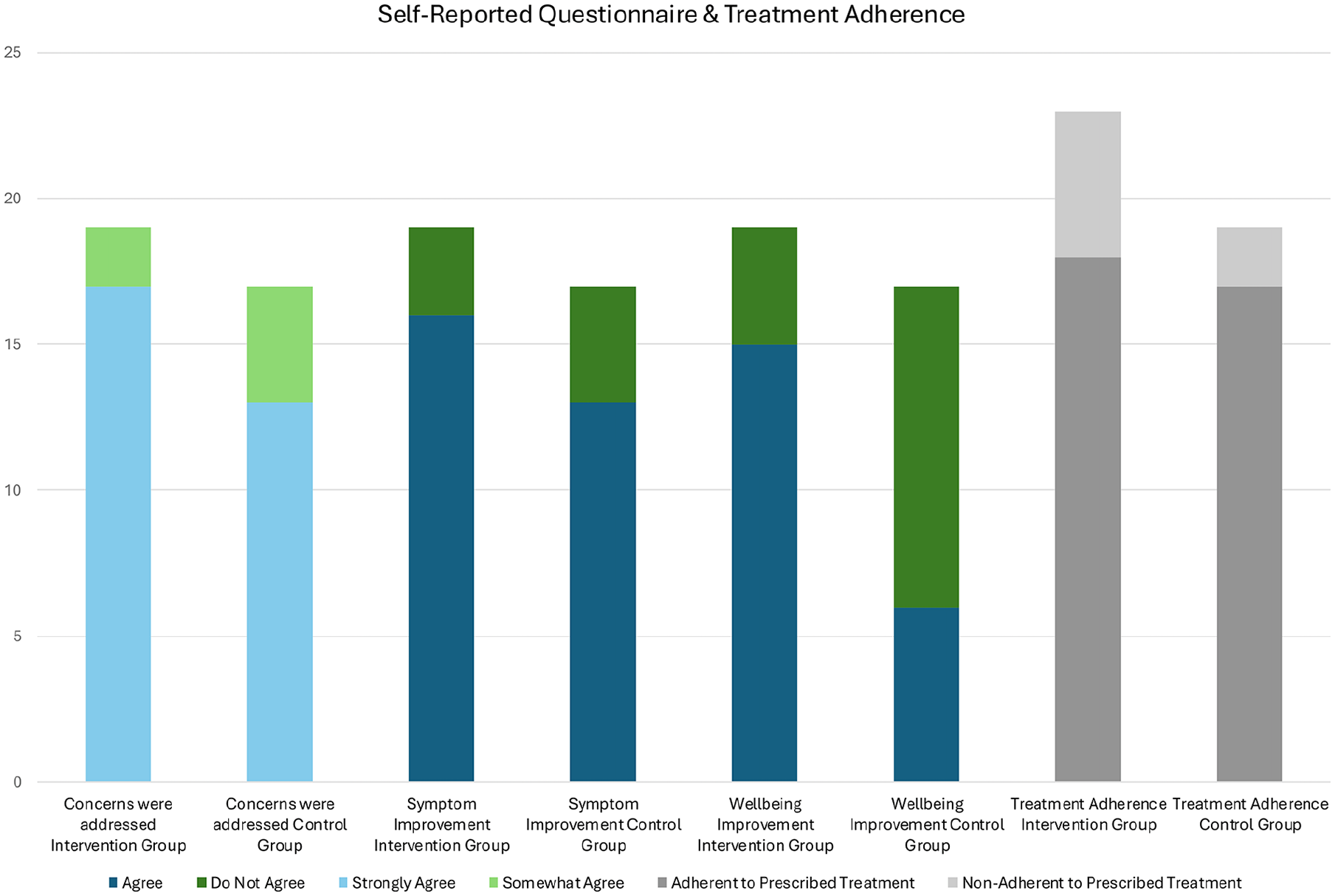
Self-reported questionnaire on symptom improvement, whether concerns were addressed, and patient well-being. Symptom improvement (P = .684) and feelings of concern being addressed (P = .391) were statistically similar. Well-being was reported to be statistically significant (P = .017). Treatment adherence was statistically similar (P = .428).
Optional surveys were completed by 82.6% (19) of patients in the intervention group and 89.4% (17) in the control group. Self-rated symptom improvement was statistically similar (P = .684) with 84.2% and 76.5% among the intervention and control groups, respectively (Figure 2). In addition, both groups, 89.5% in the intervention and 76.5% of those in the control group, reported that they strongly agreed that their dermatologic concerns were addressed in the first visit (P = .391; Figure 2). Notably, 35.3% (6) of the patients in the control group and 79.0% (15) of those in the intervention group self-reported that their well-being improved between visits; this was statistically significant (P = .017; Figure 2). Lastly, patients participated in an optional survey at the end of the study, with 17 from the control group and 19 from the intervention group. The majority of patients within both groups, 88.23% in the control and 89.47% in the intervention, indicated they strongly agreed that the instructions provided for the treatment regimen were clear. 88.23% in the control and 84.21% in the intervention groups strongly agreed that they felt comfortable talking about their symptoms and concerns. With regards to access to online educational resources, 41.18% of the control and 68.42% of the intervention groups indicated they were very likely to access online educational resources. There was no significant difference between the groups (Fisher’s exact = 0.179; 1-sided Fisher’s exact .096).
Additional findings from the optional self-reported survey were not compared, as the majority of respondents strongly agreed in both groups. This included the clarity of the instructions provided for the treatment regimen, 89.47% (17) in the intervention and 88.23% (19) in the control group. Patients also reported that they felt comfortable talking about their symptoms and concerns, with 84.21% (16) in the intervention and 88.23% (19) in the control group. Both groups, 68.42% (13) in the intervention and 41.18% (7) in the control, were similar in indicating they were very likely to access online educational resources (Fisher’s exact = .179; 1-sided Fisher’s exact .096).
Patient Demographics
The mean age of patients at the time of their consultation appointment was 53.16 years (range 21-88). Of the 51 patients analyzed, 21.56% (11) resided in rural areas greater than 50 km from the urban tertiary care centre, in which the clinic was located. Within the respective study groups, 21.05% (4) were considered rural within the control group, and 21.73% (5) were in the intervention group. The number of premenopausal patients in the control and intervention groups was 42.10% (8) and 43.47% (10), respectively. There were 57.89% (11) postmenopausal patients in the control group, 52.17% (12) in the intervention group, and 1 perimenopausal patient in the intervention group. There was not a significant difference in the proportion of patients with menopause between control and intervention groups (Fisher’s exact = 1.0; 1-sided Fisher’s = .533).
Patient Baseline Presentation and Management
Using Fisher’s exact test, there were no significant differences between the control and intervention groups at the initial visit regarding the distribution of symptoms (P = .886), previous treatments documented in the initial assessment (P = .787), the number of biopsies performed at the initial assessment (P = .468), nor the prescribed treatments at the initial assessment were similar between the control and intervention groups (P = .787; Table S1).
Review of all patients, including those excluded from the study, did not reveal a significant difference in the distribution of diagnoses (P = .994) after the initial visit. While 37.25% had more than 1 diagnosis at their initial assessment, the proportion of those with more than 1 diagnosis was not significant (P = .208). Diagnosed vulvar dermatoses included allergic and or irritant contact dermatitis (35.29%, n = 18); lichen sclerosus (29.41%, n = 15), lichen simplex chronicus (23.52%, n = 12); genitourinary symptoms of menopause (19.6%, n = 10); benign lesions (9.8%, n = 5) which included pilar cysts, epidermal inclusions cysts, and Fordyce spots; infectious etiologies (5.88%, n = 3) including secondary bacterial skin infection and condyloma acuminata; vitiligo (5.88%, n = 3); high-grade squamous intraepithelial lesion (1.96%, n = 1); hidradenitis suppurativa (1.96%, n = 18); lichen planus (1.96%, n = 18); prurigo nodularis (1.96%); and psoriasis (1.96%).
Referral Patterns
Using Fisher’s exact test, there were no significant differences (P = .5096) between the control and intervention groups regarding the time between referral and initial assessment, which were 2.37 and 2.57 months, respectively. Among the included participants (n = 42) in the intervention (n = 23) and control (n = 19) groups, most of the referrals were from family physicians 90.48% (n = 38), with the remainder of patients referred by dermatologists, 4.76% (n = 2), and gynecologists, 4.76% (n = 2). Moreover, 42.86% (n = 18) had a biopsy completed prior to their consultation appointment.
Discussion
This study aimed to assess the impact of the validated VQLI questionnaire during new patient vulvar dermatology assessments. The intervention and control groups were similar regarding demographics, referral, and clinical information. The intervention group completed the VQLI at the baseline appointment, and both the intervention and control groups then completed a follow-up VQLI. As a primary outcome, follow-up VQLI scores were compared between the intervention and control groups. Secondary outcomes compared VQLI scores within the intervention group; analyzed treatment adherence; and surveyed self-reported symptom improvement, whether patient health-related concerns were addressed, and well-being.
Completion of the baseline VQLI among the intervention group participants resulted in a trend of lower mean follow-up VQLI scores 8.3 (0, 18), compared to the follow-up scores of the control group 12.8 (0, 38); however, this difference was not statistically significant (unequal t-test P-value .1529). This may reflect that the use of the validated VQLI2,10 as a screening tool at initial consult assessments may be beneficial for patients to reflect on the impact of the disease on QoL before treatment. This may be due to factors such as sample size, a lack of comparison of the change in VQLI scores between the groups, as a baseline VQLI was not measured in the control group, and the comprehensive overall assessment in our vulvar dermatology clinic, which may not be standardized at other vulvar dermatology sites. Further studies, performed at multiple clinic locations, are required.
Notably, there was a significant difference (unequal t-test P-value < .0001) between baseline, mean VQLI, 18 (6, 36), and follow-up mean VQLI, 8.3 (0, 18), among the intervention group, which may reflect an overall improvement in the impact of vulvar disease on QoL. Furthermore, it demonstrates how completing a baseline and follow-up VQLI can provide patient information about the improvement of disease on QoL and may influence patients’ perception of their health. This is important given the current gap in capturing the impact of patients’ vulvar diseases on QoL despite its profound impact, as vulvar conditions have been shown to cause significant distress and anxiety, affecting physical functioning, everyday activities, and impacting well-being.3,5 This information may otherwise not be forthcoming in a dermatology clinic appointment due to embarrassment, time constraints, or not specifically asking about the various impacts vulvar disease has on overall health. 2 Incorporating the VQLI into routine assessment allows this information to be captured while standardizing disease monitoring and measuring outcomes. 2 Moreover, a recent study demonstrated how the VQLI was shown to correlate with clinician-rated severity of the impact of vulvar lichen sclerosus on QoL. 14 Overall, this highlights the value of incorporating the VQLI in a clinical setting when assessing and treating patients with vulvar disease. It captures information that has been found to often be missed,3,5 it correlates with clinician-rated severity, 14 and can provide information on the improvement of disease.
Most notable among the secondary outcomes was the significant difference in patient self-reported well-being (P = .017). Self-reported improvement of symptoms (P = .684) and whether patients felt their health-related concerns were addressed (P = .391) were statistically similar in both groups. The notable difference in patient self-reported well-being provides important insight and information that the treating physician can incorporate into evaluating their patient’s overall health as it relates to their vulvar disease. The greater self-reported well-being in the intervention group further supports the use of VQLI in routine assessments, particularly in the context of vulvar diseases profound impact on QoL1-4 and in the setting of most women attending a vulvar clinic having a higher baseline anxiety score compared with the general population. 4 Completing the VQLI at baseline helps standardize the encounter by allowing the treating physician to assess the overall impact on QoL that may otherwise be missed. Reassessing the VQLI in subsequent follow-up allows for comparison and thorough assessment of the patient’s disease state, treatment adherence, and QoL.
Moreover, treatment adherence (P = .428) and reported difficulties with the prescribed treatment difference as extrapolated from Question 15 of the VQLI, “Over the past month, how much of a problem has the treatment of your vulvar symptoms been (For instance messy, time consuming, expensive, inconvenient)?” (Fisher’s exact P-value = 1.000; 1-sided Fisher’s = .567) were statistically similar amongst the 2 groups. The latter may be due to a small sample size, as only 7 patients amongst the study participants (n = 42) who answered this question did not follow treatment. Nonetheless, the treatment adherence of the control group, 89.5% (17), and the intervention group, 78.3% (18), are notable when evaluating conditions where almost one-third of patients are non-compliant. 15 Use of the VQLI in routine assessments may improve overall treatment compliance, and further studies are required.
Limitations of this study include the sample size, lack of comparison of the change in VQLI scores between groups, and the nature of the global assessment in the vulvar dermatology clinics. Further studies applying the VQLI in initial vulvar dermatology assessments, monitoring disease, measuring the impact on QoL, and assessing treatment adherence are required.2,10,14 The VQLI continues to be a standard in monitoring disease, measuring outcomes, and addressing adherence.
Conclusion
The use of the VQLI in initial and follow-up vulvar dermatology assessments has the potential to detect changes in patient self-reported well-being and QoL. By allowing patients to evaluate themselves before and after dermatologic intervention, the questionnaire eased the collection of patient perspectives about disease impact. Additionally, provided patient perception and data that the attending physician can use to evaluate the patient's overall health in relation to their vulvar disease. To expedite the collection of this patient history data, consideration may be given to incorporating the VQLI into the referral process, making this information available prior to the initial appointment.
Supplemental Material
sj-docx-1-cms-10.1177_12034754251347552 – Supplemental material for Evaluating the Impact of the Vulvar Quality of Life Index for New Patient Dermatology Assessments
Supplemental material, sj-docx-1-cms-10.1177_12034754251347552 for Evaluating the Impact of the Vulvar Quality of Life Index for New Patient Dermatology Assessments by Kaylin Bechard, Anita Truong, Samuel A. J. Lowe, Pamela Mathura, Reidar Hagtvedt and Marlene T. Dytoc in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-2-cms-10.1177_12034754251347552 – Supplemental material for Evaluating the Impact of the Vulvar Quality of Life Index for New Patient Dermatology Assessments
Supplemental material, sj-docx-2-cms-10.1177_12034754251347552 for Evaluating the Impact of the Vulvar Quality of Life Index for New Patient Dermatology Assessments by Kaylin Bechard, Anita Truong, Samuel A. J. Lowe, Pamela Mathura, Reidar Hagtvedt and Marlene T. Dytoc in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-pdf-3-cms-10.1177_12034754251347552 – Supplemental material for Evaluating the Impact of the Vulvar Quality of Life Index for New Patient Dermatology Assessments
Supplemental material, sj-pdf-3-cms-10.1177_12034754251347552 for Evaluating the Impact of the Vulvar Quality of Life Index for New Patient Dermatology Assessments by Kaylin Bechard, Anita Truong, Samuel A. J. Lowe, Pamela Mathura, Reidar Hagtvedt and Marlene T. Dytoc in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
