Abstract
Microfluidic systems offer versatile biomedical tools and methods to enhance human convenience and health. Advances in these systems enables next-generation microfluidics that integrates automation, manipulation, and smart readout systems, as well as design and three-dimensional (3D) printing for precise production of microchannels and other microstructures rapidly and with great flexibility. These 3D-printed microfluidic platforms not only control the complex fluid behavior for various biomedical applications, but also serve as microconduits for building 3D tissue constructs—an integral component of advanced drug development, toxicity assessment, and accurate disease modeling. Furthermore, the integration of other emerging technologies, such as advanced microscopy and robotics, enables the spatiotemporal manipulation and high-throughput screening of cell physiology within precisely controlled microenvironments. Notably, the portability and high precision automation capabilities in these integrated systems facilitate rapid experimentation and data acquisition to help deepen our understanding of complex biological systems and their behaviors. While certain challenges, including material compatibility, scaling, and standardization still exist, the integration with artificial intelligence, the Internet of Things, smart materials, and miniaturization holds tremendous promise in reshaping traditional microfluidic approaches. This transformative potential, when integrated with advanced technologies, has the potential to revolutionize biomedical research and healthcare applications, ultimately benefiting human health. This review highlights the advances in the field and emphasizes the critical role of the next generation microfluidic systems in advancing biomedical research, point-of-care diagnostics, and healthcare systems.
Keywords
Introduction
Advancing biomedical research often depends on the use of modern problem-solving technologies that offer versatile tools and methods to enhance human convenience and health. At the forefront of these technologies is microfluidics 1 —an extraordinary science and enabling technology that precisely controls fluid behavior at micro-, nano-, and pico-scales.
In the past 3 decades, the technological and scientific advances in microfluidics led to the development of lab-on-a-chip 2 and organ-on-a-chip 3 devices. These devices comprise closed microfluidic channels, chambers, and components for various small-scale laboratory methods and analysis. For example, portable microfluidic devices can quantitatively detect nucleic acids in blood 4 or glucose levels in plasma and tears. 5 This holds the potential for affordable point-of-care diagnostics, which is a critical component of precision medicine. Biomimetic microfluidic devices can replicate lung functions 6 or cardiac tissue models 7 on a chip. This holds the promise for drug testing, understanding disease mechanisms, and identifying potential therapeutics using physiologically relevant 3D organ-on-a-chip models. Such models are increasingly important in personalized medicine.
Further advances expanded the capabilities of microfluidic systems, resulting in the development of lab-on-a-paper 8 platforms. These platforms utilize capillary action to passively control fluids within a single sheet of paper for rapid “sample-to-diagnose” approaches. For example, paper microfluidic assays are proven effective in detecting severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) infections when integrated with recombinase polymerase amplification 9 or with bioconjugated gold nanoparticles. 10 These assays showcase their multiplex gene and antibody detection capabilities, respectively. Additionally, they also offer rapid and semiquantitative measurements of liver enzymes 11 or glucose detection 12 in a fingerstick blood specimen. This functionality makes them accessible for monitoring drug-induced liver injury and diabetics in resource-limited settings, respectively.
More recently, a significant advancement in microfluidics was the development of lab-on-a-tip 13 devices that integrate and execute a multitude of cell-related applications directly into the tip of an open (non-contact) microfluidic probe. These “channel-less” microfluidic systems combine a bottom glass substrate with a top microfluidic delivery system. Moreover, by utilizing the capabilities of three-dimensional (3D) printing, they offer flexibility in design, efficiency, and versatility. For example, a herringbone microfluidic probe (HB-MFP) 14 can efficiently capture rare circulating tumor cells (CTCs), including clusters, from prostate cancer blood samples onto bioactivated glass substrates. Through affinity-based multiplexing, the HB-MFP offers insights into cellular phenotypes. In a related field, a non-contact multiphysics probe (NMP) 15 can operate within the physiological tissue environment to precisely manipulate single cells. Through controlled manipulation, electropermeabilization, transfection, and cytoplasm extraction from living single cells, the NMP enhances spatiotemporal single-cell analysis within tissue samples.
As a result, next generation microfluidic systems, equipped with advanced automation and screening capabilities, are demonstrating their potential in a wide range of biomedical research applications. These applications include, but are not limited to, the manipulation and patterning of single cells, multiplex characterization of cell phenotypes, precise cell sorting, diverse bioassays and biosensing, as well as contributions to tissue engineering, drug efficacy testing, and disease modeling.
Next Generation Microfluidic Systems: Trends
At the core of microfluidics lies the precise manipulation of fluids at the microscale, which is primarily facilitated by the laminar fluid flow. 16 Traditionally, microfluidic devices consist of closed microconduits designed for specific applications (see Figure 1). However, the field is currently undergoing dynamic shift toward innovative technological advances. For example, nowadays digital microfluidics 17 challenges standard microchannels by providing control over fluids through discrete droplets. Moreover, the integration of microfluidics with electronics, 18 photonics, 19 plasmonic, 20 magnetics, 21 nanomaterials,22,23 and advanced microscopy and spectroscopy techniques 24 enhances the accuracy in fluid manipulation, improves the precision in sensing and characterizing biological samples, and increases the overall automation and screening efficiency.
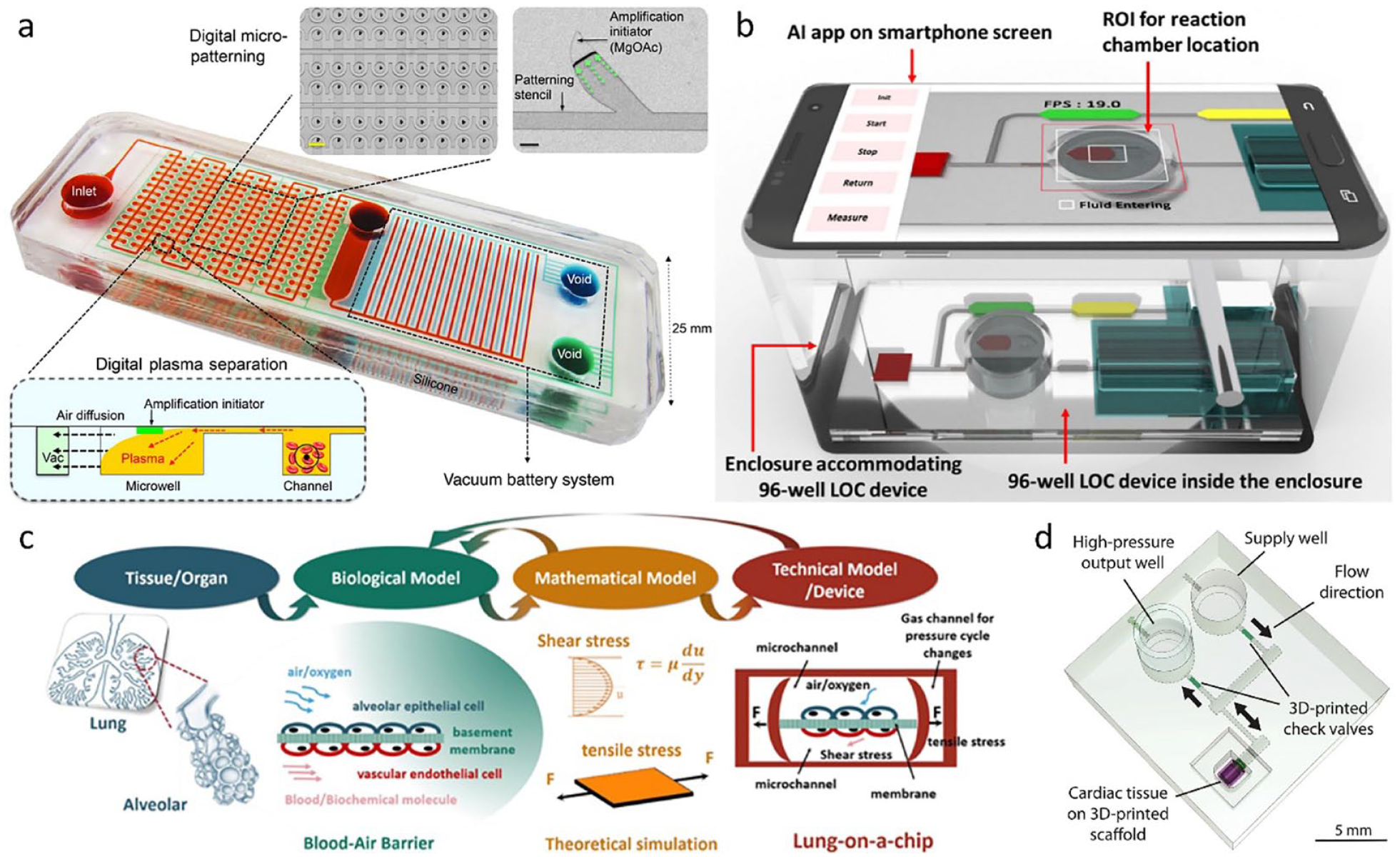
Lab-on-a-chip and organ-on-a-chip devices: (a) integrated microfluidic chip for low-cost, quantitative, and portable nucleic acid testing. (b) Microfluidic platform for immunodiagnostics integrated with artificial intelligence (AI) technology. Microfluidic systems for developing advanced models of (c) lung and (d) cardiac tissues. Reproduced from Yeh et al, 4 with permission from American Association for the Advancement of Science (a), from Bhuiyan et al, 5 with permission from American Chemical Society (b), from Li et al, 6 with permission from American Institute of Physics Publishing (c), and from Michas et al, 7 with permission from American Association for the Advancement of Science (d).
Notably, when microfluidics is integrated with wearable devices, 25 it facilitates non- to minimally invasive health and illness measurements on individuals. Whereas, when it is integrated with multifilament threads, 26 it enables the development of intelligent fabrics that continuously monitor human physiological condition in real time. Paper microfluidics 27 represents another important branch of microfluidics with applications in point-of-care diagnostics for disease detection, biomarker monitoring, and blood testing (see Figure 2). In recent years, paper-based microfluidic devices advanced into a new dimension by incorporating the art of origami (ie, paper folding) to transform from 2D microfluidics to 3D microfluidics. This innovative technology utilizes capillary action to passively “guide” 3D fluid flow on porous paper, thus eliminating the need for valves or external pumps. For example, Liu and Crooks 28 fabricated an entire 3D device using a flat sheet of paper in a single photolithographic step through origami approach. The device compatibility for chemical analysis was demonstrated in fluorescent and colorimetric assays. Zhu et al 29 developed 3D origami paper-based ratio-metric fluorescent microfluidic device for visual point-of-care detection of alkaline phosphatase.

Lab-on-a-paper devices: wearable (a) electrochemical microfluidic device and (b) antibody test assay for viral infection detection and severity monitoring, respectively. Point-of-care fingerstick devices for (c) liver function and (d) glucose level testing in blood. Reproduced from Kim et al, 9 with permission from Elsevier (a), from Boumar et al, 10 with permission from Springer Nature (b), from Pollock et al, 11 with permission from American Association for the Advancement of Science (c), and from Luo et al, 12 with permission from MDPI (d).
Microfluidic probes, 13 on the other hand, further expand the boundaries of microfluidics through innovative “wall-free” control of fluid, which makes them suitable for multiplexing. For example, Dorrigiv et al30,31 presented computer-controlled drug screening platform that utilizes pixelated MFPs to enable multiplexed delivery of multiple biochemical reagents—up to 9 distinct biochemical reagents—to 3D tumor models. This innovation significantly increased the throughput of traditional MFPs, enabling multiple testing scenarios in each experiment. Conversely, Brimmo et al 32 experimented with various aperture configurations to bio-fabricate selective multicellular patterns on an open substrate without the use of confined conduits.
In recent years, microfluidics influenced various aspects of life, from applications in smartphones to outer space. In space, microfluidics requires special forces beyond gravitational forces to function effectively. These forces include capillary flow, 33 thermocapillary (Marangoni) forces, 34 and electrolytic gas evolution.34,35 To date, various types of electrospray thrusters, 36 based on capillary and porous surface wetting emitters, successfully demonstrated functionality both on Earth and in space, among other innovative applications.37-39 These examples highlight the significant role of microfluidics in space exploration. Furthermore, microfluidic chips are developed to track the physiological indicators of astronauts, 40 identify diseases, 41 and monitor living conditions including air, water, and food quality. 42 Additionally, the Phorm iPad mini case is a real-life example of using microfluidics to create pop-up guiding bubble buttons for providing a tactile interface on touch-screen devices. 43 This innovation is particularly valuable for individuals who may have difficulty with operating touchscreens, such as blind, visually impaired, elderly, and individuals with diseases that affect fine motor skills, including arthritis and Parkinson’s disease.
Modern digital electronics typically rely on rigid semiconductor materials, which limit their physical configurability. This constraint makes flexible and bendable electronics desirable in several applications, including sensing and actuation. In this regard, microfluidics provides the necessary flexibility needed for various applications that are not achievable with rigid semiconductors. 44 For example, in the work by Cheng and Wu, 45 a deformable radiation sensor was achieved by fabricating its radio frequency antenna through soft lithography patterning of the microfluidic channel grid onto polydimethylsiloxane (PDMS), a silicone polymer. Room-temperature conductive liquid alloy, Galinstan, is then used to fill the channels to allow for stretchability and bending. 41 The same microfluidic fabrication technique was employed by Othman et al 46 to fabricate a multi-modal microfluidic bend sensor for robotic sensing applications. These microfluidic smart sensors are proven to provide valuable insights into organ stiffness during minimally invasive surgeries, as demonstrated in the preceding works by the same authors.46,47 In addition to mentioned applications, microfluidic devices provide high performance in various applications, including heat sinks to cool integrated circuits, 48 simplified solutions in agriculture to study enzyme reactions and plant responses,49,50 and in wearable devices to monitor body movements 51 or mimic human skin. 52
Materials, Fabrication, 3D Printing, and Commercialization
Characteristics of the used materials in manufacturing microfluidic devices significantly impact the fabrication cost, performance, and versatility of their functions. Traditional microfluidic devices commonly employ the PDMS due to its ease of production, biocompatibility, and optical transparency. Their fabrication is commonly based on soft lithography, 53 where master molds with microfluidic channels are initially created, followed by their replication using casting. This technique offers precise control over channel geometries and other microstructures, but is labor-intensive and time-consuming. 54 In contrast, paper microfluidic platforms employ hydrophilic cellulose- or nitrocellulose-based paper substrates due to their intrinsic capillary action characteristic. 55 This enables the passive flow of fluid within the porous 3D microenvironment of the paper, which is guided through paths fabricated via chemical modifications, wax printing, or other methods. Microfluidic probes, on the other hand, are classically fabricated by standard photo- and stereo-lithography approaches in silicon and glass substrates, but more recently, researchers started employing ultraviolet (UV)-curable, biocompatible, and transparent resins used in stereolithographic 3D printing. 56 Their fabrication based on 3D printing emerged as an innovative approach for creating customizable device designs that otherwise would be time consuming using the conventional photolithography methods.
In the last decade, the commercialization of microfluidic devices witnessed substantial growth.57,58 Standard microfluidic chips are now widely available in the market, where they provide individuals and healthcare personnel with point-of-care solutions. In particular, paper microfluidic platforms, similar to pregnancy tests, are becoming preferred tools for diagnosis of infectious diseases in resource-limited settings due to their simplicity in function, flexibility in design, portability, and low cost.
After over 3 decades of development, a new concept in microfluidics, the modular microfluidics, 59 has emerged. This innovative concept was made possible by the consistency and precision of the injection-molded bricks, such as those from the famous brand LEGO, which fit together perfectly regardless of where they are manufactured in the world. Consequently, modularity has infinite potential that eliminates the borders of creativity. For example, these bricks inspired MIT researchers to use them as the foundation for a new modular microfluidic design that enabled them to form modular microfluidic devices capable of conducting biological operations such as cell sorting, fluid mixing, and molecule filtering. 60
Applications in Biomedical Research
A key feature of microfluidics is the ability to integrate various biology, engineering, computation, chemistry, and physics related functions into a single device. In single-cell analysis, microfluidic devices enable isolation and phenotyping14,61,62 and sorting and patterning15,32,63 of rare cells, such as circulating tumor or plasma cells. These capabilities are instrumental in unraveling cellular heterogeneity and identifying biomarkers (Figure 3). In bioassays, microfluidic devices achieve high sensitivity and rapid quantification through the utilization of different phenomena and methods, such as isotachophoresis, 64 a promising approach for protein extraction, purification, and quantification.

Lab-on-a-tip devices: Non-contact microfluidic probe technologies for (a) affinity-based multiplex isolation of circulating tumor cells and clusters, and (b) spatiotemporal resolved single cell manipulation and analysis. Reproduced from Deliorman et al, 65 with permission from Springer Nature (a), and from Brimmo et al, 15 with permission from Wiley (b).
Microfluidic devices also play a pivotal role in biosensing applications by exhibiting remarkable signal amplification capabilities. 66 In point-of-care diagnostics, on the other hand, microfluidic devices enable rapid detection of diseases and infections. 67 This expedites patient care and relieves the testing burden in laboratories, especially during unprecedented pandemic times such as the Coronavirus Disease 2019 (COVID-19).68,69 The environmental implications of microfluidic technology are equally promising. Monitoring water and food quality, air pollutants, and soil contaminants is achievable with improved precision. 70
Paper microfluidics is a good example of recent innovations in microfluidics. Given the advantages, such as capillary-driven fluid flow and portability, paper microfluidic platforms enable point-of-care diagnostics to be efficiently conducted with minimal sample volumes and shorter analysis times. Additionally, textile microfluidics, which is often integrated into wearable devices, extends the reach of point-of-care diagnostics. These “smart” textiles incorporate microfluidic channels that collect and analyze bodily fluids such as sweat, 71 urine, 72 and blood. 73 In the process, the automation through microfluidics ensures seamless data collection, while the integration into clothing offers non-invasive and continuous health monitoring in real time.
Microfluidics is rapidly integrating innovative technologies, including automation and robotics, and the Internet of Things (IoT) (Figure 4). In terms of automation, labor-intensive and time-consuming experimental tasks, such as sample preparation, mixing, and detection, can now be performed in parallel, thus reducing human error and enabling high-throughput screening and analysis. For example, in drug efficacy testing, the automation in microfluidic devices allows for a large library of compounds to be screened quickly and efficiently. 74 Furthermore, the precise control of biomolecular concentration gradients in these devices enables investigation of cellular responses and behaviors including patterning, differentiation, migration, and the development of drug resistance.75,76 For example, microfluidics enables the production of plasmonic nanomaterials through aqueous colloidal synthesis within a structured composite foam lattice inside a straightforward microfluidic device. 77 This approach allows for precise control over reagent dispensing and mixing, while the organized foam structure facilitates compartmentalized nanoparticle growth. Importantly, this production method operates continuously, possesses inherent digital control, is scalable, and operates fully automated.

Microfluidic platforms integrated with Internet of Things (IoT) for healthcare applications: (Left) Paper-based electrochemical wearable sensors integrated with microfluidics and nanotechnology for combined sampling and data analysis. Reproduced from Deroco et al, 78 with permission from Wiley. (Right) A wearable patch, incorporating fluorescent probes on paper substrates and cotton thread microfluidics, is fabricated and paired with a smartphone-based fluorescence imaging module, a self-developed smartphone app, and an IoT-based model to enable multi-sensing of sweat biomarkers, including glucose, lactate, chloride, pH, and volume. Reproduced from Ardalan et al, 79 with permission from Elsevier.
In the context of the IoT, the integration of microfluidics with data networks results in “smart” devices that can transmit real-time information to remote locations for monitoring and in-depth analysis of the measurements.78,79 Microfluidic devices, with their ability to precisely manipulate small volumes of fluids and perform complex chemical and biological assays, are well-suited for creating intelligent and responsive systems. By combining microfluidics with artificial intelligence (AI), these devices can take on advanced functionalities.80,81
For example, microfluidic sensors can collect substantial data from various biological and chemical processes. AI algorithms can then be employed to analyze this data, identify patterns, and make predictions or decisions based on the collected information. This integration enables microfluidic systems to autonomously adapt, optimize processes, and respond to changing conditions using cloud-based analysis and remote monitoring. 82 As a result, the connectivity and analytical capability hold potential in fields such as healthcare, environmental monitoring, and diagnostics, where real-time insights and rapid decision-making are crucial. 83 For example, microfluidic devices integrated with environmental sensors can continuously monitor water quality and transmit data for real-time analysis. 84
In the context of the dynamic platforms, the integration of microfluidics with robotics creates platforms capable of executing even more advanced research tasks. 85 For example, a robotic arm integrated with an acoustofluidic device as its end effector induces oscillations within a capillary to create controlled flow patterns or microstreaming when placed in a liquid. 86 Moreover, self-guided movement of microrobots in fluids allows their autonomous navigation capabilities in complex environments such as blood vessels. 87
Tissue Engineering and Human-on-a-Chip
Tissue engineering merges biological cells, natural biomaterials, and dynamic biochemical and biophysical factors with microfluidic technologies to form 3D tissue constructs for drug efficacy testing, 88 disease modeling, 89 and other fundamental research.90,91 The formation of the tissue constructs relies on the growth and differentiation of various cell types, such as parenchymal and mesenchymal fibroblasts, to mimic cell-cell and cell-environment interactions within natural tissues.92,93 During this process, biomaterials provide structural integrity and mechanical support for the cells. These 3D scaffolds mimic the extracellular matrix of the target tissue for facilitating the 3D cell adhesion, migration, and organization. 94 Cellular behavior and tissue development, on the other hand, are regulated by both biochemical factors, such as growth factors, cytokines, and signaling molecules, 95 and mechanical factors, such as tissue stiffness. 96 These factors are delivered spatiotemporally to induce cellular proliferation, differentiation, and morphogenesis. 97
Merging these 3 components (ie, cells, biomaterials, and biochemical/mechanical factors) is crucial for fabricating 3D tissue constructs that mimic real organs. 98 Organ-on-a-chip devices, for this matter, are becoming increasingly successful in providing the necessary in vitro microenvironment for cells to interact with the biomaterials and get exposed to the biochemical and mechanical factors during their growth and differentiation. Nowadays, the developed organ-on-a-chip models offer accurate representation of human organs, and provide spatiotemporal organ-specific responses to drugs and diseases. 99 Human-on-a-chip systems, on the other hand, connects multiple organ-on-a-chip units (Figure 5). This integration aims to mimic how various organs influence each other and how drugs or diseases impact the entire body. 100 Importantly, this integration also has transformative implications for pre-clinical testing. Traditionally, animal testing was a necessary step prior to clinical trials. However, with the advent of the organ-on-a-chip platforms, these pre-clinical tests are increasingly replacing animals with microfluidic chips that closely replicate the responses of human body to drugs. 101 This transition not only saves animal lives but also yields outcomes that are more relevant to clinical results. As a result, it enhances both the efficiency and ethical considerations of the drug development processes.
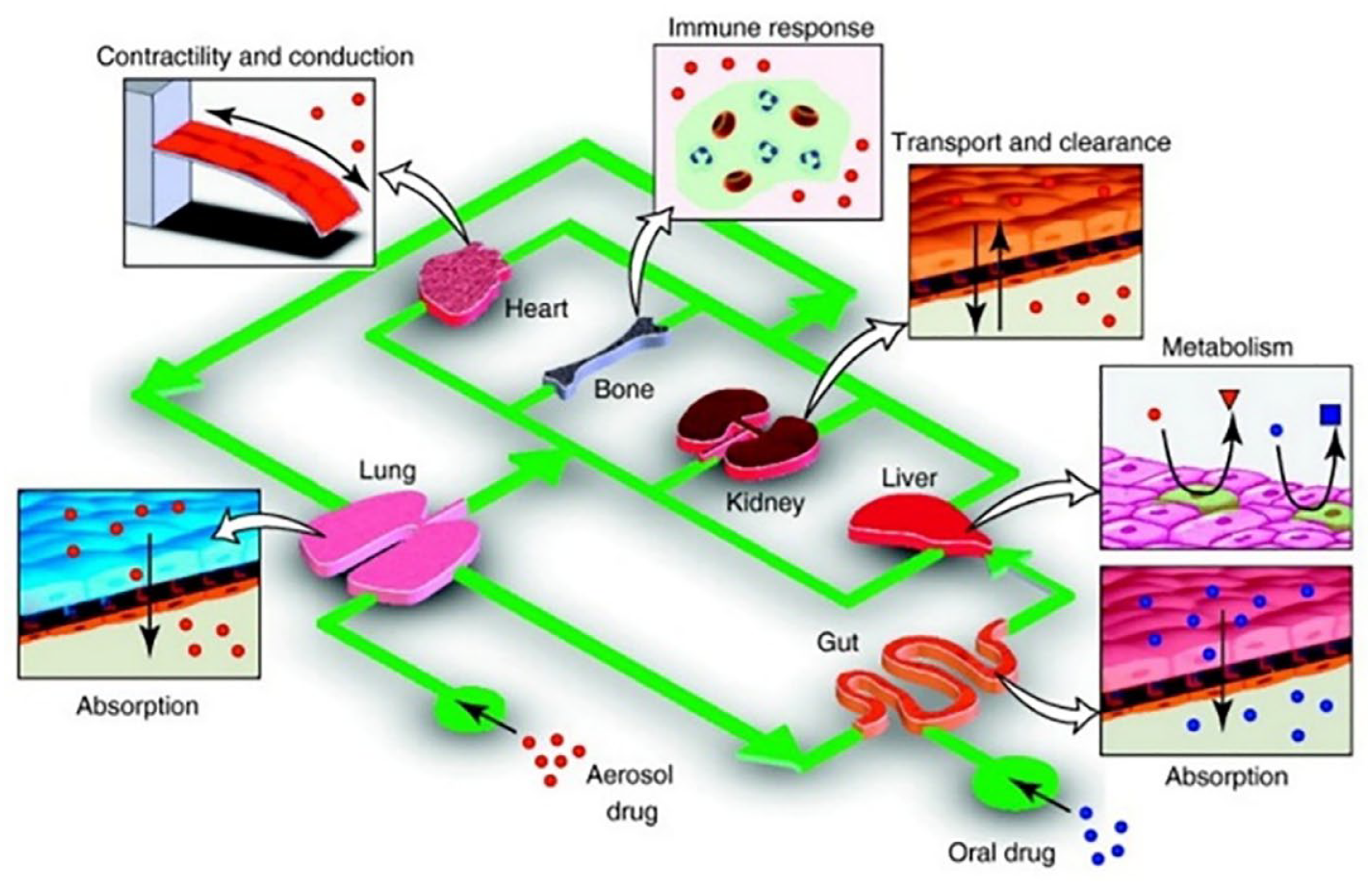
Human-on-a-chip system: The Human-on-a-chip system connects different organs within a single device through a microfluidic circulatory system. By integrating organ models like the lung, heart, gut, liver, kidney, and bone, the 4 key drug processes such as absorption, distribution, metabolism, and excretion can be simulated effectively. Reproduced from Huh et al, 100 with permission from Elsevier.
Current Challenges and Future Directions
The miniaturization of laboratory procedures and essays demands interdisciplinary collaboration between engineers, biologists, chemists, and medical experts to develop the full potential of lab-on-a-chip, organ-on-a-chip, lab-on-a-paper, and lab-on-tip devices. Whereas accurate and reproducible control of fluid flow at microscales requires detailed design, computation, fabrication, optimization, and biological testing. Particularly, the integration of conventional 3D printing with microfluidics minimizes the microfabrication workflow and enables the creation of complex designs that were previously challenging to achieve.
While the technological and scientific advances in microfluidics are exciting, several challenges still persist. One of these challenges is the physical adsorption of hydrophobic and other small molecules in PDMS microchannels that leads to altered composition of solutions and inconsistent results. 102 Another challenge is the micro-to-macro scalability. 103 The complexities of organs consisting of a number of cells are not accurately reflected by relatively small number of experimental cells. Importantly, the use of a common (standardized) medium in multiorgan-on-a-chip platforms poses challenges for the growth and differentiation of cells in certain organ tissues. 104 In addition to these challenges, shortcoming of unified standards for various aspects of microfluidics poses a significant hurdle. The International Organization for Standardization (ISO) is the global entity responsible for international standardization efforts. In this regard, microfluidics requires standards for dimensions, connections, and initial device classification due to its diverse applications, numerous technologies, and wide array of materials. 105 This diversity becomes problematic when attempting to integrate microfluidic components. Moreover, there is a pressing need for standardized testing, which may be application-specific. Some of these tests include leakage testing, 106 pressure testing, 107 and flow throughput testing. 108 Developing these test protocols must take into consideration the material of the chips as well as the temperature and pressure ranges of operation. To expedite market access, researchers have identified specific application classes with defined temperature and pressure ranges, which provide boundary conditions for the development of tests. 109 Standardized specifications for microfluidic devices include standardizing chip thickness, outer dimensions for compatibility with microplates and microscope slides, as well as port pitch and diameters. 110
The integration of microfluidics with innovative technologies holds substantial promise in overcoming many challenges in the field. For example, the 3D printing can facilitate rapid prototyping toward commercialization. 111 Advanced sensing technologies can enhance the capabilities of microfluidic systems.112,113 Whereas, advanced wearable sensors can provide real-time and precise measurements of various parameters, ranging from molecular concentrations to cellular responses. 114 Notably, droplet microfluidics can accelerate drug discovery workflow by coupling with high-throughput paper-based 3D cell culture and spheroids.115,116 This integration can expedite the assessment of drug candidates through fast compound screening, and significantly reduce reliance on animal testing by utilizing patient-specific cells.
In parallel, AI algorithms can optimize experimental protocols, automate data analysis, and predict outcomes based on large datasets, thus accelerating biological testing with improved precision in data acquisition. 81 Moreover, the use of smart materials with stimuli-responsive properties can improve the functionality and reliability of microfluidic devices. 117 Noteworthy, miniaturization can offer more accurate representations of organ function at the microscale, thus accelerating drug development, toxicity assessment, and disease modeling. 118
In the last decade, the integration of smartphones and microfluidic devices opened up new possibilities. 119 This integration enabled the utilization of microfluidic detection modules that rely on complementary metal oxide semiconductor cameras of the smartphones.120-122 More than ever, they found applications in medical and public health practice where they serve as point-of-care-diagnostic tools. 123 For example, one notable integration is the work by Schaumburg et al, 124 where a free platform called “Appuente” was developed. This platform seamlessly combines microfluidic chips, smartphones, and cloud access. It includes a mobile application for end users that provides several functions, such as chip identification and tracking, guidance and control, data processing, smart imaging, and result reporting.
In tissue engineering, the goal is to recreate the microenvironment of natural tissues to facilitate proper cell growth, differentiation, and function. This involves replicating the biochemical and biophysical factors that cells experience in their physiological environment. In this regard, organ-on-a-chip devices successfully generate human organs in microfluidic devices, which expedite drug testing and disease modeling. In the near future, multiple organ-on-a-chip models are envisioned to be interconnected within a human-on-a-chip system. When combined with automation, robotics, and disease modeling, human-on-a-chip models can provide personalized detailed view on how drugs affect the entire body of a human. 125 Excitingly, a radical step was recently taken by the U.S. Food and Drug Administration toward ending the use of animal test subjects in pharmaceutical research. 126 This game-changing decision can open doors for microfluidic systems to provide various complex in vitro multiorgan models for “real-life” drug development, toxicity assessment, and disease modeling as alternatives to animal testing.
Conclusion
Next generation microfluidic systems stand at the forefront of transformative technologies, bridging automation, screening, and manipulation with biomedical research. Presently, challenges still exist such as material compatibility, scaling, and standardization across multiple systems and procedures. However, the integration of AI, smart materials, and miniaturization is a promising approach to redefine the conventional approaches, and paves the way for creating transformation in biomedical research and applications that will revolutionize human health. Whereas the integration with 3D printing, advanced sensing, robotics, and clinical methods holds the key to diverse applications ranging from fundamental research to healthcare solutions.
Footnotes
Acknowledgements
D.S.A. acknowledges the NYU Global PhD fellowship. M.A.Q. acknowledges the financial support from NYU Abu Dhabi.
Author Contributions
M.D. and M.A.Q.: Study conception and design. M.D. and D.S.A.: Manuscript preparation including literature review, analysis, and writing. M.A.Q.: Supervision of the work and revising the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by NYU Abu Dhabi, UAE.
