Abstract
Background:
Pruritus is a common complaint in patients with end-stage renal disease. Indoxyl sulfate (IS) is a tryptophan end metabolite extremely renal excreted. Activated charcoal can interfere with IS intestinal absorption.
Objectives:
To evaluate the serum level of IS and the effect of activated charcoal on uremic pruritus.
Materials and Methods:
In all, 135 participants were divided into 2 main groups. In total, 45 normal and healthy individuals as a control group and 90 patients on regular hemodialysis; 45 of these patients had uremic pruritus and the other 45 were not complaining of uremic pruritus. Serum IS was measured. Activated charcoal was used by patients with uremic pruritus. The severity of pruritus and Dermatology Life Quality Index (DLQI) were assessed.
Results:
The serum IS was significantly elevated in uremic patients than in control subjects (P < .001) and significantly elevated in uremic patients without pruritus (P < .001). Furthermore, there were positive significant correlations between the serum IS and both severity of pruritus (P < .001) and DLQI (P < .001). After activated charcoal usage, there was a significant decrease in IS level with the improvement of pruritus and quality of life of patients.
Conclusions:
IS may play a role in uremic pruritus. Activated charcoal could be considered a treatment for uremic pruritus.
Introduction
Uremic pruritus is a distressing symptom in more than 60% of patients with end-stage renal disease (ESRD).1,2 Several mechanisms have been postulated in this complaint, yet; the data are still inconclusive.3-5 Although regular hemodialysis may result in a partial improvement in pruritus, many patients continue to complain of pruritus.6,7
Tryptophan “an essential amino acid” is converted into indole by the act of intestinal microbes and further metabolized by the liver into indoxyl sulfate (IS).8,9 Mainly the secretion of renal tubular, and bound to serum albumin, deteriorated kidney or regular hemodialysis cannot excrete IS.10,11
Aim of the Study
We aimed to investigate the role of IS in uremic pruritus and the effect of decreasing the level of IS on the severity of pruritus.
Type of the Study
A quasi-interventional study.
Subjects and Methods
Ethical Approval
This study was performed after the approval of the Institutional Review Board (IRB; approval number: RC 22-10-2022) and registered at ClinicalTrials.gov (NCT05634083). All the participants were informed about the aim and nature of the study and were not subjected to any harmful procedure. Written informed consent was taken from each participant.
Subjects
This study recruited 90 participants from both sexes older than 18 years. These participants were divided into 2 main groups; Group I included 45 apparently healthy individuals as a control group, and Group II included 90 patients with ESRD under regular hemodialysis (3 sessions per week) for more than a year at a single hemodialysis unit. These patients were equally subdivided into 2 groups: group IIA included patients complaining of uremic pruritus not improved after the hemodialysis, and group IIB included patients who did not complain of uremic pruritus. All the 3 groups were age, sex, body mass index, and socioeconomic level matched. Patients with autoimmune disorders, blood diseases, nutritional deficiency, inflammatory bowel disorders, itchy dermatological disease, hepatic disease, suffering from malignancy, under chemotherapy or receiving chemotherapy in the past 6 months, or failed renal transplantation were excluded from this study.
Methods
A detailed medical history was taken from patients regarding the cause, onset, and duration of renal failure. A complete dermatological examination was done on all participants. The severity of pruritus was assessed using the visual analog scale 12 at the initiation of the study and after 2 months of the study before the hemodialysis session. This is a 10-point scale and measures pruritus degree, course, the surface area involved, and the associated sleep disturbance, and classified as no pruritus (score 0), mild pruritus (score 1-3), moderate (score 4-7), and severe (score >7). 13
Concurrently, the Dermatology Life Quality Index (DLQI) 14 by a self-report validated Arabic version of the questionnaire. 15 This score ranged from 0 to 30, and the impact on quality of life was divided into no impact (score 0-1), small impact (score 2-5), moderate impact (score 6-10), very large impact (score 11-20), and extremely impact (score 21-30). 16
Activated Charcoal
Uremic patients in group IIA were supplied with an activated charcoal tablet (Biocarbon®, Chem.-Pharm. Fabrik GmbH, Vienna, Austria). Patients were instructed to take 2 tablets after each meal (250 mg/tablet) for 2 months. No other anti-itching therapy was used during the study. There was a follow-up of patients at every hemodialysis session and reported side effects were recorded. None of the patients were lost during the period of the study.
Laboratory Investigations
An 8 ml morning venous blood sample was taken from the control group. A comparable venous blood sample was taken from uremic patients before the hemodialysis session and from patients in group IIA after 2 months of activated charcoal intake. The blood lifted to clot, the liquid serum was separated into 2 parts; the first part was used to measure hemoglobin, albumin, calcium, phosphorus, and parathyroid hormone levels, while the second part was stored at −45° and used for measurement of IS. Measurement of IS was done using a human monoclonal antibody IS ELISA Kit (Catalog No.: MBS3802650; MyBioSource, Inc., San Diego, CA, USA).
Statistical Analysis
The demographic, clinical, and laboratory data were collected and tabulated in an Excel file. The analysis of these data was performed using IBM SPSS Statistics® version 27 (IBM Corp., Armonk, NY, USA). The P ≤ .05 was selected as a point of significance. Comparison of means was used for numerical data of normally distributed variables by analysis of variance (ANOVA) test and Independent Simple test, while categorical variable differences were analyzed with χ2 (chi-square) test. The existence of correlations between values was tested using Spearman correlation analysis.
Results
The demographic, clinical, and laboratory results are briefed in Table 1. Blood hypertension was the most common cause of renal failure (23 patients in group IIA, 23 patients in group IIB) followed by diabetes mellitus (11 patients in group IIA, 10 patients in group IIB), chronic use of nonsteroidal anti-inflammatory drugs (7 patients in group IIA, 8 patients in group IIB), urinary tract infection (3 patients in group IIA, 2 patients in group IIB), and unknown cause in the remaining patients. There was no significant difference between patients in group IIA and patients in group IIB regarding the mean duration of starting hemodialysis (6.18 ± 3.21 years in group IIA vs 5.87 ± 2.67 years in group IIB, P = .619).
Clinical and Laboratory Data of All the Participants.
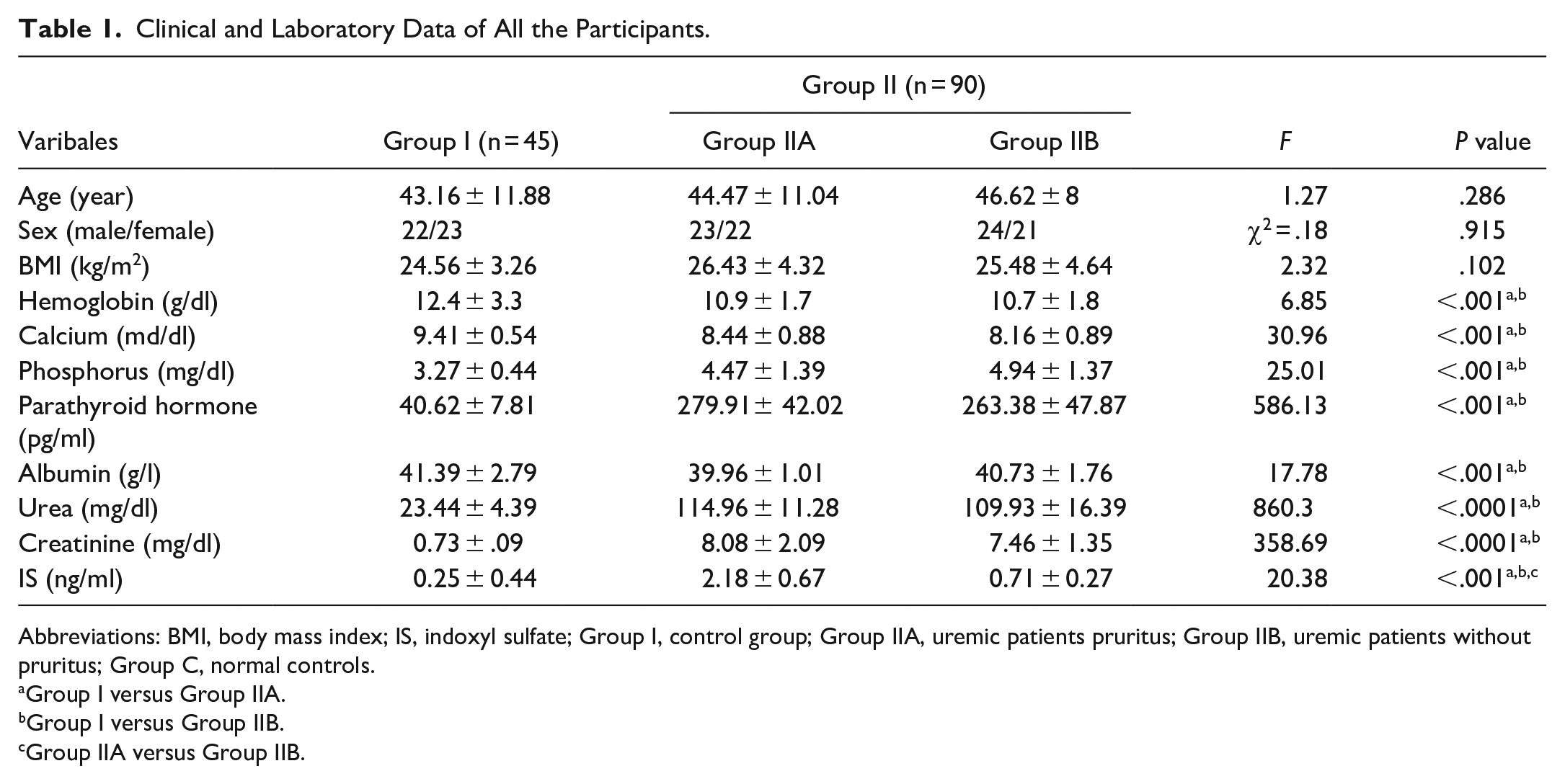
Abbreviations: BMI, body mass index; IS, indoxyl sulfate; Group I, control group; Group IIA, uremic patients pruritus; Group IIB, uremic patients without pruritus; Group C, normal controls.
Group I versus Group IIA.
Group I versus Group IIB.
Group IIA versus Group IIB.
The results of the study showed significant elevations of phosphors, parathyroid hormone, urea, creatinine, and IS, concurrently with significant decreases of hemoglobin and albumin in uremic patients than in control (Table 1). Furthermore, uremic patients with pruritus had a significantly increased serum level of IS (P < .001; Table 1). Using linear regression models for the patient with renal failure with or without pruritus for predication of pruritus, 8 different models are achieved. IS was the significant independent biomarker of the development of pruritus in uremic patients (Table 2).
Coefficients of the Linear Regression Model for Patients With Renal Failure With or Without Pruritus.
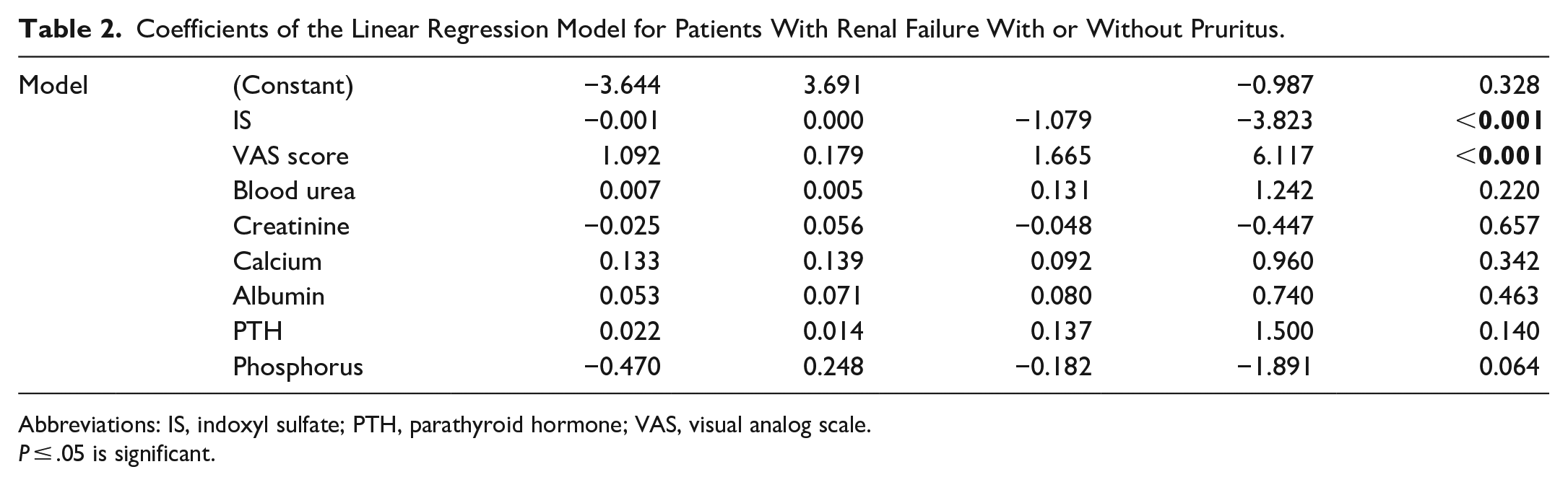
Abbreviations: IS, indoxyl sulfate; PTH, parathyroid hormone; VAS, visual analog scale.
P ≤ .05 is significant.
Regarding pruritus severity, 25 patients had severe pruritus, 15 patients had moderate pruritus, and 5 patients had mild pruritus. The effect of hemodialysis on pruritus was varied in group IIA, as 30 patients did not experience any difference before or after the hemodialysis sessions, 8 patients had a transient relief of pruritus after the hemodialysis session, and 7 patients had a temporary exacerbation after the hemodialysis session.
The result of this study showed that the effect of pruritus on quality of life was extremely impacted in 5 patients, a very large impact on 11 patients, a moderate impact on 16 patients, a small impact on 8 patients, and no or little impact on 5 patients.
The statistical analysis of the laboratory data showed a significant correlation between serum level of IS and pruritus severity (r = .92, P < .001; Table 3).
Correlation Between Laboratory Data and Pruritus Severity in Uremic Patients With Pruritus.
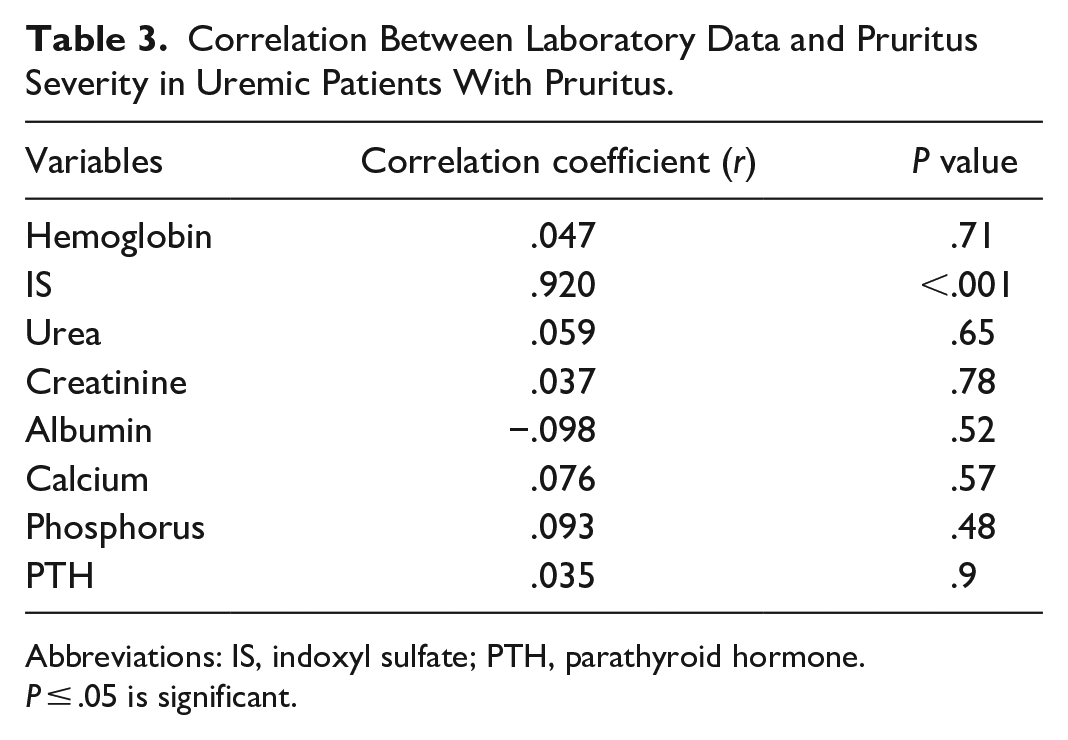
Abbreviations: IS, indoxyl sulfate; PTH, parathyroid hormone.
P ≤ .05 is significant.
In the results that showed after 2 months of activated charcoal usage, there was a significant decrease in the serum level of IS (P < .001). In addition, there was a significant decrease in pruritus severity (P < .001) and a significant decrease in DLQI (P = .032; Table 4).
Comparison Between Serum Level of IS, VAS Score, and DLQI in Patients in Group IIA Before and After 2 months of Activated Charcoal Usage.
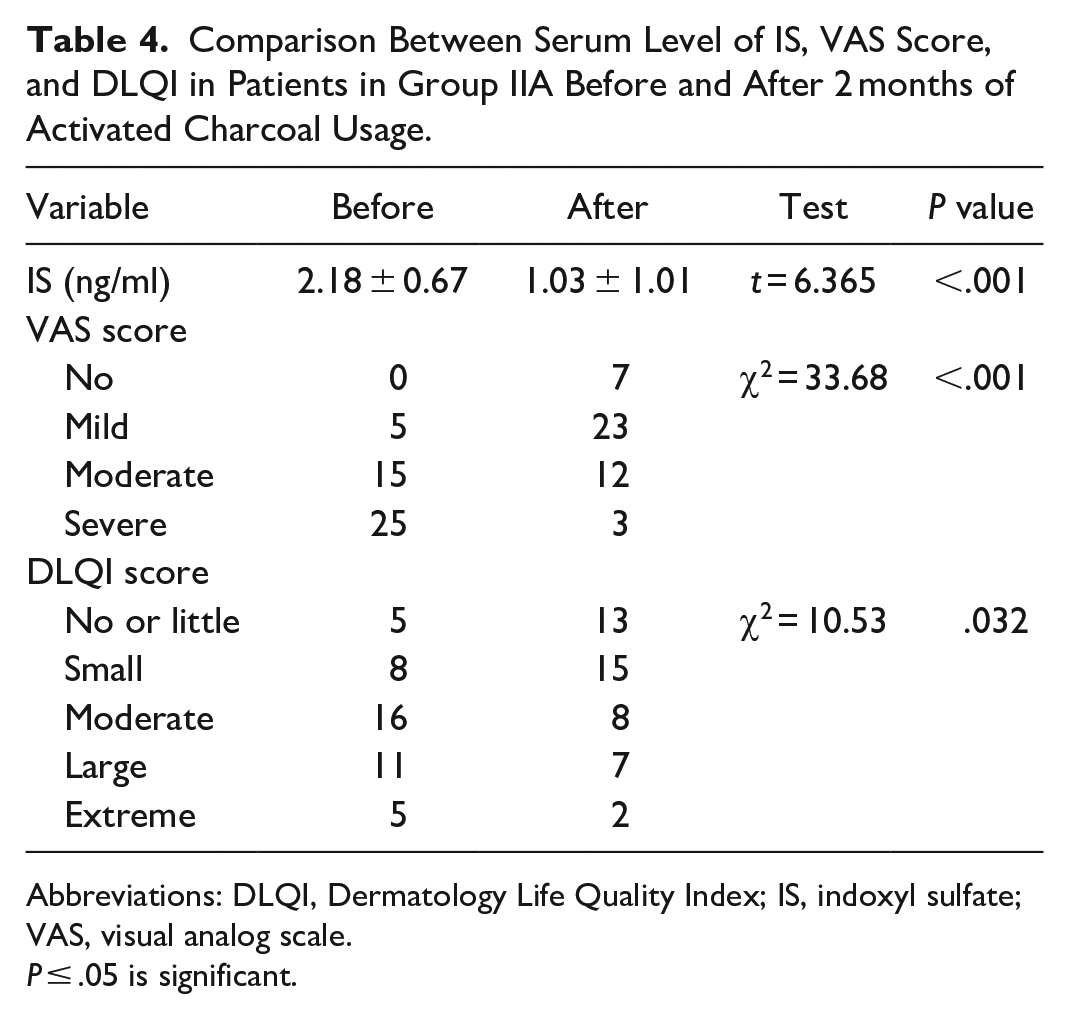
Abbreviations: DLQI, Dermatology Life Quality Index; IS, indoxyl sulfate; VAS, visual analog scale.
P ≤ .05 is significant.
During the treatment period, 3 patients in group IIA complained of transient mild diarrhea for 2 days after initiation of the activated charcoal that resolved spontaneously. No other serious side effects were noted during the period of treatment. None of the patients were dropped during the study.
Discussion
In the present study, results demonstrated the elevation of IS in uremic patients with a positive correlation with pruritus severity. Furthermore, results showed the negative impact of uremic pruritus on the quality of life of patients.
Pruritus associated with ESRD remains an unsolved problem. 17 Still, many physicians underestimate this distressing complaint.18,19 Several etiological factors had been investigated including xerosis, immunological factors, neurological changes, and endocrinal disturbance. 20
Many of the accumulative uremic toxins, like IS, formed from gut microbes and represent an inter-organismal crosstalk.21,22 Gut dysbiosis (increased number of anaerobic bacteria secondary to increased intestinal urea excretion) is a characteristic in patients with ESRD, 23 resulting in increased production of toxic metabolites such as IS. 24 This accumulation cannot be avoided by regular hemodialysis. 25
To decrease serum IS levels, 3 main pathways are studied. The first pathway is to increase the effectiveness of dialysis such as modification of dialysis technique,26,27 and adding water-soluble adsorbent,28,29 but these procedures may be accompanied by protein loss and mortality risk. 30
The second pathway is to decrease IS production. Change in gut microbes was investigated using prebiotic, probiotic, or symbiotic, however; the resulting data are unsettled.31,32
The third pathway is to decrease IS intestinal absorption. The addition of activated charcoal as an adsorbent agent has been evaluated in uremic patients, especially AST-120 (KREMEZIN®). A lot of studies demonstrated the effectiveness of AST-120 in improving ESRD-related disorders but not the restoration of renal function.33,34 However, AST-120 is not worldwide available, and the high dose needed daily (20-30 piles/day) limits its use. 35
Activated charcoal is an easily available drug and less costly than AST-120 ($10 vs $349). Two previous studies showed the benefit of activated charcoal in the amelioration of uremic pruritus in a small number of patients, without an explanation for this action.36,37 In the present study, the usage of activated charcoal resulted in decreasing pruritus severity, combined with decreasing IS and improving the quality of life of patients without serious side effects.
The role of IS in pruritus is not fully explained. IS can activate and upregulate several meditators involved in itching such as protease-activated receptor-2,38-40 cannabinoid receptor type 1,41,42 NLRP3 Inflammasome,43,44 endothelin-1,45,46 signal transducer and activator of transcription 3,47,48 astrocyte activity,49,50 NADPH oxidase,51,52 transient receptor potential vanilloid 1,53,54 Intercellular Adhesion Molecule 1 (ICAM-1),55,56 Transforming growth factor-β (TGF-β),57,58 and macrophage.59,60 In addition, IS can downregulate anti-itching mediators such as nuclear factor E2-related factor 2.61,62 Also, IS is a neurotoxin and can initiate neuroinflammation and induce cognitive impairment and behavioral abnormality,63-67 which can exacerbate itching.68,69 All these mediators contribute to non-histamine-induced itching. 70
The results of this study showed that marked improvement of pruritus is accompanied by improved quality of life of patients. Many uremic patients with pruritus suffer from sleep disturbance, poor dialysis and treatment adherence, infection, depression, and increased mortality risk. 71 None of the uremic patients dropped out during the study. The improvement in quality of life increases the patient’s adherence to the dialysis session and decreases the mortality risk. 72 Meanwhile, a minority of the patients in this study remain complaint of pruritus. This can be explained by higher doses of activated charcoal or longer periods of treatment are needed to achieve marked improvement of relief of pruritus. Moreover, these patients had xerosis and needed skin moisture.
Conclusions
IS is elevated in uremic patients with pruritus. This elevation is correlated with the severity of pruritus. Treatment with activated charcoal results in improvement of decreasing serum level of IS, improvement of pruritus, and quality of life of patients.
Limitations
This is a single-center-based study. No placebo drug was used in this study and a large-scale randomized controlled clinical trial is needed to confirm these findings.
Footnotes
Author Contributions
EMA and OHA: Conception and design of the study. EMA, JME, AAE, NHM, and OHA: Analysis of data. JME, NHM, AMM, and AEM: Interpretation of data. EMA, NHM, and AEM: Drafting the article. JME and NHM: Revising it critically for important intellectual content. All authors: The final approval of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
