Abstract
Background:
Early melanomas, dysplastic melanocytic nevi, and melanocytic tumours of uncertain malignant potential (MELTUMPs) reveal similar clinic and dermoscopic findings leading to underdiagnosis of malign lesions or unnecessary excision of benign ones. High-grade dysplastic nevi and MELTUMPs in the intermediate category should be recognized and completely excised.
Objectives:
We evaluated the diagnostic performance of pattern analysis, ABCD rule, colour, architecture, symmetry, and homogeneity algorithm, melanoma-specific structures and asymmetry of dermoscopic features in distinguishing early melanomas, high-grade dysplastic nevi, and MELTUMPs from low-grade dysplastic nevi.
Methods:
We retrospectively assessed dermoscopic images blindly to the histopathological diagnoses in a university hospital.
Results:
One hundred forty histopathologically confirmed melanocytic lesions were included (93 low-grade dysplastic nevi, 26 thin melanomas, 17 high-grade dysplastic nevi, and 4 MELTUMPs). All dermoscopic methods had poor diagnostic performance in early melanomas and intermediate melanocytic lesions. In the multivariate analyses of dermoscopic findings, the atypical pigment network (OR: 3.58, 95% CI: 1.31-9.72), asymmetry of globules (OR: 3.17, 95% CI: 1.37-7.35), streaks (OR: 6.16, 95% CI: 1.95-19.48) and homogenous structureless areas (OR: 5.92, 95% CI: 2.36-14.92) were the significant predictive factors for melanomas and intermediate melanocytic lesions. Positive predictive values of melanoma-specific structures were shiny white structures (100%), scar-like depigmentation (85.7%), blue-white veil (72.7%), atypical blotch (72.2%), and negative pigment network (71.4%).
Conclusions:
Shiny white structures, atypical network, asymmetry of globules, streaks, and homogeneous structureless areas were the most significant dermoscopic findings in discriminating early melanomas and intermediate melanocytic lesions from low-grade dysplastic nevi.
Introduction
High-grade dysplastic nevi and “melanocytic tumours of uncertain malignant potential” (MELTUMPs) fall under the WHO 2018 intermediate category and exhibit clinical and dermoscopic characteristics similar to early melanomas and low-grade dysplastic nevi, making accurate diagnosis challenging.1,2 This similarity can lead to either the underdiagnosis of malignant lesions or the unnecessary excision of benign ones. 3 Furthermore, these intermediate lesions often show low interobserver agreement among experts in clinical practice.2,4,5 Proper management involves the complete excision of high-grade dysplastic nevi and MELTUMPs, with recommended surgical margins to ensure thorough removal.4,5
Early diagnosis and excision are critical for reducing melanoma-related mortality and improving prognosis. 6 Various dermoscopic algorithms are used to differentiate melanoma from benign lesions, with reported sensitivities ranging from 78% 7 to 98% 8 depending on the evaluated parameters, dermatologists’ experience, 9 and the diagnostic difficulty of the evaluated lesions. 10 Pattern analysis 11 is based on classifying melanocytic lesions by global patterns and a distinctive combination of specific local features, while the ABCD rule focuses on asymmetry, borders, colours, and differential structures. 12 The CASH (colour, architecture, symmetry, and homogeneity) algorithm evaluates many dermoscopic features in the ABCD rule, but unlike the other algorithms, it assesses the architecture of the lesion. 8
High-grade dysplastic nevi, 1 characterized by severe cytological and architectural atypia, and MELTUMPs, 13 which show poorly defined findings in terms of malignancy, and have the potential to metastasize, pose a significant diagnostic challenge. There are limited data about the distinguishing dermoscopic findings of the intermediate lesions, especially from low-grade dysplastic nevi.
The accurate differentiation of these lesions is crucial for appropriate clinical management, as underdiagnosis can result in delayed treatment of malignant lesions, while overdiagnosis can lead to unnecessary surgical procedures and associated morbidity. This study aimed to assess the sensitivity, specificity, and diagnostic accuracy of pattern analysis, the ABCD rule, and the CASH algorithm, which include different parameters for these challenging melanocytic lesions. Additionally, we aimed to identify dermoscopic findings that can help differentiate early melanomas, high-grade dysplastic nevi, and MELTUMPs from low-grade dysplastic nevi.
Methods
Inclusion and Exclusion Criteria of Melanocytic Lesions
We included low-grade dysplastic nevi, thin melanomas, high-grade dysplastic nevi, and MELTUMPs, which were evaluated between January 2010 and June 2021 in the dermoscopy unit of a university hospital and excised due to clinical and dermoscopic suspicious findings. Polarized light dermoscopy (MoleMax HD; Derma Instruments, Vienna, Austria) was used to examine lesions. Cases obtained from histopathological databases were included when corresponding dermoscopic images of sufficient quality were available. Lesions from the face, scalp, ear, and acral and genital areas were excluded.
An experienced dermatopathologist had previously evaluated all lesions. In histopathological examinations, a lesion with architectural disorder showing nuclear variation prominent in a larger minority of the nevus cells characterized by enlargement of nuclear size, coarse granular chromatin, or peripheral condensation, hyperchromatism, and prominent nucleoli, was diagnosed as a high-grade dysplastic nevus. If the lesion with architectural disorder displayed prominent nuclear variation in a small minority of nevus cells with hyperchromatism and prominent nuclei (random atypia), it was diagnosed as a low-grade dysplastic nevus. 1 Melanocytic lesions showing dermal melanocytic proliferations that exhibit ill-defined architectural and cytological atypia in the epidermal component and dense lymphohistiocytic cell infiltration around the dermal mass were classified as MELTUMP. 14 HMB45, Ki-67, Melan-A, and SOX-10 immunohistochemical stains were used to assess biopsy specimens with the diagnosis of MELTUMPs.
Dermoscopic Assessment
Two experienced dermatologists (C.A. and T.İ.) blinded to histological diagnosis classified each lesion as benign or malign using 3 algorithms: pattern analysis, the ABCD rule, and the CASH algorithm. Cohen’s kappa analysis was used to determine the level of agreement between the 2 dermatologists. In addition, the presence or absence of predefined dermoscopic features, colour of lesions, asymmetry in at least 1 axis of shape, dermoscopic structures, and colour distribution of lesions were retrospectively analyzed. In cases of disagreement, a third experienced investigator (SA) provided a final classification.
Equivocal melanocytic lesions were subdivided into 2 groups; the excision-recommended group (n = 47), including thin melanomas, high-grade dysplastic nevi, and MELTUMPs, and the excision-not-required group, including low-grade dysplastic nevi (n = 93).
Statistical Analysis
Descriptive statistics included mean, standard deviation, median, minimum, maximum, frequency, and ratio values. The normality of variables was tested using the Kolmogorov-Smirnov and Shapiro-Wilk tests. Quantitative independent data were analyzed using the Mann-Whitney U test, while qualitative independent data were analyzed using the chi-squared test or Fischer’s exact test when appropriate. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value were calculated for 3 dermoscopic algorithms and dermoscopic features. Kappa tests assessed concordance. Univariate and multivariate logistic regression analyses identified significant predictors for distinguishing thin melanomas, MELTUMPs, and high-grade dysplastic nevi from low-grade dysplastic nevi. Receiver operating characteristic (ROC) curve analysis quantified the accuracy of dermoscopic algorithms and features in recommending lesion excision. An ROC curve visualizes the performance of a diagnostic test by plotting its sensitivity on the y-axis against the false-positive rate on the x-axis. The area under the ROC curve (AUC) is used to measure how effectively a parameter can differentiate between 2 diagnostic groups (excision-recommended group/excision-not recommended group). Analyses were conducted using IBM SPSS Statistics 28.0.0, with P-values <.05 considered statistically significant.
Results
Characteristics of Patients and Lesions
A total of 140 equivocal melanocytic lesions of 130 patients (58 women and 72 men) were included in the study. The mean age of the patients was 39 ± 16 years with a range of 18 to 78 years. Of the 140 melanocytic lesions, 93 were low-grade dysplastic nevi (Figure 1a), 17 were high-grade dysplastic nevi (Figure 1b), 4 were MELTUMPs (Figure 2a) and 26 were thin melanomas (Figure 2b). Histopathological features are given in Figure 1c and d and Figure 2c and d. Of melanomas, 7 were in situ, and 19 were Stage IA with a mean Breslow thickness of 0.61 ± 0.24 mm (range: 0.1-1 mm). Only one melanoma had a Breslow thickness equal to 1 mm. Most of the lesions were located on the posterior trunk (51.4%), with the remaining on the abdomen (14.3%), on the pectoral area (13.6%), on the extremities (19.3%) and on the neck (1.4%). Regarding the special nevus sites, 16 of the 19 lesions on the pectoral area were located on the breast (in 16 male patients). None of these lesions were located on the areolar area. There was no statistical difference between the excision-recommended group and the excision-not-required group regarding localizations (P > .05).
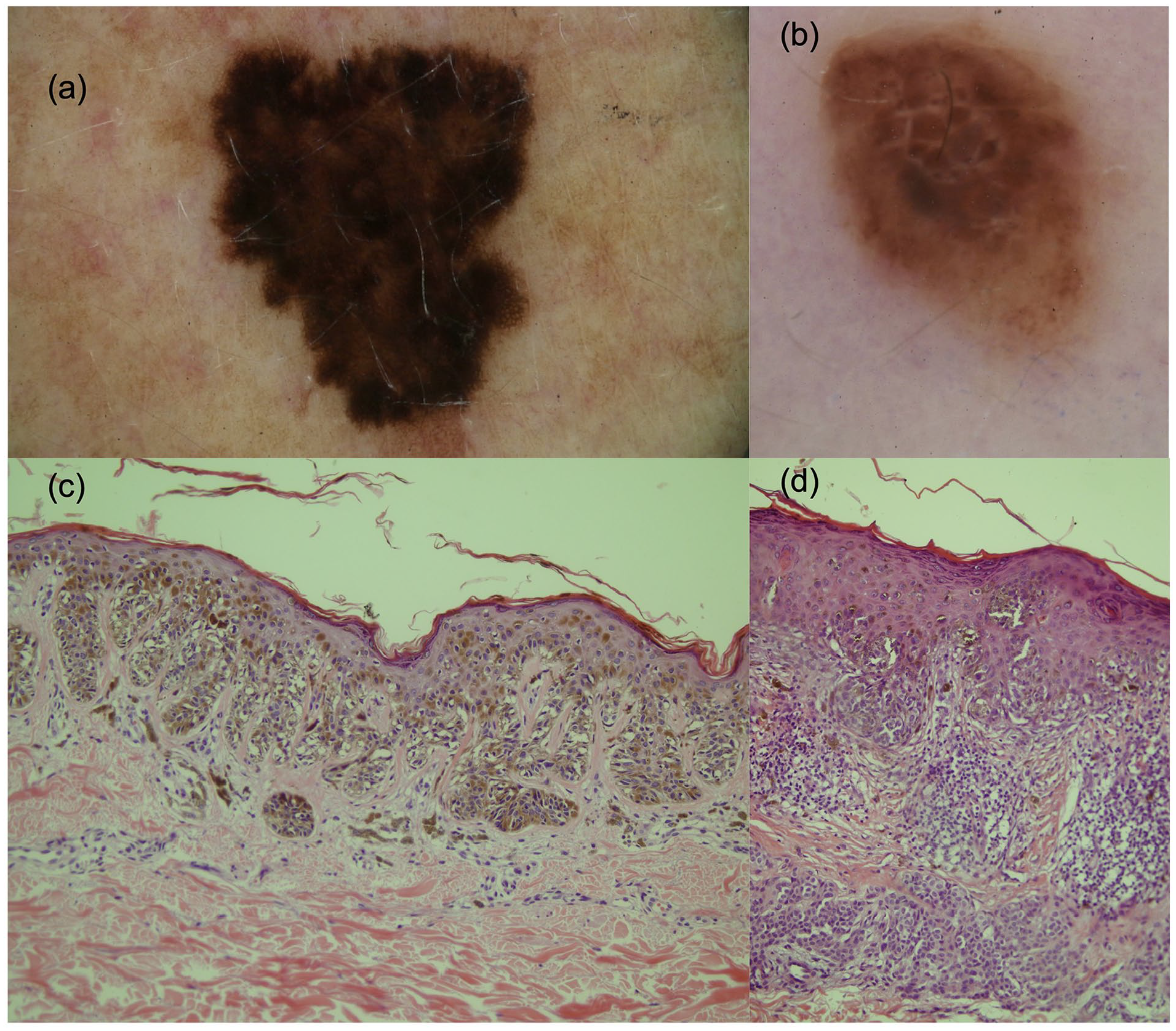
(a) A low-grade dysplastic nevus diagnosed as malign in pattern analysis, the ABCD rule, and the CASH algorithm. (b) A high-grade dysplastic nevus diagnosed as malign in pattern analysis and the CASH algorithm, and as suspicious in the ABCD rule (total dermoscopic score was between 4.75 and 5.45). Shiny white streaks are seen in the centre of the lesion. (c) Histopathology of the low-grade dysplastic nevus in (a) showing nevus cells with cytological atypia showing hyperchromatism and prominent nuclei in a small minority of cells in the epidermal component (haematoxylin and eosin stain, original magnification ×20). (d) Histopathology of the high-grade dysplastic nevus in (b) showing nevus cells with severe cytological and architectural atypia (haematoxylin and eosin stain, original magnification ×20).
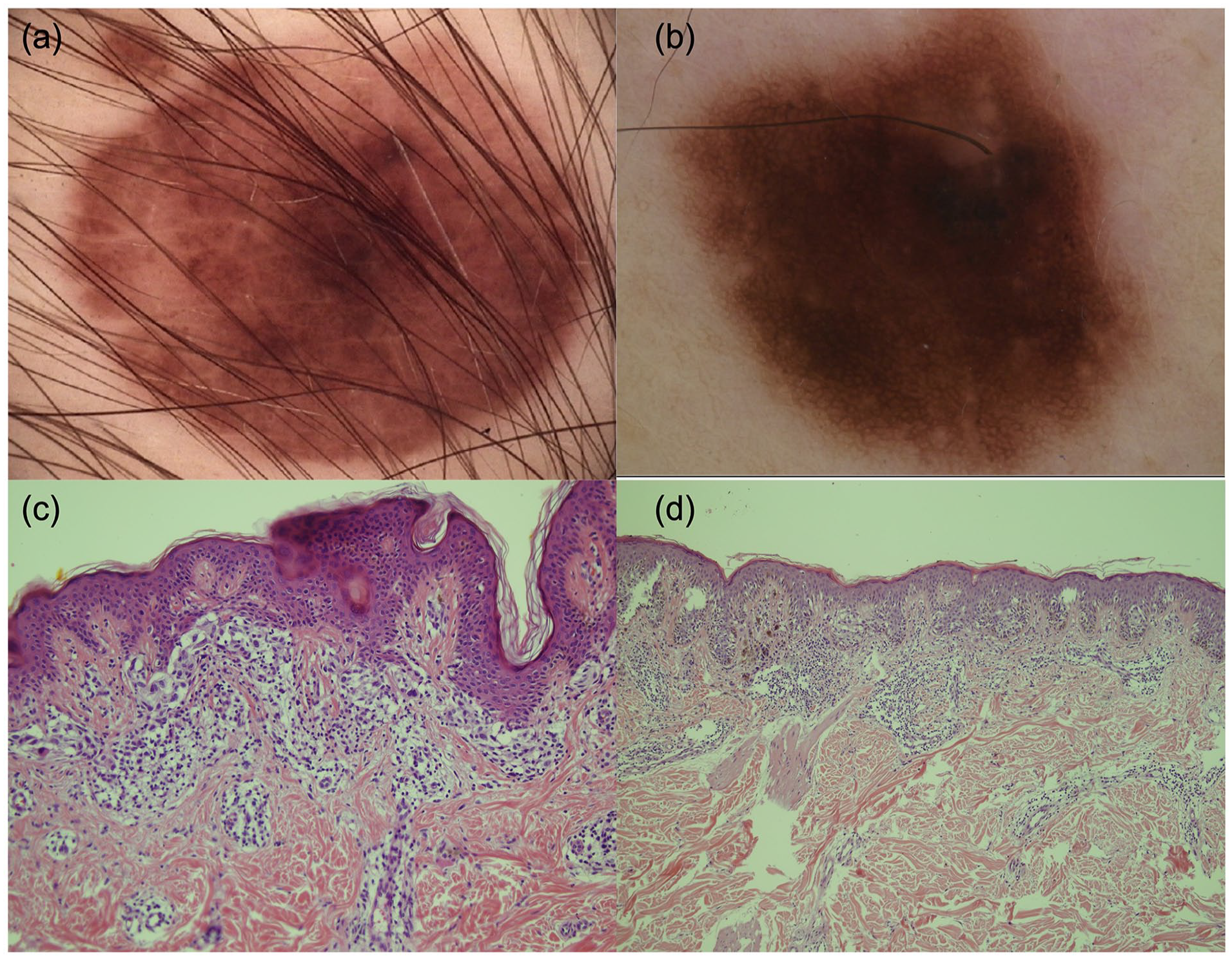
(a) A MELTUMP diagnosed as malign with all algorithms. Asymmetric homogenous structureless areas are seen. (b) A melanoma diagnosed as malign in pattern analysis and the CASH algorithm while as benign in the ABCD rule. (c) Histopathology of the MELTUMP in (a), with dermal and epidermal components and marked cytologic and architectural atypia. Mitosis is absent in the dermal component, and there is a band-like, very dense lymphohistiocytic cell infiltration including plasma cells in the dermis around and at the base of the lesion. The dermal component is generally mature, but focal loss of maturation and marked nuclear pleomorphism are seen. Ki-67 proliferative index is 5% to 6%. There are no pagetoid melanocytes in the epidermis with Melan-A and HMB45. Staining stratification (decreased staining toward the dermis) was found in the dermal component with HMB45 (haematoxylin and eosin stain, original magnification ×20). (d) Histopathology of the superficial spreading melanoma in (b) with a Breslow depth 0.5 mm (haematoxylin and eosin stain, original magnification ×10). Immunohistochemistry is positive for protein HMB45, SOX-10, and Melan-A.
Dermoscopic Algorithms
The agreement between the 2 dermatologists was fair in pattern analysis and the ABCD rule of dermoscopy (κ = 0.342, P < .001; κ = 0.354, P < .001, respectively). A moderate agreement (κ = 0.499, P < .001) existed between them using the CASH algorithm.
Of the 140 equivocal melanocytic lesions, 71 were correctly diagnosed by pattern analysis (50.1%) compared with 91 of 140 (65%) and 85 of 140 (60.7%) by the ABCD rule and the CASH algorithm, respectively. Table S1 shows that pattern analysis had a higher sensitivity (93.6%) and a lower specificity (29%) than the ABCD rule (sensitivity: 68.1%, specificity: 64.4%) and the CASH algorithm (sensitivity: 72.3%, specificity: 54.8%). At the ABCD score cut-off value of 5.45, sensitivity was 51.1% and specificity was 75.3% in differentiating melanocytic lesions.
ROC Analysis
The AUC was 0.667 (95% CI: 0.571-0.763) for the ABCD rule; 0.636 (95% CI: 0.540-0.732) for the CASH algorithm; and 0.613 (95% CI: 0.519-0.707) for pattern analysis (Figure S1a). [The AUC ranges from 0.5 (equivalent to chance) to 1 (indicating perfect discrimination). AUC values between 0.6 and 0.7 suggest that the performance of the diagnostic test is poor.]
AUC for at least 1 axis asymmetry of globules was 0.637 (95% CI: 0.538-0.735); at least 1 axis asymmetry of streaks was 0.622 (95% CI: 0.519-0.725); and at least 1 axis asymmetry of homogeneous structureless areas was 0.658 (95% CI: 0562-0754) (Figure S1b). The total number of melanoma-specific structures had an AUC of 0.784 (95% CI: 0.705-0.862).
Pattern Analysis
Between the 2 groups, the frequency of diffuse reticular pattern, patchy reticular pattern, peripheral reticular with central hypopigmentation, homogeneous pattern, peripheral globules/starburst pattern, peripheral reticular with central globules pattern, 2-component pattern, and nonspecific pattern did not differ (P > .05). The rate of peripheral reticular with central hyperpigmentation pattern in the excision-recommended group (8.5%) was significantly lower than in the excision-not-required group (24.7%) (P = .022). The rate of multicomponent pattern in the excision-recommended group (46.8%) was significantly higher than in the excision-not-required group (24.7%) (P = .008).
Melanoma-Specific Structures
The median number of melanoma-specific structures was significantly (P < .001) higher in the excision-recommended group (median: 2; range: 0-10) than in the excision-not-required group (median: 1, range: 0-4). The frequencies of melanoma-specific structures in melanocytic lesions are shown in Figure 3. It was determined that shiny white streaks (crystalline structures) were seen only in thin melanomas, high-grade dysplastic nevi, and MELTUMPs.
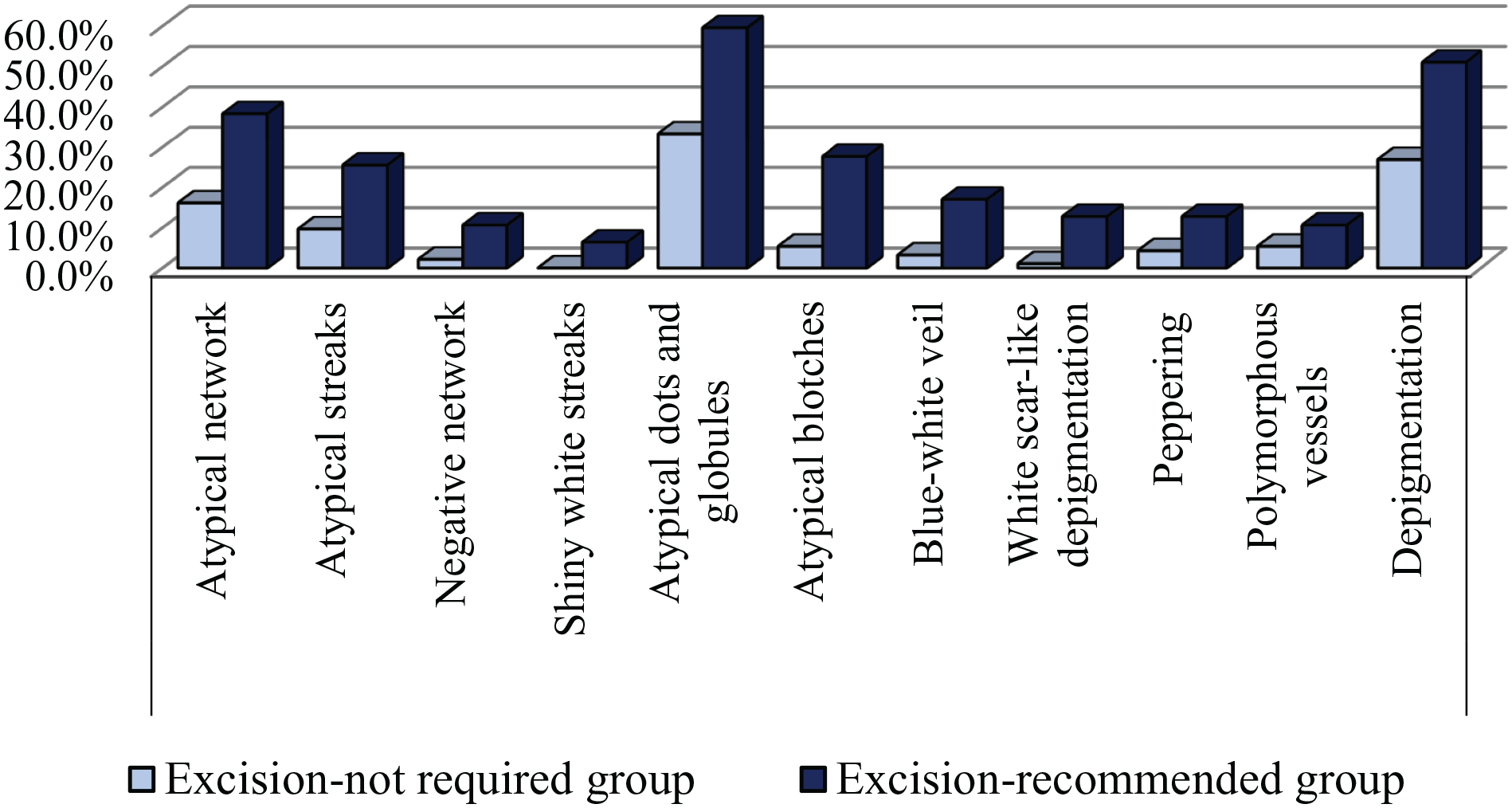
Frequency of melanoma-specific structures and depigmentation in the study groups. In the excision-recommended group, the rate of the atypical network, atypical streaks, negative pigment network, atypical dots and globules, atypical blotch, blue-white veil, white scar-like areas, and depigmentation were statistically significantly higher than in the excision-not-required group. Shiny white streaks (crystalline structures) were seen only in the excision-recommended group.
In the excision-recommended group, the rate of the atypical network (38.3% vs 16.1%, P = .004), atypical streaks (25.2% vs 9.7%, P = .013), negative pigment network (10.6% vs 2.2%, P = .03), atypical dots and globules (59.6% vs 33.3%, P = .003), atypical blotch (27.7% vs 5.4%, P < .001), blue-white veil (17% vs 3.2%, P = .004), white scar-like areas (12.8% vs 1.1%, P = .003) and depigmentation (51.1% vs 26.9, P = .005) were significantly higher than in the excision-not-required group.
The ratio of peppering and polymorphic vessels did not differ significantly between the 2 groups.
Colour, Structure, and Border Asymmetry
Melanocytic lesions were evaluated regarding at least 1 axis asymmetry of colour distribution, pigment network, dots, globules, streaks, homogeneous structureless areas, and borders. There was no significant difference in the rates of at least 1 axis asymmetry of colour distribution, pigment network, dots, and the border between the 2 groups. The rates of at least 1 axis asymmetry in globules (59.6% vs 32.3%, P = .002), streaks (34% vs 9.7%, P < .001) and homogeneous structureless areas (68.1% vs 36.6%, P < .001) were significantly higher in the excision-recommended group than in the excision-not-required group. The frequencies of white (23.4% vs 4.3%, P = .001), red (29.8% vs 16.1%, P = .006), blue/grey (59.6% vs 39.8%, P = .027), and black colours (51.1% vs 28%, P = .007) were significantly (P < .05) higher in the excision-recommended group than in the excision-not-required group.
Univariate and Multivariate Analysis
Table S2 provides the univariate analysis of dermoscopic features, related odds ratios, and the 95% confidence intervals.
In the multivariate analyses, the atypical pigment network (OR: 3.58, 95% CI: 1.31-9.72), at least 1 axis asymmetry of globules (OR: 3.17, 95% CI: 1.37-7.35), streaks (OR: 6.16, 95% CI: 1.95-19.48), and homogenous structureless areas (OR: 5.92, 95% CI: 2.36-14.92) were the significant independent predictive factors for excision-recommended lesions (Table S2).
Discussion
This study demonstrates that all 3 dermoscopic methods—pattern analysis, the ABCD rule, and the CASH algorithm—have similar diagnostic performances, which are generally poor for discriminating low-grade dysplastic nevi from thin melanomas, and intermediate melanocytic lesions. Pattern analysis had the highest sensitivity and lowest specificity. In literature, pattern analysis was reported to be the best (sensitivity, 83.5%; specificity, 83.4%) among the ABCD rule, Menzies and 7-point checklist in distinguishing melanoma from benign pigmented melanocytic lesions. 11 In addition, pattern analysis was superior to ABCD and the 7-point checklist method in differentiating thin melanomas from atypical melanocytic nevi regarding sensitivity (85.4%), specificity (79.4%), and PPV (79.6%). 7 However, similar to our findings, pattern analysis failed to discriminate early melanomas and melanocytic nevi in a study by Skvara et al. 15 The diversity of results in the literature and our study may be attributed to the variety of melanocytic lesions with different diagnostic challenges in the control groups.
In our study, using the conventional cut-off point of the ABCD score (total dermoscopic score >4.75), the specificity was higher (68.1%) than the CASH algorithm. In line with our findings, the ABCD score demonstrated a specificity of 59.4%, outperforming the CASH algorithm in distinguishing melanoma from melanocytic nevi in Carrera et al’s study. 9
The CASH algorithm, differing from other algorithms by evaluating lesion architecture, showed higher sensitivity than the ABCD score in our study. 8 The CASH algorithm was also superior to pattern analysis in distinguishing low-grade dysplastic nevi (specificity). A comparative study of dermoscopic algorithms revealed that both the ABCD rule and the CASH algorithm demonstrated higher sensitivities (91.6% each) in distinguishing between melanoma and benign melanocytic nevus than those in our study. 10 This variance may be attributed to the absence of common melanocytic nevi in the control group and the composition of the malignant group in our study, which included thin melanomas, MELTUMPs, and high-grade dysplastic nevi.
Our study identified several significant dermoscopic features that independently predicted excision-recommended lesions. These included asymmetry of globules, streaks, and homogeneous structureless areas. Asymmetry is a dermoscopic criteria evaluated in algorithms such as the ABCD rule, the Menzies method, “Chaos and Clues” and the CASH algorithm.16,17 When we consider the asymmetry weight factor is 1.3 in the ABCD rule, it appears that asymmetry contributes significantly to the diagnosis of melanoma. The colour asymmetry was found to be statistically higher in melanomas and dermoscopic atypical melanocytic nevi than in the benign melanocytic lesions by Seidenari et al. 18 Although colour asymmetry was more common in the excision-recommended group than in the low-grade dysplastic nevi, the difference was not statistically significant in our study. It is probably because low-grade dysplastic nevi often exhibit colour asymmetry, making them hard to differentiate from thin melanomas and intermediate nevi. The colour asymmetry could serve as a valuable dermoscopic characteristic for differentiating melanomas from melanocytic nevi with no dysplasia.
In a previous study evaluating 200 melanomas, of which 67% had <1 mm Breslow thickness or were in situ, homogeneous structureless areas were determined as the most common dermoscopic finding. 19 In another study, light brown structureless areas were determined to be the most important and reliable dermoscopic finding in distinguishing thin melanomas from atypical melanocytic nevi. 7 Similarly, the asymmetric distribution of homogeneous structureless areas was associated with excision-recommended lesions in our study.
Shiny white streaks were only found in early melanomas and intermediate melanocytic lesions, not in low-grade dysplastic nevi. Our study emphasizes the significance of shiny white streaks in a recent study, indicating that their presence increases the likelihood of diagnosing melanoma when assessing such intermediate lesions. Their study included high-grade dysplastic nevus, MELTUMP, superficial atypical melanocytic proliferations of unknown significance (SAMPUS), and atypical Spitz tumour. 20 Our results also support previous studies showing that the rates of atypical pigment network,21-23 shiny white streaks, 21 and blue-white veil21-23 were significantly higher in early melanomas than in atypical melanocytic nevi.
Depigmentation is characterized by a loss of pigment network, which is lighter brown and differs from skar-like depigmentation. In a study by Dika et al, 24 it was the only feature associated with thin melanomas on lower extremities that were difficult to distinguish from common melanocytic nevi. Our study also found a significantly higher frequency of this feature in the excision-recommended group than in low-grade dysplastic nevi.
There are several limitations in this study. First, the sample size of MELTUMPs and high-grade dysplastic nevi were small. Second, our control group only included low-grade dysplastic nevi; no common melanocytic nevi or other pigmented skin lesions existed. This might lead to the underestimation of the dermoscopic algorithms and indicators. Third, only one experienced dermatopathologist evaluated the lesions. These lesions are challenging to diagnose through histopathological examination, and there is often a low level of agreement among experts. In addition, an immunohistochemical panel including anti-BRAF-mutated protein VE1 antibody, immunostain for BAP1, and anti-PRAME immunostain that is recommended2,5 to make a further differential diagnosis in intermediate lesions could not be performed because they were not available in our university laboratory.
The dermoscopic features of melanomas compared with those of benign or atypical melanocytic nevi are well documented. Our study adds to the existing literature by demonstrating the accuracy of dermoscopic methods and highlighting specific findings that distinguish more complex melanocytic lesions, including early melanomas, intermediate melanocytic lesions, and low-grade dysplastic nevi. Shiny white structures, scar-like depigmentation, blue-white veil, atypical blotch, and negative pigment network stand out in distinguishing early melanomas and intermediate melanocytic lesions from low-grade dysplastic nevi. Atypical network, asymmetry of globules, streaks, and homogeneous structureless areas were the independent dermoscopic indicators. Intermediate lesions were orphaned previously, and they were usually excluded from the study samples, but in real life, these lesions need to be diagnosed and excised. Therefore, the dermoscopic findings in our study can support the dermoscopic diagnosis in these challenging lesions.
In conclusion, the diagnostic performance of dermoscopic methods in distinguishing low-grade dysplastic nevi, thin melanomas, and intermediate melanocytic lesions is limited. In decreasing order, the highest positive predictive values were shiny white structures, scar-like depigmentation, blue-white veil, atypical blotch, and negative pigment network. Atypical network, asymmetry of globules, streaks, and homogeneous structureless areas were the independent dermoscopic indicators in discriminating these lesions. These dermoscopic findings may help distinguish early melanomas and intermediate lesions from the challenging low-grade dysplastic nevi in real life.
Supplemental Material
sj-docx-1-cms-10.1177_12034754251325508 – Supplemental material for Diagnostic Performance of Dermoscopy for Distinguishing Early Melanomas and Intermediate Melanocytic Lesions From Low-Grade Dysplastic Nevi
Supplemental material, sj-docx-1-cms-10.1177_12034754251325508 for Diagnostic Performance of Dermoscopy for Distinguishing Early Melanomas and Intermediate Melanocytic Lesions From Low-Grade Dysplastic Nevi by Ceylan Avcı, Meltem Kaşıkçı, Banu Lebe, Sevgi Akarsu and Turna İlknur in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-2-cms-10.1177_12034754251325508 – Supplemental material for Diagnostic Performance of Dermoscopy for Distinguishing Early Melanomas and Intermediate Melanocytic Lesions From Low-Grade Dysplastic Nevi
Supplemental material, sj-docx-2-cms-10.1177_12034754251325508 for Diagnostic Performance of Dermoscopy for Distinguishing Early Melanomas and Intermediate Melanocytic Lesions From Low-Grade Dysplastic Nevi by Ceylan Avcı, Meltem Kaşıkçı, Banu Lebe, Sevgi Akarsu and Turna İlknur in Journal of Cutaneous Medicine and Surgery
Footnotes
Author Contributions
C.A. and M.K. collected the data. B.L. evaluated pathology slides C.A. performed the statistical analysis and wrote the first draft of the manuscript. All authors in the study contributed all the details for the manuscript including the study concept and design, interpretation of data; critical review of important intellectual content, effective participation in the research guidance, critical review of the literature, and final approval of the final version of the manuscript.
Data Availability Statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Clinical data were assessed after the approval of the local, noninterventional studies’ ethics committee (date: July 28, 2021; number: 2021/22-14).
Consent to Participate
Non-applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
