Abstract
Purpose
This study aimed to evaluate the influence of the triglyceride-glucose index (TyG index) on clinical outcomes and to develop nomogram models for predicting early neurological deterioration (END) and long-term prognosis in acute ischemic stroke (AIS) patients following intravenous thrombolytic (IVT) therapy.
Methods
We conducted a multi-center retrospective cohort study involving 333 AIS patients treated with IVT. The short-term and long-term outcomes were defined as the occurrence of END and 90-day prognosis. Multivariate logistic regression was used to develop nomogram models for forecasting these clinical outcomes.
Results
Patients in the high-TyG group exhibited significantly higher risks of END (P = 0.0010) and poor 90-day outcomes (P = 0.0012). Independent risk factors for END included a lower baseline NIHSS score, delayed door-to-needle time (DNT), reduced ASPECTS score, elevated TyG index, higher potassium (K+) levels, and incomplete Willis artery. Additionally, a higher initial NIHSS, increased TyG levels, presence of END, and a history of hypertension were predictors of poor prognosis. Based on the identified risk factors, two nomogram models yielded AUC values of 0.746 and 0.849 for predicting END and poor prognosis, respectively. NIHSS scores, TyG index, and admission glucose levels (Glu) emerged as prognostic indicators across all patients, while higher mean platelet volume (MPV) and history of stroke were identified as novel risk factors for poor prognosis in NO-END group.
Conclusion
A higher TyG index correlates with poor clinical outcomes in AIS patients post-IVT. The nomograms combining the TyG index with various factors enhanced risk prediction for END and poor prognosis.
Keywords
Backgrounds
Alteplase-based intravenous thrombolysis constitutes the benchmark for treating acute ischemic strokes. 1 The WAKE-UP and EXTEND trials, together with the implementation of emergency ‘green channel’ systems, have significantly expanded the accessibility of intravenous thrombolytic therapy (IVT), resulting in substantial enhancements in functional outcomes for patients with acute ischemic stroke (AIS).2,3 Early neurological deterioration (END) is a prevalent complication, occurring in about 5% to 40% of AIS patients following IVT. This complication exacerbates neurological deficits, extends hospital stays, and is linked to poor long-term outcomes.4–6 Therefore, there is an urgent need for valid markers and efficient tools that can forecast the onset of END and subsequent poor long-term prognosis.
The Triglyceride-Glucose (TyG) index serves as a convenient indicator of chronic insulin resistance (IR), being derived from readily available fasting measures of glucose (Glu) and triglycerides (TG).7,8 Recent research has highlighted the TyG index as a significant risk factor for the incidence and progression of ischemic stroke.9–12 Some studies have also suggested an association between an elevated TyG index and the development of END13,14 and a poor long-term prognosis.15–18
Although the TyG index has been extensively studied in AIS, its prognostic value in IVT-treated patients remains undetermined. This multicenter cohort study systematically evaluated the longitudinal relationship between TyG index and the risk of END, as well as poor prognosis in AIS patients receiving IVT. Additionally, we developed two comprehensive nomogram prediction models incorporating the TyG index and multi-dimensional variables to precisely estimate the occurrence of END and poor 90-day prognosis.
Materials and Methods
Study Patients and Treatment
A retrospective, multicenter investigation was conducted among all individuals who were admitted to the stroke units at Shanghai Tongji Hospital affiliated to Tongji University and The Fourth Affiliated Hospital affiliated to Guangzhou Medical University from January 2021 to June 2024. The investigation obtained ethical approval from the Ethics Review Committee of Tongji Hospital in Shanghai (No. K-2020 021) and the Ethical Review Board of the Fourth Affiliated Hospital of Guangzhou Medical University (No. 2024-H-022). In alignment with national regulations and institutional guidelines, no written informed consent was solicited from participants. The inclusion parameters for the study were: (1) individuals diagnosed with AIS and administered IVT with tissue plasminogen activator; (2) subjects 18 years of age or older; (3) patients having blood glucose and TG measurements within 24 h post-admission; (4) patients who underwent a pre-treatment computed tomography (CT) scan; (5) patients who received cerebral computed tomography angiography (CTA) or magnetic resonance angiography (MRA), as well as a head magnetic resonance imaging (MRI) during their hospital stay; and (6) patients with comprehensive hospitalization and follow-up data available. The exclusion criteria comprised: (1) patients not receiving IVT treatment due to time constraints or medical contraindications; (2) patients undergoing concurrent mechanical thrombectomy; (3) patients with preexisting disability or prior stroke-related functional impairment (Baseline mRS ≥ 2); (4) patients with images of insufficient quality for accurate evaluation; and (5) patients with incomplete data or those who were unavailable for follow-up. Ultimately, 333 cases satisfied the inclusion criteria. A flowchart depicting the patient selection process was provided in Figure 1. Flow Chart of Patient Selection. AIS = Acute Ischemic Stroke, IVT = Intravenous Thrombolysis, Glu = Glucose, TG = Triglycerides, CTA = Computed Tomography Angiography, MRA = Magnetic Resonance Angiography
Multi-Dimensional Dataset
We meticulously compiled a comprehensive dataset on patient characteristics. This included demographic information (including age and sex); previous medical history (eg, stroke, diabetes); clinical indictors such as National Institute of Health stroke scale (NIHSS), 19 systolic and diastolic blood pressures; imaging biomarkers like the Alberta Stroke Program Early CT (ASPETS) Score to evaluate the size of infarction; and relevant laboratory markers (blood glucose levels, TG, total cholesterol, etc.).
The imaging assessment was conducted by a team of four neuroradiologists, each boasting at least 5 years of collective expertise. The ASPECTS represents a standardized ordinal scale that gauges the extent of early ischemic alterations on CT images, ranging from 0 (signifying complete infarction) to 10 (absence of infarction). 20 The Willis Artery status was determined as complete, partially complete, or incomplete via CTA or MRA. 21 Additionally, the degree of white matter hyperintensities was quantitatively measured using the Fazekas scale, with scoring ranging from 0 to 6 points. 22
SPAN-100 Index and THRIVE Score
The SPAN-100 index, a composite score derived from the sum of age and the NIHSS score, serves as a quantitative indicator of stroke severity, with higher values signifying more pronounced stroke manifestations. A threshold of ≥100 signifies a positive SPAN-100 index, indicating a greater level of stroke severity. 23 The Health Risks in Vascular Events (THRIVE) scoring system, on the other hand, encapsulates a comprehensive assessment of AIS patients by incorporating five key factors: NIHSS score, age, hypertension, diabetes, and atrial fibrillation. 24 This scoring system is designed in such a way that higher scores correspond to a more adverse prognosis.
TyG Index
The TyG index was calculated as: Ln [TG (mg/dL) × Glu (mg/dL)÷2]. The fasting venous blood samples were drawn to measure TG and Glu within the first 24 h on admission.
Outcomes
The short-term outcome of this examination was the emergence of END, evidenced by a nominal NIHSS score escalation of at least two points during the initial 24-h period. The long-term outcome was the subject’s functional status at the 90-day mark (90d-mRS) with a unfavorable outcome denoted by a mRS score within the range of 3 to 6 according to established criteria.1–3
Statistical Analysis
Participants were categorized into groups based on the presence or absence of END and poor 90-day prognosis. The NO-END group underwent a detailed subgroup analysis to identify prognostic risk factors. Categorical data were presented as frequencies and percentages, and differences across groups were evaluated via the chi-squared test. For continuous variables, medians along with interquartile ranges (IQRs) were reported, and the Mann-Whitney U test was employed for their analysis. Subgroup analysis, stratified by TyG index levels, employed the chi-square test to examine the discrepancies in the incidence of END and the likelihood of poor 90 day-prognosis between the Low-TyG and High-TyG groups. The Forward Stepwise Method of logistic regression and restricted cubic splines (RCS) was utilized to evaluate the association between the included factors and the risk of END as well as the likelihood of a poor prognosis. Nomogram models were constructed based on the outcomes of the multivariate logistic regressions. Receiver Operating Characteristic (ROC) analysis was conducted to assess the diagnostic efficacy, while Decision Curve Analysis (DCA) was employed to evaluate the clinical utility. Calibration curves were used to assess the accuracy of the models, and the Delong test was applied to compare the ROC curve performances. All statistical analyses were performed using R software version 4.0.3, with all P-values presented as two-tailed, and a P < 0.05 considered statistically significant.
Results
Baseline Characteristics in Patients within END and 90 Day-Prognosis Subgroups
Baseline Characteristics for END and 90-Day Prognosis Groups

IQR-interquartile range; NIHSS-National Institute of Health stroke scale; ASPECTS-Alberta Stroke Program Early CT Score; PT-Prothrombin Time; APTT-Activated Partial Thromboplastin Time; TT-Thrombin time; INR-International Normalized Ratio; PDW-Platelet Distribution Width; MPV-Mean Platelet Volume; NLR-Neutrophil-to-lymphocyte Ratio; K+-Potassium; Na+-Sodium; Cl-Chloride; Glu-Glucose on admission; HbA1c-Glycated hemoglobin; TG-Triglycerides; CHOL-Total cholesterol; LDL-Low-density lipoprotein; HDL-High-density lipoprotein; UA-Uric acid; TyG-Triglyceride-glucose index; END-Early neurological deterioration. THRIVE-The Health Risks in Vascular Events. For continuous variables, the Mann Whitney U test was used. For categorical variables, chi-square test was used. *Statistical significance: P < 0.05.
Subgroups Analysis Based on TyG Level for END and Poor 90 Day-Prognosis
In the absence of consensus cutoff values for the TyG index in stroke populations, patients were stratified into two groups based on the median TyG index value: the Low-TyG group (≤7.12) and the High-TyG group (>7.12). Importantly, individuals in the High-TyG group were identified to be at a significantly higher risk of developing into END (P = 0.0010, see Figure 2A) or poor 90-day prognosis (P = 0.0012, see Figure 2B). Figure 2C depicted the grouping flow of END occurrence and 90-day prognosis in patients from different TyG subgroups, intuitively showing that AIS patients post IVT in the High-TyG group were more likely to develop END and exhibit unfavorable prognosis. Approximately 39.67% patients with END experienced a poor 90-day outcome, compared to 24.83% of those from NO-END group. Comparisons of the Occurrence of END (A) and Poor 90-Day Prognosis (B) Between the Low-TyG and High TyG Groups. Sankey Diagram Illustrating the Distribution and Interconnections Among TyG Index Groups, END Categories, and 90-Day Prognosis Categories (C). TyG = Triglyceride-Glucose Index; END = Early Neurological Deterioration; NO-END = Without Early Neurological Deterioration; Good = Good 90-Day Prognosis; Poor = Poor 90-Day Prognosis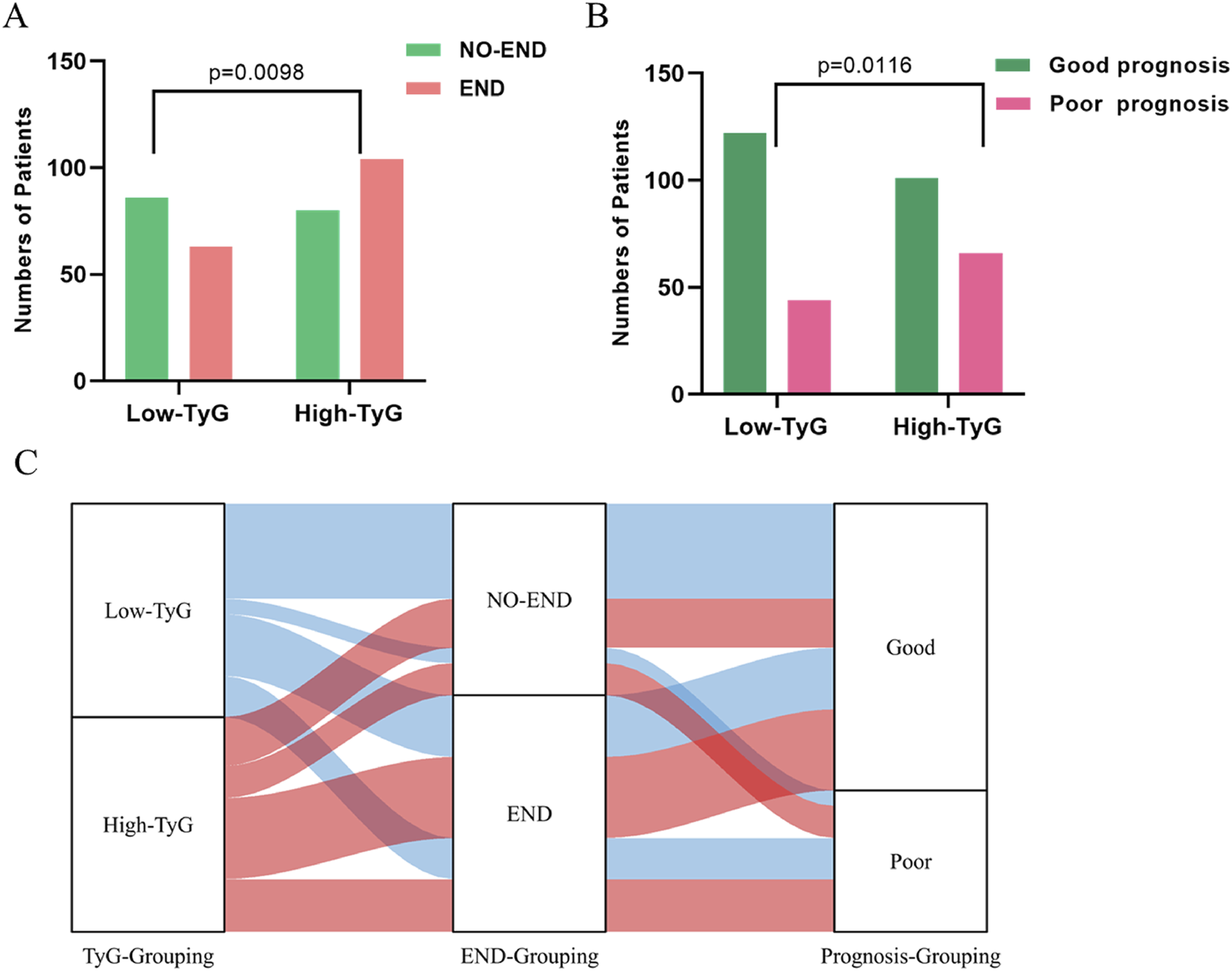
Early Neurological Deterioration
Predictive Model for END Prediction (Nomogram END Model)
Due to the significant multicollinearity observed between triglycerides (TG) and the TyG index (Pearson r = 0.704, P < 0.001), TG was excluded from the subsequent multivariate logistic regression analysis. The analysis revealed several independent predictors of END, as outlined below: the NIHSS score at admission (OR 0.914 [95% CI 0.879-0.950], P < 0.001), DNT (OR 1.008 [95% CI 1.001-1.015], P = 0.035), ASPECTS score (OR 0.812 [95% CI 0.682-0.966], P = 0.019), Willis Artery integrity (OR 1.658 [95% CI 1.145-2.399], P = 0.007), K+ (OR 1.714 [95% CI 1.053-2.791], P = 0.03), and TyG index (OR 1.850 [95% CI 1.233-2.755], P = 0.003). The results of this analysis were presented in Figure 3A, and these factors were then integrated into the Nomogram END Model (Figure 3B). Multivariable Logistic Regression Analyses With Respective Odds Diagrams Assessing Independent Factors of END (A). Nomogram END Model for Predicting the Probability of END at 24 h (B). Calibration Plots of the Nomogram (C). The AUCs of Willis, DNT, TyG, K+, NIHSS Score, ASPECTS and Nomogram Model to Predict END for Patients With AIS Following IVT (D). Decision Curve Analysis Evaluating the Independent Predictors and the Nomogram Model (E). DNT-Door-To-Needle Time; TyG-Triglyceride-Glucose Index; K+-Serum Potassium Level; NIHSS-National Institute of Health Stroke Scale; ASPECTS-The Alberta Stroke Program Early CT Score; IVT-Intravenous Thrombolytic Therapy
Results of DeLong’s Test for ROC Curves Between the Nomogram END Model and Independent Predictors for END

NIHSS-National Institute of Health stroke scale; TyG-Triglyceride-glucose index; DNT-Door-to-needle time; ASPECTS-Alberta Stroke Program Early CT Score.
Potential Nonlinear Analysis for Prognostic Factors of END
The analysis employed restricted cubic spline models (RCS) to elucidate the possible nonlinear correlations between the occurrence of END and pertinent indicators. Our findings indicated a nonlinear declination in the risk of END for patients, particularly when NIHSS surpassed 5, as demonstrated in Figure 4A (P < 0.001). A significant nonlinear reduction in the likelihood of END was also observed when the ASPECTS was greater than 6 (P = 0.047), as depicted in Figure 4B. Furthermore, there was a linear increment in the risk of developing into END as the TyG index,K+ level and DNT increased. Cubic Regression Model Illustrating the Relationship Between the National Institute of Health Stroke Scale (NIHSS) Score at Admission and the Door-To-Needle Time (DNT) in Predicting the Occurrence of END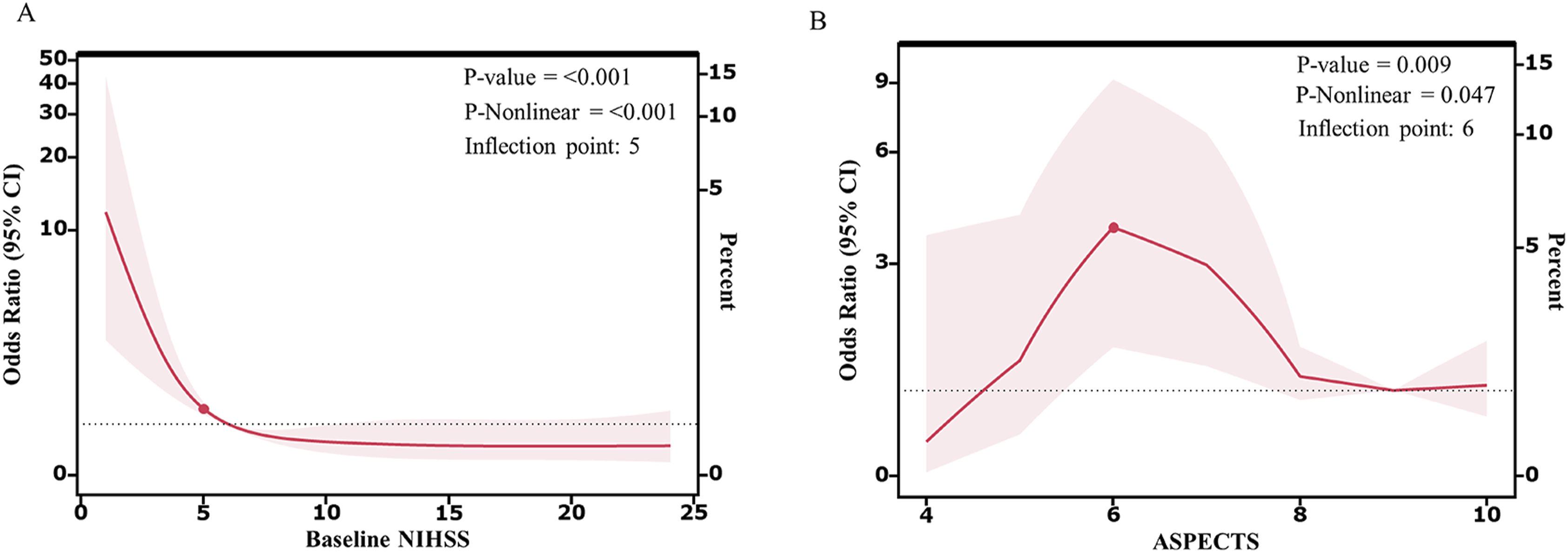
90-Day Prognosis
Predictive Model for 90 Day-Prognosis Prediction (Nomogram Prognosis Model)
Significant multicollinearity was observed between the TyG index and its component variables - triglyceride (TG: Pearson r = 0.704, P < 0.001) and glucose (Glu: Pearson r = 0.549, P < 0.001). Therefore, both TG and Glu were excluded from the subsequent multivariate logistic regression analysis. The regression revealed several independent predictors of a poor prognosis, including the NIHSS score at the time of admission (OR 1.267 [95% CI 1.190-1.349], P < 0.001), TyG index (OR 1.752 [95% CI 1.093-2.808], P = 0.02), END (OR 0.156 [95% CI 0.075-0.325], P < 0.001), and a history of hypertension (OR 1.267 [95% CI 1.190-1.349], P < 0.001). The findings were presented in Figure 5A, and these significant factors were integrated into the Nomogram Prognosis Model (Figure 5B). Multivariable Logistic Regression Analyses, Accompanied by Odds Ratio Plots, Examining the Independent Predictors of Poor 90-Day Prognosis (A). Nomogram Prognosis Model Designed to Estimate the Probability of a Poor 90-Day Outcome (B). Calibration Curve of the Nomogram, Validating its Predictive Accuracy (C). The AUCs for the NIHSS, Hypertension, END, TyG and the Nomogram Model in Predicting Poor Outcomes for Patients With Acute Ischemic Stroke (D). Decision Curve Analysis is also Presented to Evaluate the Clinical Utility of the Independent Predictors and the Nomogram Model (E). NIHSS = National Institute of Health Stroke Scale; END = Early Neurological Deterioration; TyG = Triglyceride-Glucose Index
Results of DeLong’s Test for ROC Curves Between the Nomogram Prognosis Model and Independent Predictors for 90-Day Prognosis
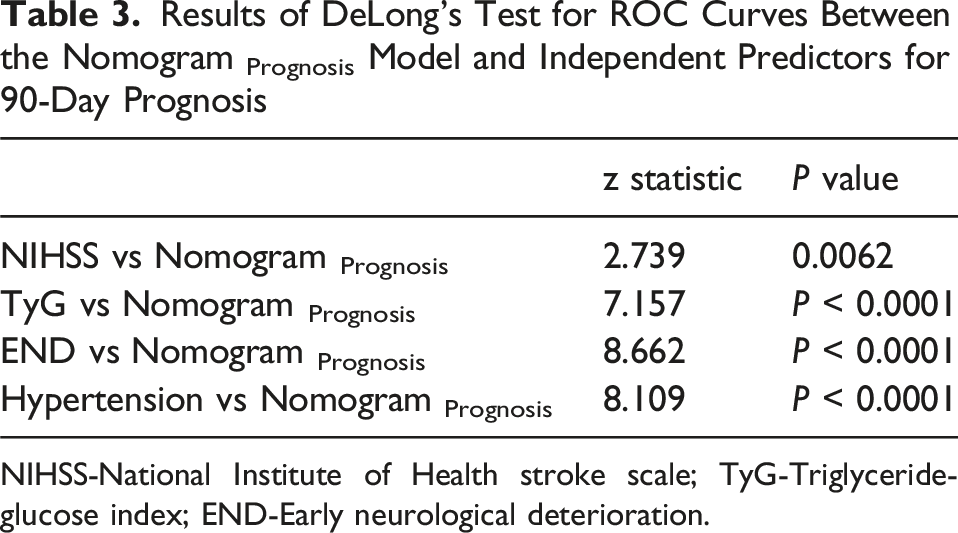
NIHSS-National Institute of Health stroke scale; TyG-Triglyceride-glucose index; END-Early neurological deterioration.
Potential Nonlinear Analysis for Predictive Factors of 90-Day Prognosis
We further performed RCS analyses to assess the associations of NIHSS score and TyG index with poor 90-day outcomes in AIS patients. The analysis revealed a significant non-linear relationship between NIHSS score and poor outcome risk (P = 0.002; Figure 6A), with a sharp increase in risk observed when NIHSS scores exceeded 6. In contrast, the TyG index exhibited a linear positive association with poor outcome risk (P = 0.630), indicating that higher TyG values corresponded to progressively greater risks. (A)Figure Depicting a Cubic Regression Analysis Showing the Relationship Between the National Institute of Health Stroke Scale (NIHSS) Score at Admission and the 90-Day Prognosis Outcomes. (B) The AUCs for SPAN-100 Binary, SPAN-100, THRIVE and Nomogram Prognosis Model, Evaluating Their Performance in Identifying Poor Prognostic Risks for Patients With Acute Ischemic Stroke
Comparisons with the Prior Classic Scoring Tools
To rigorously evaluate the prognostic performance of our newly developed Nomogram Prognosis model, we established reference 90-day poor outcome models based on the established SPAN-100 and THRIVE scoring systems within our AIS study population. The distribution of patients’ SPAN-100 and THRIVE scores was presented in Table 1. We developed three distinct models: one utilizing a binary variable of the SPAN-100 (SPAN-100 Binary), another employing the continuous variable version of the SPAN-100 (SPAN-100), and the third using the THRIVE score (THRIVE). In predicting the 90-day poor prognosis, the SPAN-100 Binary yielded an AUC of 0.563 (95% CI, 0.529-0.596); the SPAN-100 achieved an AUC of 0.698 (95% CI, 0.637-0.760); THRIVE followed closely with an AUC of 0.747 (95% CI, 0.692-0.802); and the Nomogram Prognosis model demonstrated the highest prognostic accuracy with an AUC of 0.849 (95% CI, 0.807-0.892) (Figure 6B). The Delong test confirmed that there were statistically significant differences in the performance of the various ROC curves (P < 0.05), thus displaying the added value of our Nomogram Prognosis model in prognostic evaluation.
90d-Prognostic Subgroup Analysis in NO-END Group
Results of Univariable Regression Analysis for Predictors of Poor Prognosis in Patients From NO-END Group
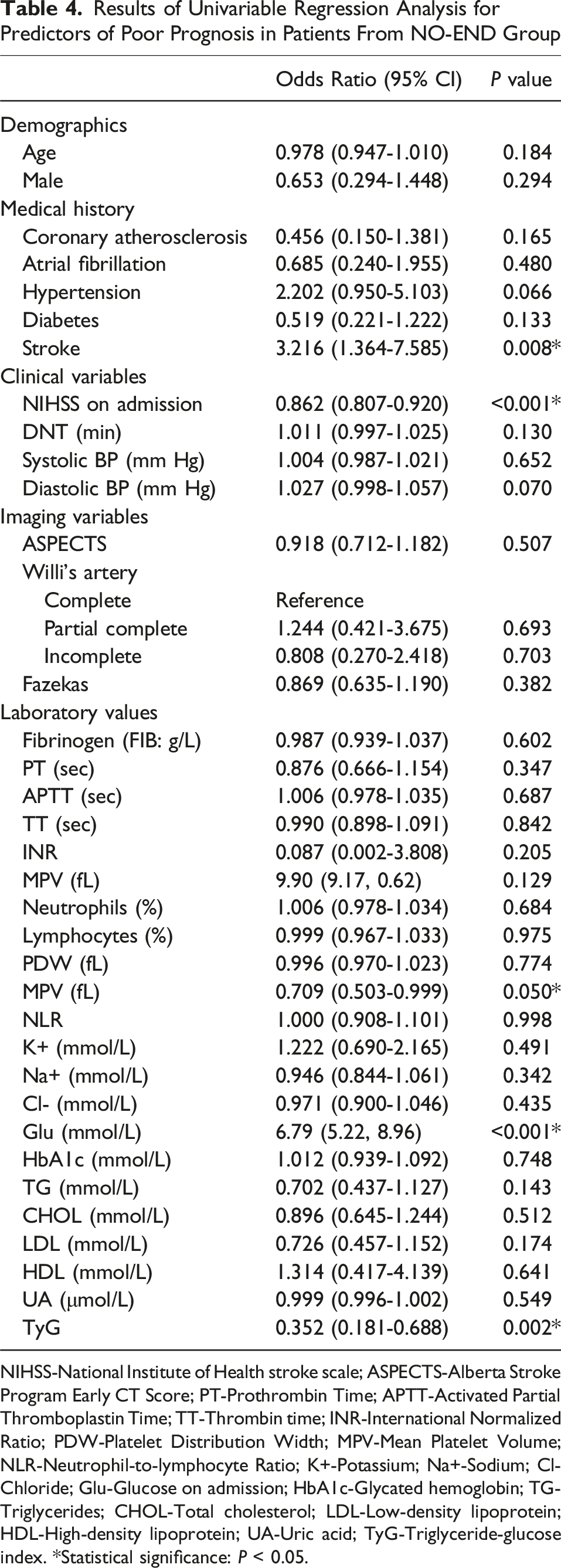
NIHSS-National Institute of Health stroke scale; ASPECTS-Alberta Stroke Program Early CT Score; PT-Prothrombin Time; APTT-Activated Partial Thromboplastin Time; TT-Thrombin time; INR-International Normalized Ratio; PDW-Platelet Distribution Width; MPV-Mean Platelet Volume; NLR-Neutrophil-to-lymphocyte Ratio; K+-Potassium; Na+-Sodium; Cl-Chloride; Glu-Glucose on admission; HbA1c-Glycated hemoglobin; TG-Triglycerides; CHOL-Total cholesterol; LDL-Low-density lipoprotein; HDL-High-density lipoprotein; UA-Uric acid; TyG-Triglyceride-glucose index. *Statistical significance: P < 0.05.
Discussions
The triglyceride-glucose (TyG) index has emerged as an innovative biomarker for insulin resistance (IR) assessment. Growing evidence supports its prognostic value in AIS, demonstrating consistent associations with stroke recurrence, neurological deterioration, poor functional outcomes, and all-cause mortality.13–18 A prospective observational study 14 established the TyG index as an independent risk factor for both cervicocerebral atherosclerosis progression and post-ischemic major adverse cardiovascular events. Additional studies have shown that elevated TyG index values correlate with increased in-hospital mortality in severe stroke cases 15 and higher rates of early stroke recurrence. 16
Our dataset revealed that patients with a high TyG index are at greater risk of experiencing END and exhibit a higher risk of poor 90-day prognosis for AIS post IVT therapy. Baixiang Zhang and colleagues demonstrated that increasing TyG index was associated with a higher risk of END and a lower probability of early neurological improvement (ENI) in patients with AIS treated with intravenous thrombolysis. 13 The study by Emma M. S. Toh et al further highlighted that TyG index was significantly associated with 90-day mortality (OR: 2.12, 95% CI: 1.39-3.23, P = 0.001), poor functional outcome (OR: 1.41, 95% CI: 1.05-1.90, P = 0.022), and negatively associated with ENI (OR: 0.68, 95% CI: 0.52-0.89, P = 0.004). 14 Our research expanded upon previous findings by exploring the associations between the TyG index and END, as well as the 90-day outcomes. We furtherly enhanced the analysis by incorporating multi-dimensional imaging variables and laboratory metrics, and by creating predictive nomogram models within a well-defined patient cohort. In addition, the study conducted by Jia Wang and colleagues focused on a cohort of patients who were not administered intravenous thrombolysis, yet their findings aligned closely with our own. 15 These findings indicated that TyG can be served as a potent predictive indicator for AIS patients across various treatment modalities.
This study revealed an intriguing pattern: while diabetes history prevalence showed no significant differences between the END cohort and 90-day prognosis cohort, substantial differences emerged in TyG index values. We propose two explanations for this finding: First, real-time biochemical markers like the TyG index provide more accurate reflections of patients’ current metabolic status than historical medical records. Second, insulin resistance (IR), characterized by reduced sensitivity and responsiveness to insulin, has been established as a pivotal indicator of type 2 diabetes mellitus (T2DM). IR often occurring several years before the onset of the disease. 25 These results highlight the TyG index’s superior prognostic value over traditional diabetes history.
The TyG index serves as a reliable and accessible surrogate marker for IR in routine clinical practice, the potential mechanisms by which the IR influences the onset of END and 90-day prognosis are multifaceted: Firstly, IR may elevate the risk of END and poor prognosis by accelerating atherosclerosis, interfering with platelet function, and exacerbating modifiable risk factors for AIS, such as hypertension, diabetes, and dyslipidemia. 26 Secondly, inflammation plays a pivotal role in the pathogenesis of brain injury associated with AIS. Research indicated that IR can stimulate the recruitment of monocytes and activate proinflammatory macrophages, leading to the production of monocyte chemoattractant protein. This interaction between inflammation and IR creates a vicious cycle that can adversely affect the occurrence of END and the prognosis of AIS. 27
Our study also identified several other risk factors that are closely associated with the development of END, including NIHSS score, ASPECTS score, DNT, potassium levels, and the integrity of the Willis artery. A comprehensive review by Benjamin D. has underscored the potential significance of potassium intake in both the etiology of stroke and its management, as indicated by a convergence of evidence from animal research, epidemiological studies, and clinical trials. 28 The ASPECTS score functions as a semi-quantitative index of infarct size and emerges as a pivotal prognostic marker for AIS. 29 In consonance with this, Vynckier et al have reported that a lower NIHSS score at hospital admission (aOR 0.81, P = 0.006), the presence of capsular warning syndrome (aOR 7.00, P < 0.001), ventral pontine infarction (aOR 3.49, P = 0.008), and imaging-confirmed hypoperfusion lesions (aOR 2.13, P = 0.026) are predictive of END in individuals with lacunar stroke. 30 Nonetheless, the aforementioned study merely stratified risk factors without developing a predictive model. Drawing on this, we conducted a comprehensive analysis of imaging, clinical, and laboratory parameters to construct an objective predictive model for END with an AUC of 0.746, which held practical utility in clinical settings.
Building on our END prediction, we further forecasted the 90-day prognosis. Additional predictors of the 90-day outcome include the baseline NIHSS score, blood glucose levels, the occurrence of END, and hypertension—factors that aligned with a broad spectrum of scientific literature.31,32 We have developed another Nomogram Prognosis model with the objective of accurately predicting the long-term functional outcomes for AIS patients treated with IVT. The nomogram model created by Yinglei Li and co-workers included analogous predictors (such as NIHSS, DNT, and baseline blood glucose) for 90-day prognosis and demonstrated a robust diagnostic performance, with an AUC of 0.822 (95% confidence interval: 0.769 to 0.875). 33 Ke-Jia Zhang and colleagues have devised a personalized nomogram model for forecasting poor functional outcomes, incorporating BNP, NIHSS scores, and baseline glucose levels, which yielded an AUC of 0.764. 34 Our study distinguished itself from earlier researches by integrating a more extensive range of variables, particularly the TyG index and neuroimaging data, leading to the predictive nomogram models showing superior performance with AUC of 0.849. Moreover, our results revealed that the Nomogram Prognosis surpassed the performance of the SPAN-100 and THRIVE scores, which were established and validated as dependable prognostic indicators for AIS patients.
The subgroup analysis revealed that mean platelet volume (MPV) served as a prognostic indicator for 90-day outcomes in patients who have not experienced END. An elevated MPV can enhance platelet adhesion and aggregation, promoting atherosclerosis and increasing the likelihood of thrombotic events following plaque rupture, indicative of a more advanced pre-thrombotic condition.35–37 Concurrently, higher MPV levels are linked to reduced bleeding times, which may diminish the therapeutic effectiveness of antiplatelet and thrombolytic treatments.38,39 Therefore, for patients without END, it is crucial to closely monitor MPV levels and to identify any early signs of adverse prognostic developments.
Our study has several limitations. Firstly, the scope of our population sample may be limited, and our findings would benefit from broader validation. Secondly, the study did not comprehensively document the factors such as the pre-existing use of lipid-lowering medications or the presence of complications. The omission of these potential confounders from our statistical analysis may have contributed to bias in our results. Thirdly, the retrospective design of our study inherently possessed certain methodological shortcomings.
Conclusion
The TyG index serves as a surrogate marker for IR and is prognostically linked to END and poor 90-day outcomes in patients with AIS treated with IVT. By integrating the TyG index with a panel of other complex variables within nomograms, the accuracy of predicting END and poor long-term prognosis in AIS patients post thrombolysis has been significantly enhanced. These predictive models were developed to offer evidence-based decision support, particularly for AIS patients presenting near the terminal time window for IVT or those with borderline contraindications.
Footnotes
Ethical Considerations
The research obtained ethical approval from the Ethics Review Committee of Tongji Hospital in Shanghai (No. K-2020 021) and the Ethical Review Board of the Fourth Affiliated Hospital of Guangzhou Medical University (No. 2024-H-022).
Author Contributions
Lai Wei, study concept and design, analysis and interpretation of data, drafting the manuscript. Zhihua Wu, acquisition and interpretation of data, manuscript revision. Xiang Zhou, acquisition and interpretation of data, manuscript revision. Zhifeng Liu, acquisition and interpretation of data, manuscript revision. Xiaoyan Wu, acquisition and interpretation of data, manuscript revision. Kangwei Zhang, methodology, analysis and interpretation of data, manuscript revision, critical review of manuscript for intellectual content. Peijun Wang, analysis and interpretation of data, manuscript revision, funding acquisition, critical review of manuscript for intellectual content. Haoyi Ye, study concept and design, interpretation of data, funding acquisition, critical review of manuscript for intellectual content. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Major project of the National Natural Scientific Foundation of China (Grant No. 82227807), the Research Project of Shanghai Municipal Health Commission (Grant No. 2022JC017), the National Key R&D Program of the Ministry of Science and Technology (Grant No. 2022YFC2009904), the Guangzhou Municipal Science and Technology Project (Grant No. 2025A03J4017) and the Research Project of Guangdong Health Information Net Association (Grant No. HX-202407-0002).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
