Abstract
Objective
To investigate the association between the Dietary Inflammatory Index (DII) and stroke risk among hypertensive adults, as well as all-cause mortality post-stroke, utilizing data from the National Health and Nutrition Examination Survey (NHANES).
Methods
This cross-sectional analysis included 7,590 hypertensive participants (stroke group: N=609; non-stroke group: N=6,981). DII was derived from 28 dietary components. Participants were stratified into DII tertiles: Q1 (lowest), Q2 (moderate), and Q3 (highest). Weighted multivariable logistic regression assessed associations between DII (continuous and categorical) and stroke prevalence. Restricted cubic splines (RCS) evaluated non-linearity. Subgroup analyses identified effect modifiers. Cox proportional hazards regression modeled associations of DII and its components with all-cause mortality in the stroke cohort.
Results
Stroke patients exhibited significantly higher DII scores than non-stroke controls (P <0.05). Each 1-unit increase in DII was associated with a 13% elevated stroke risk (Odds Ratio (OR)=1.13, 95%CI: 1.04–1.22, P =0.006). Compared to Q1, Q3 had a 68% higher stroke risk (OR=1.68, 95%CI: 1.22–2.32, P=0.002). RCS confirmed significant non-linearity (P<0.001). Antihypertensive medication modified this association (P-interaction =0.042). Among stroke patients, DII demonstrated a U-shaped association with mortality (P-trend =0.048): Q2 had the lowest mortality, while Q1 and Q3 showed poorer survival. Component analysis revealed higher β-carotene scores associated with increased mortality risk (Hazard Ratio (HR)=1.44, 95%CI: 1.01–2.04), whereas higher vitamin A scores correlated with reduced risk (HR=0.68, 95%CI: 0.47–0.99).
Conclusion
This cross-sectional study identifies a significant, dose-response association between elevated DII and increased stroke risk in hypertensive adults, suggesting that reducing dietary inflammatory load holds preventive potential. Moreover, β-carotene and vitamin A show opposing associations with post-stroke mortality, reflecting the complexity of nutritional inflammation and informing precision nutrition strategies for stroke.
1. Introduction
According to the 2019 Global Burden of Disease (GBD) study, stroke remains the second leading cause of death worldwide. Projections indicate that stroke-related mortality will reach 7.8 million by 2030, with an estimated 23 million new cases of ischemic stroke—the most prevalent subtype, accounting for approximately 71% of all strokes.1,2
Multiple factors, including hypertension, dyslipidemia, diabetes mellitus, smoking, and excessive alcohol consumption, are well-established as significant risk factors for stroke. 3 Among these, hypertension is consistently recognized as the predominant modifiable risk factor,4,5 conferring a stroke risk up to 9.7 times higher than that in normotensive individuals. 6 The core pathophysiological mechanism linking hypertension to stroke involves sustained, chronic systemic inflammation.7,8 Studies consistently demonstrate elevated levels of various inflammatory biomarkers in hypertensive patients. These inflammatory mediators collectively heighten stroke risk by promoting atherogenesis, activating immune cells, and directly impairing vascular endothelial function.9-11 Crucially, inflammatory cytokines released by activated immune cells and damaged tissues can infiltrate the brain parenchyma, triggering localized vascular inflammation and neuroinflammation, thereby further amplifying susceptibility to stroke.12,13 Therefore, inflammation plays a pivotal pathogenic role in both the initiation and progression of stroke among individuals with hypertension.
Given that inflammation is a core driver in the pathogenesis of atherosclerosis and stroke, 14 and considering the central role of dietary intake in modulating systemic inflammation—where different dietary patterns exhibit significant variation in their inflammatory potential 15 —targeting diet presents a compelling avenue for intervention. Evidence indicates that pro-inflammatory diets elevate circulating concentrations of established inflammatory biomarkers, such as complement C3, C-reactive protein, tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6). 16 This suggests substantial potential for anti-inflammatory dietary strategies in both stroke prevention and management. The Dietary Inflammatory Index (DII) serves as a validated tool to quantify the overall inflammatory potential of an individual’s diet. It provides a composite score reflecting the diet’s pro-inflammatory or anti-inflammatory tendency, derived from systematically assessing the intake levels of numerous bioactive food components known to influence inflammatory processes (higher scores indicate greater pro-inflammatory potential; lower scores indicate stronger anti-inflammatory potential). Constructed through systematic reviews of the scientific literature, the DII incorporates dietary factors with clinically validated effects on inflammatory markers, and its reliability and accuracy have been confirmed in validation studies. 17 Application of the DII in epidemiological research has consistently revealed associations between diet-induced inflammation and the development of various chronic diseases, including cardiovascular disease, cancer, obesity, diabetes mellitus, and inflammatory bowel disease.18-20
Although multiple epidemiological studies have investigated the association between the DII and cardiovascular disease risk, and subgroup analyses in some suggest a positive trend linking higher DII scores (indicating a more pro-inflammatory dietary pattern) with increased stroke incidence risk,21-23 the specific impact of dietary inflammatory potential on stroke outcomes remains inadequately elucidated within the high-risk population of hypertensive adults. This gap is particularly pronounced concerning the influence of DII and its specific dietary components on mortality among hypertensive individuals with established stroke. Consequently, this study aims to leverage data from the nationally representative National Health and Nutrition Examination Survey (NHANES) to rigorously examine the association between DII scores and stroke occurrence risk, specifically among hypertensive adults. Our findings will provide crucial epidemiological evidence elucidating the role of diet-related inflammation in stroke risk within this vulnerable population. This evidence is vital for identifying modifiable dietary risk factors and informing the future development of targeted dietary intervention strategies, potentially facilitating early risk mitigation and improving clinical outcomes.
2. Materials and Methods
2.1. Study Design
NHANES is a nationally representative cross-sectional survey spearheaded by the National Center for Health Statistics (NCHS). It systematically assesses the health and nutritional status of the non-institutionalized U.S. population. NHANES employs a complex, multi-stage, stratified probability sampling design, annually recruiting approximately 5,000 participants to ensure accurate representation of the U.S. population’s demographic diversity. Comprehensive health data across all age groups is collected through standardized interviews, physical examinations, laboratory tests, and detailed dietary assessments. All of the NHANES data is accessible to the public and can be downloaded freely through: https://www.cdc.gov/nchs/nhanes/index.htm.
This analysis leveraged data from seven consecutive NHANES cycles (2005–2018). The initial merged dataset comprised 70,190 respondents. To focus specifically on the hypertensive population, we first excluded individuals lacking hypertension diagnosis information or confirmed to be non-hypertensive (N = 52,288). Subsequently, participants meeting any of the following criteria were further excluded: (1) missing dietary data precluding DII score calculation (N = 3,102); or (2) missing information on stroke diagnosis status or key covariates (N = 7,210). Following this selection process, the final analytical sample consisted of 7,590 hypertensive participants. Among these, 609 had a history of stroke, while 6,981 were stroke-free. The detailed participant selection flowchart is presented in Figure 1. A detailed flow chart of participant recruitment
2.2. DII
DII scores were computed using dietary data from NHANES. NHANES collects dietary information through standardized protocols involving an initial in-person 24-hour dietary recall at the Mobile Examination Center (MEC) followed by a secondary telephone interview. For this study, mean intake values from both recall-representing habitual consumption of foods and beverages, excluding supplements and medications, were utilized for DII calculation. The DII framework, established by Shivappa et al, 24 quantifies a diet’s overall inflammatory potential. Although the original system incorporates 45 dietary parameters, its methodology permits valid score derivation with fewer than 30 components. Given constraints in NHANES food composition data, we calculated DII scores based on 28 available components.
Computation followed standard methodology via the nhanesR R package: Individual intake of each component was standardized (typically via z-score transformation), centered by subtracting global reference means, and weighted according to empirically derived effect strengths and directions (pro-/anti-inflammatory) on six core inflammatory biomarkers (IL-1β, IL-4, IL-6, IL-10, TNF-α, CRP) as documented in the literature. Weighted scores for all 28 components were summed to generate the final DII score, where higher values indicate greater pro-inflammatory potential and lower values reflect anti-inflammatory properties. 25 In primary analyses, DII was treated as a continuous variable. To evaluate potential nonlinear associations, participants were stratified into DII tertiles (Q1-Q3).
2.3. Hypertension Definition
Blood pressure measurements adhered to the American Heart Association (AHA) standardized protocol. Participants rested quietly for 5 minutes before undergoing three consecutive readings at 30-60 second intervals, with mean systolic (SBP) and diastolic (DBP) blood pressure values calculated from these measurements. Hypertension was defined per the *2017 AHA/ACC Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults 26 through fulfillment of either clinical or historical criteria: clinical hypertension was determined as SBP ≥130 mmHg or DBP ≥80 mmHg, while historical hypertension required self-reported physician diagnosis confirmed by affirmative response to “Have you ever been told by a doctor or health professional that you have hypertension?”. Participants meeting either criterion were categorized as hypertensive.
2.4. Stroke Definition
Stroke status was ascertained using self-reported data from the NHANES medical conditions questionnaire. Participants responded to the item: “Has a doctor or other health professional ever told you that you had a stroke?” Those answering affirmatively were categorized into the stroke group, while negative respondents comprised the non-stroke group.
2.5. Mortality Ascertainment
Mortality outcomes were determined by linking participant records to the National Death Index (NDI) database maintained by the Centers for Disease Control and Prevention (CDC) (https://www.cdc.gov/nchs/data-linkage/mortality-public.htm). Follow-up duration extended from the NHANES interview date until the date of death or December 31, 2019 (the final NDI update date), whichever occurred first. All-cause mortality encompassed every death event recorded in the NDI and was identified using International Classification of Diseases, Tenth Revision (ICD-10) codes. 27
2.6. Covariates
Consistent with established evidence, this study adjusted for multidimensional covariates significantly associated with hypertension and stroke. These encompassed sociodemographic characteristics (gender, age, race/ethnicity, education level, marital status, family income-to-poverty ratio [PIR]), clinical and biochemical indicators (body mass index [BMI, kg/m2], total cholesterol [mg/dL], high-density lipoprotein cholesterol [HDL-C, mg/dL]), health behaviors, and disease status. Health behaviors included smoking status defined as current/former smoking (lifetime consumption ≥100 cigarettes based on questionnaire item SMQ020) and alcohol consumption (≥12 standard drinks annually from item ALQ101). Disease status comprised physician-diagnosed comorbidities via self-report: diabetes (DIQ010=“Yes”, excluding gestational diabetes), high cholesterol level (BPQ080=“Yes”), coronary heart disease (MCQ160C=“Yes”), and cancer history (MCQ220=“Yes”). Antihypertensive medication use (BPQ040A=“Yes”) was additionally included.
2.7. Statistical Analysis
Leveraging the NHANES sampling design, all analyses employed SAS survey procedures with appropriate sampling weights to generate nationally representative estimates. Continuous variables are presented as mean ± standard deviation (SD), while categorical variables are summarized as frequencies (percentages). Group comparisons between stroke and non-stroke participants utilized independent samples t-tests for normally distributed continuous variables, Wilcoxon rank-sum tests for non-normally distributed variables, and chi-square tests for categorical variables.
Multivariable logistic regression models evaluated the association between the DII and stroke prevalence. A progressive adjustment strategy was implemented: Model 1 (crude) included no covariates; Model 2 adjusted for sociodemographic factors (gender, age, race/ethnicity, education, marital status, PIR); Model 3 further incorporated clinical indicators (BMI, total cholesterol, HDL-C) and health factors (smoking status, alcohol consumption, diabetes, high cholesterol level, coronary heart disease, cancer history and taking prescription for hypertension (TPH)). To test for non-linear associations, restricted cubic spline analysis with three knots at the 10th, 50th (median), and 90th percentiles of the DII variable was conducted, with all models incorporating the complex NHANES survey design (including survey weights, strata, and cluster variables) to ensure national representativeness. The statistical significance of the non-linear relationship was evaluated by the P value for non-linearity (P-nonlinear).
In the cohort of hypertensive stroke patients (N=609), this study employed Cox proportional hazards regression to examine the associations between individual DII components (dichotomized at the median) and all-cause mortality. Variables with P <0.50 in univariate analyses were included in a multivariable Cox regression model to assess their independent effects. Results are reported as hazard ratios (HR) with 95% confidence intervals (CI).
Prespecified subgroup analyses across 14 strata (e.g., gender, age, race) evaluated potential effect modification. Likelihood ratio tests comparing models with and without DII-by-stratum interaction terms determined the statistical significance of modifying effects.
All analyses were conducted using R software (v4.4.1; R Foundation). Two-sided P-values <0.05 defined statistical significance.
3. Results
3.1. Study Population Characteristics
Baseline Characteristics of the Study Participants Grouped by Hypertensive Stroke Status
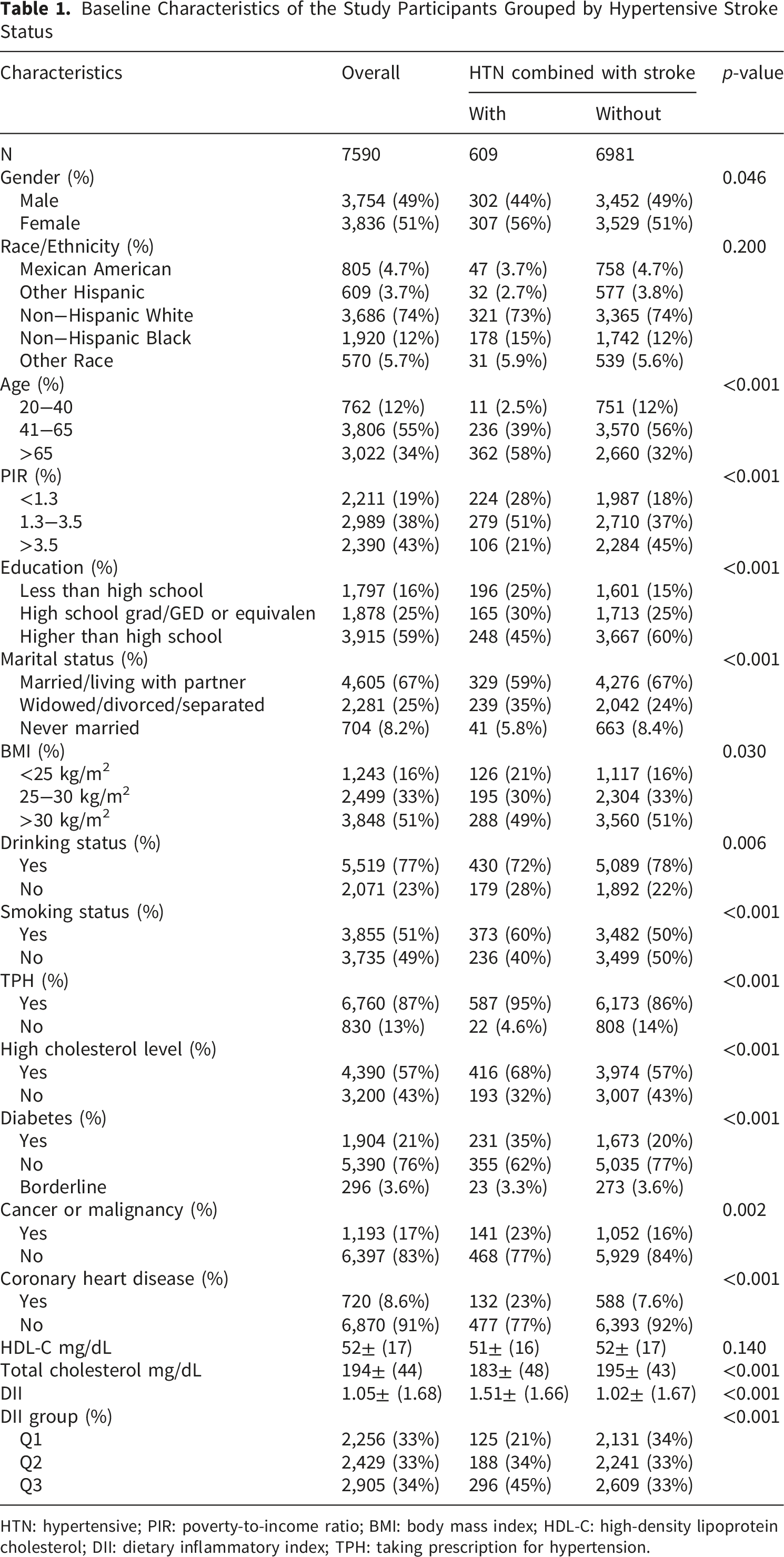
HTN: hypertensive; PIR: poverty-to-income ratio; BMI: body mass index; HDL-C: high-density lipoprotein cholesterol; DII: dietary inflammatory index; TPH: taking prescription for hypertension.
3.2. Association Between DII and Stroke Risk in Hypertensive Adults
Weighted Logistic Regression Analysis on the Association Between DII and Hypertensive Stroke Patients
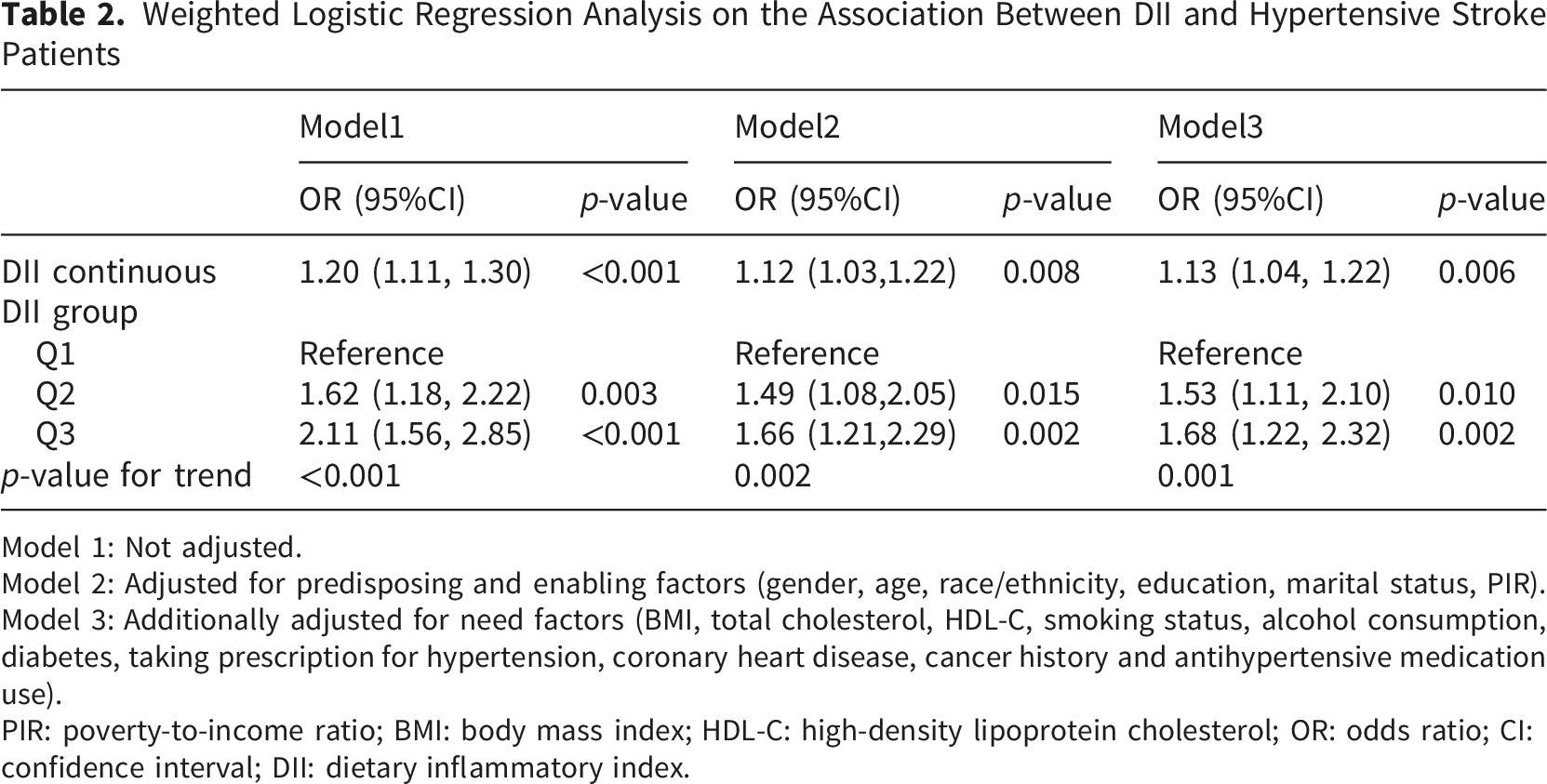
Model 1: Not adjusted.
Model 2: Adjusted for predisposing and enabling factors (gender, age, race/ethnicity, education, marital status, PIR).
Model 3: Additionally adjusted for need factors (BMI, total cholesterol, HDL-C, smoking status, alcohol consumption, diabetes, taking prescription for hypertension, coronary heart disease, cancer history and antihypertensive medication use).
PIR: poverty-to-income ratio; BMI: body mass index; HDL-C: high-density lipoprotein cholesterol; OR: odds ratio; CI: confidence interval; DII: dietary inflammatory index.
3.3. RCS Analysis
To characterize the functional form of the DII-stroke relationship, restricted cubic splines were incorporated into the fully adjusted model (Model 3). This analysis revealed a significant nonlinear positive association between DII and hypertensive stroke (test for nonlinearity: P <0.001), with the dose-response curve illustrating progressively elevated risk beyond mid-range DII values, as illustrated in Figure 2. Restricted cubic spline curve for the association between DII and hypertensive stroke
3.4. Subgroup Analyses
Prespecified subgroup analyses stratified by sex, age, race/ethnicity, PIR, education, marital status, BMI, smoking, alcohol consumption, TPH, high cholesterol level, diabetes, cancer history, and coronary heart disease are detailed in Figure 3. Interaction analysis revealed significant effect modification by antihypertensive medication status (P-interaction =0.042). Notably, the DII-stroke association was substantially stronger among participants not TPH (OR=1.59, 95% CI: 1.14–2.22) compared to medication users. These findings demonstrate that the relationship between pro-inflammatory diets and stroke risk in hypertensive adults is significantly modified by antihypertensive treatment status. Subgroup analyses of the association between dietary inflammatory DII and hypertensive stroke
3.5. Association Between DII and All-Cause Mortality in Hypertensive Stroke Patients
Kaplan-Meier survival analysis revealed significant mortality differences across DII tertiles in hypertensive stroke patients (log-rank P =0.048; Figure 4). Participants with moderate DII scores Q2 demonstrated the most favorable survival outcomes, whereas both Q1 and Q3 DII tertiles exhibited comparatively poorer survival trajectories. Survival curves by DII tertiles (Q1, Q2, Q3) in hypertensive stroke Patients
3.6. Univariable and Multivariable Analyses of DII Components and Mortality
To identify specific dietary factors influencing survival outcomes in hypertensive stroke patients, preliminary univariable Cox regression screening (Supplemental Table 1) informed subsequent multivariable modeling. The adjusted Cox proportional hazards model revealed divergent effects: higher β-carotene scores were associated with a 44% increased mortality risk (HR=1.44, 95% CI: 1.01–2.04, P =0.043), whereas elevated vitamin A scores demonstrated a protective effect with 32% risk reduction (HR=0.68, 95% CI: 0.47–0.99, P =0.047), as illustrated in Figure 5. Multivariable Cox regression analysis of the 28 individual DII components with all-cause mortality in hypertensive stroke patients
4. Discussion
Leveraging the nationally representative National Health and Nutrition Examination Survey (NHANES) database, this study comprehensively investigated the association between dietary inflammatory potential—quantified via the Dietary Inflammatory Index (DII) constructed from 28 inflammation-modulating components—and stroke risk among hypertensive adults, along with post-stroke all-cause mortality. Our principal findings demonstrate significantly elevated DII scores in stroke survivors compared to stroke-free counterparts, with a graded increase in stroke incidence paralleling rising DII levels. Both unadjusted and fully adjusted logistic regression models consistently established higher DII as an independent risk factor for stroke in hypertension. Restricted cubic spline analysis further revealed a significant nonlinear dose-response relationship between DII and stroke risk. Notably, taking prescription for hypertension emerged as a significant effect modifier in subgroup analyses. Regarding prognosis, all-cause mortality among hypertensive stroke patients exhibited a non-monotonic association with DII: optimal survival occurred at moderate DII levels (Q2), whereas both anti-inflammatory (Q1) and pro-inflammatory (Q3) extremes demonstrated poorer outcomes. Importantly, multivariable Cox regression analysis of individual DII components identified β-carotene and vitamin A as significant independent predictors of mortality with opposing effects—higher β-carotene scores increased mortality risk by 44% (HR=1.44), while elevated vitamin A conferred a 32% protective reduction (HR=0.68). This work provides novel insights into the differential impacts of specific pro- and anti-inflammatory dietary components on survival in hypertensive stroke patients.
These findings align robustly with extant evidence linking the DII to cardiovascular disease (CVD) pathogenesis. As substantiated by Hariharan et al’s systematic review, 19 elevated DII correlates with increased risks of obesity, type 2 diabetes, and CVD. Multiple studies28,29 further confirm DII-mortality associations in CVD populations. Wang et al 30 demonstrated that modulating pro-inflammatory food intake effectively reduces 10-year CVD risk. Specifically, Shivappa et al 31 documented a positive DII-coronary heart disease mortality. Concerning hypertension and stroke, observational evidence consistently associates higher DII with significantly elevated hypertension risk. 32 For stroke subtypes, patients with elevated DII exhibit a higher prevalence of large-artery atherosclerotic stroke compared to those with minimal dietary inflammation (P =0.050). 33 Huang et al 34 established DII as an independent stroke risk factor among U.S. adults, noting a nonlinear positive association between baseline inflammatory diet patterns and stroke incidence. Multivariable risk analyses further confirm high DII and physical inactivity as significant independent stroke risk determinants. 35 Environmental factors (e.g., lead exposure) 36 and micronutrient intake (e.g., folate) 37 may further modulate stroke risk through interactions with dietary inflammation or mediation pathways.
The complex nonlinear dynamics characterizing the DII-stroke risk relationship warrant particular attention. Emerging evidence indicates threshold-dependent effects, where each unit increase below DII 2.0 significantly elevates stroke risk, yet paradoxically associates with risk reduction beyond this inflection point. 38 Our mortality findings—demonstrating poorer prognosis at both anti-inflammatory (Q1) and pro-inflammatory (Q3) extremes compared to moderate DII (Q2)—further substantiate this intricate pattern, suggesting a U-shaped association between dietary inflammation and post-stroke survival. Biologically, sustained pro-inflammatory diets (high DII) drive chronic low-grade inflammation through elevated TNF-α, IL-1, IL-2, IFN-γ, and vascular cell adhesion molecule-1 (VCAM-1), 39 compromising endothelial function and accelerating atherogenesis, thrombosis, and metabolic dysregulation—collectively increasing recurrent events and mortality risks. Conversely, overly restrictive anti-inflammatory diets (low DII) may incur nutritional deficits impairing tissue repair and immune function during post-stroke hypermetabolism, while excessive suppression of inflammation could hinder essential recovery processes like angiogenesis and necrotic tissue clearance, potentially increasing vulnerability to fatal complications such as pneumonia or urinary tract infections. Thus, moderate DII levels likely represent an optimal equilibrium that avoids chronic vascular injury from excessive inflammation while providing balanced nutritional support, yet permitting regulated inflammatory responses necessary for tissue repair and immune defense—a biological reconciliation explaining the observed U-shaped mortality pattern.
A pivotal finding emerged from our component-specific Cox regression analyses of 28 DII constituents: despite their metabolic kinship as vitamin A precursors, β-carotene (HR=1.44, 95% CI: 1.01–2.04) and vitamin A (HR=0.68, 95% CI: 0.47–0.99) exerted opposed effects on mortality in hypertensive stroke patients. This divergence likely stems from distinct post-stroke metabolic fates and biological activities. While our observed detriment of β-carotene aligns with studies identifying it as a cardiovascular risk factor,40,41 Farashi’s meta-analysis 41 conversely associates vitamin A with reduced stroke mortality and implicates β-carotene deficiency as hazardous—partially contradicting our results. The protective mechanism of vitamin A may involve its active metabolite, all-trans retinoic acid (ATRA), modulating neuroinflammation, particularly through promoting beneficial N2 neutrophil polarization and suppressing neutrophil extracellular trap (NET) formation via STAT1 signaling. Exogenous retinoids demonstrably enhance neurological recovery post-ischemia.42,43 As a natural derivative, retinoic acid (RA) exhibits well-established anti-inflammatory and anti-proliferative properties, potently inhibiting vascular smooth muscle cell migration and dedifferentiation to attenuate atherosclerosis. 44 Moreover, RA maintains blood-brain barrier integrity during inflammatory challenges. 45 Conversely, β-carotene’s paradoxical risk elevation remains mechanistically enigmatic. Though some studies link β-carotene-rich foods to reduced mortality, 46 meta-analyses of dietary antioxidants often show linear dose-response relationships with maximal benefits at lower intakes, 47 potentially reflecting biological saturation or measurement artifacts. Emerging evidence challenges β-carotene’s safety profile: both the CARET trial 48 and Prince et al 49 reported increased cardiovascular mortality with supplementation, suggesting context-dependent toxicity in compromised populations.
As far as we know,the findings of this study are based on a U.S. population from NHANES. Dietary patterns, food sources of inflammatory components, and lifestyle factors vary considerably across cultures and geographic regions. Therefore, the generalizability of our results to non-U.S. populations, such as Asian or African cohorts, may be limited. Future studies in diverse populations are needed to confirm these associations and inform culturally tailored dietary recommendations.
However, several limitations warrant consideration. First, the cross-sectional design inherently precludes causal inference regarding DII and stroke risk in hypertension. Second, the DII calculation relied on only 28 of the original 45 dietary components due to data availability constraints in NHANES. This partial computation may reduce the comprehensiveness of the inflammatory potential assessment and could introduce measurement error compared to the full DII. Third, stroke ascertainment was based on self-reported physician diagnosis without neuroimaging or medical record verification. This may introduce misclassification bias and precludes differentiation between ischemic and hemorrhagic subtypes, which have distinct pathophysiologies and may respond differently to dietary influences. However, prior validation studies within NHANES have indicated acceptable accuracy of self-reported stroke for epidemiological analyses. Fourth, reverse causation is possible, as stroke survivors may alter their dietary habits after diagnosis. Thus, the observed DII scores may reflect post-stroke dietary changes rather than pre-stroke exposure. The cross-sectional design cannot disentangle the temporal sequence. Fifth, although we adjusted for several key covariates, data on physical activity and total dietary energy intake were not included in the final models due to complexities in NHANES variable harmonization across cycles. Their absence may contribute to residual confounding. Additionally, as a secondary analysis of the NHANES survey, no prior sample size or power calculation was performed. The sample size was determined by the available data meeting inclusion criteria.
To address these limitations, future investigations should prioritize prospective cohort designs to establish causal relationships between DII and stroke in hypertension. Refining dietary assessment methodologies is imperative—either by developing expanded DII versions incorporating broader food components and processing-level data, or by integrating novel inflammatory indices within NHANES. Mechanistic studies must delineate biological pathways linking pro-inflammatory diets to cerebrovascular pathogenesis. Additionally, research should evaluate DII’s role in secondary stroke prevention, examining associations with recurrence risk and functional recovery among survivors. Crucially, randomized controlled trials (RCTs) are needed to test whether DII-lowering interventions reduce systemic inflammation, improve vascular endothelial function, attenuate blood pressure variability, and ultimately prevent stroke events in high-risk hypertensive populations. Finally, comparative analyses across racial/ethnic and socioeconomic strata will inform culturally tailored precision nutrition strategies to mitigate disparities.
5. Conclusion
Utilizing a large nationally representative NHANES sample, this study demonstrates that elevated DII scores constitute a key modifiable risk factor for stroke in hypertensive adults, while revealing a U-shaped association between post-stroke survival and dietary inflammation levels. Crucially, we identified divergent prognostic impacts of specific DII components—β-carotene increasing mortality risk and retinol (vitamin A) conferring protection. These findings underscore the critical importance of modulating dietary inflammation equilibrium in hypertension and stroke management. Our work lays essential groundwork for future prospective studies to verify causality, refine DII assessment methodologies, and elucidate the distinct biological mechanisms underlying β-carotene and retinol effects. Ultimately, this evidence supports developing precision nutrition strategies targeting inflammatory balance optimization for stroke prevention and post-stroke outcome improvement.
Supplemental Material
Supplemental Material - Assessing the Association of the Dietary Inflammatory Index With Stroke Risk and All-Cause Mortality Among Hypertension Patients: A NHANES Cross-Sectional Study (2005-2018)
Supplemental Material for Assessing the Association of the Dietary Inflammatory Index With Stroke Risk and All-Cause Mortality Among Hypertension Patients: A NHANES Cross-Sectional Study (2005-2018) by Amu Jike, Shuming Wang, Erha Munai, Ping Xiong, Lvmeng Song, Yinchuan Cheng and Shun Li in Journal of Central Nervous System Disease.
Footnotes
Ethical Considerations
The study protocol was approved by the NHANES Institutional Review Board and was performed by the Declaration of Helsinki, with all NHANES participants providing signed informed consent.
Author Contributions
AMJK conceived and designed the study, and drafted the manuscript; SMW and YCC performed all data analyses and interpreted the results; PX prepared the tables and data; EHMN revised the manuscript and made necessary modifications to enhance logic and readability; LMS supervised all aspects of the work. SL provided valuable suggestions during the revision of the article. SL will cover the article processing charge, which is essential for broadening the dissemination of the findings and conclusions of our work. All authors contributed to the article and approved the submitted version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The current study was supported by grants from the Sichuan Medical Science and Technology Innovation Research Association (YCH-KY-YCZD2024-054), the Health China·BuChang ZhiYuan Public welfare projects for Heart and brain health of Zhongshe Social Work Development Foundation (HIGHER2024056), the Affiliated Hospital of North Sichuan Medical College (2019ZD001), the North Sichuan Medical College (CBY23-ZDA01) and the Bureau of Science and Technology Nanchong City (19SXHZ0321).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
