Abstract
Pityriasis rubra pilaris (PRP) is a rare, inflammatory papulosquamous skin disease with unknown exact etiology. Historically, PRP has been challenging to diagnose, especially during the acute phase, and to treat, due to its unclear pathogenesis. To better inform clinical practice, a literature review was conducted employing a broad search strategy to capture PRP-related published studies between January 1, 2012 to October 31, 2022. Two hundred twenty-one studies were identified, which were categorized into 9 themes: (1) potential causes and triggering factors, (2) comorbidities, (3) diagnostic difficulties, (4) genetics, (5) clinical manifestations and laboratory values, (6) treatment, (7) treatment-related adverse events, (8) quality of life, and (9) other. COVID-19 infection, COVID-19 vaccination, and malignancy were the most commonly reported potential triggering factors. Misdiagnosis is very common during the early acute stages. Pathogenesis and genetic studies have further implicated caspase recruitment domain family member 14 (CARD14) mutations in the development of familial PRP (Type V) and have underlined the overlap between psoriasis and PRP. To date, there are currently no specific and validated scoring systems or tools to assess the severity of PRP. While large, randomized trials are still lacking, biologic agents remain the most effective therapy.
Introduction
Pityriasis rubra pilaris (PRP) is a rare, inflammatory papulosquamous disorder with an estimated incidence of 1 per 5000 dermatologic patients. 1 Key clinical features of PRP include follicular keratotic papules, orange-red erythematous plaques, and waxy-looking palmoplantar keratoderma with red-orange hue. Histopathology findings include alternating orthokeratosis and parakeratosis, hypergranulosis, suprapapillary plates, broad rete ridges, and superficial perivascular infiltrate. 2 In adult populations, PRP commonly begins on the head and neck and spreads cephalocaudally, while in pediatric populations the disease may begin on the trunk or lower half of the body. PRP has been traditionally classified into 6 subtypes, differentiated by age of onset, morphologic and clinical presentation, course of disease, and associated medical conditions. 3 Classical adult (Type I) PRP is the most common type, with a spontaneous remission rate of 80% in 3 to 5 years. 4 Atypical adult (Type II) has a more chronic course with additional features of ichthyosiform scales and areas of eczematous patches and plaques as well as alopecia. Classical juvenile (Type III) PRP is similar in presentation to Type I, but has a pediatric age of onset. Circumscribed juvenile (Type IV) PRP is the most common type in the pediatric population, and is characterized by well-demarcated hyperkeratotic erythematous follicular papules mostly on the elbows and knees. Atypical juvenile (Type V) PRP, known as familial PRP, has been potentially linked to Caspase Recruitment Domain Family Member 14 (CARD14) gene mutations. 5 HIV-associated (Type VI) PRP is associated with nodular acneiform lesions, lichen spinulosus-like lesions, and erythroderma. This type is often resistant to therapy and has poor prognosis. 6
PRP has been a challenging disease to diagnose and treat, given its rarity and unclear specific pathogenesis and etiology. In the past decade, there has been an increase in research, highlighting potential etiologies, diagnostic data, and treatment options. For instance, there have been increasing reports of PRP-like eruptions potentially related to infections and vaccines, particularly Coronavirus Disease 2019 (COVID-19) vaccines. 7 Given the breadth of new data, we aimed to synthesize and categorize PRP studies published in the last 10 years, in order to highlight novel findings and to inform future clinical practice and research.
Materials and Methods
Literature Search
A scoping review of literature was conducted using CINAHL (via EBSCOhost), Cochrane Library, Embase (via OVID), MEDline (via OVID), PubMed, and Web of Science databases with the date limits of January 1, 2012 to October 31, 2022. A broad search strategy was employed: (pityriasis rubra pilaris) OR (Devergie’s disease) OR (lichen ruber acuminatus) OR (lichen acuminatus) OR (pityriasis rubra pilaire) OR (lichen ruber pilaris). Any relevant Subject Headings were also added. Additional studies were identified through references of included studies if applicable. This scoping review was carried out in accordance with the PRISMA Extension for Scoping Reviews (PRISMA-Scr) checklist and used the methodological framework outlined by Arksey and O’Malley. 8 The protocol can be found on Open Science Framework (https://osf.io/kh6bu/?view_only=4f7f630c297c4bc7962bdc9c28469240). Our completed checklist is available as Supplementary Figure S6.
Study Identification and Selection
Two thousand sixty-six studies were returned and were de-duplicated manually. Title, abstract, and full-text screening were carried out by 2 reviewers (TZ and LK) using Covidence. Covidence is a literature screening and data extraction tool. A third reviewer MA performed spot-checking and acted as a third opinion if screening discrepancies could not be resolved.
Inclusion and Exclusion Criteria
All study designs were included except literature reviews. Studies were excluded if: (a) study was a literature review, (b) study was not published in English, (c) only an abstract or conference proceeding could be found, and (d) focus or content of the article did not include PRP.
Data Extraction
Studies were categorized based on theme. Nine broad themes were identified: (1) potential causes and triggering factors, (2) comorbidities, (3) diagnostic difficulties, (4) genetics, (5) clinical manifestations and laboratory findings, (6) treatment, (7) treatment-related adverse events, (8) quality of life (QoL), and (9) other. Some studies fell under multiple themes. This was performed individually by 2 separate reviewers, and a consensus was reached. Characteristics extracted from all studies is presented in Supplementary Table S1. Data extraction for items were performed using an Excel sheet and was done separately by 2 reviewers. Any discrepancies were discussed until conflicts were resolved.
Results
Two hundred twenty-one studies were included in this review. A breakdown of study identification and screening is shown in Supplementary Figure S1.
Potential Causes and Triggering Factors
Fifty-three studies reported on potential causes and triggering factors that resulted in PRP-like eruptions or subsequent PRP diagnosis. The most commonly cited factors were COVID-19 vaccination (n = 12), malignancy causing paraneoplastic PRP eruption (n = 10), and COVID-19 infection (n = 5). Data is presented in Supplementary Table S2.
Comorbidities
Six case reports were found to have focused on outlining novel comorbidities with PRP presentation. Three were found before onset of PRP diagnosis, including: bullous pemphigoid, primary biliary cholangitis, and membranous nephropathy. Two were found simultaneously with PRP including: mesangial proliferative glomerulonephritis and rickets. One report yielded polyarthritis after the onset diagnosis of PRP.
Three cross-sectional survey studies collected data on comorbidities with PRP. Halper et al 9 found that the most common comorbidities included: allergic rhinitis (17.9%), depression (16.9%), unspecified cardiovascular disease (13.8%), non-melanoma skin cancer (9.7%), type 2 diabetes mellitus (8.8%), malignancy other than skin cancer (8.5%), hypothyroidism (8.2%), and irritable bowel syndrome (7.2%). Ross et al 10 found that the most common comorbidities were: hypothyroidism (20%), dyslipidemia (18%), skin disorder other than PRP (14%), malignancy (10%), and type 2 diabetes mellitus (8%). Ji-Xu et al 11 reported that 8.2% of PRP patients had concurrent malignancy, excluding non-melanoma skin cancer.
Genetics
Sixteen studies investigated CARD14 mutations and their relationship to development of sporadic or familial (Type V) PRP. Nine of 16 (56%) studies provided evidence that CARD14 mutations may be implicated in the development of type V PRP. Three (19%) studies provided evidence that CARD14 mutations were not pathogenic in individuals with non-familial PRP. One (6%) study examined CARD14 mutations in the context of CARD14-associated papulosquamous eruptions (CAPE), concluding that CARD14 mutations play a role in the development of PRP phenotype eruptions. One (6%) study linked CARD14 mutations with non-specific inflammation pathways, suggesting a possible role in PRP pathogenicity. One (6%) study provided evidence that CARD14 mutations play a role in PRP development, but did not specify PRP subtype. One (6%) study hypothesized the link between CARD14 mutations and PRP pathogenesis.
Pathogenesis
Four studies focused on outlining the pathogenic pathways of PRP, such as signalling cascades related to immunologic function.
Kamata et al 12 used immunohistochemical staining of tissue biopsies, finding that bleomycine hydrolase, caspase-14, and calpain I staining were markedly decreased in PRP affected areas compared to normal skin.
Strunck et al was a case-control that compared protein levels in PRP patients with age- and sex-matched controls. The study found that two IL-23/TH17 pathway proteins, IL-17C and CCL20, were upregulated in tissue samples from PRP patients compared to healthy controls. Furthermore, levels seemed to correlate with PRP disease severity and responsiveness to IL-17A blockade treatment. 13
Al-Hage et al 14 employed immunohistochemical analysis of tissue from PRP patients to demonstrate an increased presence and activity of plasmacytoid dendritic cells, highlighting their potential role in PRP.
Shao et al 15 used global transcriptomic profiling to show that differentially expressed genes in PRP overlap with psoriasis, especially the upregulation of phospholipase A2 primarily expressed in the epidermis.
Diagnostic Difficulties
Eight studies were case reports where individuals with PRP were initially misdiagnosed with a different skin condition or initial diagnosis was inconclusive. Five based their diagnosis on clinical presentation and histopathology: psoriasis (n = 1), transient acantholytic dermatosis (n = 1), erythema annulare centrifugum (n = 1), pemphigus foliaceus (n = 1), and unspecific annular erythematous plaques (n = 1). Three based their diagnosis on clinical presentation only: psoriasis (n = 2) and follicular eczema (n = 1). Of these 8 studies, diagnosis was modified due to subsequent repeat biopsies (n = 4), or reassessment and/or second opinion of the initial biopsies (n = 4).
One case report outlined a PRP patient where biopsy showed overlapping histologic features between PRP and pemphigus vulgaris, obfuscating diagnosis. 16 The authors considered the possibility of pemphigus vulgaris, but ultimately diagnosed the patient with PRP due to absence of bullae, absence of other clinical features of immunobullous disorders, and PRP histopathologic findings.
One case series highlighted the biopsy findings of 3 PRP patients, commenting that histopathologic features of lichen nitidus may be rarely found in biopsies of PRP lesions. 17
Four studies collected data on the percentage of misdiagnosed PRP patients, and what the initial diagnosis was. Initial misdiagnosis rates ranged from 60.70% to 91.30%. Data is presented in Supplementary Table S3.
Clinical Manifestations and Laboratory Findings
Twelve case reports described novel clinical manifestations or dermoscopic findings seen in PRP. By topic, the studies reported on: dermoscopic and/or histopathologic findings (n = 3), unusual distribution of lesions (n = 3), erythema gyratum repens-like morphology (n = 2), Kaposi’s varicelliform eruption (n = 1), lichen planopilaris-like scarring pattern on scalp (n = 1), trachyonychia nail changes (n = 1), and bullous morphology (n = 1). The 3 studies describing an unusual distribution of PRP lesions were: cyclical lesions on inguinal and scrotal regions (n = 1), persistent lesions on the antecubital and popliteal areas (n = 1), and widespread eruption after a chronic localized disease suggestive of mixed type III/IV disease (n = 1).18-20
Two case reports outlined typical clinical and dermoscopic findings in PRP patients.
Two case series reported on the most common clinical findings. One reported that scaling of the skin on the body and scalp, as well as pruritus, were the most common symptoms. The other study showed that skin scaliness and palmar-plantar hyperkeratosis or thickening were the most common features.
Two studies highlighted common histopathological findings. One retrospective study included 32 PRP cases, finding that variable acanthosis (100%) and follicular plugging (100%) in the epidermis, vascularity in the dermis (91%), perivascular pattern of inflammation (100%), and lymphocyte infiltration (100%) were the most common histopathological features. The second retrospective study investigated histopathology findings in PRP biopsies with acantholysis. 21 The study found that only 5/33 biopsies showed acantholysis.
One retrospective study reported on dermoscopic findings. The most common features were presence of a central hair, follicular plugs, and perifollicular yellow/orange halos.
One retrospective study reported on the most common clinical and histopathological findings. The most common clinical features were pruritus and hair loss. The most common histopathological findings were alternating orthokeratosis and parakeratosis in both vertical and horizontal directions, as well as superficial perivascular lymphohistiocytic infiltrate in the dermis.
One prospective study investigated the use of trichoscopy to diagnose erythrodermic conditions. Of the 49 individuals included, 3 had PRP. PRP lesions were characterized by dotted vessels, white and yellow scale, and peripilar casts.
Two case reports highlighted associated laboratory findings with PRP. The 2 separate findings were: blood eosinophilia and subclinical hypothyroidism. In the eosinophilia case report, the authors performed a laboratory workup of the patient, finding abnormally high absolute eosinophil count and a subsequent negative eosinophilia workup. 22 The patient underwent treatment for PRP, and the eosinophil count returned to normal value after therapy, suggesting that eosinophilia may be related to PRP. Lastly, one retrospective study identified 28/142 individuals with PRP had concurrent blood or tissue eosinophilia, but additional investigations were not discussed. 23
Treatment
A total of 94 studies reported on topical, systemic, or biologic treatments for PRP. Seventy-two studies were case reports, 18 were case series/retrospective analyses, 1 was a prospective cohort study, 2 were single-arm clinical trials, and 1 was a patient survey.
A total of 192 individuals with PRP were treated, composing of type I (n = 93), type II (n = 11), type III (n = 12), type IV (n = 15), type V (n = 5), type VI (n = 1), and unspecified PRP type (n = 55).
Five methods of collecting treatment outcomes were identified. Four were non-PRP specific measurement tools: Body Surface Area (BSA), Psoriasis Area and Severity Index (PASI), Physician Global Assessment (PGA), and Dermatology Life Quality Index (DLQI).24-26 The last method of collecting outcome data was the physician’s judgment, without the use of a specified measurement tool. The most commonly used outcome measure was physician judgment (n = 68), BSA (n = 18), DLQI (n = 11), PASI (n = 8), and PGA (n = 6). In total, 27/107 studies utilized outcome measurement tools, with 18 of these studies employing 2 or more outcome measures.
Due to the refractory nature of PRP, individuals were classified based on the most recent primary treatment received. Data for the magnitude of responsiveness to treatment, as well as mean and range for time to response was collected. Magnitude of response was broken down into 4 categories: significant response (SR), partial response (PR), minimal or no response (NR), and not specified (NS).
If the study did not specify an outcome measurement tool, it was considered to be assessed by physician judgment. SR was defined as: a description stating that the patient had “almost complete clearance,” “complete clearance,” “significant response,” or a “marked response” after treatment. PR was defined as: a “moderate response” or “partial response” to treatment. NR was defined as: “no response” or “minimal response” or “poor response” to treatment. Treatment outcomes assessed by physician judgment are presented in Supplementary Tables S4 and S5.
When an outcome measurement tool was employed, improvement from baseline was assessed based on score. If the study specified the use of an outcome measurement tool, then SR was defined as: a ≥75% improvement of the patient’s PRP symptoms from baseline. PR was defined as: a ≥25% to ≤75% improvement. NR was defined as: a <25% improvement. Treatment outcomes assessed by validated tools are presented in Table 1.
Treatment Outcomes Assessed with Outcome Measurement Tools (Systemic and Biologic Therapies).
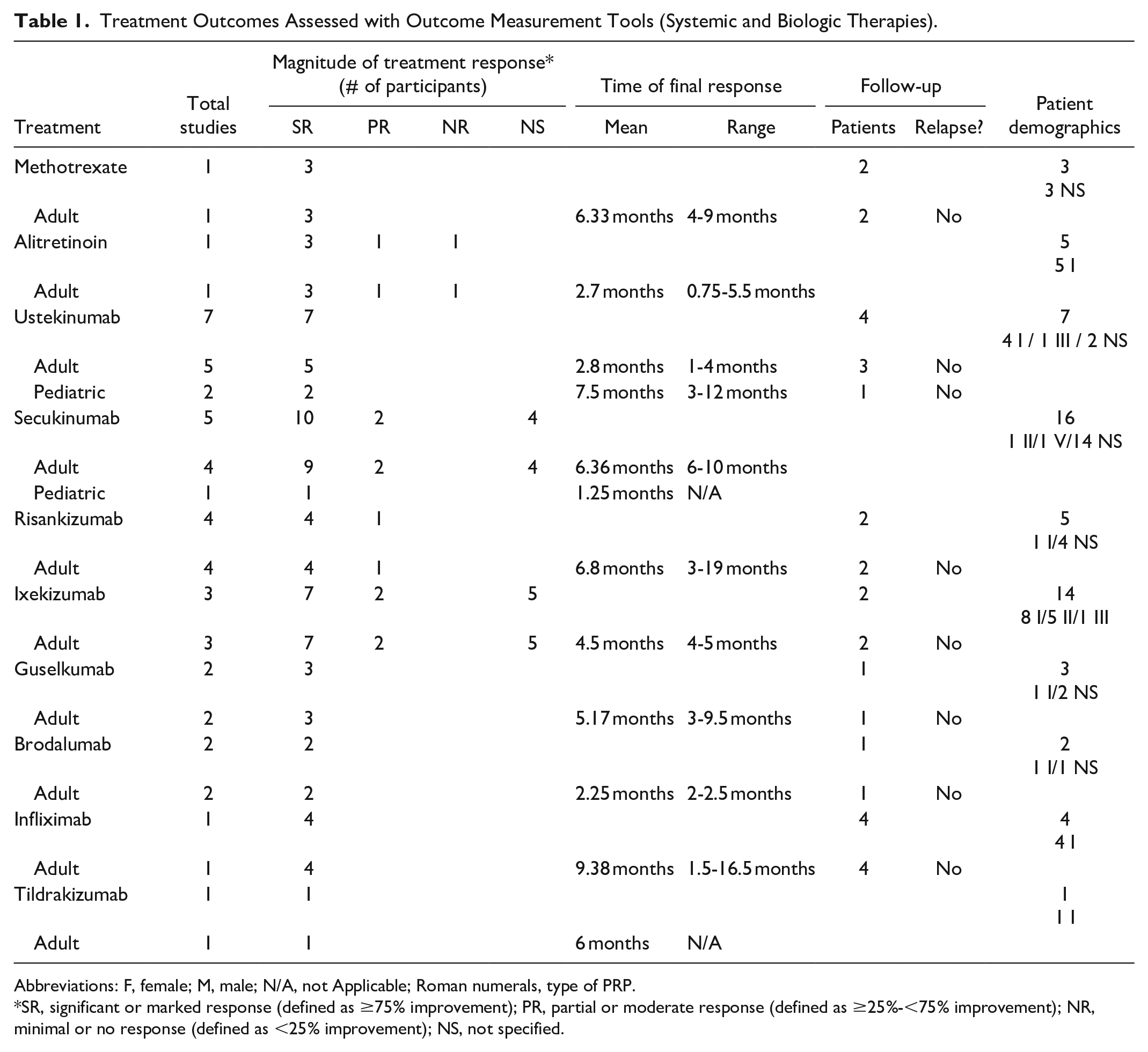
Abbreviations: F, female; M, male; N/A, not Applicable; Roman numerals, type of PRP.
SR, significant or marked response (defined as ≥75% improvement); PR, partial or moderate response (defined as ≥25%-<75% improvement); NR, minimal or no response (defined as <25% improvement); NS, not specified.
Treatment outcome, assessed either with a tool or without, will be discussed together. A total of 7 studies investigated topical treatments. The studied treatments were: calciptriol (n = 1), mometasone furoate (n = 1), tretinoin (n = 1), hydrocortisone (n = 1), unspecified topical corticosteroids (n = 1), combination corticosteroid and calcipotriene (n = 1), and multiple corticosteroids (n = 1).
The most reported oral systemic agents were acitretin (n = 12), followed by methotrexate (n = 6) and apremilast (n = 6). For individuals treated with acitretin (n = 32 participants), 59.4% achieved SR, 25% achieved PR, 3.1% had NR, and in 12.5% treatment response was unspecified. For methotrexate (n = 10 participants), 80% achieved SR, and in 20% treatment response was unspecified. For apremilast (n = 6 participants), 83.3% achieved SR, and was unspecified in 16.7% of cases.
The most studied biologics were ustekinumab (n = 19), secukinumab (n = 10), and adalimumab (n = 7). For individuals receiving ustekinumab (n = 24 participants), 83.3% achieved SR, 8.3% had PR, while 9.1% had NR to treatment. For secukinumab (n = 21 participants), 71.4% achieved SR, 9.5% had PR, and in 19.0% treatment response was unspecified. For adalimumab (n = 8 participants), 75% achieved SR, 12.5% had PR, and 12.5% had NR to treatment. All biologic agents were used due to refractory PRP, or when first-line systemic therapy was contraindicated.
A total of 73 individuals treated with biologic agents were reported in case studies (n = 42), case series (n = 13), or single-arm clinical trials (n = 2). Approximately 33% (n = 24/73) of participants received biologics in combination therapy with other oral systemic agents, 36% (n = 26/73) were on monotherapy, and 32% (n = 23/73) were unspecified. Of the 24 subjects who received combination therapy, 14 received biologics with acitretin, 5 with methotrexate, 3 with cyclosporine, and 2 with both acitretin and methotrexate.
Twenty-six individuals were bio-experienced before demonstrating an effective clinical response to a different biologic agent. Sixteen had failed 1 biologic agent, 6 had failed 2 biologics, and 4 had failed 3 biologics.
Treatment-Related Adverse Events
Four case reports and 1 case series reported on adverse events related to biologic or systemic agents. Biologic agents involved were: ustekinumab, ixekizumab, secukinumab, and adalimumab. The adverse events associated with ustekinumab were: meningitis associated with meningococcal/herpes simplex virus 2 (HSV-2) infection in one patient, and acute heart failure in another patient. An erythema gyratum repens-like eruption adverse event occurred with ixekizumab (n = 1) and secukinumab (n =1 ). The adalimumab-related adverse event was an erythroderma exacerbation.
The one systemic agent reported was acitretin (n = 1), and was associated with drug-induced liver injury.
Quality of Life
Two studies used Skindex-29, 36-Item Short Form (SF-36) survey, and DLQI to investigate factors relating to QoL in PRP patients. 27 Skindex-29 is a 3-dimensional questionnaire on dermatology-specific QoL. 28
Eastham et al 29 found that PRP was associated with worse Skindex-29 scores in functioning, symptoms, and emotions categories compared to patients with other dermatological conditions like: vulvodynia, atopic dermatitis, pemphigus vulgaris, psoriasis, acne vulgaris, cutaneous T-cell lymphoma, rosacea, alopecia, vitiligo, actinic keratosis, and non-melanoma skin cancer. 29 Furthermore, Skindex-29 and DLQI scores were significantly correlated.
Ji-Xu et al reported that DLQI was significantly reduced in patients with PRP. Furthermore, the study authors reported that respondents experienced PRP-related depression (87.6%), anxiety (21.4%), and suicidal ideation (7.3%) over the previous 2 weeks.
Other
One retrospective chart review described PRP drug survival rate and adverse effects. 30 Drug survival was characterized by the study to reflect a drug’s performance in routine medical practice. The study found that median survival at 600 days was 78% for methotrexate, 40% for biologics, and 32% for retinoids. Failure due to adverse effects was most common with retinoid use (54%).
Two case reports described interesting anecdotal findings with positive Koebner phenomenon of a previous PRP eruption due to a hot water burn (n = 1) and an unclear origin (n = 1).
One case report supports classifying facial discoid dermatosis (FDD) as a localized type of PRP limited to the face. 31 The rationale was the shared features between FDD and PRP, such as the salmon-colored papules and plaques, the prolonged course of disease, and the similar histopathologic findings, which suggest both are closely related.
One case report showed that treatment of HIV with antiretroviral therapy for 3 months attenuated PRP symptoms and resolved lesions in a patient with type VI PRP. 32
One case report showed that optical coherence tomography and laser reflectance confocal microscopy can be used to monitor in vivo response to therapy. 33
Discussion
Etiology and Triggering Factors
The pathophysiology of PRP is still not fully understood, lending to the uncertainty of what causes or triggers the disease. The most commonly reported potential triggering factors identified in this review were COVID-19 vaccination, malignancy, and COVID-19 infection.
COVID-19 and vaccinations are potential causes and should be considered by clinicians when encountering PRP or PRP-like eruptions.
Another potential cause could be an underlying malignancy, suggesting that PRP might be a paraneoplastic eruption. Various dermatologic eruptions or conditions have been linked to possible underlying malignancy. 34 In this review, malignancy was the second most reported cause for PRP. Moreover, an additional 3 case series identified by this review found that concurrent malignancy was reported in 8.2% to 10% of individuals with PRP. To our knowledge, there are no recent studies detailing the types of malignancies associated with PRP eruption. This review identified 9 types of cancer: chronic lymphocytic leukemia, cholangiocarcinoma, prostate cancer, myelodysplastic syndrome, lung adenocarcinoma, undifferentiated metastatic squamous cell carcinoma, colon cancer, papillary urothelial carcinoma, and cerebral meningioma. Our review suggests a possible association between malignancy and PRP, and clinicians should be aware that PRP or PRP-like eruptions may be linked to an unidentified cancer.
Diagnostic Considerations
Despite the well-described and distinct clinical and histopathological features of PRP, diagnosing PRP can often still be a dilemma. From a clinical features perspective, this is especially true in the acute phases of PRP presentation, which may overlap with other papulosquamous, eczematous, immunobullous, connective tissue disease, viral, and drug-induced eruptions. 4
At the histopathological level, PRP has overlapping features with other conditions like psoriasis. However, PRP is typically distinguished from other conditions by the presence of hypergranulosis either focally or confluently, alternating orthokeratosis and parakeratosis both vertically and horizontally, thickened suprapapillary plates, shoulder parakeratosis, dilation of hair follicles, and presence of a keratotic plug. 35 Additional features, such as a lack of polymorphonuclear leukocytes and Munro microabscesses, may serve as a clue toward alternative diagnoses like psoriasis.
Our review identified interesting histopathologic reports in the literature. Although not a constant feature, focal acantholysis has been traditionally well-reported, and can be a helpful clue toward PRP diagnosis if interpreted within the appropriate context. 36 One case report highlighted the possibility of extensive acantholysis in PRP, involving both follicular and interfollicular epithelium with a suprabasal split and the formation of a tombstone pattern resembling pemphigus vulgaris. Another rare finding was the presence of lichenoid infiltrate, which had been exceptionally reported in the literature. 37 However, a recent case series described typical lichen nitidus findings in 3 PRP cases. Whether these lichen nitidus changes are just incidental findings or rare but distinctive features of PRP remains unclear. Although no explanations could be found to elucidate reports of acantholysis or lichenoid features, our findings underscore the importance of clinicopathological correlation when dealing with PRP.
Accurate initial diagnosis of PRP remains a challenge. Our review found that misdiagnosis rates ranged from 60.7% to 91.3% based on 4 studies. PRP was most commonly misdiagnosed initially as psoriasis. Other misdiagnoses include eczema, drug reaction, viral exanthems, contact dermatitis, pityriasis rosea, and other dermatoses. Several measures can be performed to confirm initial diagnosis and increase accuracy, such as dermoscopy, regular follow-ups in short intervals, and repeat biopsies. 38 A recently published study reported 2 cases where individuals initially diagnosed with atopic dermatitis were refractory to initial therapeutic efforts, resulting in repeat biopsy and correct diagnosis of PRP. 39 Given the difficulty of differentiating PRP from other dermatoses, especially during the early stages, clinicians should always consider different diagnoses and repeat skin biopsies for treatment-resistant papulosquamous eruptions.
Pathogenesis and Genetics
Four studies were identified to have investigated the pathogenesis pathway for PRP, finding that there is dysregulation of proinflammatory pathways (eg, IL-23/Th-17) and disruption of epidermal and skin barrier function. Moreover, there is evidence of significant overlap between psoriasis and PRP pathogenesis, supported by the success of psoriasis biologic agents when used for PRP treatment. 14 Further research should focus on identifying key molecules and cytokines involved in the pathogenesis of PRP. This could provide therapeutic targets for drug development and expand options for treatment.
The genetic studies identified provide evidence that CARD14 mutations were associated primarily with familial PRP (Type V), while there was a paucity of evidence to suggest that the same family of mutations are implicated in other types of PRP. CARD14 has been shown to play a role in activating nuclear factor kappa B, a proinflammatory mediator.40,41 This review bolsters existing evidence that CARD14 mutations are associated with familial PRP, similar to certain types of psoriasis.42,43 Individuals with a family history of PRP-like eruptions may benefit from genetic testing to inform current and future care.
Interestingly, one study reported on CARD14 mutations in patients exhibiting a spectrum of clinical features ranging from predominantly psoriasis to predominantly PRP. A previous study proposed the term “CAPE,” or CARD14-associated papulosquamous eruptions, that describe individuals with CARD14 mutations that exhibit features of psoriasis and familial PRP. 44 The overlap of CARD14 mutations and clinical manifestations of psoriasis and familial PRP suggest shared pathogenic pathways and could be a topic for future exploration.
Treatment Considerations
Systemic and biologic therapy options were effective in the majority of reported cases. In the cases where systemic therapies such as retinoids and classical immunosuppressants have failed to achieve a desired response, biologics have been shown to be an effective second-line intervention for refractory PRP. Previously conducted studies investigating systemic treatment options for PRP have come to similar conclusions.1,45 Biologic therapy shows promising preliminary data to be significantly effective in treating severe and refractory PRP. However, to date, there has yet to be a randomized controlled trial. Future research should aim to investigate the efficacy and feasibility of different therapies to treat PRP.
A formal treatment algorithm for PRP has yet to be established, potentially due to a variety of factors such as a lack of high-quality studies and the rarity of the disease. A previous study utilizing an informal literature search and clinical expertise proposed the use of systemic retinoids as first-line, followed by classical immunosuppressants, and then possible escalation to biologics. 46 Based on our literature review, systemic therapies seem to be effective in treating PRP on initial presentation. In the cases where PRP is refractory, transition to biologics has been shown to lead to an effective therapeutic response. Furthermore, 35.6% (n = 26/73) of patients included in our review transitioned through various biologics prior to a significant response to treatment, suggesting that patients may benefit from switching to a different targeted drug if the initial response to biologics is unsatisfactory. The main biologics identified by this review mainly targeted Th17-mediated and proinflammatory pathways, namely IL-17, IL-12/IL-23, and TNF-α. Many of these biologics are mainstay treatments for psoriasis. Despite emerging evidence of the applicability of biologics, feasible use of biologics for the treatment of PRP remains low. Many individuals will need to attempt multiple first-line or second-line treatments prior to escalation to biologics, and with the increasing cost of biologics, access to these medications will probably decrease. 47 This potential deficit in care, specifically for patients with refractory or severe PRP, may need to be a topic to consider in the future.
When reporting the magnitude of response to treatment, there was high heterogeneity in reporting methods. Only 27/107 studies specified the use of an outcome measurement tool, specifically BSA, DLQI, PASI, and PGA. Currently, there is no standardized or validated scoring system to measure PRP severity. Our review highlights that treatment response is often measured with tools not validated for PRP. Future efforts to report treatment outcomes for individuals with PRP should incorporate a validated, objective measurement tool to improve the ability to interpret and compare results, especially when a scoring system to assess PRP severity does not currently exist.
The most recently added category of type VI PRP is characterized by its association with HIV. In type VI PRP, antiretroviral therapy has been previously shown to attenuate cutaneous symptoms of PRP. 48 This review provides additional evidence from a recently reported case that antiretroviral therapy can aid in treating this type of PRP.
Quality of Life
The PRP patient experience has been given little attention to date. Only 2 studies focused on the QoL of the patient. A recently published study found that depression is common after diagnosis with PRP, along with a considerable impact on daily living. 5 Additional studies should be conducted to outline the range of impacts PRP has on an individual’s life, allowing clinicians and patients an improved understanding of the expected disease course and support needed.
Limitations
An in-depth analysis of treatment effect for interventional studies, as well as reasons for misdiagnosis in patients, were not fully explored by this review.
Conclusion
This review underlines the significant evolution of the clinical landscape of PRP in the past decade. Our review has highlighted clinical findings and considerations for future research:
PRP or PRP-like eruptions may be triggered by COVID-19, vaccination, or underlying malignancy.
PRP is often misdiagnosed, is difficult to distinguish especially during early presentation, and repeat biopsies should be considered for patients with unusual papulosquamous eruptions.
While biologic therapy remains the most effective modality for treating PRP, additional studies are needed to validate their safety and efficacy.
Further research is needed to outline the exact pathogenetic pathways of PRP, which appears to have an overlap with psoriasis.
Supplemental Material
sj-docx-1-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-1-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-2-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-2-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-3-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-3-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-4-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-4-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-5-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-5-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-6-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-6-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-docx-7-cms-10.1177_12034754231223159 – Supplemental material for Updates on Pityriasis Rubra Pilaris: A Scoping Review
Supplemental material, sj-docx-7-cms-10.1177_12034754231223159 for Updates on Pityriasis Rubra Pilaris: A Scoping Review by Ted Zhou, Abdullah Al Muqrin and Mohannad Abu-Hilal in Journal of Cutaneous Medicine and Surgery
Footnotes
Acknowledgements
We would like to express our sincere appreciation to Lauren Kim (LK) for her invaluable assistance in screening abstracts and full texts for the review process.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
Study was performed in compliance with ethical standards.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
