Abstract
Background
Acne vulgaris is a common dermatological condition that greatly impacts patients’ self-confidence. Ongoing research is conducted to explore new treatment modalities. Silymarin owns special characteristics that qualify it as a possible treatment for acne vulgaris.
Objective
We evaluated the efficacy and safety of silymarin cream as a new therapeutic option against salicylic acid peels in the treatment of mild to moderate acne vulgaris.
Methods
A split-face, comparative, Quasi-experimental clinical trial included 30 patients with acne vulgaris. Salicylic acid 30% peels were applied as an office procedure to one half of the face every 2 weeks for 3 months. Topical silymarin 1.4% cream was prescribed as a home treatment, twice daily, to the other half of the face for 3 months. The results were evaluated using the Global Acne Grading System (GAGS), photographic evaluation, and patient self-assessment scale. The adverse effects during treatment were recorded. The sample size was calculated by Stata/IC 16.1.
Results
After treatment, a significant reduction of GAGS was noted on both sides of the face, with an insignificant difference between both treatments. The comparative photographic evaluation and patient self-assessment scale were also insignificant. Hyperpigmentation was recorded in 2 cases on the salicylic acid-treated side. No side effects for silymarin cream were observed.
Conclusion
Topical silymarin cream 1.4% showed comparable results to Salicylic acid 30% peels. It can be considered a promising safe treatment modality for mild to moderate acne vulgaris.
Introduction
Acne vulgaris is a skin disease that affects the pilosebaceous unit. It manifests clinically as noninflammatory lesions (open and closed comedones), and inflammatory lesions (papules, pustules, nodules) in the affected skin. Eventually, it may cause scarring. 1 The frequency of acne among adolescents is 80% or more, making it an extremely prevalent skin condition. The prevalence ratio of males to females is approximately 1: 1.81. 2,3 While the majority of patients experience mild to moderate acne, 20% suffer severe acne that mostly leaves scars. 4 Anxiety, poor self-image, and depression are among the psychological adverse events in acne vulgaris with a detrimental effect on patients’ quality of life. 5
Treatment of acne aims to decrease both the noninflammatory and inflammatory lesions of acne. Thus, it helps reduce the incidence of possible complications such as scarring and improves the overall appearance. 6
Treatment of acne is challenging. Concerning the rise in antibiotic resistance globally, there is a shift away from antibiotics as a monotherapy for acne. Given how common acne is, especially in teenagers, there is still a significant demand for innovative treatments. 7 To address the various stages of acne pathogenesis, a multimodal strategy using a variety of products is advised. 8 Different treatment modalities had been used for acne vulgaris, of which peeling is one of the commonest.
Salicylic acid (SA), a beta-hydroxy acid, is considered a peeling agent of preference in active acne owing to its sebostatic and potent keratolytic actions. 9 It is capable of dissolving the intercellular cement substance, thereby decreasing corneocyte adhesion. 10 Additionally, it has well-known anti-inflammatory characteristics, which contribute to its acceptance and effectiveness for acne patients. 11 Salicylic acid is a great superficial peeling agent for patients with dark skin, considering that other peeling agents have been linked to post-inflammatory pigment changes. The capacity to self-neutralize, to decrease acne-related post-inflammatory pigmentation, and the high safety profile are all advantages of SA. 12 When applied repeatedly, SA improves both noninflammatory and inflammatory acne lesions. The most typical adverse reactions for SA are dryness, erythema, and a burning sensation. 10
Silymarin is extracted from Silybum marianum seeds. Due to its potent regenerative abilities, it is typically utilized as a hepatoprotective agent. Currently, silymarin is used in cosmetic and dermatological preparations because of its anti-inflammatory, immunomodulatory, and antioxidant effects. 13 The anti-inflammatory action of silymarin is exerted by reducing the expression of tumor necrosis factor-α (TNF-α), interleukin-1α (IL-1α), 12-O-tetradecanoylphorbol-13-acetate, and myeloperoxidase activity. Additionally, it suppresses the action of the UVB-induced cyclooxygenase and lipoxygenase, which inhibits the synthesis of prostaglandin metabolites. 14 Furthermore, silymarin was reported to reduce melanin synthesis by inhibiting tyrosinase’s ability to oxidize L-dihydroxyphenylalanine (L-DOPA). It also decreases the expression of the tyrosinase protein, hence its use in the treatment of melasma. 15 Silymarin has been used in oral and topical preparations for different dermatologic conditions including atopic dermatitis, rosacea, acne, melasma, psoriasis, melanoma, and non-melanoma skin cancers. In addition, it has been used in wound healing, anti-aging therapy, and cosmeceuticals. 16,17
For acne vulgaris, both the inflammatory reaction and oxidative stress play a major role in disease pathogenesis. As a result, topical antioxidant therapies like silymarin could be a helpful therapeutic option. 18
Herein, we aimed to evaluate the efficacy and safety of topical silymarin 1.4% cream as a novel therapy versus SA 30% peels as a well-known, effective, safe treatment in acne vulgaris.
Materials and Methods
We conducted a comparative, split-face, Quasi-experimental clinical trial. Thirty adult patients of both sex, older than 18 years of age, with mild to moderate acne vulgaris, of different disease durations, were included in the study. The skin phototype of all patients was III to IV. The diagnosis of acne vulgaris was made clinically based on the typical clinical features of the disease. Exclusion criteria were uncertain diagnosis of acne vulgaris, severe acne, patients under treatment with contraceptive pills or any kind of systemic or topical acne medication (e.g., isotretinoin, antibiotics, topical products) during the last month, patients with a history of hypertrophic or keloid scar formation, active bacterial, fungal, or herpetic infection, photosensitivity, hypersensitivity to salicylic acid, pregnancy, uncooperative patient and patient with unrealistic expectations. The experimental design was approved by the institutional Ethics and Research Committee (N: IRB17101388). Informed consent was obtained from each patient before participation in the study and for publication of the images.
All patients were subjected to history taking, including name, age, sex, duration of acne, and previous treatment. Most patients reported previous use of topical preparations for their acne at home and few reported systemic antibiotic use. Acne severity was evaluated using Global Acne Grading System (GAGS) 19 ; the total severity score is the summation of the subscores of 6 regions. Each score is calculated by multiplying the factor (2 for forehead, 2 for each check, 1 for nose, 1 for chin, 3 for both chest and back) by the most predominant lesion in each area (1 for 1 or more comedones, 2 for 1 or more papules, 3 for 1 or more pustules, and 4 for 1 or more nodules). Grading of the severity of acne is according to the total score; Mild acne: a score of 1-18, Moderate: 19-30, Severe: 31-38, and very severe: >39.
The studied patients underwent facial chemical peeling using SA 30% applied to the right half of the face every 2 weeks and used topical silymarin 1.4% cream to the left half twice daily as a home treatment. The study duration was 3 months. Salicylic acid peel was performed by washing the face, degreasing the skin with alcohol, and applying SA with a cotton ball to one half of the face. A white precipitate on the skin with a burning, stinging sensation disappearing within 4 minutes, was considered an endpoint. The face was then washed, and emollients and sunscreen were prescribed after treatment. Preparation of Silymarin cream was done with the following formula: glycerin 5 g, stearic acid 15 g, H2O 79 g, Tween‐80 1%, KOH 0.72 g, and sodium benzoate 0.1%. Then Silymarin, as 1.4%, was added. 20 At the follow-up visits, patients were requested to confirm compliance with the twice-daily application of Silymarin cream at home.
Patients were assessed before treatment and after 3 months by two main criteria; photographs using a digital camera (Canon Digital IXUS 850 IS, China) and clinical response assessed through grading by GAGS. The photographic assessment was done by two-blinded dermatologists after treatment on both sides of the face as follows; poor, good, very good, or excellent improvement. The safety of drugs was evaluated by comparison of side effects rates. Patient self-assessment grades were used as follows; 0 indicates no improvement, 1: 1% to 25% improvement, 2: 26% to 50%, 3: 51% to 75%, and 4: 76% to 100% improvement.
Sample Size Calculation
It was calculated using Stata/IC 16.1 after reviewing the literature. A sample size of 30 achieves at least 90% power to show a difference of 8.55 between the null hypothesis that the mean before and after treatment are 17.55 and our hypothesis that the mean after treatment with salicylic acid 0% is 9.00 with estimated standard deviations of 10.40 before treatment and 7.64 after treatment with a level of significance equal to 0.05 using a paired student t-test then we added 6 patients taking into consideration a 20% drop rate.
Statistical Analysis
Data analysis was done using IBM-SPSS (Statistical Package for the Social Sciences) version 24.0 (IBM-SPSS Inc., Chicago, IL, USA) after verification and coding of data by the researcher. Test of significances: Chi-square/Fisher’s Exact/Monte Carlo Exact test was used to compare the difference in the distribution of frequencies among different groups. Student t-test was calculated to test the mean differences in continuous variables between groups, while paired sample t-test was used to compare the mean before versus after treatment. Two-way repeated measure analysis was calculated to test the mean differences of the data that follow a normal distribution and had repeated measures (between groups, within groups, and overall difference). Significant test results were considered when the p value ≤ .05.
Results
The current study included 30 patients with mild to moderate acne recruited from the outpatient clinic of the Dermatology department. The findings of the current work showed that the age of the participants ranged between 18 and 26 years old with a mean of 20.77 ± 2.7 years old. The majority (90%) were females (n = 27). The severity of acne according to GAGS demonstrated that two-thirds (63.3%) of patients had mild acne (n = 19), and about one-third (36.7%) had moderate acne (n = 11). Some baseline socio-demographic and clinical characteristics of the studied patients are shown in Table 1.
Some Socio-Demographic and Clinical Data of Included Patients.
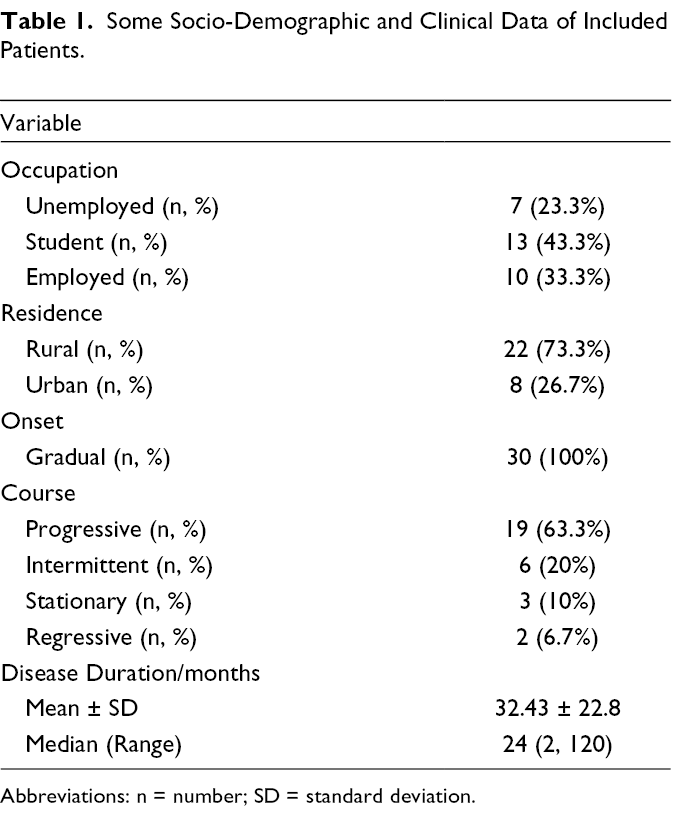
Abbreviations: n = number; SD = standard deviation.
Regarding improvement, the GAGS decreased significantly after treatment on both sides (p < .001) as shown in Table 2 (Figures 1 and 2). On the right side of the face treated with SA 30% peeling, the score of acne decreased from a mean of 9.67 ± 3.1 pre-treatment to 5.30 ± 2.7 after treatment. Likewise, the left side of the face treated with silymarin 1.4% cream showed a score reduction from a mean of 8.90 ± 3.5 pre-treatment to 4.83 ± 2.5 post-treatment. However, the difference between both sides was insignificant (p = .377). In addition, photographic evaluation of the improvement after treatment revealed an insignificant difference (p = .322) between both groups (Table 3). Patient self-assessment after treatment is shown in Table 3 and also found an insignificant difference between the right and left sides of the face (p = .496).
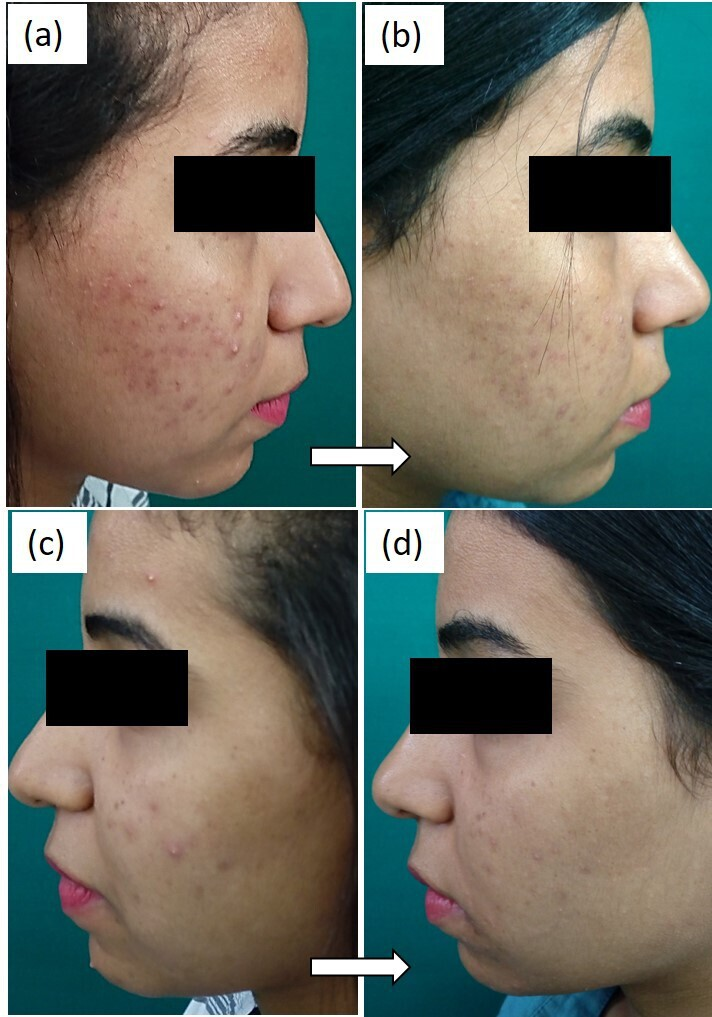
Treatment with SA 30% for the right facial side: (
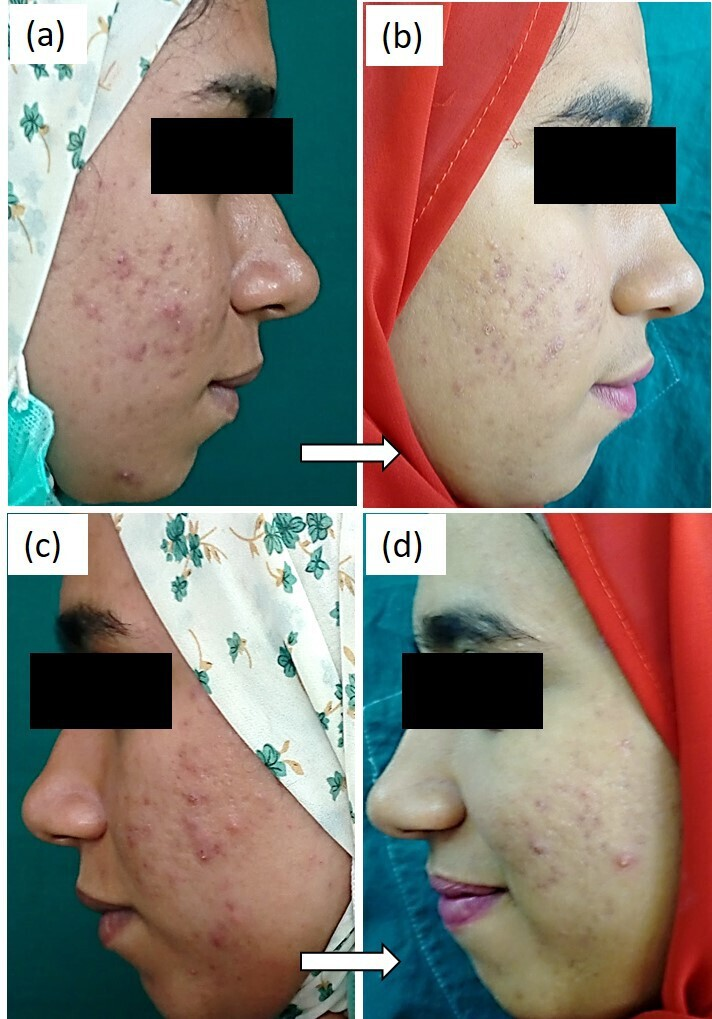
Treatment with SA 30% for the right facial side: (
Effect of Treatment on GAGS.
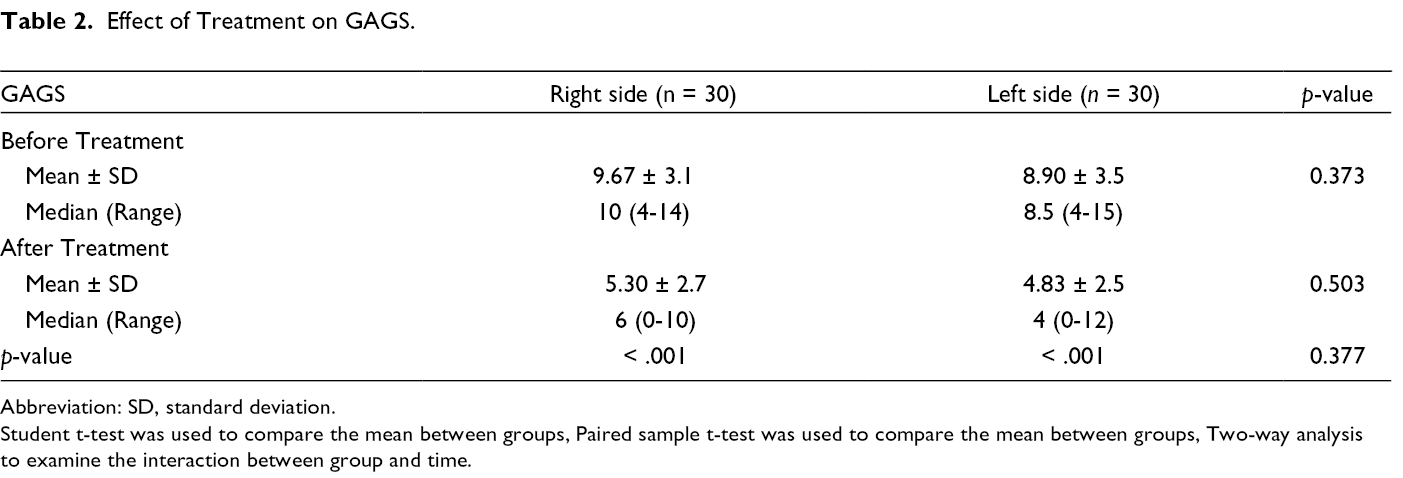
Abbreviation: SD, standard deviation.
Student t-test was used to compare the mean between groups, Paired sample t-test was used to compare the mean between groups, Two-way analysis to examine the interaction between group and time.
Photographic Assessment and Patient Self-Assessment After Treatment.
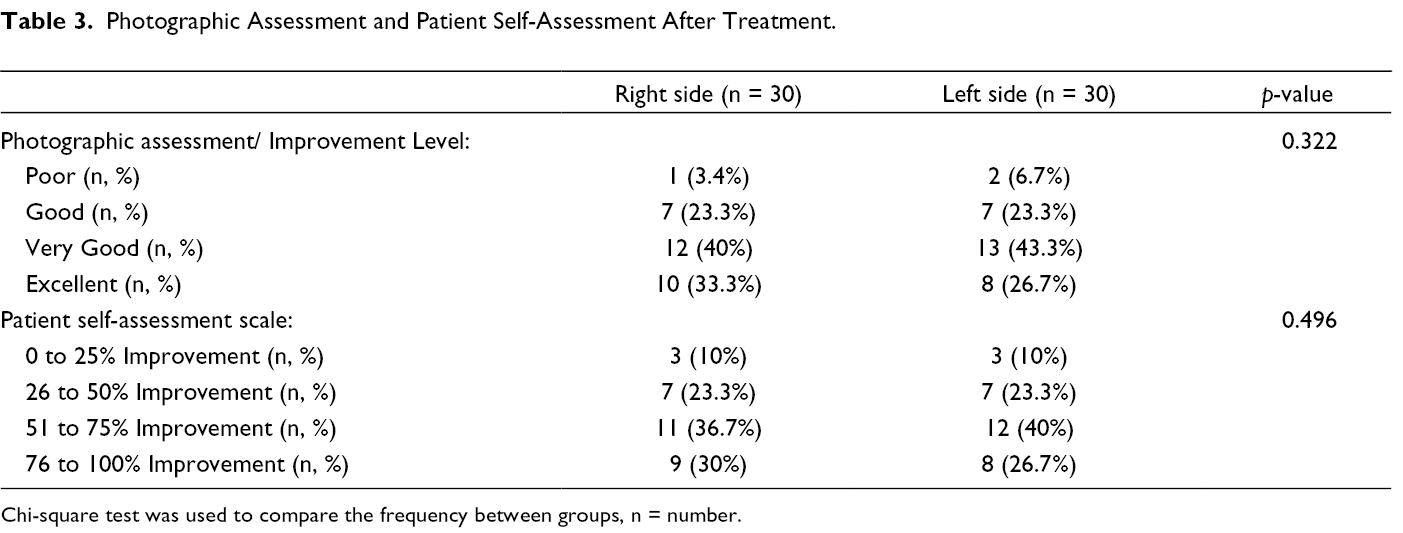
Chi-square test was used to compare the frequency between groups, n = number.
The adverse effects among the studied groups were evaluated. On the right side
Discussion
Acne vulgaris is a chronic inflammatory skin disease that typically involves the face, chest, and back. 21 There is a wide variety of therapeutic options that are available for acne. These are mainly topical, systemic, and procedural treatments. To the best of our knowledge, this is the first study to use pure topical silymarin 1.4% cream in acne vulgaris therapy.
The present study revealed the efficacy of SA 30% peeling in the treatment of acne vulgaris. As evaluated by GAGS, the severity of acne was significantly reduced after treatment with SA 30% peeling for one side of the face every 2 weeks for 3 months (p < .001). These results agreed with the results of Abdel Meguid et al. 22 who included 20 patients, Fitzpatrick skin Types III to V with facial acne. Twenty-five percent of Trichloroacetic acid (TCA) was applied to the right half of the face and SA 30% to the left half at 2 week intervals for 2 months. The total number of lesions before and after treatment showed significant improvement on both sides (p < .000). Our results of SA 30% peeling also agreed with the results of Dayal et al. 23 ; they included 50 patients with mild-to-moderate acne. The patients were randomly divided into two groups and received either 30% SA peels or 45% mandelic acid peels at 2 week intervals for 6 sessions. Both agents were found to be effective in treating mild-to-moderate acne according to Michaelsson Acne Score (MAS) (p < .001 in both groups).
Silymarin was found to retain both anti-inflammatory and antioxidant properties. 18 Compared to healthy skin, acne-prone skin is known to have higher levels of oxidative stress and lower amounts of antioxidants. 24 Recent research indicates that lipid peroxidation induces inflammation and fosters the growth of acne-causing bacteria. Because of this, topical antioxidant therapies like silymarin can aid in the disruption of such pathogenesis. 25
The present study revealed a promising efficacy of topical silymarin 1.4% cream in the treatment of acne vulgaris. The severity of acne, as evaluated by GAGS, was significantly reduced after treatment with silymarin cream for one half of the face twice daily for 3 months (p < .001). Kim and colleagues 26 investigated 22 patients with mild to moderate acne. They prescribed 0.5% silymarin-containing antioxidant serum, twice daily for 4 weeks, as an adjuvant treatment for participants while using their anti-acne medications. The acne lesion counts, modified GAGs, and Global evaluation of acne scale were all significantly reduced (p ≤ .001). Besides, Lynch and co-workers 25 used a serum containing 0.5% silymarin, 15% vitamin C, 0.5% ferulic acid, and 0.5% SA in 53 subjects with mild-to-moderate acne, who applied it once daily for 12 weeks. It showed a 76% average reduction in the lipid-peroxidation index compared to the baseline, proposing the formula to be a possibly effective topical antioxidant therapy for blemish-prone, oily skin.
The beneficial effect of silymarin was also supported by Al-Anbari et al.
27
Another randomized, controlled trial was performed by Shie et al. 18 on 60 patients with acne vulgaris. Three groups, each of 20 patients, were treated with oral silymarin, oral doxycycline, or a silymarin/doxycycline combination. The response to silymarin was insignificantly different from doxycycline in terms of GAGS (p = .260) but was significantly less according to the Acne Severity Index (ASI) (p = .021). In addition, the silymarin/doxycycline combination showed a more favorable response against doxycycline alone without a significant difference (p = .9 for ASI, p = .5 for GAGS).
The assessment of possible adverse effects during treatment in the present study revealed their absence in the group of silymarin cream. Post-treatment hyperpigmentation was detected in 6.7% of patients in the SA group without a significant difference between both treatments. In agreement with our results, Nofal et al. 20 reported the absence of adverse effects of 2 concentrations of topical silymarin cream in the treatment of melasma. They included 44 female patients with melasma divided into 3 groups, each of 14 patients. Groups 1 and 2 were treated with twice daily application of 0.7% and 1.4% silymarin cream respectively. Group 3 received 4% hydroquinone cream, once daily and showed a significant difference in incidence of adverse effects compared to silymarin cream. However, the relatively small sample size in that study and our study could have affected the occurrence and detection of side effects by silymarin cream.
No sufficient information is available about silymarin safety in pregnancy. Animal studies showed controversy regarding the teratogenicity of oral silymarin in mouse and rat models. 28,29 As human studies are lacking, oral and topical silymarin are currently not supposed to be used during pregnancy. Multiple research works considered silymarin as one of the natural galactagogues that increase milk production in humans. 30,31 Although that effect is of low evidence, those studies did not report any adverse effects in infants. 32 Also, a recent animal study on mice documented the absence of adverse effects of silymarin on the offspring during breastfeeding. 29
In the current study, there was no statistically significant difference in the therapeutic response between topical silymarin 1.4% cream and SA 30% peeling in the treatment of mild to moderate acne vulgaris (p = .377). This could be possibly explained by the fact that both drugs act through more than one mechanism in treating acne lesions.
In conclusion, silymarin 1.4% cream was proven effective and safe in the treatment of mild to moderate acne vulgaris with comparable results to SA 30% peeling as a well-known therapy.
Limitations of the present study included a relatively small sample size, and lacking a measure of treatment responses in different acne lesions (inflammatory and noninflammatory), as well as a follow-up period to detect acne recurrence. Further comparative, clinical trials on topical silymarin either as a mono- or adjunctive therapy in acne vulgaris, and searching for the optimum concentration are recommended in future studies.
Footnotes
Ethics Approval
The experimental design was approved by the institutional Ethics and Research Committee of Assiut University (N: IRB17101388). Informed consent was obtained from each patient before participation in the study and for publication of the images.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
