Abstract

To the Editor,
Current guidelines recommend targeted systemic treatments for managing moderate-to-severe atopic dermatitis (AD). 1 Despite the promising efficacy of Janus kinase inhibitors (JAKi) in randomized clinical trials (RCTs), their use has been associated with increased incidence of infections, such as herpes zoster (HZ). 2 This systematic review and meta-analysis characterizes infections observed with JAKi treatment in patients with AD.
Following PRISMA guidelines, MEDLINE and EMBASE databases were searched on April 13th, 2023 using English keywords for AD and JAKi (see Supplemental File 1). We included RCTs that reported infection incidence rates in patients treated with JAKi for moderate-to-severe AD. Risk of bias was evaluated for each study using the Cochrane tool for assessing risk of bias (RoB 2). 3 A fixed-effect meta-analysis using Peto’s odds ratios (OR) was performed using R (v.4.3.1, The R Foundation), to quantify the risk of HZ, upper respiratory tract infection (URTI), and serious infections with JAKi treatment in patients with AD compared to placebo. An I 2 statistic was used to assess heterogeneity across studies.
Fourteen articles met our eligibility criteria (Supplemental Figure 1; Supplemental Table 1). The weighted mean age of participants treated with JAKi and control agents was 35.6 and 35.0 years respectively. Based on a pooled meta-analysis using data from 11 studies, JAKi treatment in patients with AD increased the risk of HZ infections compared to placebo (Peto OR: 2.22, 95% confidence interval [CI]: 1.30-3.77; I2 = 0) (Figure 1). However, subgroup analysis showed no significant difference in the risk of HZ between individual JAKi drugs and placebo (Figure 1). Similarly, despite the risk of URTI also increasing with JAKi use overall (Peto OR: 1.31, 95% CI, 1.05-1.64; I2 = 0) among 14 studies, no difference in the rate of URT infections was observed with individual JAKi agents compared to placebo in the subgroup analysis (Figure 2). Additionally, there was no increase in the risk of serious infections with JAKi treatment overall or with specific JAKi drugs (Peto OR: 0.79, 95% CI: 0.53-1.19; I2 = 0) (Supplemental Figure 2).
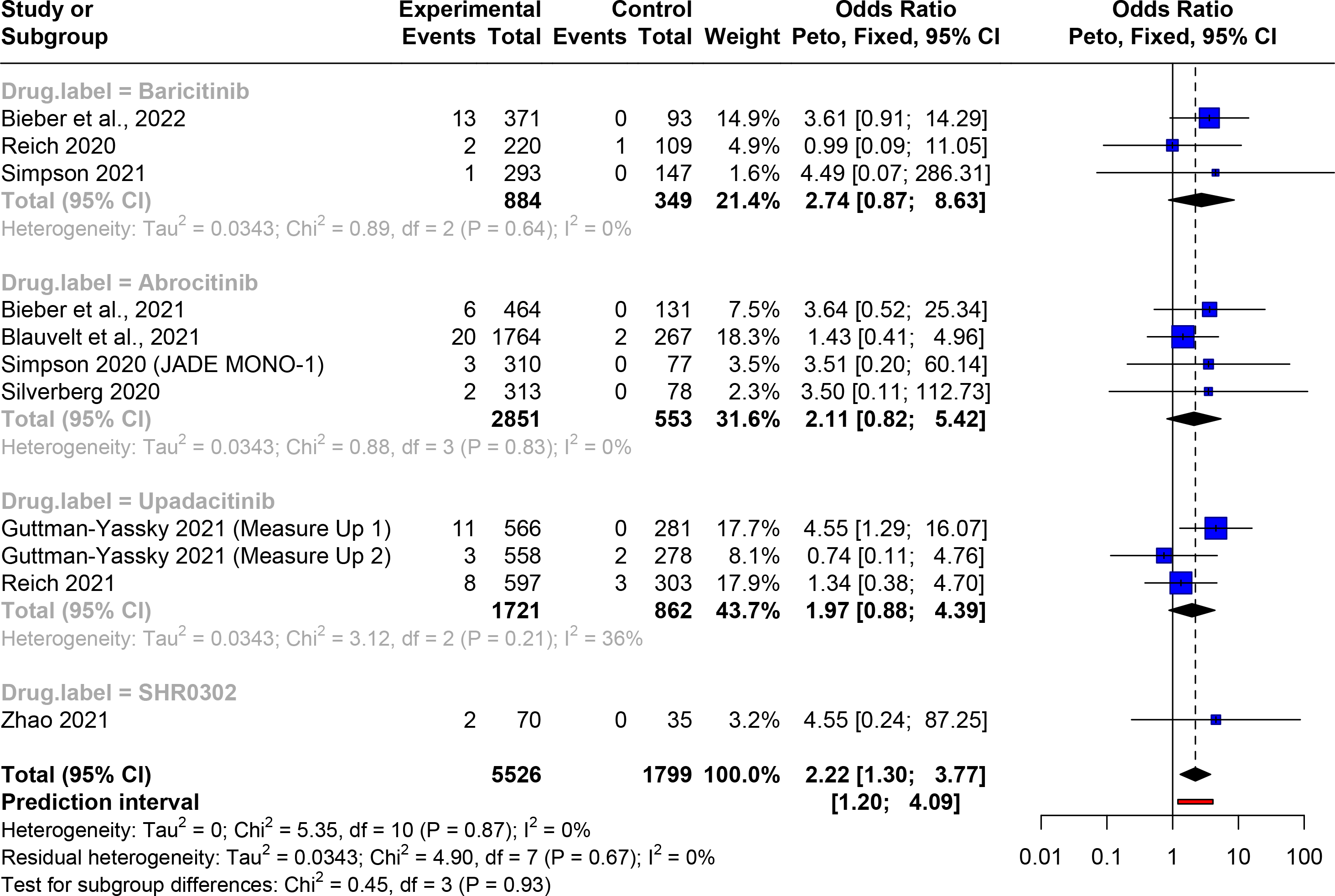
Forest plot of the risk of herpes zoster infection in patients with atopic dermatitis treated with a) abrocitinib vs. placebo b) baricitinib vs. placebo c) upadacitinib vs. placebo d) SHR0302 vs. placebo. Abbreviations: confidence internal, CI.
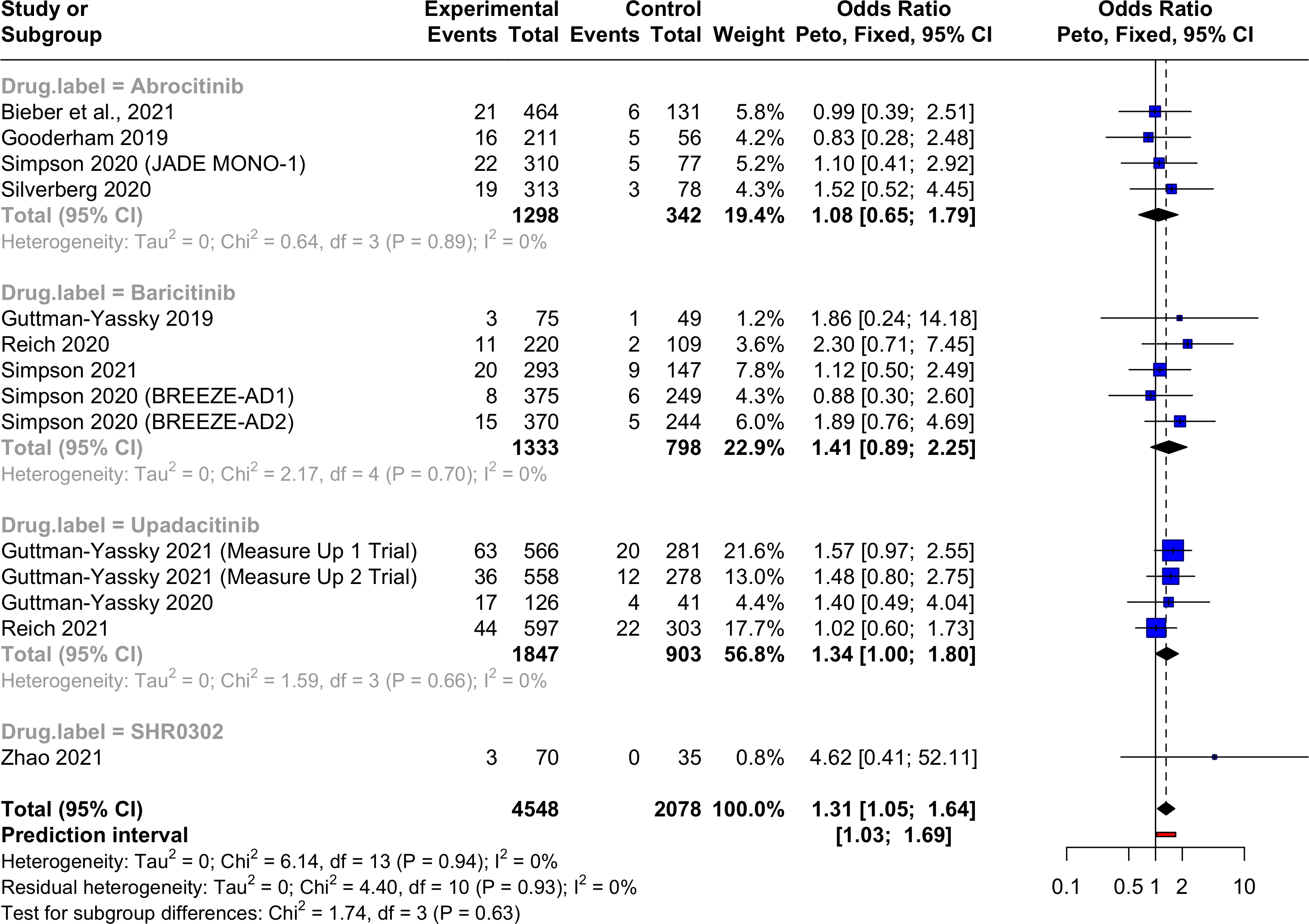
Forest plot of the risk of upper respiratory tract infection in patients with atopic dermatitis treated with a) abrocitinib vs. placebo b) baricitinib vs. placebo c) upadacitinib vs. placebo D) SHR0302 vs. placebo. Abbreviations: confidence internal, CI.
AD pathology is associated with an overproduction of Th2-mediated cytokines, which bind to immune cells and peripheral sensory neurons to further drive inflammation and pruritis. JAKi agents are promising treatment options since they block several intracellular signaling pathways involved in the regulation of Th2 cytokines. 4 However, JAKi treatment may affect other JAK-STAT-dependent cytokines that are involved in the differentiation of T cells, thus weakening the immune response to intracellular pathogens. 5 This may place patients at increased risk of infections such as HZ and URTI. This systematic review was limited by the small sample sizes for each drug, making subgroup analyses challenging. Furthermore, the reporting of concomitant treatments with corticosteroids and other immunosuppressive agents was inconsistent across studies and further sensitivity analysis could not be conducted.
Our findings support an association between JAKi treatment in AD patients and HZ and URT infections. Additional longer-term studies are needed to further elucidate the effect of JAKi treatment on infection risks and assess the comparative risk between different JAKi drugs and other drug classes.
Supplemental Material
online supplementary file 1 - Supplemental material for The Rate of Infections With Janus Kinase Inhibitor Treatment for Atopic Dermatitis: A Systematic Review and Meta-Analysis
Supplemental material, online supplementary file 1, for The Rate of Infections With Janus Kinase Inhibitor Treatment for Atopic Dermatitis: A Systematic Review and Meta-Analysis by Nawar Tarafdar, Muskaan Sachdeva, Natnaiel M. Dubale, Ciaran Smythe, Yuliya Lytvyn, Khalad Maliyar, Jorge R. Georgakopoulos, Asfandyar Mufti and Jensen Yeung in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Yeung has been a speaker, consultant, and investigator for AbbVie, Allergan, Amgen, Astellas, Boehringer Ingelheim, Celgene, Centocor, Coherus, Dermira, Eli Lilly, Forward, Galderma, GSK, Janssen, Leo, MedImmune, Merck, Novartis, Pfizer, Regeneron, Roche, Sanofi Genzyme, Takeda, UCB, Valeant, and Xenon. The other authors do not have any conflicts of interest.
Funding
The authors received no financial support for the research, author-ship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
