Abstract
Psoriatic arthritis (PsA) can affect a diverse range of anatomical sites and its heterogeneous presentation contributes to misdiagnosis and delayed treatment with conventional and biologic disease-modifying antirheumatic drugs (DMARDs). Up to 15% of psoriasis (PsO) patients affected by PsA remain undiagnosed. Early detection and referral to a rheumatologist are crucial to optimize care and minimize irreversible erosive joint damage. To improve the rheumatology referral process, the authors propose a risk stratification tool to identify and triage patients with possible psoriatic arthritis. With the aim of ultimately assisting in early treatment initiation, this risk stratification algorithm can be used in both dermatology and primary care clinics. It is based on the Psoriasis Epidemiology Screening Tool (PEST) combined with the ClASsification criteria for Psoriatic Arthritis (CASPAR). This article intends to provide a rationale for further prospective studies whose objective would be to validate this screening algorithm.
Introduction
Psoriatic arthritis (PsA) can affect numerous anatomical sites including peripheral joints, axial skeleton, and entheses in addition to the diverse skin and nail findings. Its heterogeneous presentation and widely varying degree of disease severity can delay treatment initiation with conventional or biologic disease-modifying antirheumatic drugs (DMARDs). 1,2
In more than 75% of cases, cutaneous lesions precede the development of musculoskeletal symptoms. 2 Dermatologists and primary care providers thus play an essential role in the early identification of PsA. 3,4 It is estimated that approximately 25 to 30% of psoriasis (PsO) patients have concurrent PsA disease. 5 Furthermore, up to 15% of PsO patients affected by PsA remain undiagnosed. 5,6
Early detection and referral to a rheumatologist are imperative to minimize irreversible erosive joint damage. 7 -9 As biologics agents are increasingly used to treat PsO, selecting a medication that addresses both rheumatologic and cutaneous disease implies the recognition of concurrent inflammatory arthritis in the patient with PsO.
The Psoriasis Epidemiology Screening Tool (PEST) is a self-administered PsA screening tool (Figure 1) which has demonstrated superior or comparable sensitivity, and relative specificity compared to other screening tools such as the Psoriatic Arthritis Screening and Evaluation (PASE), the Early Arthritis for Psoriatic Patients (EARP), the Toronto Psoriatic Arthritis Screen (ToPAS), the Psoriasis Arthritis Screening Questionnaire (PASQ) and the CONTEST questionnaire. 10 -13 The PEST’s sensitivity ranges from 60% to 94% and its specificity from 66% to 78% in different studies. 10 -13 Compared to other aforementioned screening tools which can include more than 10 items, the 5-question PEST’s rapidity of completion provide an important advantage. 14 Once screening is completed, physicians can apply the ClASsification criteria for Psoriatic Arthritis (CASPAR) (Table 1) to selected patients to identify those with the highest likelihood of PsA and thus hasten appropriate referrals to rheumatology.
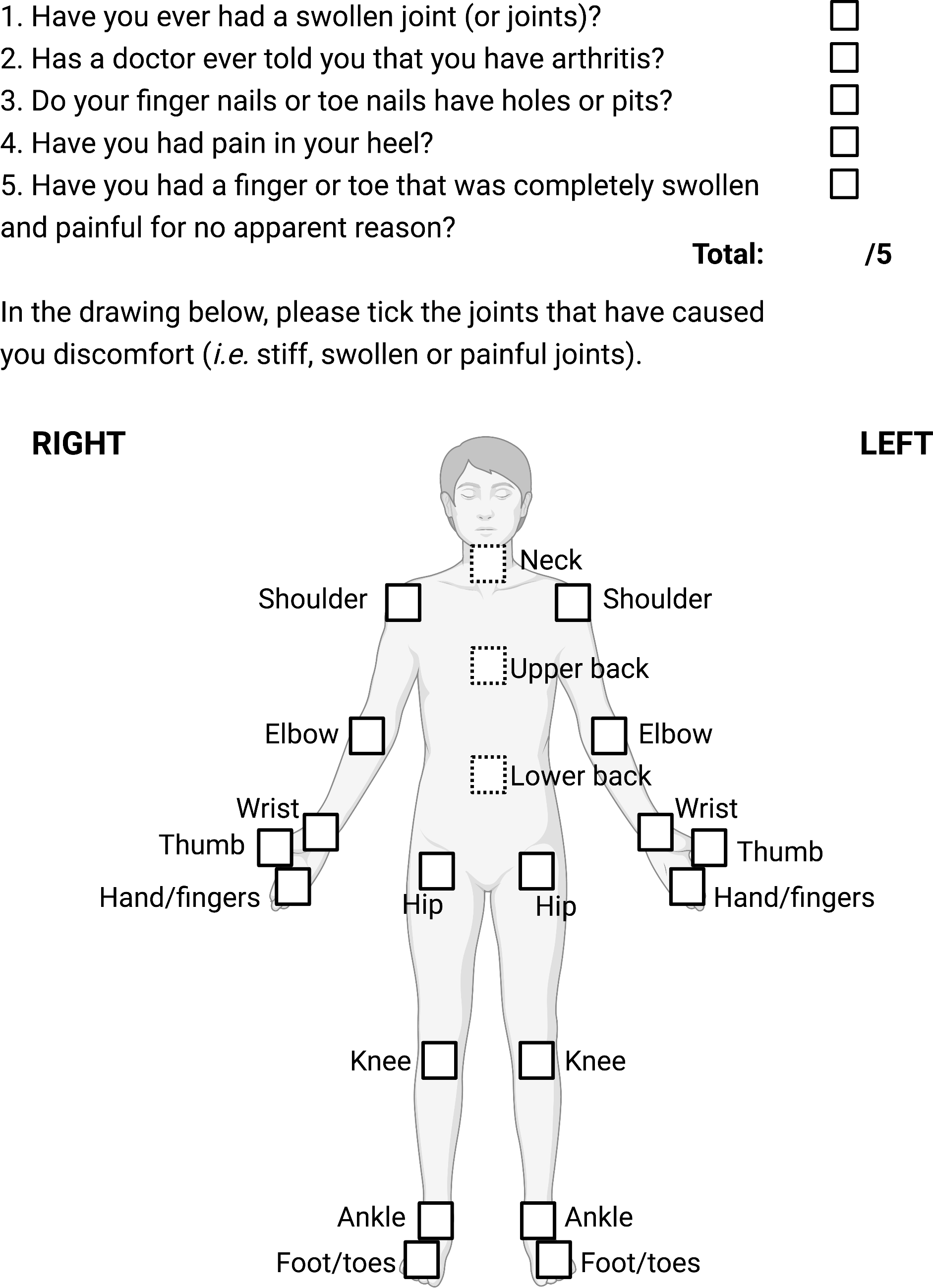
The Psoriasis Epidemiology Screening Tool (PEST). Adapted from: Ibrahim GH, Buch MH, Lawson C, Waxman R, Helliwell PS. Evaluation of an existing screening tool for psoriatic arthritis in people with psoriasis and the development of a new instrument: the Psoriasis Epidemiology Screening Tool (PEST) questionnaire. Clin Exp Rheumatol. 2009;27(3):469-474.
the ClASsification Criteria for Psoriatic Arthritis (CASPAR)

Risk Stratification Strategy
The authors propose a twostep risk stratification tool based on the PEST score combined with the CASPAR criteria. To facilitate early referral and treatment initiation, sensitivity prevails over specificity. Consequently, the authors recommend using a PEST score of ≥2 to identify patients with suspected PsA. Besides, it is the set threshold score to initiate a referral to rheumatology (Figure 2), as the cut-off of a higher score of ≥3 can decrease the PEST sensitivity to 60% based on select studies. 12,13 To mitigate unnecessary referrals to rheumatologists, the highly-specific CASPAR criteria (specificity of >98%) 15,16 can subsequently be used to further categorize patients. In fact, those with a moderate or high risk of concurring PsA would be evaluated within a timeframe of 6 months or 3 months, respectively. To complete the CASPAR criteria evaluation, a rheumatoid factor level and radiographs of the wrists, hands, ankles, and feet in search of juxta-articular new bone formation are required (Table 2). Radiographs of other painful joints can be obtained to facilitate the rheumatologist’s evaluation. Patients who score ≥3 on the CASPAR criteria are considered to have a high suspicion for PsA. They should be evaluated within 3 months and should be sent for work-up in preparation for immunosuppressive therapy with laboratory tests as defined in Table 2: complete blood count, creatinine, liver function tests, urinalysis, hepatitis B and C serologies, as well as a tuberculin skin test or IGRA (Interferon-Gamma Release Assay). Hepatitis A, human immunodeficiency virus (HIV) and strongyloides serologic testing can be requested if risk factors are present. A chest radiograph should be obtained if none was performed in the last year. C-reactive protein levels and anti-CCP antibodies are helpful as well. Considering the cardiovascular adverse effects of certain classes of medication, a lipid profile can also be added to the work-up. Patients who score ≤1 on the PEST questionnaire can be considered of low suspicion for PsA. However, repeated screening with the PEST is encouraged at least yearly or whenever joint pain symptoms are present bearing in mind that PsA can develop at any time point in the PsO disease course, and its initial presentation can be subtle. 1,2
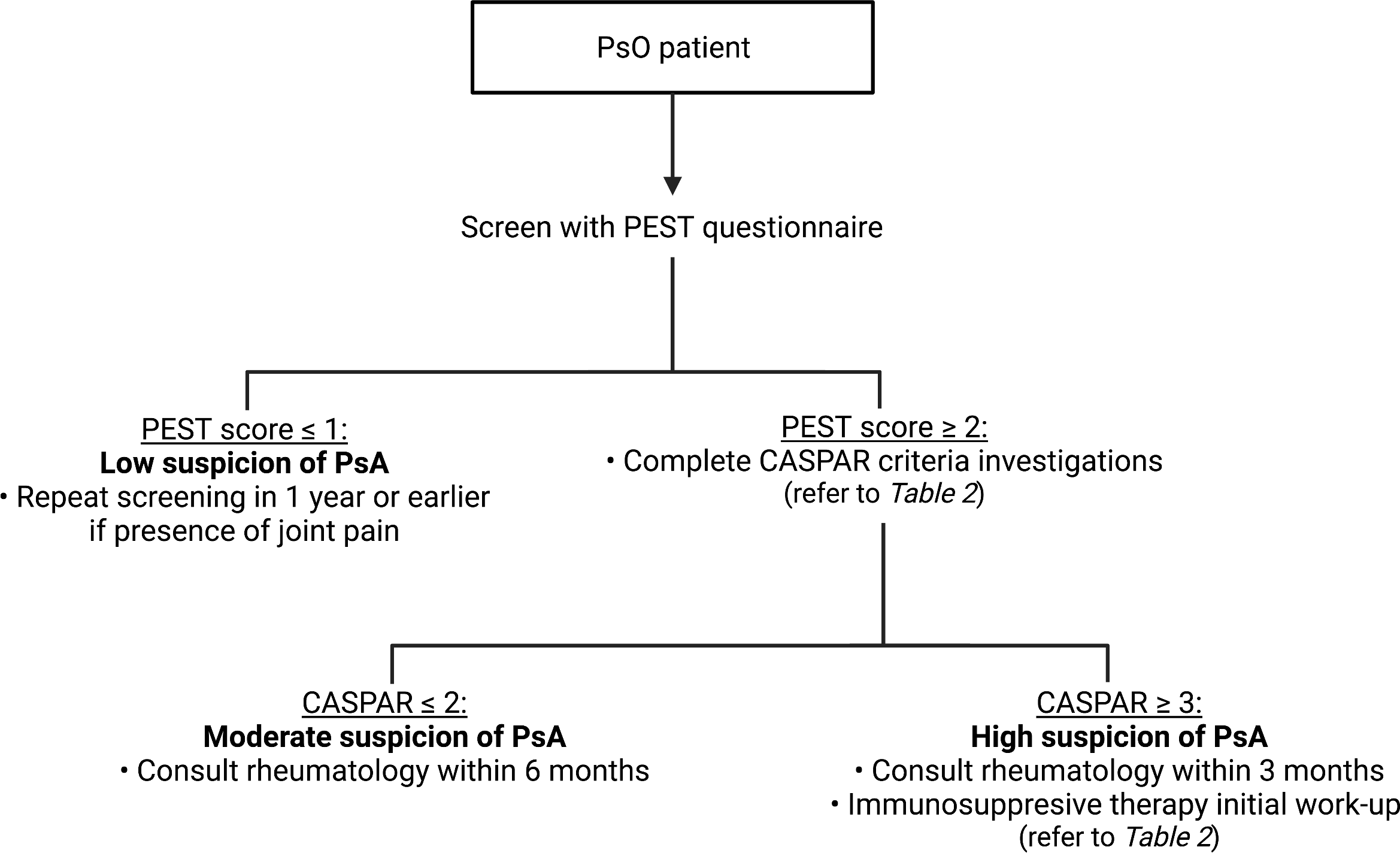
Risk stratification algorithm for screening and referral of patients with suspected PsAAbbreviations: CASPAR, ClASsification criteria for Psoriatic Arthritis; PEST, Psoriasis Epidemiology Screening Tool; PsA, psoriatic arthritis; PsO, psoriasis
Investigations Included in the Risk Stratification Algorithm
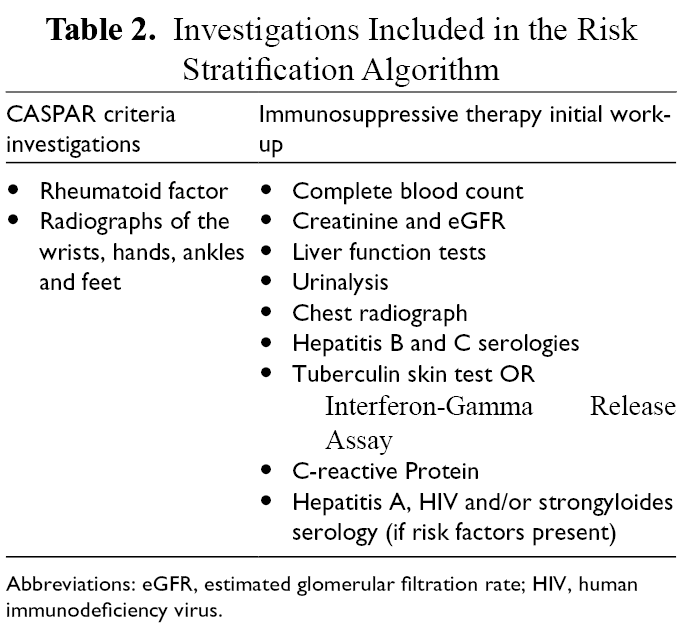
Abbreviations: eGFR, estimated glomerular filtration rate; HIV, human immunodeficiency virus.
Conclusion
The PEST questionnaire and the CASPAR criteria have both been individually studied and validated for screening and detection of PsA disease. Further prospective studies are needed to validate the authors’ proposed combined PEST/CASPAR screening algorithm which has been developed with the goal of optimizing early referral and early treatment of PsA patients. We aim to contribute to its validation by integrating this risk stratification algorithm in our research center and associated clinics to generate data that support its utilization.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
