Abstract
Background:
Adalimumab is a biologic used in the treatment of hidradenitis suppurativa (HS). Therapeutic drug monitoring (TDM) has emerged as a potential strategy to optimize treatment efficacy, yet its utility in HS remains underexplored. Our aim was to assess the utility of TDM in HS patients with suboptimal adalimumab response by investigating the prevalence of antidrug antibodies and subtherapeutic drug levels.
Methods:
A cross-sectional study of 62 patients with suboptimal response to adalimumab was conducted at a dermatology clinic in Toronto, Ontario. Data on adalimumab serum trough levels, autoantibody status, and demographics were collected. Patients were divided into therapeutic (≥10.6 μg/mL) and subtherapeutic (<10.6 μg/mL) drug categories based on trough level.
Results:
Of 51 patients on adalimumab 40 mg weekly, 32 patients (62.7%) had therapeutic drug levels and 19 (37.3%) had subtherapeutic levels. In the 11 patients on adalimumab 80 mg weekly, 7 patients had therapeutic drug levels (28.19 μg/mL) and 4 had subtherapeutic levels (mean 3.26 μg/mL). Autoantibodies were detected in 21.06% of patients with subtherapeutic drug levels on adalimumab 40 mg weekly. There was a significant association between Hurley stage and drug level (P = .015) in patients on adalimumab 40 mg weekly.
Conclusions:
In HS patients with suboptimal response to standard adalimumab dosing, a significant number of patients have subtherapeutic drug levels with a minority of those having anti-adalimumab antibodies. TDM can be helpful in identifying HS patients with subtherapeutic drug levels and without antidrug antibodies who could potentially benefit from dose escalation.
Keywords
Introduction
Hidradenitis suppurativa (HS) is a debilitating inflammatory skin condition characterized by recurrent nodules, abscesses, sinus tracts, and scarring in flexural sites. 1 The pathogenesis is multifactorial, likely involving genetic, bacterial, hormonal, and immune-related factors. Risk factors for HS include smoking, mechanical stress, obesity, and family history, while comorbidities include depression, hyperlipidemia, diabetes, hypertension, chronic kidney disease, respiratory disease, spondyloarthropathy, and inflammatory bowel disease (IBD).2-5
Levels of the cytokine tumour necrosis factor-alpha (TNF-α) are elevated in lesions of HS, providing a basis for the use of biologics in the treatment of severe HS. 6 Adalimumab, a TNF-α inhibitor, is the first-line systemic treatment for patients with moderate-severe HS. 7 Adalimumab is also used in other inflammatory conditions including IBD, psoriasis, and rheumatoid arthritis.8-10 For HS, adalimumab is given as a weekly 40 mg injection, after a loading dose of 160 mg (day 1) and 80 mg (day 15). 9 This contrasts with the dosing regimen given for psoriasis and Crohn’s disease, which is 40 mg given every 2 weeks.9,10
Based on the PIONEER I and II trials, ~42% to 59% of patients achieved Hidradenitis Suppurativa Clinical Response (HiSCR) by week 12, defined as a 50% reduction in absolute nodule count with no increase in abscess or draining tunnel count relative to baseline.11,12 The reasons why some patients with HS respond to adalimumab while others do not, or experience a loss of response over time, remain unclear. Various hypotheses have been proposed to explain these differences, including mechanistic failure of the drug, the development of bacterial biofilms, and the presence of subtherapeutic drug levels. 13 Low drug levels may occur for a variety of reasons including antidrug antibody (ADA) formation, suboptimal dosing, increased drug clearance, body weight variation, and genetic polymorphisms.13-15
Therapeutic drug monitoring (TDM) is the measurement of the serum drug concentration immediately before the next scheduled dose (trough), and screening for the development of ADAs.13,16 Evidence from other inflammatory diseases has demonstrated that TDM may enable optimization of treatment outcomes through dose optimization.10,16,17 As such, the primary objective of our study was to determine what proportion of HS patients with suboptimal clinical response to adalimumab have subtherapeutic drug levels, and secondly, what factors are associated with these subtherapeutic levels.
Materials and Methods
A cross-sectional study of 62 patients was conducted at the dermatology clinic at Sunnybrook Health Sciences Centre (Toronto, ON, Canada). Patients included had moderate-severe HS, were on adalimumab 40 or 80 mg weekly maintenance dosing, had suboptimal clinical response, and had TDM results available. TDM was only performed when a patient was deemed to have suboptimal response after a minimum of 3 months duration on therapy. Suboptimal response was determined based on clinical assessment by the treating dermatologist and was defined as an increased number of inflammatory nodules and/or sinus tracts compared with pre-adalimumab assessment. Subtherapeutic drug levels were defined as <10.6 µg/mL. This level was determined based on pharmacokinetic analysis of the PIONEER I and II trials showing that mean serum adalimumab level in responders ranged from 10.3 to 10.6 μg/mL compared with nonresponders, which ranged from 6.93 to 7.23 µg/mL. 13 Per testing laboratory protocol, antidrug antibodies were only tested for when drug level was undetected (≤0.024 µg/mL). Patients who were on 80 mg of adalimumab had previously been on 40 mg but were dose-escalated due to suboptimal response and subtherapeutic drug levels. All patients who were dose-escalated did not show prior evidence of antidrug antibodies.
Blood was collected once from patients prior to receiving the next scheduled dose (trough level) and was sent to Gamma Dynacare Medical Laboratories using 2 enzyme-linked immuno-sorbent assay kits, IDKmonitor® Adalimumab drug level (Immundiagnostik, Bensheim, Germany) and Promonitor®-Anti-ADL (Progenika Biopharma, Derio, Spain).18,19
Electronic medical records were reviewed to obtain demographic data, antibody status, and drug levels. Inflammatory comorbidities were defined as any of the following: pyoderma gangrenosum, inflammatory arthritis, psoriasis, or IBD. Differences in demographic data and clinical characteristics between the groups were examined using Fisher’s exact test and chi-squared test whenever appropriate. Patients on 40 and 80 mg weekly were analyzed separately. A P < .05 was considered statistically significant.
The STROBE checklist was used as a guidance in formulating the manuscript. The study protocol complied with the principles of the Declaration of Helsinki, and it was approved by the institutional review board.
Results
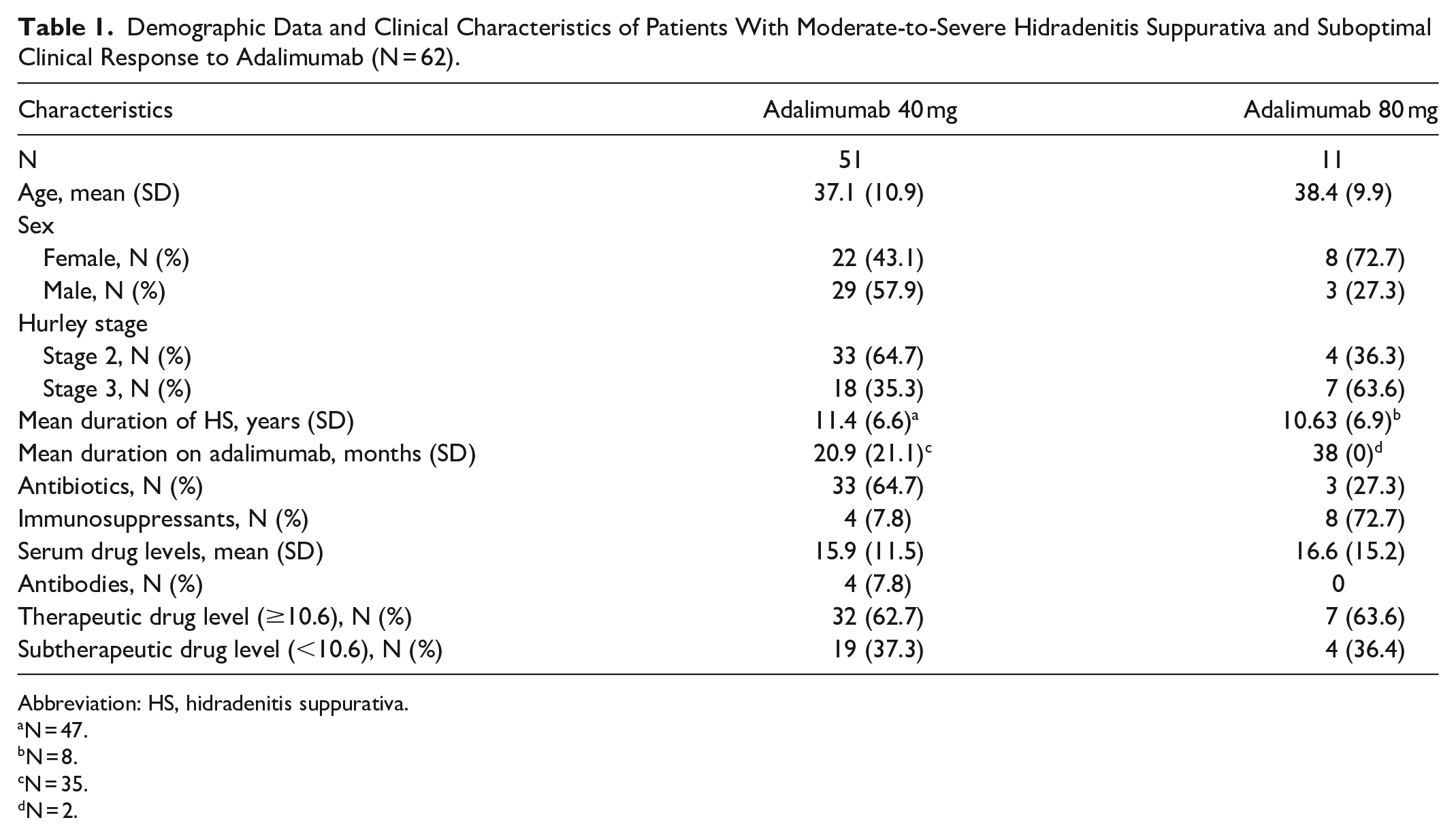
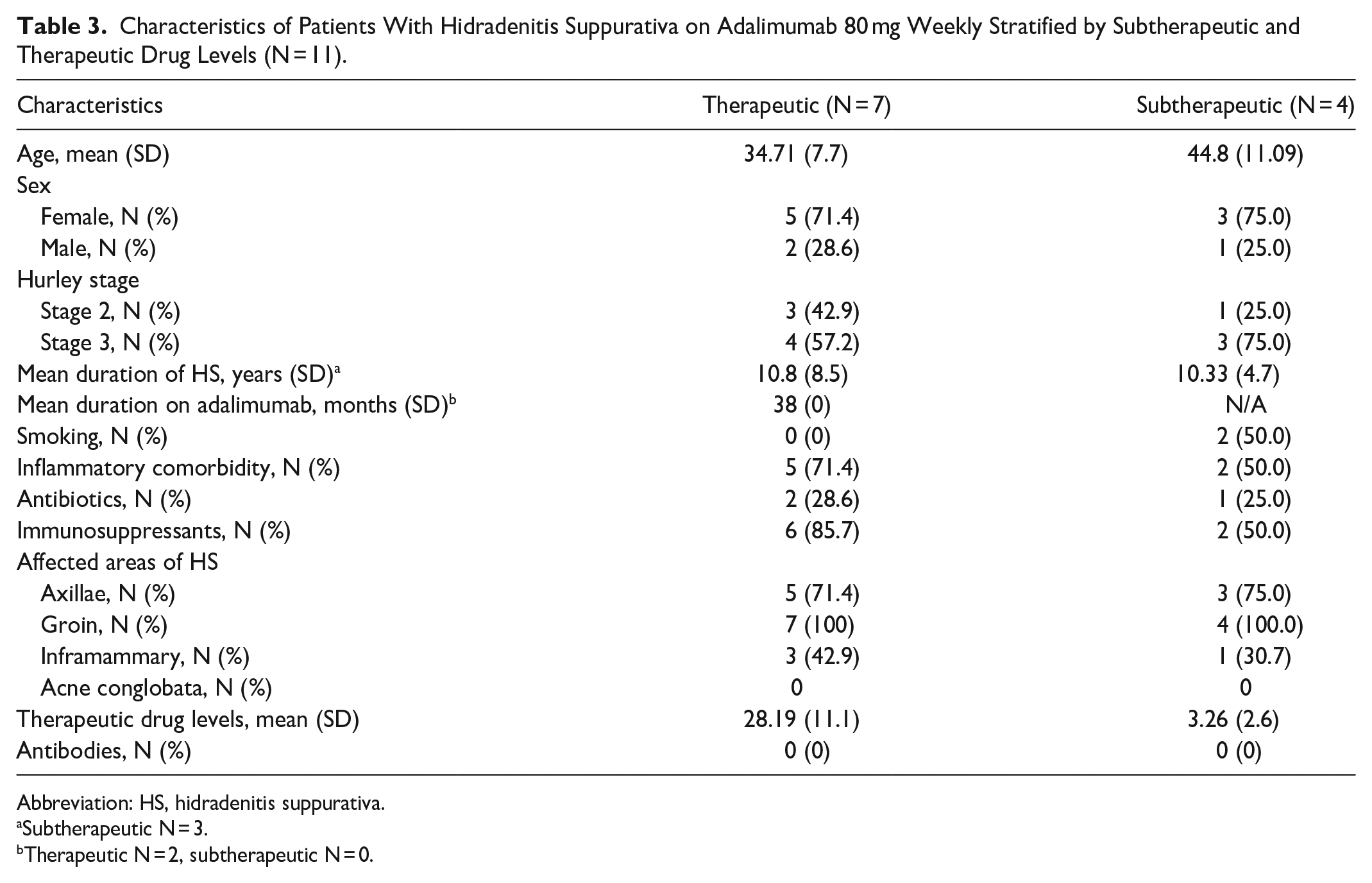
Demographic information for all 62 patients is presented in Table 1. In 51 patients on adalimumab 40 mg, and 11 patients on adalimumab 80 mg, the mean serum adalimumab concentrations were 15.97 μg/mL (SD 11.5) and 16.63 μg/mL (SD 15.2), respectively. In the adalimumab 40 mg group, 32 patients (62.7%) had therapeutic drug levels (mean 22.26 μg/mL) and 19 (37.3%) had subtherapeutic levels (mean 4.16 μg/mL). In the adalimumab 80 mg group, 7 (63.6%) patients had therapeutic drug levels (mean 28.19 μg/mL) and 4 (36.4%) had subtherapeutic levels (mean 3.26 μg/mL). Antidrug antibodies were present in 4 patients (21.1%) on 40 mg dosing and only in those with subtherapeutic levels. No patients in the 80 mg group had antidrug antibodies detected.
Demographic Data and Clinical Characteristics of Patients With Moderate-to-Severe Hidradenitis Suppurativa and Suboptimal Clinical Response to Adalimumab (N = 62).
Abbreviation: HS, hidradenitis suppurativa.
N = 47.
N = 8.
N = 35.
N = 2.
Patients were further stratified based on therapeutic and subtherapeutic drug level status, and associated characteristics are outlined in Table 2 (40 mg) and Table 3 (80 mg). There was no significant association between drug level and smoking, sex, inflammatory comorbidities, concomitant antibiotic use, immunosuppressant use, or anatomic location of HS. However, there was a significant association between Hurley stage and drug level (P = .015) for those on adalimumab 40 mg weekly. In the 40 mg cohort, most patients with therapeutic drug levels were Hurley stage 2 (78.1%), and most patients with subtherapeutic drug levels were Hurley stage 3 (57.9%).
Characteristics of Patients With Hidradenitis Suppurativa on Adalimumab 40 mg Weekly Stratified by Subtherapeutic and Therapeutic Drug Levels (N = 51).

Abbreviation: HS, hidradenitis suppurativa.
40 mg therapeutic N = 28.
40 mg therapeutic N = 25, subtherapeutic N = 10.
Characteristics of Patients With Hidradenitis Suppurativa on Adalimumab 80 mg Weekly Stratified by Subtherapeutic and Therapeutic Drug Levels (N = 11).
Abbreviation: HS, hidradenitis suppurativa.
Subtherapeutic N = 3.
Therapeutic N = 2, subtherapeutic N = 0.
Discussion
While TDM is a routine practice in other inflammatory conditions, its role in the treatment of HS is still largely unknown. Due to limited systemic treatment options, it is worthwhile investigating if TDM and dose optimization can improve treatment outcomes for patients with HS.
Out of 62 patients with HS who had suboptimal clinical response to adalimumab, over one third had subtherapeutic drug levels. These findings are slightly different from the findings of a smaller case series by Abdalla et al who found 42% of nonresponders to adalimumab had subtherapeutic drug levels. 15 Although the authors used a <6 μg/mL threshold for subtherapeutic levels, which does not reflect the pharmacokinetic evidence in HS patients, a higher proportion of their patients had subtherapeutic drug levels. Similarly, a recent study of 67 patients with HS showed that of their 19 responders to adalimumab, 63% maintained therapeutic drug levels, while most suboptimal responders had subtherapeutic drug levels (58% and 69% for primary and secondary suboptimal responders, respectively). 20 These findings underscore the importance of considering serum drug levels when assessing a patient’s response to adalimumab treatment.
Previous pharmacokinetic analyses have shown that adalimumab has a dose-response relationship, with improved clinical response at higher doses.13,21 Therefore, TDM and subsequent dose optimization in those with suboptimal clinical response and subtherapeutic drug levels is a logical next step. 16 Indeed, 2 small case series have shown that dose intensification to 80 mg weekly can improve clinical outcomes in HS.22,23 Additionally, a recent retrospective case series investigated HS patients with suboptimal response to adalimumab 40 mg weekly to see whether dose escalation to 80 mg weekly would improve clinical response and drug levels. 21 Out of 16 patient’s dose-escalated, 7 improved clinically with corresponding increases in their drug levels from 7.8 to 18.7 μg/mL. Interestingly, those who were dose-escalated and did not clinically improve had similar increases in their drug levels, indicating that higher drug levels do not always correlate with improved clinical response.
There are a few reasons why the dosing of adalimumab may need to be higher in HS than in other inflammatory conditions. 10 Firstly, compared with psoriasis and IBD, HS patients are known to have a higher drug clearance of adalimumab. 13 Additionally, it is well known that higher adalimumab levels are needed to satisfy more stringent outcome criteria, such as fistula healing in Crohn’s disease. 10 A 2014 national expert consensus on IBD suggests target adalimumab drug levels of 8 to 12 µg/mL, and not discontinuing until levels of 10 to 15 µg/mL are achieved.10,24 Comparatively, the suggested adalimumab therapeutic window for patients with psoriasis is ~3.2 to 7.0 μg/mL.24-26 HS likely requires higher serum drug levels based on its significant inflammatory burden, similar to what may be seen in fistulizing Crohn’s disease. 13
Regarding the cause for low drug levels, ~20% of our patients with subtherapeutic drug levels had ADAs. This is lower than prior estimates showing that 56% of nonresponders with subtherapeutic drug levels had ADAs. 15 Phase I and II clinical trials found an overall low rate of ADAs in HS patients on adalimumab, ranging from 3.2% to 10.1%. 13 The presence of antibodies is an important factor to assess as dose optimization is unlikely to improve drug levels or clinical response in those with ADAs. When antibodies are present in low titers, but drug levels are still detectable (a scenario not detected by our laboratory protocols), addition of an immunosuppressant may be tried to suppress antibody formation.10,16 Interestingly, no ADAs were detected in our 80 mg cohort despite subtherapeutic levels. However, over 70% of this group were on concomitant immunosuppressants, which could explain this finding. Additionally, this cohort would have had previous negative testing for ADAs as a requirement to be dose-escalated to 80 mg and therefore may reflect selection bias for a group that is inherently less likely to develop immunogenicity.
Finally, we found that higher Hurley stage was associated with subtherapeutic drug levels. One explanation for this is that a higher inflammatory burden functions to decrease drug levels itself. Alternatively, because patients have lower drugs levels to begin with, their disease goes untreated, and they progress to more advanced disease with a higher Hurley stage. Interestingly, there is evidence, which suggests that an elevated C-reactive protein (a nonspecific inflammation marker) results in higher drug clearance and lower adalimumab drug levels. 13 Overall, our study suggests that high disease burden rather than ADAs is the main cause of subtherapeutic drug levels in most patients with suboptimal clinical response.
Limitations to our study include a small sample size and, due to a lack of standardized guidelines, our chosen threshold of <10.6 µg/mL as subtherapeutic drug level makes it hard to compare with other studies that may use a different threshold. Another major limitation is the lack of standardized criteria (such as the HiSCR) for defining patient response or nonresponse to adalimumab. Further studies are required to establish standardized cut-offs for adalimumab drug levels in the treatment of HS.
In conclusion, despite lack of standardized guidelines, these findings suggest that TDM is a useful tool in patients with suboptimal clinical response to adalimumab to identify those who may benefit from subsequent dose escalation.
Footnotes
Author Contributions
Hannah Stirton: conducting a research and investigation process, specifically performing the experiments, or data/evidence collection; preparation, creation, and/or presentation of the published work, specifically writing the initial draft (including substantive translation). Raed Alhusayen: ideas; formulation or evolution of overarching research goals and aims; oversight and leadership responsibility for the research activity planning and execution, including mentorship external to the core team; critical review, commentary, or revision—including pre- or post-publication stages.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Sunnybrook Research Institute (no. SUN-2647) on June 2020.
Consent for Publication
The Ethics Committee of the Sunnybrook Research Institute waived the need for patient consent for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this noninterventional study.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
