Abstract
Raman spectroscopy is a group of analytical techniques, currently applied in several research fields, including clinical diagnostics. Tissue-mimicking optical phantoms have been established as an essential intermediate stage for medical applications with their employment from spectroscopic techniques to be constantly growing. This review outlines the types of tissue phantoms currently employed in different biomedical applications of Raman spectroscopy, focusing on their composition and optical properties. It is therefore an attempt to present an informed range of options for potential use to the researchers.
Introduction
Raman spectroscopy (RS) is a group of techniques based around the analysis of inelastic (Raman scattered) light resulting from the interaction between light and matter. Raman scattered light provides information on the molecular bond vibrations, which can be exploited for the acquisition of sample-specific molecular fingerprints. Due to its non-invasive nature, Raman spectroscopy has been employed in different modes to address various biomedical issues over the last few decades. Some of them include cancer screening, 1 pathogen identification,2,3 body fluid analysis, 4 transfusion blood assessment, 5 disease diagnosis,6-8 drug analysis, 9 or even as a process analytical tool for cell therapies. 10
The continuous need for tissue-like samples in the standardization and development of spectroscopic and other optical applications combined with the limited availability of human tissue due to ethics reasons, has led to the adoption of tissue-mimicking phantoms or tissue models. The optical tissue phantoms are synthetic models mimicking not only human tissue properties, but also light propagation and interactions through it. This presents a significant quality for spectroscopic techniques, which have recruited tissue phantoms over time for (a) feasibility and reproducibility testing, (b) optimization, and (c)calibration reasons.
Tissue phantoms are essentially diffuse matrices of various composition, geometry, and optical properties. They exist in many different forms but are generally divided into those of solid, hydrogel, and liquid state. Their common feature is that absorption, scattering, and other optical properties can be controllably modeled during fabrication. A different category of tissue phantoms is the ex vivo animal tissue, which is often used in spectroscopy studies for a more realistic approach. Although animal phantoms cannot be easily controlled in terms of optical properties, they provide an inherent heterogeneity in tissue composition which is very similar to the human one.
The flexibility and properties of tissue phantoms have rendered them an important part of technology development and translation process. Over the past few decades, tissue phantoms have been extensively used for ex vivo measurements preceding the implementation of any optical technique in clinical settings. 11 In this review, we attempt to outline most of the tissue phantoms employed for biomedical applications in Raman spectroscopy, focusing on three different aspects: (a) most commonly used tissue phantom types, (b) phantom design, and (c) biomedical applications in the field of Raman spectroscopy involving optical tissue phantoms. We hope that this review will inform researchers on the current applications of synthetic and animal tissue phantoms, facilitating their choice in future research.
Types of Phantoms
The most commonly used phantoms are liquid tissue phantoms, solid silicone or polymer phantoms, gelatin/ agarose phantoms, and animal phantoms (Figure 1). As the different types of phantoms have been extensively described in comprehensive reviews before,12-14 they are only briefly mentioned below.

Liquid tissue phantoms are the easiest and most flexible in fabrication, as they can be easily controlled and managed in terms of volume and consistency. Because of the latter, the optical properties of liquid phantoms can be adjusted just by changing the relative proportions of absorbing and scattering agents in the phantom solution. The liquid phantoms are commonly made of mixing a commercially available lipid emulsion (Intralipid, Liposyn) and added absorber (eg, ink) in a container. The container material may vary depending on the application and the possibility of light reaching the container walls. The liquid nature of the phantoms allows for significant flexibility in sample measurements, such as incorporation of Raman scattering or fluorescence features, even after the fabrication process has been completed. Another major advantage of liquid phantoms over the solid ones is the uncomplicated and rapid synthetic procedure.
On the other hand, solid phantoms can be time-consuming and not flexible in fabrication, as their optical properties cannot easily change by adding or removing components to the matrix. Solid phantom samples have been made using bulk matrices of different degrees of transparency, such as polymers, silicone, and wax. Although polymer-based phantoms have been reported to be sometimes instable in size during polymerization, 16 they are generally considered to be preserved better and for a longer period of time compared to animal tissue or liquid tissue phantoms. A special category of semi-solid tissue phantoms are the hydrogel-based ones, including agarose and gelatin matrices. Both phantom types have been extensively used for imaging 12 and assessed for biocompatibility. 17 Since hydrogel phantoms contain water as their main component, evaporation of the solvent can take place, changing the dimensions and optical properties of the phantom within a short period of time. As such phantoms are also prone to bacterial growth, preservatives may be used for lifetime extension.
Animal phantoms are the most realistic choice as they accurately simulate the heterogeneity, mechanical properties, and consistency of the human tissue. Ex vivo animal tissue is commonly used in Raman spectroscopy not only for their intrinsic scattering and absorbing properties but also for their morphology and chemical composition. In combination with their morphological complexity and presence of autofluorescence induced by chromophores which are also found in human tissue, ex vivo animal tissue can serve as a highly accurate model for light propagation. 18 Despite their benefits, animal tissue phantoms are difficult to be controlled in terms of sample composition for reproducible measurements as their optical properties cannot be adjusted precisely. In addition, reduced tissue absorption is expected in the excised animal tissues, due to decrease in blood volume during removal. However, animal phantoms are useful as an intermediate stage prior to human or in vivo animal studies, as they allow researchers to investigate healthy and diseased tissues in ways that would be inaccessible in a human patient due to ethics.
Phantom Design
Optical properties
While photons propagate through tissues, they are being scattered or absorbed by the tissue molecules. During scattering, which is more commonly taking place at interfaces of tissue components with different refractive indices, light photons interact with matter and change their direction, with (inelastic scattering) or without (elastic or diffuse scattering) losing energy. The more scattering the medium is, the more the photons will deviate from the original forward direction (Figure 2). In the case of absorption, the photons will be absorbed only when the energy of the photon matches the energy gap between the initial and the final energy states of the atom or molecule. How efficiently the photon will travel through the tissue, depends on the fraction of light which will be absorbed or scattered by it, which is described principally by the scattering and absorption coefficients. In biological tissues, absorption and scattering are commonly taking place in the same sample with the effect of absorption to be much stronger than one would expect as a result of the extra optical path length.
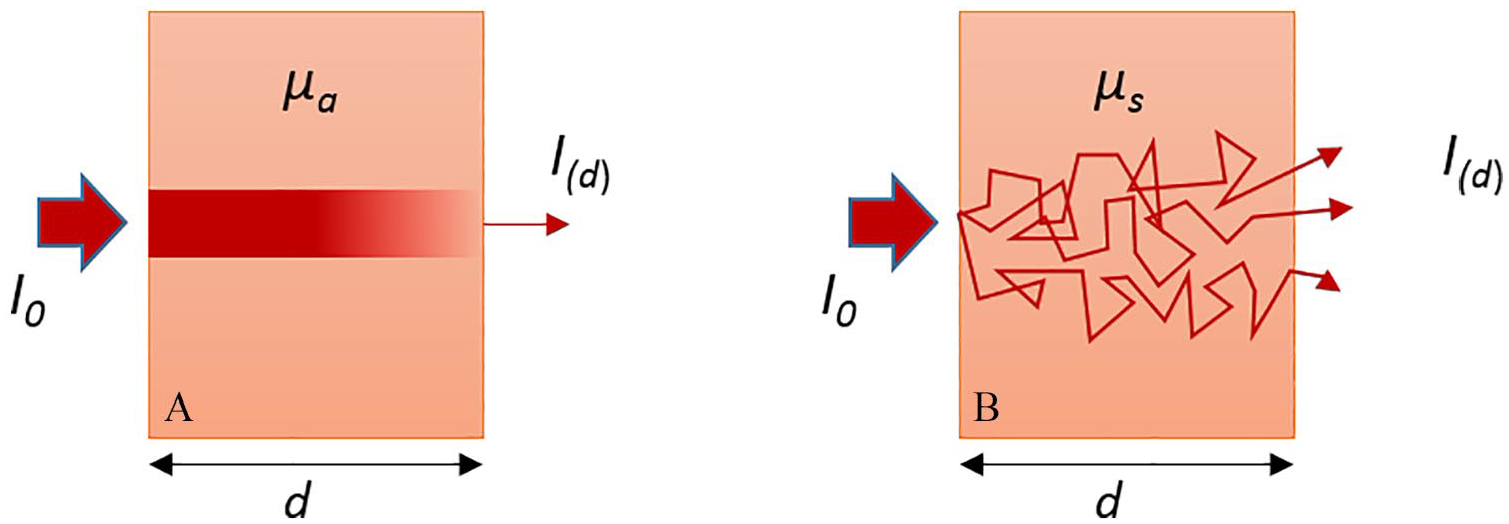
Transmission of light through an absorbing (A) and diffusely scattering (B) medium. 19
The scattering coefficient (μs) depends on the probability of the medium per unit length to redirect the incident photons into new directions and therefore prevent the forward on-axis transmission of light. The average distance that a photon travels between scattering events is called mean free path length. Similarly, the absorption coefficient (μα) describes the probability of the medium per unit length to absorb the photons. 20 Because in biological tissues the light is not scattered in all possible directions but is mainly forward directed (anisotropic scattering),21,22 it is convenient to define a mean scattering angle which is described by the value of anisotropy (g). 23 To be more precise in the description of scattering in biological tissues, scattering coefficient and anisotropy are combined into the expression of reduced scattering coefficient (μ's) which is defined as
The optical properties described above (μα, μs, μ's and g) are characteristic of tissue and vary for different types of human tissue. To appropriately simulate visible or infrared light propagation in a certain tissue, it is necessary to reproduce its optical properties at the measuring wavelength. By replicating optical properties, we simulate co-dependent parameters such as the concentration of various chromophores inside the tissue (absorption coefficient), and the form, size and concentration of the scattering components in the tissue (scattering coefficient). 24
Commonly used scatterers for the fabrication of tissue phantoms include fat emulsions such as Intralipid and Liposyn, milk, TiO2, latex, and polystyrene microspheres. Absorbing media include different types of inks (India ink, black ink, red ink), dyes (blue dye, naphthol green dye, nigrosin, indocyanine green), and biological absorbers (hemoglobin, β-carotene, melanin). The desirable optical properties in the phantom volume can be achieved by mixing the correct proportions of scattering and absorbing agent.
Below we present typical concentrations of scatterer and absorber media, commonly used in tissue phantom fabrication, in correlation with the optical properties of characteristic human tissues (Figure 3). The reduced scattering and absorption coefficients are based on experimental data of healthy human tissue at 830 nm for different skin layers, 25 brain,26,27 breast, 28 cranial bone, 29 liver, 30 muscle, 31 stomach wall mucosa, 32 colon, 33 prostate, 34 skull, 26 and whole blood. 35 Optical properties of the scattering and absorbing media included in the plot were also derived from the literature for nigrosin, 36 TiO2, 37 milk, 38 Liposyn, 39 hemoglobin, 40 indocyanine green (ICG), 39 Intralipid, 41 and India ink. 42 The exact values used in the figure can be found in Supplementary Material (S1).
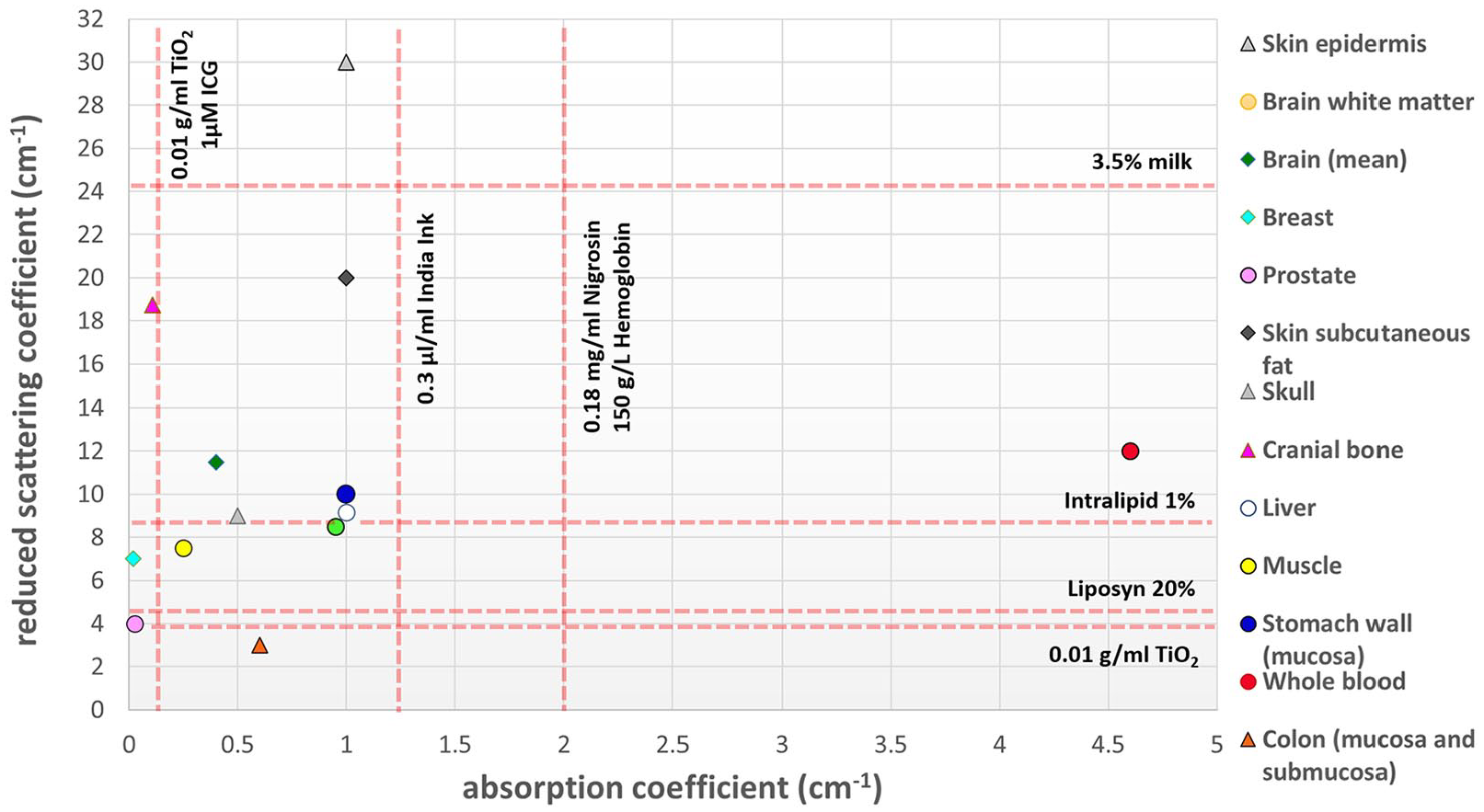
Reduced scattering and absorption coefficients for typical concentrations of scattering and absorbing agents commonly used in tissue phantom fabrication, in comparison with optical properties of different types of healthy human tissues at 830 nm.
Geometry and chemical composition
Even if a phantom exhibits optical properties equivalent to a certain tissue, the morphology of the phantom is a significant feature to match the tissue in terms of the anatomical shape. For cases where feasibility evaluation of a spectroscopic technique is required, the macroscopic geometry of the sample should be ideally reproduced in the tissue phantom.
Another prerequisite for the phantoms to fully simulate the respective tissue is to match the chemical properties. This is especially important in Raman spectroscopy because of the sensitivity of the technique to tissue chemistry on a molecular level. The chemical composition of a tissue is particularly challenging to reproduce in a phantom. This has led to the introduction of ex vivo animal tissue, or animal phantoms.
Liquid and solid tissue phantoms both lack realistic complexity that can be otherwise found in mammalian tissue. To further improve the realistic complexity of such phantoms, multiple layers of identical or different optical parameters and Raman-scattering features have been introduced to the bulk matrix. An example of the latter is well-represented by mineral inclusions, commonly used to enhance the biological importance of phantoms simulating rich-calcification tissues (eg, breast). Inclusions of hydroxyapatite (HAP), a type of apatite synthesized calcification powder, are often added inside the phantom to represent pathological calcifications. 43 Other strong scattering materials, such as calcium carbonate, trans-stilbene or polymers, can also be used, especially when there is a need of multiple inclusions with different scattering and distribution within the simulated tissue. Liquid tissue phantoms allow for easy addition or removal of inclusion in the sample volume. In contrary, robust phantoms cannot easily incorporate inclusion features following fabrication, but allow for the presence of a more realistic surrounding tissue such as in the case of animal phantoms.
Tissue Phantoms in Biomedical Applications
Optical tissue phantoms have a wide and diverse range of biomedical applications in the field of Raman spectroscopy (Table 1), and especially deep RS and Raman fiber probe measurements, where the studies mainly focus around lesion detection in cancer and bone disease diagnosis, or glucose sensing in blood. Numerous applications of tissue phantoms can be also found in the field of surface-enhanced Raman spectroscopy (SERS), where the signal of the molecule of interest can be significantly enhanced using nanoparticles. This review outlines most of the aforementioned applications, with an emphasis on the types of phantoms employed in each case.
Types and characteristics of tissue phantoms in Raman spectroscopy studies with a focus on biomedical applications.

Abbreviations: BMSB, 1,4-bis(2-methylstyryl)-benzene; BPE, trans-1,2-bis(4-pyridyl)-ethylene; COD, calcium oxalate (dihydrate); COHAP, carbonate substituted hydroxyapatite; COM, calcium oxalate monohydrate; CS, chondroitin sulfate; DCM, dichloromethane; DMD, digital micro-mirror device; FEP, fluorinated ethylene-propylene copolymer; FORS, frequency offset Raman spectroscopy; HAP, hydroxyapatite; Hb, hemoglobin; MPR, magnetic resonance imaging–Photoacoustic imaging–Raman imaging; OCT, optical coherence tomography; PCL, polycaprolactone; PDMS, polydimethylsiloxane; PE, polyethylene; PET, polyethylene terephthalate; PS, polystyrene; PTFE, polytetrafluoroethylene; PVA, polyvinyl alcohol; RS, Raman spectroscopy; SERS, surface-enhanced Raman spectroscopy; SERRS, surface-enhanced resonance Raman scattering; SESORS, surface-enhanced spatially offset Raman spectroscopy; SORS, spatially-offset RS; SWNTs, single walled carbon nanotubes; TRS, transmission RS.
Bone studies
Optical phantoms of osteochondral tissues are regularly used to simulate Raman measurements on either bone or soft tissue. Such phantoms are employed for the study of bone diseases or system optimization and characterization. In these studies, by probing the bone below soft tissue, Raman spectroscopy can provide useful information on (a) bone demineralization relevant to osteoporosis or other metabolic bone diseases, and (b) bone changes induced by rheumatoid arthritis or monitoring of bone graft osseointegration.
Raman tomography is a modality providing Raman signal combined with spatial and distribution information of the sample. The setup arrangements either consist of multiple excitation and collection fibers surrounding the sample, or by acquiring signal of a rotating sample in a transmission mode. Tissue phantoms have been employed in both modalities to assist the recovery of Raman signals from subsurface bone tissue. Schulmerich et al acquired computed tomographic (CT) data from skin and bone animal tissue. The CT data were then calibrated against scattering phantoms of agarose matrix with a Teflon sphere inclusion, in order to reconstruct a canine limb through diffuse Raman tomography.44,45 Apart from calibration reasons, bone tissue phantoms have also been employed for optimization and system development in Raman tomography. In a fiber optic coupled Raman system for measurements on musculoskeletal tissues, gelatin tissue phantoms with a mineral inclusion have been used for the optimization of the position of excitation and collection fibers.46,47 Esmonde-White et al 46 have measured phantoms of human lower and upper extremities with a variety of fiber optic probe configurations for specific anatomical sampling locations. It was shown that although the transcutaneous Raman signal of the bone could be recovered from 1 to 3 cm of soft tissue, the signal significantly depends on the phantom and therefore the anatomical location geometry. A follow-up study confirmed the feasibility of transcutaneous bone Raman measurements using Raman tomographic imaging, by testing tissue phantoms with mineral inclusions in the range of physiological HAP concentration in bone (Figure 4). 48 The system response was assessed through the contrast between the mineral and background (surrounding tissue) signal, whereas a multichannel detection version of the technique demonstrated multifold sensitivity and highly accurate position recovery of the inclusion. 49

(A) Photograph of a tissue phantom with a mineral “bone” inclusion (HAP) embedded in the centrifuge material (Cytop) and placed in the fiber holder of a Raman tomographic imaging setup. (B) Raman signal acquired with the collection fibers, for different HAP concentrations of the “bone” phantom inside its plastic centrifuge tube.
A different simulation of osteochondral interfaces has been achieved with scattering gelatin-based phantoms consisted of HAP and chondroitin sulfate to mimic bone and cartilage, respectively. Measurements on tissue phantoms of different optical properties which were conducted using fiber-optic Raman spectroscopy, 50 indicated that osteoarthritis parameters, such as optical scattering and thickness of cartilage, significantly affect photon propagation in the tissue and the subsequent bone signal recovery.
In other Raman fiber optic studies, rat tissue phantoms have acted as the intermediate stage for assessing the quality of implanted grafts.51-53 The gelatin-based phantoms were constructed in line with CT scans and in identical morphology to a rat leg (Figure 5). The phantoms incorporated scattering (Intralipid), absorption (Hemoglobin), and fluorescence (Hemoglobin and Hydroxyapatite) properties, which allowed calibration of the system’s response (position of the fibers) for the assessment of bone quality during graft osseointegration. 47

(A) Rat tissue phantom reconstructed from a rat leg model. (B) Rat tissue phantom reconstructed as in (A) but with hemoglobin and Intralipid omitted to make the bone layer visible. (C) Rat tissue phantom transcutaneous Raman spectrum of the bone layer showing major Raman bands (average signal recovered from 47 individual collection fibers). 47
Deep Raman spectroscopy, an emerging field of RS mainly consisting of spatially offset RS (SORS) and transmission RS (TRS), has utilized tissue phantoms to demonstrate feasibility of acquiring Raman signals from different sampling depths. The technology is based on decoupling the excitation from the collection area and has been extensively applied in the biomedical field.54,55 In the case of applications related to bone, SORS has been employed to non-invasively assess the mineralization of bone underneath skin, in tissue engineering scaffolds implanted in a sheep bone defect. Dooley et al 56 conducted measurements on 3D printed composite scaffolds with polycaprolactone (PCL) and hydroxyapatite (HA) microparticles, for which the concentration of HA varied to simulate different degrees of mineralization. The study demonstrated feasibility of the approach and a promising monitoring tool for bone healing. Last, tissue phantoms have also been used for the optimization of SORS setups, such as in the case of a mice skull which was wrapped in porcine tissue and filled with agarose gel and a polytetrafluoroethylene (PTFE) sheet inside. 57 The measurements were analyzed to assess the optimum spatial offset for the detection of the PTFE inclusion within the skull.
Breast cancer studies
Raman spectroscopy has been extensively applied to breast cancer diagnostics as an analytical tool. Its application has been reinforced by the presence of breast calcifications, small calcium deposits in human breast which can be benign or related to cancer. Their chemical composition varies and can be either calcium oxalate (type I) or calcium phosphate (type II), mainly hydroxyapatite. 58 Due to their high clinical significance, detection of calcifications has given rise to the employment of breast tissue phantoms in Raman spectroscopy, with calcification-like inclusions of a high Raman cross-section compound (hydroxyapatite, trans-stilbene, etc) to represent a malignant element in the phantom matrix.
In transmission Raman measurements conducted by Kerssens et al, 43 the phantom inclusions consisted of various concentrations of either calcium oxalate or calcium phosphate (standing for type I and type II calcifications, respectively) contained in quartz cuvettes which were in turn buried in porcine tissue. In a follow-up study, the selection of the optimum excitation wavelength for the optimization of the setup was achieved through TRS measurements on animal phantoms with inclusions of higher Raman cross-section, such as trans-stilbene. 59 The animal tissue chosen in such studies was porcine shoulder tissue, due to its composition of mixed fat and protein, similar to that of human breast.43,60-62 In further deep Raman studies, researchers employed liquid tissue phantoms of Intralipid solution and India ink, adjusting their optical properties to match those of the calcification-rich tissues of breast and prostate.19,42,63 The origin of the Raman signal of the calcification-like inclusion within the phantom volume was studied as a function of the phantom optical properties in a transmission mode, with the aim to inform future design of Raman probes for cancer diagnosis.
Phantoms of chicken and human breast tissue with calcification-material inclusions were employed in different modes of RS, such as spatially offset Raman spectroscopy 64 and picosecond Kerr-gated, 65 respectively, in order to assess the feasibility of calcification detection within the tissue. It is worth mentioning recent efforts that have been made with liquid and solid phantoms using combined sample differential transmittance (ie, differential attenuation of Raman photons at different wavelengths due to optical properties) and deep Raman spectroscopy, toward the non-invasive prediction of the depth of inclusions representing cancer lesions within turbid matrices.66,67
Skin studies
Various skin optical phantoms have been employed for calibration of Raman spectroscopic systems for potential skin cancer detection. Liu et al fabricated an agarose two-layer phantom with scattering similar to dermis and epidermis, introducing polystyrene microspheres in the matrix. Each phantom layer contained either urea or potassium formate as a Raman scatterer. The Raman signal ratio of the two was assessed to evaluate the performance of a snapshot depth sensitive Raman system. The system was evaluated on the basis of its ability to acquire signals from different depths of tissue phantoms, as well as ex vivo animal and in vivo human (fingernail) tissue. 68 Multimodality skin tissue phantoms have also been employed in studies to demonstrate the effectiveness of combined system approaches, such as in the case of a photo-acoustic Raman probe. 69 The multi-layer agarose phantom employed in the study, contained Intralipid and nigrosin dye to simulate skin tissue with a malignant tumor, represented by a trans-stilbene inclusion. All of the studies have yielded positive results on potential depth localization of skin tumors and assessment of cancer margins, paving the way for future skin cancer diagnostics.
In the field of skin diagnostics, phantoms have been used for system calibration to correct Raman signal in terms of both optical properties (spectral distorting from elastic scattering) and sample depth. For the correction of in-depth Raman signals in confocal Raman spectroscopy, Roig et al fabricated polydimethylsiloxane (PDMS) scattering skin phantoms of known optical properties with 70 and without absorption. 71 In the latter and more realistic approach, the measurements allowed for signal correction on skin phantoms consisting of multiple layers, each one of them representing a different skin layer (dermis, epidermis, stratum corneum) with distinct optical properties. Other studies including resonance Raman measurements on silicone skin phantoms combined with Monte Carlo simulations 72 have suggested correction functions for the Raman signal of carotenoids (a main contributor in skin absorption) in skin, without a priori knowledge of the tissue optical properties. 73
Blood studies
The non-invasive measurement of blood glucose has become a subject of continuous research in the field of bioanalytics. Tissue phantoms have significantly supported the research development toward that direction. Research groups under M. Feld, R. Dasari, and later I. Barman have employed liquid tissue phantoms of different optical properties and varying glucose concentration in Raman measurements. Importantly, their research was focused on correcting the acquired glucose Raman signal in terms of contribution due to skin optical properties, which were in turn simulated through the tissue phantoms. More specifically, Dingari et al 74 used tissue phantoms to establish that varying optical properties heavily affect the prediction accuracy of blood glucose detection in sample-to-sample variability. In a follow-up study, the researchers employed phantoms with different glucose concentrations to initially calibrate and then demonstrate that Turbidity-Corrected Raman Spectroscopy (TCRS) combined with least-squares analysis 75 or Support Vector Machines (SVM) 76 is able to recover useful spectral information by correcting the signal for intensity and shape distortions due to optical properties’ effects. Previous studies by the same research group, utilizing intrinsic Raman spectroscopy, also focused on correcting turbidity-induced variations (ie, spectral variations caused by absorption and scattering), however with the prerequisite of already known diffuse scattering coefficient in the employed phantoms. 77 Datasets from the same study were later used in the application of wavelength selection for linear partial least squares (PLS) and nonlinear calibration support vector regression (SVR), with the latter to exhibit the best predictive accuracy for transcutaneous blood glucose detection. 78 Addressing the spectral variations due to photo-bleaching in blood measurements where autofluorescence is usually encountered, Barman et al 79 have employed shifted subtracted Raman spectroscopy combined with partial least squares to increase the prediction accuracy of glucose detection in liquid phantoms, where the fluorescence has been previously induced with indocyanine green.
In a different study, Chaiken et al have used gelatin phantoms containing bovine erythrocytes and hemoglobin as a fluorescence agent, highlighting that interpatient blood qualities such as hematocrit and microcirculation may introduce higher variability in Raman measurements compared with glucose levels. The Raman spectrometer was calibrated using the employed phantoms for non-invasive in vivo measurements of blood glucose in a human fingertip. 80 Also for calibration reasons, epoxy resin solid phantoms have been fabricated using TiO2 and red ink as a scatterer and an absorber, respectively. 81 The researchers of the study achieved to design phantoms exhibiting not only optical properties similar to those of human fingernail, but also simulating the underlying skin tissue with several different layers including glass capillaries filled with red ink (blood) (Figure 6).

(A) The side view of a fingernail tissue phantom with capillaries. Red lines show upper and lower layer (75 capillaries per layer) of the designed capillary system. (B) Magnified image of the tissue phantom (three holes after red ink testing). (C) Raman spectrum of the tissue phantom basic material (epoxy resin). 81
In the category of blood equivalent tissue phantoms, it is worth mentioning the case of washed red blood cells suspended in saline instead of plasma 82 and matched in optical properties to human blood. 83 Rather than assess or optimize an instrument performance, those phantoms serve to eliminate inter-patient variability during measurements, as plasma carries most of the blood chemical variability.
SERS applications
SERS is a modified Raman technique where the sample is being absorbed onto a nano-colloidal metallic surface (typically silver or gold). 84 Due to the surface plasmon resonance of the metallic nanostructures, Raman signals of specific molecules can be amplified, leading to increased detection sensitivity of the analyte.
Numerous studies have included the simplest type of liquid tissue phantoms, in order to determine the depth of detection of the nanoparticles (NPs) for different Raman imaging systems. The approach consisted of Raman measurements from a capillary or Tygon tube with NPs immersed in varying depth of a scattering agent (ie, Intralipid).85-87
Hydrogel tissue phantoms made of Matrigel, polyvinyl alcohol (PVA), and agarose also play a significant part in SERS measurements for biomedical applications. Matrigel tumor phantoms stained with nanoparticles have been employed to demonstrate feasibility of imaging on a rat esophagus with a Raman endoscope. 88 Moran et al used PVA phantoms to study the effect of NPs aggregation on the SERS signal intensity. The NPs Raman signal has been shown to increase with NPs aggregation. 89 It is worth mentioning here that although not commonly used, PVA gels are able to match in optical properties soft tissues, by undergoing freezing and thawing cycles. 90
Among hydrogel phantoms, the agarose-based are the most commonly employed ones in several SERS studies, for example in assessing the repeatability and stability of a SERS sensor array for pH measurements. 91 In the field of non-invasive blood glucose monitoring, researchers have used them to demonstrate proof-of-concept SERS measurements using an Ag-coated microneedle.92,93 The agarose skin-mimicking phantoms were enhanced with Intralipid and nigrosin to mimic human skin. The approach was later employed in combination with resonance enhancement, to detect malaria in blood. 94 Chenet al 95 have composed an excellent review on the applications of SERS through skin. Other SERS applications include employment of tumor phantoms (breast cancer cells in agarose medium) for the development of a plasmonic nanoprobe utilized for in vivo photothermal therapy in tumors. 96 Agarose tissue phantoms have been used for NP-assisted cancer tissue scanning (ie, surgical margins), more specifically to assess the NP detection threshold, 97 the depth of penetration, 98 and their photothermal effect. 99 To better simulate the optical properties of the tissue, scattering (Liposyn) and/or absorbing (India ink) agents were added in the phantoms of the last two studies.
The combination of SERS and SORS (SESORS) has quickly led to the most clinically relevant applications, as it allows for non-invasive and highly sensitive probing of nanoparticle-conjugated biomolecules in different sampling depths. Both tissue and animal phantoms have been employed in SESORS measurements, especially in studies focused on brain tumor detection. Gel phantoms made of an agarose matrix with optical properties equivalent to brain tissue and infused with NPs were contained in a skull to demonstrate feasibility of the SESORS approach in brain cancer.100-103
On their end, animal tissue phantoms in SERS studies have served as models for the evaluation of the maximum penetration depth in tissues through which a distinct signal from the SERS-active NPs could be obtained. In those studies, one must consider the multiple parameters affecting the assessment of penetration depth using nanoparticles, such as concentration and degree of conjugation achieved. With a combination of SERS and deep Raman spectroscopy (SORS and transmission),104,105 researchers have used NPs to probe a localized inclusion (ie, cancer lesion) in animal tissues and evaluate the NP maximum penetration depth. A recent application of combined SERS, SORS and transmission Raman in porcine tissue, has yielded high accuracy not only in terms of NPs’ signal detection but also of their depth prediction. 106 Other applications of combined SERS and deep Raman spectroscopy in porcine tissue phantoms include multiplexed imaging, 107 combined Raman tomography, 108 and drug detection. 109 Stone et al 110 have specifically combined transmission RS with SERS to improve the detection limit of resonance (SERRS) active nanoparticles in animal tissue, leading toward the prospect of small tumor identification. Nicolson et al 57 used plasmon resonance active nanoparticles, leading to the first in vivo application of SESORRS (Surface-Enhanced Spatially Offset Resonance Raman Spectroscopy) for the detection of brain tumor in rats after the SORS signal has been optimized in equivalent agarose tissue phantoms.
Last, it is worth mentioning a multimodal approach of combined photoacoustic and Raman imaging, where Shi et al111,112 employed a special case of phantoms to evaluate SERS imaging for the detection of breast cancer cells tagged with NPs. The cells targetted with nanoparticles were suspended in rat blood flowing within an acrylic capillary tube, achieving in that way a more realistic blood microvasculature imaging (Table 1).
Closing Remarks
There is a large number of optical tissue phantoms covering a wide range of shapes, geometries, and optical properties. Due to their flexibility in design, time-efficiency and low-cost fabrication, optical tissue phantoms are now a standard part of instrumentation for studies translating into a clinical environment. However, even if their geometry mimics the tissue of interest accurately, the phantoms are only able to simulate the absorption and scattering of the tissue on a large scale. Their composition and lack of heterogeneity do not allow for accurate representation of the complicated optical properties arising from cellular and subcellular features within the tissue. For that reason, although tissue phantoms are an indispensable part of research development and validation, ex vivo tissue has also been introduced in their place.
Advances on tissue engineering and 3D printing hold great promise for further development of tissue phantom technology. The rapidly evolving field of 3D printing can support increasing refinement of the phantom composition, contributing to a more realistic structure. Although 3D phantoms in Raman Spectroscopy are not very common, an excellent review of their qualities and limitations in other imaging techniques has been composed by Filippou and Tsoumpas. 141
Together with ex vivo tissue, tissue engineering can become an established alternative to the standard tissue phantoms, serving the needs of current technology and applications. One of these challenges consists of the employment of multiple approaches within the same study. Optical and spectroscopic techniques are constantly combined with standard clinical imaging (magnetic resonance imaging, X-rays, optical coherence tomography) to further advance biomedical research. As part of this development, the efficiency in cost and time is pushing toward the fabrication of multimodality phantoms. Although this is a desirable concept, it is also highly challenging as different techniques require different properties integrated into the phantoms.
Further research is required to take full advantage of tissue phantoms as a useful tool not only for the better understanding of biological systems, but also for the more efficient development of diagnostic and therapeutic approaches.
Supplemental Material
Supplementary – Supplemental material for Tissue Phantoms for Biomedical Applications in Raman Spectroscopy: A Review
Supplemental material, Supplementary for Tissue Phantoms for Biomedical Applications in Raman Spectroscopy: A Review by Martha Z Vardaki and Nikolaos Kourkoumelis in Biomedical Engineering and Computational Biology
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is co-financed by Greece and the European Union (European Social Fund- ESF) through the Operational Programme “Human Resources Development, Education and Lifelong Learning” in the context of the project “Reinforcement of Postdoctoral Researchers - 2ndr Cycle” (MIS-5033021), implemented by the State Scholarships Foundation (IKΥ).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MV and NK outlined the scope of the review, and MV developed the initial manuscript draft. All authors participated in developing the final manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

