Abstract
The IL-17 pathway is a potential therapeutic target shown to be implicated in hidradenitis suppurativa (HS), however, it remains unclear whether evidence from mechanistic studies may translate into clinical practice. This systematic review summarizes available treatment outcomes of IL-17 inhibitors in patients with HS. Embase, MEDLINE, PubMed, and clinicaltrials.gov were comprehensively searched on February 26, 2021 to include 16 original studies representing 128 patients with HS (mean age: 36.5 years; age range: 21-47 years; male: 50.0%). Treatment outcomes were reported for the following biologics: secukinumab (n = 105), brodalumab (n = 22), and ixekizumab (n = 1). Patients were classified as responders or non-responders according to achievement of a positive response/improvement based on criteria established for each included study. For secukinumab 57.1% (n = 60/105) of patients were responders in a mean response period of 16.2 weeks and 42.9% (n = 45/105) were non-responders; for brodalumab, 100.0% (n = 22/22) of patients were responders within 4.4 weeks; and the one patient treated with ixekizumab was a responder within 10 weeks. In conclusion, IL-17 inhibitors may serve as an effective therapeutic target in approximately two-thirds of patients with HS and can be considered in those who are refractory to other treatment modalities. We also stress the importance of consistent outcome measures to enhance evidence synthesis, decrease reporting bias, provide potential for future meta-analysis, and ultimately improve clinical outcomes for patients with HS.
Introduction
Hidradenitis suppurativa (HS), also known as acne inversa, is a chronic cutaneous disorder characterized by inflammatory nodules, abscesses and fistulae, with lesions most commonly occurring in the groin, inframammary folds and axillae. 1 HS tends to present in the second decade of life, occurs predominantly in females and affects approximately 1% of the population. 1,2 HS has a detrimental impact on physical, social and emotional aspects of life, extending into peoples’ ability to work and complete activities of daily life. 3 -6 A recent study found that HS may have a greater impact on general health when compared to depression, type 2 diabetes, hypertension, congestive heart failure and myocardial infarction. 7
Therapeutic options for HS have been limited to adalimumab, the only approved biologic for this indication. 1,6 However, recently, as summarized in the expert opinion by Rosales et al. (2020) and systematic review by Frew at el. (2020), an increasing number of promising biologic therapies have been reported to lead to favorable outcomes in HS treatment. 1,6 The IL-17 pathway is a potential therapeutic target shown to be implicated in hidradenitis suppurative (HS) as shown by several experimental studies. For example, Schlapbach et al. (2011) found a 30 fold increase in gene expression of IL-17 and clusters of IL-17 producing T-helper cells in HS lesional tissue as compared to normal skin. 8 Additionally, Matusiak et al. (2017) found elevated serum levels of IL-17 in HS patients. 9 However, it remains unclear whether evidence from mechanistic studies may translate into clinical practice. 6 The objective of this systematic review is to comprehensively summarize treatment outcomes of IL-17 inhibition in patients with HS. This information will be important to physicians managing patients with HS.
Methods
Guidelines from the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA), were followed for this systematic review. 10
Search Strategy
A literature search utilized Embase, MEDLINE and PubMed databases as well as clinicaltrials.gov on February 26, 2021 using variations of pre-determined keywords, (“hidradenitis suppurativa” or “acne inversa” or “verneuil’s disease” or “pyoderma fistulans significa”) and (“IL 17” or “ixekizumab” or “brodalumab” or “secukinumab”). No date or language restrictions were used.
Eligibility
Articles were included if they reported (i) treatment outcomes of HS patients receiving IL-17 inhibitors, using validated or quantifiable scales, (ii) were observational or experimental studies, with human subjects, and iii) had data in the English language. Examples of non-validated or quantifiable scales which were excluded include “clinical response” and “discharge and nodule counts.”
Data Screening
Title and abstract screening and full text review were completed by two independent researchers (N.K. and A.M.) while conflicts were resolved by discussion with a third reviewer (J.Y.) Relevant articles were manually checked to identify any additional studies.
Quality Appraisal
After full text review, quality appraisal of included studies was assessed independently by two reviewers (N.K. and Y.L.) using the Joanna Briggs Institute (JBI) critical appraisal tools. 11 Any conflicts which arose were resolved by discussion between the two reviewers. Guided by the JBI manual for evidence synthesis and based on the number of criteria met for each type of study (Supplemental File 1-4), authors grouped studies into the following categories: (1) good quality, (2) moderate quality, and (3) poor quality. Authors decided to exclude any studies that were of poor quality. 11
Data Extraction
Data extraction was completed by two researchers (S.A. and A.R), data verified by another researcher (N.K.) and conflicts resolved by discussion with a third researcher (A.M.) Data were extracted on study and patient characteristics, HS information, biologic information and treatment outcomes.
Definitions
Treatment outcomes of IL-17 inhibitors collected included the following:
Responder: defined as a patient achieving a positive response/improvement based on criteria established for each included study in the review. For example, achieving the Hidradenitis Suppurativa Clinical Response (HiSCR). Table 1 provides a summary of each study’s interpretation of response.
Nonresponder: defined as no response/improvement based on criteria established for each included study in the review. For example, not achieving HiSCR (Table 1).
Mean response period: defined as the mean time between treatment initiation and response/improvement of HS.
Summary of Response Measures to Quantify Treatment Outcomes.
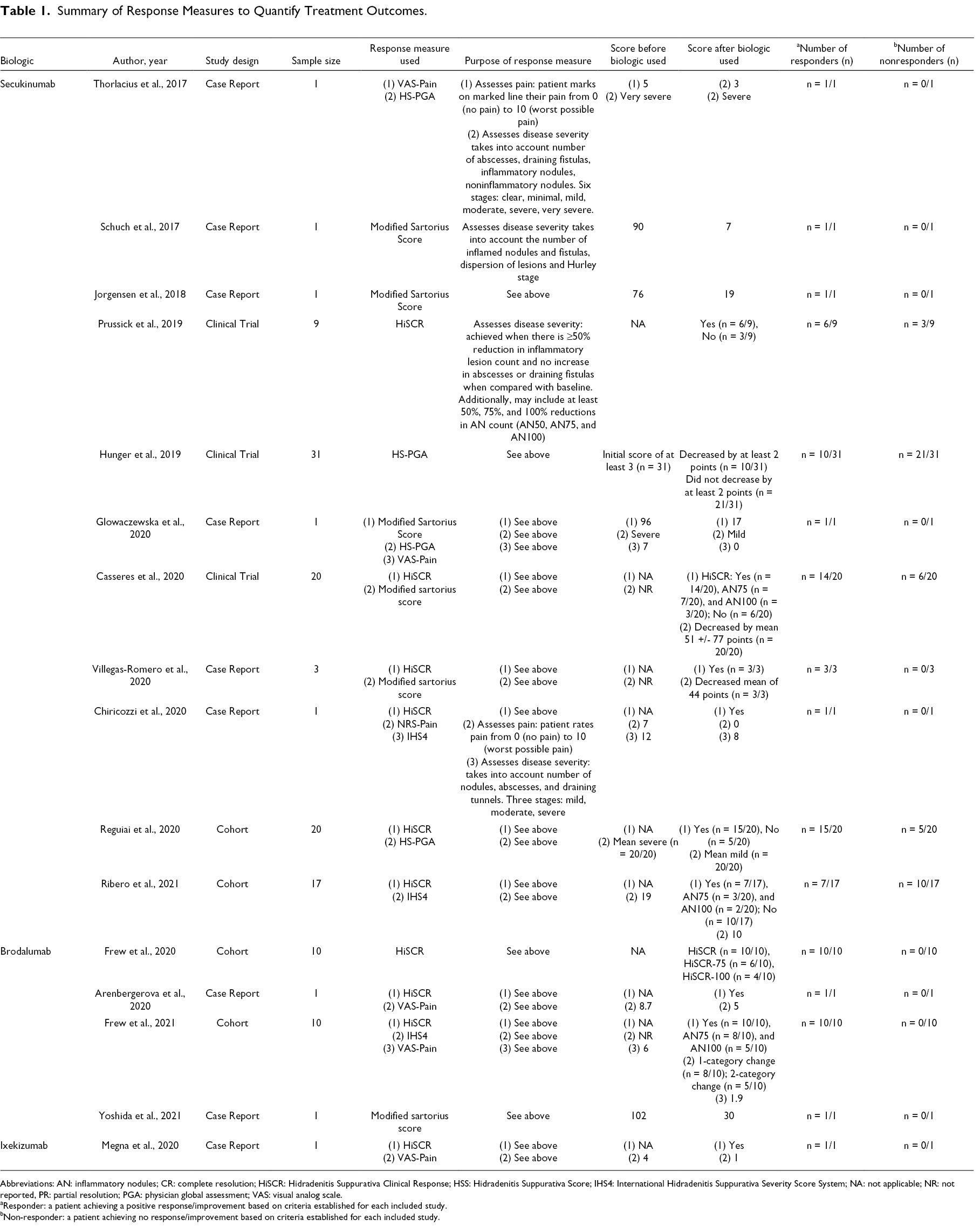
Abbreviations: AN: inflammatory nodules; CR: complete resolution; HiSCR: Hidradenitis Suppurativa Clinical Response; HSS: Hidradenitis Suppurativa Score; IHS4: International Hidradenitis Suppurativa Severity Score System; NA: not applicable; NR: not reported, PR: partial resolution; PGA: physician global assessment; VAS: visual analog scale.
aResponder: a patient achieving a positive response/improvement based on criteria established for each included study.
bNon-responder: a patient achieving no response/improvement based on criteria established for each included study.
Level of Evidence
The Oxford Centre for Evidence-Based Medicine 2011 Levels of Evidence were used to determine level of evidence for included articles. 12 Due to the heterogeneity of data, only descriptive analysis was performed, no meta-analysis was completed.
Results
Study and Patient Characteristics
Of the 311 studies identified, 36 full texts were assessed for eligibility and 16 met inclusion criteria, representing 128 patients (mean age: 36.5 years; age range: 21-47 years; male: 50.0%) (Supplemental File 5-6). 13 -28 Studies included 9 case reports, 4 cohort studies and 3 clinical trials. Level of evidence analysis yielded 9 studies with a level of evidence of 5, 4 studies with level of evidence of 2, and 3 studies with level of evidence of 1. Treatments outcomes were reported for the following biologics: secukinumab (n = 105), brodalumab (n = 22) and ixekizumab (n = 1).
Outcome Measures
Table 1 summarizes the outcome measures reported in included studies to quantify treatment outcomes. In total, 17 different outcome measures were used to mark treatment outcomes of HS in patients on IL-17 inhibitors (mean per study: 4; median per study: 4; range: 1 to 9). Most commonly, HiSCR and the Dermatology Life Quality Index were reported in 68.8% (n = 11/16) studies each, the Modified Sartorius Score (MSS) in 56.3% (n = 9/16) studies each, and the International Hidradenitis Suppurativa Severity Score System (IHS4) in 50.0% (n = 8/16) studies (Supplemental File 7). Next, the pain Visual Analog Scale (VAS) was reported in 37.5% (n = 6/16) studies, the HS-Physician Global Assessment (HS-PGA) in 25.5% (n = 4/16) studies and the Global Disease Assessment VAS was reported in 13.3% (n = 3/16) studies. Further, the pain/utility/handicap VAS and three other outcome measures were reported in 12.5% (n = 2/16) studies each, and finally the Hidradenitis Suppurativa Severity Index (HSSI) and five other outcome measures were reported in 6.3% (n = 1/15) studies each.
Secukinumab
In 105 patients who received secukinumab, available data showed that 56.3% (n = 27/48) of patients were Hurley Stage 3, 37.5% (n = 18/48) Hurley Stage 2, 4.2% (n = 2/48) Hurley Stage 1, and 2.1% (n = 1/48) Refined Hurley Stage 2C. Overall, 57.1% (n = 60/105) of patients were responders in a mean response period of 16.2 weeks (Supplemental File 8) and 42.9% (n = 45/105) were non-responders. Overall, 69 adverse events occurred in 34.3% (n = 36/105) of patients. Most commonly, adverse events included gastrointestinal upset (n = 13), self-resolving upper respiratory tract infections (n = 8), nasopharyngitis (n = 7), cutaneous effects (n = 6), back pain (n = 5) and headaches (n = 4). Concomitant medications were reported in only 2.9% (n = 3/105) of patients. These included intralesional triamcinolone; dapsone; or hydroxychloroquine, oral steroids, acetylsalicylic acid and pregabalin in 33.3% (n = 1/3 each).
Brodalumab
Of the 22 patients who received brodalumab, available data showed that 25.0% (n = 3/12) were Hurley Stage 3% and 75.0% (n = 9/12) Hurley Stage 2. Overall, 100.0% (n = 22/22) of patients were responders in a mean response period of 4.4 weeks. Overall, one patient (4.5%) experienced upper respiratory tract infection after receiving brodalumab. Additionally, concomitant medications were reported in 0.0% (n = 0/22) of patients.
Ixekizumab
Finally, one patient with Hurley Stage 2 disease was treated with ixekizumab, was classified as a responder in 10 weeks of treatment without adverse events. No concomitant medications were reported.
Discussion
This systematic review comprehensively summarizes treatment outcomes of IL-17 inhibitors in patients with HS. In total, 16 studies, representing 128 patients, were included. Approximately two-thirds of patients with HS treated with IL-17 inhibitors were classified as responders (64.8%, n = 83/128), suggesting that the IL-17 mechanistic pathway is an effective therapeutic target in HS. Additionally, findings highlight the heterogeneity of HS outcome measures in the literature.
While the exact pathogenesis of HS is unknown, the IL-17 pathway has been demonstrated to play a role in the associated immunopathological process. 1 In normal functioning skin, IL-17 is important for barrier function by inducing inflammatory cytokines, chemokines, antimicrobial peptides and metalloproteinases. 29 However, dysregulation promotes inflammatory processes by which IL-17 activates keratinocytes promoting a feedforward loop. 30 In HS patients, elevated levels of IL-17A/C/F transcripts in lesional tissue, IL-17A in serum, and CD4+ IL-17 + T cells in lesional and perilesional tissue have all been reported. 6,8,9,31 -35 Additionally, Th17 cells, characterised by production of IL-17, 36 have been found to be rich within HS lesional tissue, 8 and mesenchymal stem cells to secrete increased levels of IL-17A. 37 IL-17A and IL-17F work synergistically with other cytokines to increase inflammatory cytokine and chemokines. 38 IL-17C, upon stimulation by IL17A and IL-17F elevates the production of inflammatory cytokines, including IL-17A and IL-17F by Th17 cells, creating an inflammatory feedback loop. 30,39,40 Further, IL-17 induces the proteins LL37/cathelicidin, S100A7, S100A8, and S100A9 which are involved in cytokine and chemokine expression and keratinocyte proliferation, also found in HS patient lesional tissue and serum. 41 -43 Interestingly, IL-17 overexpression has not only been found in lesional and perilesional tissue in HS patients, but also in unaffected skin, suggesting subclinical inflammatory processes occur prior to development of active lesional tissue. 41
IL-17 inflammatory effects can be inhibited by therapeutic binding to IL-17RA, IL-17A, or IL-17F. 30 Specifically, secukinumab and ixekizumab have been shown to bind to IL-17A, inhibiting part of the cytokine and inflammatory cascade, 13 however, still allowing IL-17F to signal the receptor. 30 Our results support these mechanistic pathways with 57.5% (n = 61/106) of patients classified as responders. Brodalumab binds to the IL-17RA, blocking a wider range of IL-17 isoforms including IL-17A, IL-17C, and IL-17F. 26,30 One hundred percent (n = 22/22) of patients were responders to brodalumab in our analysis, supporting its wider range of involvement in the IL-17 pathway. With 64.8% (n = 83/128) of patients responding to IL-17 therapies, the clinical response observed in our review further substantiates the role of IL-17 in HS pathogenesis and the need to explore this target. Clinical trials assessing the role of IL-1, IL-17, IL-23, IL-12/23, IL-36, and complement C5a blockade, along with JAK inhibitors, are under way and their results are likely to provide greater insight into both clinical and mechanistic aspects of HS. 30
Heterogeneity of reported outcome measures must be discussed, as comparing treatment outcomes requires consistent and valid outcome measures. 44 The current gold standard outcome measure for assessment of therapeutic options for HS is the HiSCR, 44 defined as a reduction in inflammatory lesion count (≥50%), which includes abscesses and inflammatory nodules, and no increase in abscesses or draining fistulas. 45 The HiSCR has been validated for assessment of treatment efficacy, 45 however, the HiSCR does not take into account draining fistulae in advanced disease, may account for high placebo response rates in evaluating therapeutic options, has poor inter-rater reliability, and may be effected by patient characteristics including Body Mass Index , smoking status, antibiotic use, and draining tunnels. 44 -46
After the introduction of the HiSCR, several other outcome measures such as the HS-PGA and the IHS4 have been developed. In fact, there are 30 or more outcome measures for HS, 47 however, their validation is incomplete or of low quality; 48 data regarding comparison of outcome measures within a common cohort is lacking; 44 and many lack data regarding minimal clinically important differences, 49 leading to variation in their use. One study which assessed inter-rater reliability of HS outcome measures found that the HS-PGA had moderate inter-rater reliability and the IHS4 had fair inter-rater reliability. 48 For the MSS and HSSI, intervals for limits of agreement were very wide relative to scale ranges. 48 Heterogeneity of outcome measures provides the potential for selective reporting leading to outcome reporting bias, 47 limits evidence synthesis and potentially impacts the quality of our results.
Additionally, disease processes in Hurley Stage 2 versus Stage 3 disease must be considered. Different treatment outcomes between Hurley Stage 2 and 3 have been reported for adalimumab, highlighting the potential for pathogenic heterogeneity between stages to be implicated in variable responses to therapeutics. 46,50,51 It is suggested that this may be due to TNF-α being the dominant therapeutic target in Hurley Stage 1 and 2, with Th17 pathways emerging with increasing severity of disease. 52 However, current data suggesting different response between stages presents sample sizes too small for statistical significance. 46 Unfortunately, data is lacking in included studies on how many patients of each Hurley Stage were responders. Thus, our results are unable to provide insight into the potential for pathogenic heterogeneity between stages to be implicated in variable responses to IL-17 inhibitors. We encourage future studies to report how many patients of each stage are responders.
As discussed above, a major limitation of this review includes heterogeneity of included outcome measures, potentially leading to outcome reporting bias, limiting evidence synthesis, and impacting the quality of our results. Additional limitations of this review include small samples sizes, which may limit the generalizability of our findings; and publication bias, with positive findings being more frequently published, potentially inflating the improvement shown in our results.
Despite these limitations, this review provides important clinical conclusions. First, IL-17 inhibitors may serve as an effective therapeutic target in approximately two-thirds of patients with HS and could be considered in those who are refractory to other treatment modalities. Additionally, as only 2.3% (n = 3/128) of patients received concomitant treatment, we believe reported outcomes can be attributed to IL-17 inhibition. Our findings also stress the importance of consistent outcome measures to enhance evidence synthesis, decrease reporting bias, provide potential for future meta-analysis, and ultimately provide patients with the most effective treatments. Further large-scale trials are needed to confirm our findings.
Supplemental Material
Online supplementary file 1 - Supplemental material for Treatment Outcomes of IL-17 Inhibitors in Hidradenitis Suppurativa: A Systematic Review
Supplemental material, Online supplementary file 1, for Treatment Outcomes of IL-17 Inhibitors in Hidradenitis Suppurativa: A Systematic Review by Nadia Kashetsky, Asfandyar Mufti, Shaikhah Alabdulrazzaq, Yuliya Lytvyn, Muskaan Sachdeva, Abdullah Rahat and Jensen Yeung in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Jensen Yeung has been a speaker, consultant, and investigator for AbbVie, Allergan, Amgen, Astellas, Boehringer Ingelheim, Celgene, Centocor, Coherus, Dermira, Eli Lilly, Forward, Galderma, GSK, Janssen, Leo, Medimmune, Merck, Novartis, Pfizer, Regeneron, Roche, Sanofi Genzyme, Takeda, UCB, Valeant, and Xenon. Ms. Kashetsky, Dr. Mufti, Dr. Alabdulrazzaq, Mrs. Lytvyn, Ms. Sachdeva, and Mr. Rahat have nothing to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
