Abstract
The field of cancer neuroscience has rapidly evolved, shedding light on the complex interplay between the nervous system and cancer, with a particular focus on the relationship between the central nervous system (CNS) and gliomas. Recent advancements have underscored the critical influence of CNS activity on glioma progression, emphasizing the roles of neurons and neuroglial cells in both the onset and evolution of gliomas. This review meticulously explores the primary communication pathways between the CNS and gliomas, encompassing neuro-glioma synapses, paracrine mechanisms, extracellular vesicles, tunneling nanotubes, and the integrative CNS-immune-glioma axis. It also evaluates current and emerging therapeutic interventions aimed at these pathways and proposes forward-looking perspectives for research in this domain.

Keywords
Introduction
Gliomas are primary brain tumors that arise from glial cells, the supportive cells of the nervous system. These tumors are classified based on the type of glial cell involved in the malignancy, including astrocytes, oligodendrocytes, and ependymal cells. Astrocytomas originate from astrocytes, which provide structural and metabolic support to neurons.1,2 Glioma represents one of the most challenging malignancies in neuro-oncology.3,4 Characterized by heterogeneous cellular composition and a high degree of invasiveness, gliomas pose significant therapeutic challenges, primarily due to their intricate interaction with the tumor microenvironment (TME), in which neurons and glial cells constitute significant components.5,6 The complexity of these tumors, especially high-grade variants like glioblastoma multiforme (GBM), is further compounded by their remarkable ability to adapt and resist current treatment modalities.7-9 Patients with high-grade gliomas confront alarmingly high rates of mortality and disability, coupled with an exceptionally grim prognosis. 10 In conclusion, for high-grade gliomas, there is still a lack of exhilarating breakthroughs in both basic research and clinical treatment. 10 This prompts a reconsideration of the mechanisms driving the development of glioma from a novel standpoint.
In the past few years, cancer neuroscience, a burgeoning discipline aiming to elucidate the intricate interplay between the nervous system and cancers, has sparked immense interest among researchers.11-14 People gradually recognize that neuronal activity exerts a substantial role in regulating cancer progression, especially for brain tumors like glioma. The central nervous system (CNS) is primarily composed of neurons and neuroglial cells. Recent research has elucidated that these cellular components significantly contribute to the pathogenesis of gliomas. 15 Neurons can influence glioma cells by forming synaptic connections or by secreting signaling molecules that act in a paracrine manner on receptors on glioma cells, ultimately regulating the growth and invasion of gliomas.11-14 In addition to neurons, normal astrocytes within the glioma microenvironment can secrete various chemokines to modulate the microenvironment, such as by recruiting immune cells, thereby impacting glioma progression. 16 The neuroglial cells mainly include astrocytes, microglial cells, and oligodendrocytes. While the majority of research has traditionally concentrated on neurons, it is becoming increasingly evident that neuroglial cells also play indispensable regulatory roles in the physiological processes underlying glioma development. These cells, far from being mere supportive elements, actively participate in the modulation of tumor progression, influencing both the TME and the dynamic interactions within. 16 Astrocytes, comprising approximately 50% of brain cells, have been demonstrated to foster glioma growth. 17 The regulatory mechanisms of CNS over gliomas demand specialized pathways for execution, a fact worthy of recognition. However, to date, there has been no comprehensive review solely dedicated to systematically summarizing and analyzing these pathways. Additionally, given the widespread attention and ongoing depth of research in cancer neuroscience, new knowledge and perspectives continue to emerge. Hence, timely updates are essential to reflect the evolving landscape. Therefore, in this review, we synthesize the latest research findings to systematically delineate the primary communication pathways governing CNS-glioma interactions.
This review aims to dissect these intricate communication pathways, providing a comprehensive overview of the current understanding and the therapeutic implications therein. The CNS-glioma communication pathways primarily include synapses, paracrine pathways, extracellular vesicles, tunneling nanotubes, CNS-immune-glioma axis. More importantly, we provide a concise summary of existing and potential treatment strategies, aspiring to establish a foundation for future clinical translation. Therefore, this review is not just an academic exercise, it bears direct implications for patient outcomes.
CNS-glioma communication pathways
Previous studies have confirmed the existence of diverse communication pathways between CNS and glioma cells. Through these pathways, neural activity facilitates substance exchange and information communication with glioma cells, thereby influencing glioma development. 18 The evident impact of these communication pathways underscores their crucial role in glioma development.
Synapses
Synapses refer to structures that exist between two neurons or between a neuron and other effector cells for the transmission of information. The predominant type is chemical synapses, although there are also electrical synapses known as gap junctions.
19
As research on gliomas progresses, synaptic structures have also been found between glioma cells and other tissue cells.
20
Monje et al,
21
through single-cell transcriptomic analysis, discovered extensive expression of glutamate receptor genes and synaptic-related genes in malignant glioma cells. Further, they confirmed that neurons can indeed form structural synapses with glioma cells.21,22 Specifically, under electron microscopy, researchers clearly and intuitively observed the presence of synaptic ultrastructures in human glioma tissues. To determine whether these synaptic structures possess electrophysiological functions, researchers stereotactically injected GFP-labeled glioma cells into the CA1 region of the hippocampal circuit in mice. After sufficient engraftment and growth, whole-cell patch-clamp recordings were performed on GFP + glioma cells in acute hippocampal slices. Using various pharmacological tools, it was demonstrated that the excitatory postsynaptic currents (EPSCs) of glioma cells are dependent on neuronal action potentials and are mediated through AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) receptors.21,22 Overall, the researchers provided strong evidence for the existence of synaptic structures in gliomas through single-cell transcriptomic data from patient-derived tumor tissues and electron microscopy experiments. More importantly, using a preclinical animal model in which human glioma cells were xenografted into the mouse brain, they employed electrophysiology, two-photon calcium imaging, and optogenetics to robustly demonstrate that these glioma-neuron synaptic structures have intact electrophysiological functions.21,22 This groundbreaking discovery provided evidence that synaptic connection represents a vital pathway for communication between healthy neural cells and glioma cells (Figure 1). Synaptic structure acts as a key communication pathway between CNS and glioma. The neuron-glioma synapse is a vital structure enabling neurons to regulate glioma progression. The gap junction, also referred to as the electrical synapse, significantly contributes to the formation of glioma cell network and communication between astrocytes and glioma cells.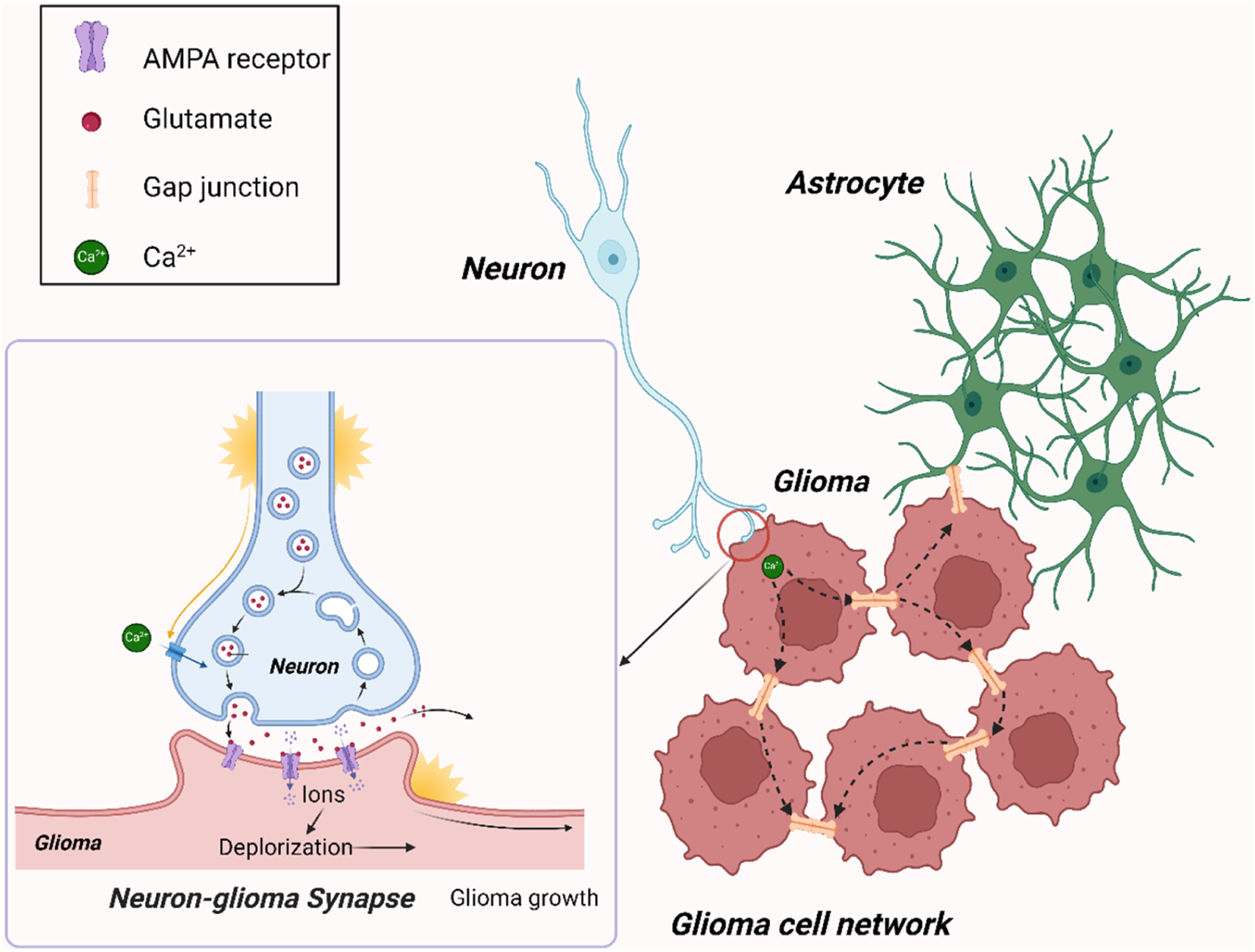
In neuron-glioma synapse (NGS), neurons serve as the presynaptic component, while glioma cells act as the postsynaptic component, receiving signals from neurons. It is worth noting that synaptic structures between neurons and glioma cells with the opposite direction have not been discovered to date. 23 Neuronal activity can stimulate the presynaptic membrane to release neurotransmitters, primarily including glutamate. Within NGS, these neurotransmitters engage with the calcium-permeable AMPA receptors on glioma cells, initiating a depolarization response (Figure 1). It was confirmed that excitatory postsynaptic currents in glioma cells are contingent upon neuronal action potentials. This event activates downstream signaling pathways, ultimately enhancing the proliferation and invasion of glioma cells21,22 (Figure 1). However, the precise mechanisms by which depolarization facilitates the progression of glioma remain to be fully elucidated. 24 Excitingly, when AMPA receptors are blocked, glioma growth is significantly inhibited.21,22,25 This indicates that synapse, as an essential communication pathway between neurons and gliomas, deserves more attention.
Outside of the chemical synapses formed between neurons and glioma cells, there also exist what are known as electrical synapses, or gap junctions, between healthy astrocytes and glioma cells (Figure 1). 26 Gap junctions are vital structures for direct substance exchange and information communication between cells, with the key molecules comprising this structure being connexin (CX), where the most common and crucial one is CX43. 27 Through these structures, cells can achieve bidirectional and rapid signal transmission. Research has reported that knocking down the expression of CX43 in glioma cells blocks the formation of gap junction channels with astrocytes, significantly inhibiting the invasion of glioma cells. 28 As for the underlying mechanisms, some discoveries have been made to date. Astrocytes, integral to CNS and significant players within the TME of gliomas, can facilitate the intercellular propagation of calcium signals with glioma cells via gap junctions. 29 This interaction, to some extent, promotes the growth and migration of gliomas. 30 An in vitro study also demonstrated that the gap junctions between glioma and astrocytes significantly enhance glioma invasiveness. 31 Additionally, this research found that the gap junctions between glioma and astrocytes allow for the passage of microRNAs (miRNAs), indicating that miRNAs can be transferred from glioma cells to astrocytes through these gap junctions. Specifically, after miR-5096 was transferred from glioma cells to astrocytes, the invasiveness of glioma cells increased significantly. 31 This also suggests that gap junctions play a crucial role in reshaping the glioma microenvironment. Gap junctions can also form between glioma cells. Glioma cells can form gap junctions by membrane protrusions, creating a glioma cell network (Figure 1). These protrusions are known as tumor microtubes (TMs).32,33 Calcium ions or other toxic metabolic products produced by glioma cells after receiving radiotherapy and chemotherapy can diffuse within the glioma cell network through gap junctions, thus diluting toxicity and avoiding cell death. 33 Therefore, this network is a significant factor contributing to radiotherapy and chemotherapy resistance, as well as the suboptimal outcomes of surgical treatments.33,34 Interestingly, glioma cells that are not connected to other cells do not exhibit significant radiotherapy and chemotherapy resistance. Since glioma cells can form synapse-like structures with neurons or glial cells, the glioma cell network can integrate into neuronal networks and astrocytic networks, receiving extensive regulation from the CNS. Independent of NGS, neuronal activity can increase the extracellular potassium concentration of glioma, leading to the depolarization of glioma cells, which subsequently promotes glioma growth. 21 However, the mechanistic details within this process remain obscure. Strikingly, this process is markedly potentiated by the presence of gap junctions within the glioma cell network. In fact, current research suggests that since gap junctions are a core mechanism of glioma networks, the growth rate of gliomas significantly decreases when treated with gap junction inhibitors, such as meclofenamate.21,22 This suggests that targeting core components of electrical synapses, such as CX43, holds important clinical value for improving the GBM treatment.
Currently, there is no definitive evidence indicating the formation of glutamatergic connections resembling NGS between healthy astrocytes and glioma cells. But a study has revealed a unique subset of astrocytes, possibly amplifying the functions of astrocytes. 35 These astrocytes can release glutamate through a neuron-like Ca2+-dependent exocytosis mechanism, influencing neuronal circuits, reminiscent of glutamatergic synaptic structures. 35 This groundbreaking discovery indicates the possibility that there may exist a glutamatergic connection like NGS linking astrocytes and glioma cells, which needs further validation. Therefore, the communication pathways between gliomas and CNS may be more complex than initially envisioned.
The degree of involvement of synaptic signaling is closely associated with the clinical characteristics of glioma patients. One study categorized glioma samples into low-neural and high-neural subtypes based on the methylation profiles of neural cells. 36 The study found that high-neural gliomas exhibit upregulated expression of genes related to synaptic integration, and patients with this subtype had significantly shorter overall survival and progression-free survival. Similarly, another preclinical study using a mouse model discovered that discrete glioma cells located at the tumor periphery can mimic neuronal characteristics, form synaptic structures with surrounding normal neurons, and thereby invade and spread throughout the brain. 29 Overall, synaptic structures are crucial for communication between the CNS and glioma cells. Neurons or neuroglial cells establish connections with cancer cells through this structure, regulating proliferation, invasion, and migration.
Paracrine pathways
The synaptic structure functions as a pivotal conduit for communication between the CNS and gliomas, albeit it is not the sole pathway. Neurons, being integral constituents of the TME, possess the ability to modulate the secretion of signaling molecules, either from their own sources or from other cells. 37 Enhanced neuronal activity can stimulate the production of these signaling molecules, capable of infiltrating the TME and subsequently influencing glioma progression. Such a regulatory mechanism is termed neuronal activity-dependent paracrine pathways. Paracrine signaling encompasses the release of cellular products that do not enter the bloodstream but instead exert their effects on neighboring target cells through diffusion, enabling intercellular communication. 38 This mode of signal transmission can modulate the growth and functionality of the target cells. 39
Neuroligins (NLGN) represent a family of trans-synaptic cell adhesion molecules that bridge the synaptic cleft. They interact with presynaptic Neurexin, playing a crucial role in modulating synaptic functions.
40
Neuroligin 3 (NLGN3) is a neuronal activity-dependent signaling molecule that plays a significant role in the development of gliomas.
41
NLGN3 is primarily secreted by neurons and oligodendrocyte precursor cells as a mitogen, with the latter being the primary source.
42
Neuronal activity promotes the production of soluble NLGN3 (sNLGN3). Venkatesh et al41,43 have found that during neuronal excitation, the A Disintegrin and Metalloproteinase 10 (ADAM10) is released, which acts on NLGN3 proteins expressed on postsynaptic cells. This enzymatic action results in cleavage and the release of sNLGN3 into the extracellular matrix. Subsequently, glioma cell receptors recognize sNLGN3, promoting focal adhesion kinase (FAK) phosphorylation and activating a series of signaling pathways downstream of FAK, which includes SRC kinase cascade, PI3K-mTOR cascade, and SHC-RAS-RAF-MEK-ERK cascade (Figure 2). The activation of these pathways consequently leads to glioma progression. Beyond activating oncogenic pathways, NLGN3’s action on glioma cells also involves the upregulation of a multitude of synapse-related genes, including NLGN3, which promotes the formation of NGS.
43
This intriguing finding highlights the pivotal role of NLGN3 in bridging the two neuro-glioma communication pathways: the synaptic and paracrine pathways. In short, NLGN3 exerts its effects on glioma cells through a paracrine mechanism, resulting in cancer-promoting outcomes. This study also identified a significant negative correlation between NLGN3 expression levels and patient survival rates. In addition to facilitating glioma progression, neuronal activity also drives glioma initiation through paracrine pathways, with NLGN3 is a pivotal molecular player. For individuals with the neurofibromatosis 1 (NF1) cancer predisposition syndrome, optic pathway gliomas (OPGs) are relatively common tumors, with the majority being diagnosed during childhood.44,45 In the mouse model of OPGs harboring NF1 mutations, Pan et al observed that light stimuli lead to increased activity in retinal ganglion cells (RGCs), which in turn promotes the secretion of ADAM10 from the optic nerves. This release of ADAM10 subsequently boosts the production of NLGN3, playing a pivotal role in the initiation, progression, and maintenance of OPGs (Figure 2). NLGN3 is considered crucial in this mechanistic pathway.
46
This underscores the significance of the paracrine pathway as an important pathway through which neurons regulate glioma progression. Paracrine pathways mediate the CNS-glioma interaction. Upon stimulation, neurons release the ADAM10 protease, which targets NLGN3 on the postsynaptic cells at synapses. This enzymatic process cleaves NLGN3 into sNLGN3, which is then released into the extracellular matrix. Glioma cells, through their receptors, detect sNLGN3, initiating downstream signaling pathways that encourage tumor growth. Similarly, BDNF activates the TrkB receptor on glioma cells, leading to the activation of oncogenic signaling pathways and the formation of NGS. Moreover, olfactory and visual stimuli can accelerate glioma progression via paracrine factors like IGF-1 and NLGN3, highlighting the complex interplay between CNS activity and tumor development.
In fact, within the paracrine pathway, neurons or other neural cells can secrete various signaling molecules.11,41,43,47 Apart from NLGN3, substances such as brain-derived neurotrophic factor (BDNF) also play important roles. 41 In normal physiological process, BDNF functions by binding to its cognate receptor, tyrosine kinase B (TrkB). Upon this interaction, BDNF initiates a series of intracellular signaling cascades, including the activation of pathways such as extracellular signal-regulated kinase (MEK-ERK), phosphatidylinositol 3-kinase (PI3K), and phospholipase C (PLC). These signaling pathways are instrumental in fostering neuronal survival, augmenting synaptic plasticity, and facilitating neurogenesis. 48 The secretion of BDNF is highly dependent on neuronal activity. 49 However, a recent study has found that glioma cells hijack the neurophysiological functions of BDNF to promote their own progression. 50 Specifically, glioma cells harness neuronal activity-regulated BDNF signals via TrkB receptors, initiating three principal signaling cascades: the MAPK–ERK, PI3K–AKT, and CAMKII calcium pathways. This interplay directly fosters glioma progression through the three cancer-promoting pathways. Concurrently, BDNF-TrkB signaling facilitates the trafficking of AMPA receptors to the postsynaptic membrane via CAMKII calcium signaling, thereby contributing to the establishment of a more extensive and robust network of NGS (Figure 2). This discovery underscores a profound interdependence between paracrine signaling and synaptic dynamics within the neuro-glioma dialogue. Consequently, both NLGN3 and BDNF play a synergistic role in the formation and strengthening of NGS. Since BDNF is released by both neurons and neuroglial cells, it remains unclear which source is more important for glioma development. Furthermore, Chen et al 47 found that olfactory sensory neuron prompts second-order neurons (mitral/tufted cells) to release insulin-like growth factor-1 (IGF-1). IGF-1, acting through the paracrine pathway, influences glioma cells, thereby initiating processes of cell malignancy and proliferation (Figure 2). However, the specific mechanisms through which IGF-1 signaling facilitates the advancement of glioma remain unclear.
In addition to paracrine signals regulated by neurons, research has revealed that under hypoxic conditions, astrocytes appear to influence the development of gliomas through paracrine signaling as well. 51 Specifically, when cocultured with the purified extracellular matrix produced by hypoxia-induced astrocytes, the proliferation of gliomas is significantly enhanced. While the specific substances involved and the mechanisms remain unknown, this undoubtedly highlights the diverse regulatory pathways through which astrocytes control glioma growth.
In summary, paracrine pathway represents a crucial pathway for CNS-glioma communication. This pathway encompasses a diverse array of signaling molecules, and due to the physiological diversity of various brain regions, gliomas occurring in different anatomical locations may be regulated by entirely distinct paracrine factors. Further exploration is needed to uncover these nuances.
Tunneling nanotubes
In the complex communication network between CNS and gliomas, a unique structure known as tunneling nanotubes (TNTs) has garnered increasing attention from researchers in recent years. TNTs share some similarities with the previously mentioned tumor microtubes, as both exhibit tubular extensions initiated from the cell membrane towards distant ends.
52
However, TNTs are thinner than TMs, cover longer distances (up to 100 μm), and have open ends at both terminals. Moreover, there are distinctions in the transported cargo between the two. TNTs have the ability to transfer large molecules, including proteins, nucleic acids, cellular structures like mitochondria and lysosomes.53-55 TNTs, constituting the fundamental framework of F-actin, include structural proteins such as GAP43
56
and GFAP,
57
playing crucial roles in facilitating mitochondrial transfer. Within the context of GBM, TNTs are generated under conditions of oxidative stress,
58
chemotherapy, and radiation, potentially serving as a self-protective mechanism for tumor tissues. Current research predominantly focuses on the transport of mitochondria through TNTs between the CNS and GBM cells. GBM cells, upon acquiring mitochondria from astrocytes, exhibit enhanced respiration and increased ATP synthesis. This metabolic shift is characterized by elevated levels of glutamate, α-ketoglutarate, glutathione, and essential amino acids, shielding GBM cells from oxidative stress. Furthermore, the increased number of GBM cells in the G2/M proliferation phase, along with enhanced self-renewal capabilities, may be linked to the upregulation of the transcription factor OCT4 and the involvement of mitochondria in regulating the JAK/STAT anti-apoptotic pathway
57
(Figure 3). Temozolomide (TMZ) chemotherapy is a crucial treatment for GBM following surgery or radiation. However, due to rapid development of resistance, the median survival of patients is only 14.6 months.
59
The above mitochondrial transport process also leads to the enhanced resistance of GBM cells to TMZ. The transfer of O(6)-methylguanine-DNA methyltransferase (MGMT), a drug-resistant enzyme, via TNTs may also explain the increased drug resistance.
60
Another study
61
corroborates this and reveals that the dihydroorotate dehydrogenase (DHODH) inhibitor Brequinar can reverse resistance induced by mitochondrial transfer, though related clinical trials are still under evaluation. TNTs serve as an emerging communication pathway between astrocytes and glioma. F-actin, GAP43, and GFAP are involved in the establishment of TNTs between glioma cells and astrocytes, facilitating bidirectional mitochondrial transport. This transport modulates the phenotypes of both cell types: glioma cells experience heightened ATP production and metabolic activity, accompanied by higher intracellular levels of antioxidants (glutamate and glutathione) and increased proliferation due to OCT4 upregulation. Concurrently, astrocytes undergo metabolic reprogramming, adopting a tumor-like metabolism to thrive in the hypoxic TME. The transfer of drug-resistance enzymes via TNTs may account for the resistance to TMZ chemotherapy.
In addition to the established transport of mitochondria from astrocytes to GBM cells through TNTs, there is also evidence of mitochondria transfer from GBM cells to astrocytes. This reciprocal exchange influences astrocytes to utilize glutamine as their main energy source, adapting their metabolic patterns to resemble tumor cells under hypoxic conditions 58 in the TME (Figure 3). In summary, TNTs provide a communication pathway between glioma cells and healthy astrocytes. The exchange of substances between them often leads to cancer cell metabolic reprogramming, drug resistance, and invasive phenotypes. Considering the characteristics of intercellular substance transfer via TNTs, exploiting TNTs may offer a therapeutic approach for GBMs. Research has reported that using engineered microglial cells to carry drugs can deliver the drugs to GBM cells through TNTs, achieving inhibition of GBM growth. 62 These studies expand our considerations for GBM treatment. On one hand, targeting the key component of TNTs, actin, to sever the connection between GBM cells and the surrounding environment may yield therapeutic effects. On the other hand, utilizing TNTs as a drug delivery pathway, without severing them, may significantly improve GBM treatment. Therefore, TNTs may represent a promising therapeutic target for GBMs.
Extracellular vesicles
Extracellular vesicles (EVs), secreted by various cells within the body, carry a diverse range of substances, including proteins, lipids, and nucleic acids, facilitating intercellular communication and material transfer.
63
Based on their size, content, and formation mechanisms, these vesicles can be classified into three main types: apoptotic bodies (1000-5000 nm), microvesicles (50-1000 nm), and exosomes (30-150 nm).
64
Due to their unique advantages such as membrane structure plasticity and biocompatibility in the glioma microenvironment, EVs serve as carriers of “cargo,” establishing a communication bridge between the CNS and glioma cells, playing a crucial role in the occurrence of gliomas (Figure 4A). EVs serve as a crucial bridge linking CNS and glioma. (A) An overall illustration of the communication among neurons, neuroglial cells, and glioma cells mediated by EVs. (B) The communication between astrocytes and GBM cells mediated by EVs involves GBM-derived EVs carrying lncRNA-ATB being internalized by healthy astrocytes. This process inhibits the expression of miRNA-204-3p in astrocytes, prompting their transformation into reactive astrocytes. GBM-derived EVs also transport CD147 to reactive astrocytes, enhancing MMP secretion, which facilitates extracellular matrix degradation and tumor invasion. Conversely, astrocyte-derived EVs carrying miR-124 exhibit an anti-tumor effect, while reactive astrocyte-derived EVs transporting TGF-β, MMP, and MGMT-mRNA support tumor progression. (C) The EV-mediated communication among neurons, GBM cells, and GSCs includes neuron-derived EVs transporting miR-184-3p to GSCs, inducing mesenchymal transition. Neuron-derived EVs can also transport miR-200c-3p to microglial cells, inducing M2 polarization. GBM-derived EVs can desynchronize neuronal activity. (D) The interaction between GAMs and GBM cells mediated by EVs involves GBM-derived EVs transporting miR-504 to GAMs, inducing M1 polarization with increased TGF-α and CD86 expression, which exhibits an anti-tumor effect. GAMs uptake of lnc-TALC, miR-304-5p, and Circ-0012381 from GBM-derived EVs upregulates CD163, IL-1, IL-10, and C5a expression, contributing to M2 polarization. M2 TAMs secrete TGF-β, IL-1, IL-10, further promoting GBM’s malignant progression.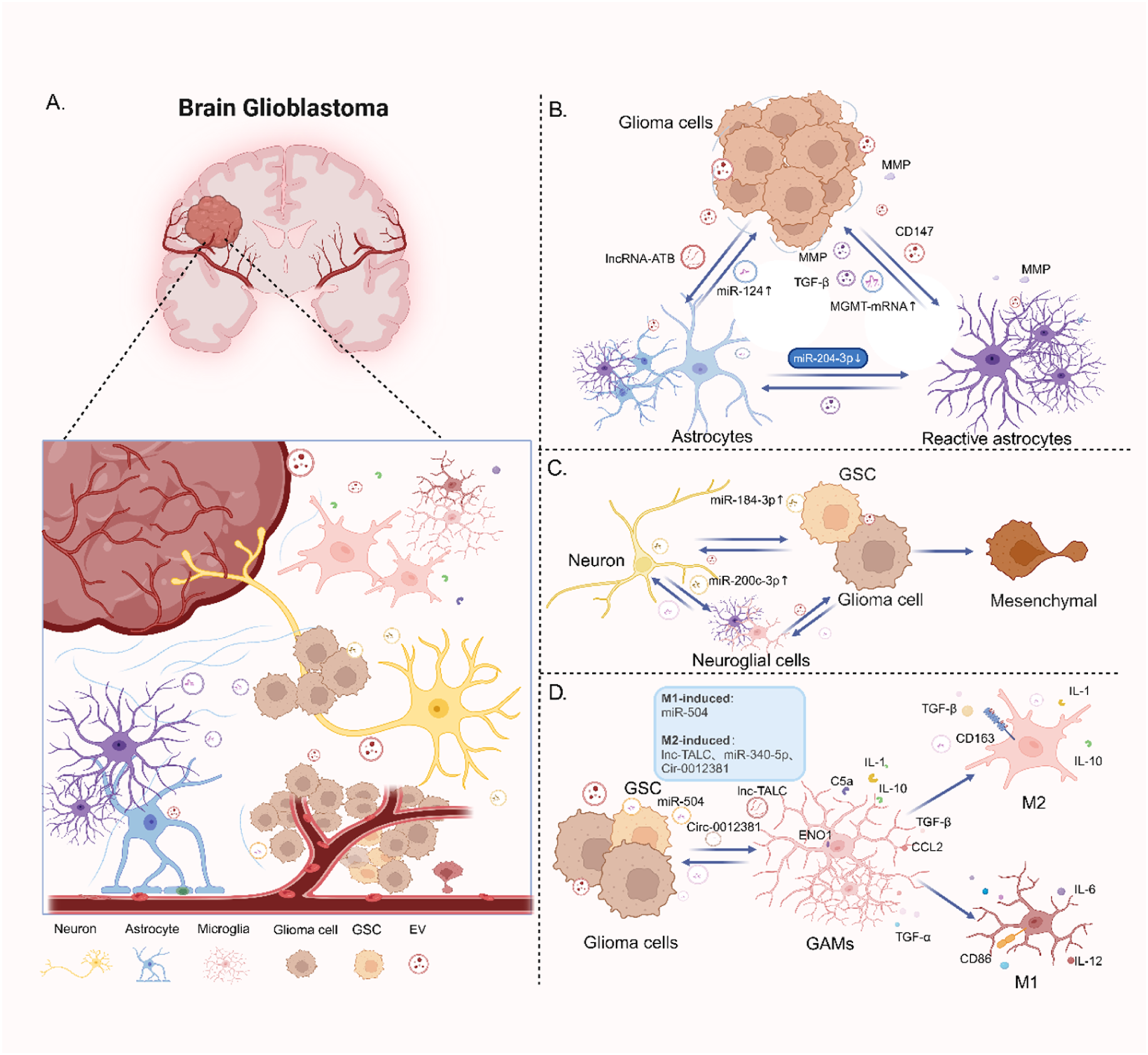
Astrocytes, being one of the most abundant cells in the brain and a vital component of the CNS, play a crucial role in maintaining brain homeostasis. They migrate towards tumor borders to isolate undamaged tissue from affected brain regions. 65 However, increasing evidence has confirmed the significant role of glioma-derived EVs in forming a complex communication network with astrocytes. 30 In vitro experiments have revealed that exposure to GBM-derived EVs can profoundly alter the intracellular signaling and cytokine profiles of normal astrocytes, driving them towards an enhanced tumor-supportive phenotype. 66 Hallal et al 67 discovered that GBM-derived EVs induce extracellular matrix (ECM) remodeling and blood-brain barrier (BBB) breakdown to promote GBM invasion by increasing the formation of filopodia in normal astrocytes. Subsequent research revealed that this mechanism is associated with increased expression of MYC and downregulation of p53 in astrocytes, further highlighting the potential of astrocytes to undergo transformation into cancer cells. Glioma stem cell-derived mRNA, carried by EVs, undergoes horizontal transfer between gliomas and astrocytes, promoting the transformation of astrocytes towards a tumor-supportive phenotype through guiding protein synthesis and metabolic reprogramming. 68 In recent years, non-coding RNA has been found to play a crucial role in the progression of gliomas. Bian et al 69 discovered that EVs derived from glioma cells, when taken up by astrocytes, deliver long non-coding RNA activated by transforming growth factor-β (LncRNA-ATB) to astrocytes, which downregulates miRNA-204-3p to enhance the activation of astrocytes. Activated astrocytes further intensify TGF-β secretion, accelerating glioma cell migration and invasion (Figure 4B). This facilitates the recruitment of new tumor cells into glioma tissue and maintains an immunosuppressive TME. Additionally, glioma-derived EVs carry the CD147 protein, activating the JNK signaling pathway in astrocytes, thereby promoting the generation of matrix metalloproteinases (MMP) and accelerating extracellular matrix degradation, ultimately enhancing tumor cell invasion 70 (Figure 4B).
In addition to astrocytes, communication between microglial cells and glioma cells is also significantly reliant on EVs. Following the uptake of glioma-derived EVs, certain gene expressions in microglial cells undergo alterations. Microglial cells, as resident cells in the brain and a major infiltrating cell population in glioma, are not only essential components of the CNS but also play a crucial role in the formation of immunosuppressive TME in gliomas, promoting immune escape and tumor resistance. 71 Previous research 72 has confirmed that tumor-associated microglial cells, upon internalizing GBM-derived EVs, experience reduced gene expression involved in perceiving tumor cells. This diminished perception capability leads to a compromised anti-tumor immune response. Additionally, MMP14, one of the three significantly upregulated MMPs (Mmp12, Mmp13, Mmp14) in these glioma-associated microglial cells, can activate the extracellular matrix degradation activity of MMP2. This activation is favorable for tumor spread and migration, although the specific mechanism remains unknown. 72 Zhang et al 73 discovered that the circular RNA Circ_0012381 is exclusively present in irradiated GBM cells and the microglia that phagocytized EVs from irradiated GBM cells. Circ_0012381, through upregulating the expression of TGFβ, CCL2, IL-1, IL-10, and CD163 in microglial cells, promotes M2 polarization and reduces their phagocytic capacity. This polarization might be associated with sponging with miR-340-5p, though the specific mechanisms remain unclear (Figure 4D).
More importantly, the communication between GBM and microglial cell, facilitated by EVs, contributes to GBM resistance to TMZ. Li et al 74 analyzed TMZ-resistant GBM cells and found that GBM-derived EVs transmit TMZ-related GBM recurrence lncRNA (lnc-TALC). This lncRNA, by binding to the ENO1 protein in microglial cells, activates the p38 MAPK signaling pathway, enhancing the release of the DNA damage repair-related cytokine C5a. This not only drives the M2 polarization of microglial cells but also promotes tumor cell resistance to TMZ. Blocking this crosstalk could become a novel therapeutic strategy for GBM. As research advances, the interaction between GBM-derived EVs and microglial cells not only promotes tumorigenesis but sometimes can also act as a means for drug delivery, exerting anti-tumor effects. For instance, miR-504 has been proven to be a tumor suppressor, inhibiting the stemness and mesenchymal transformation of GBM stem cells (GSCs). 75 EVs secreted by GSCs overexpressing miR-504 induce an increase in the expression of M1-related markers CD86 and TNF-α in microglial cells (Figure 4D), promoting anti-tumor immunity. 75
EVs not only serve as crucial means of communication between glial cells and GBM cells but also act as a significant link between neurons and GBM cells. Research indicates that EVs derived from GBM cells possess neurotoxicity, capable of inducing neuronal apoptosis or necrosis. 76 Renza et al 77 discovered that the electrical activity of neurons can be influenced by EVs originated from GBM cells. These EVs can induce synchronized electrical activity in primary cultured neurons. Interestingly, when GBM-derived EVs act on mature, synchronized neurons, the synchronicity of neuronal activity is disrupted (desynchronized). The existence of this dual effect suggests that EVs, as a communication pathway, can regulate a wide range of cellular functions, leading to various outcomes.
Just as EVs derived from GBM cells can influence the CNS, neurons or neuroglial cells can release EVs to regulate the occurrence and development of GBM. Astrocytes often play a dual role in either resisting or promoting GBM growth. This duality primarily depends on the modification of GBM cells on astrocytes and the regulatory factors carried by astrocyte-derived EVs. For instance, existing studies indicate that EVs released by healthy astrocytes can, through miR-124 mediation, decrease the expression of regulators associated with GBM cell morphology and migration, thereby resisting the growth and invasion of GBM. 78 During the development of GBM, astrocytes are induced into an activated state and transform into reactive astrocytes (RAS) as the tumor progresses. 78 Yu et al 79 observed a significant increase in the quantity of MGMT mRNA, which is carried by RAS-derived EVs. These EVs, when absorbed by MGMT-negative glioma cells, inhibit apoptosis (Figure 4B) and enhance the chemotherapy resistance of GBM cells. Similar to astrocytes, microglia-derived EVs also exhibit a dual regulatory mechanism on GBM. On one hand, the anti-glioma effect of small EVs derived from microglia is mediated by miR-124. It acts simultaneously on glioma cells and astrocytes, inhibiting the release of the neurotoxic factor glutamate by gliomas and inducing the expression of the glutamate transporter protein Glt-1 in astrocytes to increase glutamate uptake, thereby achieving a protective effect for neurons, maintaining the brain’s defense function. 80 On the other hand, in the TME of GBM, where glioma-associated microglia (GAM) predominantly polarize towards the M2 phenotype, M2 microglia-derived EVs promote GBM proliferation and migration by regulating the expression of tumor-related proteins and reducing cell apoptosis 81 (Figure 4D). Moreover, the reciprocal interaction between microglia and GBM cells is crucial in the tumor immune microenvironment (TIME). Neuronal activity not only regulates GBM through the generation of mitogens and neuron-glioma synaptic communication but also directly or indirectly drives GBM processes through neuron-derived EVs. A recent study 82 revealed that EVs secreted from neurons can act on GSCs, inducing GSCs to undergo mesenchymal transformation by activating the miR-184-3p/RBM15/DLG3/p-STAT3 pathway, contributing to the malignant progression and drug resistance. In addition to their direct effects on tumor cells, neuron-derived EVs can indirectly promote the formation of the tumor-adapted microenvironment by regulating the components within GBM TME. Li et al 83 found that neuronal activity upregulates miR-200c-3p levels in their secreted EVs. When taken up by microglia, these EVs induce epigenetic changes and activating the ERK pathway in microglia, resulting in their polarization towards the M2 phenotype (Figure 4C). This discovery opens the door to a new mechanism through which neuronal activity regulates the TIME of GBM.
In summary, GBM-derived EVs are continuously secreted and act on the CNS. Similarly, EVs derived from CNS cells also influence the progression of GBM. Therefore, EVs have become a crucial pathway mediating the communication between CNS and glioma.
CNS regulates TIME of glioma
GBM exhibits a unique immune microenvironment characterized by highly immunosuppressive cell infiltration, 84 hence referred to as an “immune-cold tumor” in immunology. 85 The TIME of GBM features high levels of immunosuppressive pro-tumor immune cells, such as regulatory T cells (Tregs), M2 macrophages, myeloid-derived suppressor cells (MDSC), and dysfunctional T cells. 86 Moreover, neurons and neuroglial cells, as primary components of the CNS, also participate in the formation of GBM TIME. 87 The specific mechanisms by which neurons and neuroglial cells contribute to the formation and maintenance of TIME remain incompletely understood. Therefore, gaining a deeper understanding of the CNS regulatory mechanisms on TIME and elucidating the evolution and chemotaxis of these immune-related cells may provide crucial insights for overcoming immune therapeutic challenges in GBM.
In the TIME of gliomas, a study has identified that NF1 gene mutations in neurons can increase RGC activity. Increased neuronal activity can stimulate the production of Midkine, a neuro-axonal growth-promoting factor, which, in turn, activates T cells to produce CCL4, stimulating microglial cells to secrete critical growth factor CCL5 required for glioma cell growth (Figure 5), this cascade promotes the further development of gliomas ultimately.88-90 By establishing the neuron-immune-glioma axis, these studies offered potential targets for glioma therapy. Additionally, in vitro studies revealed that neurons can also induce M2 polarization of microglial cells by releasing exosomes, thereby promoting the formation of an immunosuppressive microenvironment.
91
Mechanisms of CNS-TIME crosstalk in glioma. Neurons can promote M2 polarization in GAM by secreting EVs containing miR-200c-3p. Nf1 gene mutations in neurons can stimulate the secretion of the Midkine factor, inducing CD8+ T cells to release CCL4, further influencing GAM and promoting the expression of the tumor growth factor CCL5. Upregulation of the STAP1 gene in GAMs, through the IL-6/STAT3 pathway, further enhances the expression of the M2 characteristic protein ARG1. Additionally, M2 polarization can be achieved through the EVs carrying miR-504 secreted by glioma cells. GAMs also facilitate the transformation of astrocytes into reactive astrocytes. These transformed reactive astrocytes not only release anti-inflammatory factors such as TGF-β, G-CSF, and IL-10 but also secrete chemotactic factors like CCL2 and CCL7, promoting the recruitment of TAMs and upregulating the expression of PD-L1. All these processes contribute to the maintenance of an immunosuppressive TIME.
Not only can neuronal activity regulate the TIME of glioma, but astrocytes, as essential components of CNS, also play a regulatory role. Tumor-associated astrocytes (TAAs) in the TME of GBM, especially along the optic nerve pathway, play a significant role in shaping the immune microenvironment through various mechanisms. This includes regulating microglial cell activity, recruiting inflammatory cells to CNS, and even engaging in metabolic cascades. Research has revealed that the coexistence of microglial cells and astrocytes in the GBM TIME can induce astrocytes to transform into an anti-inflammatory phenotype, leading to the substantial release of anti-inflammatory cytokines such as TGFβ, IL-10, and G-CSF 92 (Figure 5). This promotes the formation and maintenance of an immunosuppressive microenvironment in GBM. 92 Interestingly, a subsequent study by Perelroizen et al 93 analyzed RAS in the GBM TIME and found that their pro-cancer phenotype is mainly enriched in metabolic processes, immune regulation, and cell proliferation pathways. In the TIME of GBM, tumor-associated macrophages (TAM) mainly involve brain-resident microglial cells and macrophages from the periphery.94,95 RAS, by secreting chemotactic factors like CCL2 and CCL7, directly control the recruitment of TAM to the GBM TIME, simultaneously elevating the expression of PD-L1 of TAM (Figure 5), consequnently contributing to the formation of immunosuppressive TME. 93 This reprograms microglial cells to facilitate their transformation into an immunosuppressive phenotype, thus reshaping the TIME to weaken anti-cancer immune responses and promote glioma progression. Excitingly, the depletion of RAS significantly impedes glioma growth.
GAM, a bridge between the CNS and immune systems, are crucial in connecting CNS and gliomas. The M2 phenotype of GAM is associated with positivity for enzyme arginase 1 (ARG1). 96 A recent study 97 confirmed differential expression of signal transducing adaptor protein 1 (STAP1), a signaling junction protein involved in tumor development and immune response, which is particularly elevated in GAM surrounding malignant glioma cells. This suggests its association with the malignant progression of gliomas. STAP1 activates IL-6/STAT3 in GAM, increasing ARG1 expression, promoting GAM M2 polarization, and reducing their phagocytic ability. This polarization is interdependent on interactions with other cells and microglial cells in the TIME, especially mediated by EVs73,74,83 (Figures 4C and 5). Furthermore, in addition to expediting M2 polarization, GAMs can activate the RAS’s JAK/STAT signaling pathway, inducing a specific anti-inflammatory phenotype, further contributing to the formation of an immunosuppressive microenvironment. 92
In conclusion, there exists a highly intricate connection between the nervous and immune systems, critical for regulating the occurrence and development of gliomas. In-depth exploration of the neuro-immune-glioma axis holds practical significance for understanding the pathogenic mechanisms of gliomas and improving current glioma treatment strategies.
Therapeutic strategies targeting CNS-glioma crosstalk
Recent advances in cancer neuroscience have provided fresh perspectives on therapeutic strategies for cancer treatment. Notably, drugs that target pivotal molecules involved in the interactions between the CNS and gliomas have undergone preclinical testing, yielding promising results. This section delves into the drugs identified through preclinical research or clinical trials that have demonstrated therapeutic efficacy.
Targeting synapses
From the perspective of cancer neuroscience, targeted interventions against key molecules within synaptic structures is emerging as a promising approach in glioma therapy. Consequently, the development of drugs that inhibit glutamate or its receptors has become a significant treatment strategy. For instance, perampanel, an FDA approved medication for epilepsy, acts as an AMPA receptor antagonist. 98 Since AMPA receptor significantly mediates the CNS-glioma communication, studies have indicated that prolonged administration of perampanel can markedly decrease glioma proliferation in mouse models, showcasing its anti-cancer properties. 22 Moreover, BBB is very permeable for perampanel. 99 Therefore, perampanel is gaining much attention from clinical researchers. Recently, a clinical trial investigating perampanel’s therapeutic effect on recurrent or progressive GBM has been initiated in Germany. 100 This is a multi-center and phase II study, assessing NGS connectivity and tumor growth rate changes after treated with perampanel. For breast-to-brain metastatic cancer, within the pseudo-tripartite synapse, the NMDA receptor on breast cancer cells is an important target. Glutamate released by neurons acting on NMDA receptors could accelerate breast cancer migration and invasion in the brain. 101 For various CNS diseases, numerous drugs targeting NMDA receptors have been developed. For instance, ketamine, targeting human NMDA receptors, has been used as a novel antidepressant with promising results. 102 In the case of GBM, research has revealed that NMDAR signaling mediates DNA methylation, leading to TMZ resistance. 103 Drugs targeting NMDAR, such as memantine and its derivatives, have been proven to have some anti-glioma effects and are commonly used in clinical settings. 104 Additionally, combining memantine with other drugs may have unexpected effects.
Glioma cells connect with each other through TMs and gap junctions to form a cellular network, as previously detailed in their impact on glioma resistance to radiotherapy and chemotherapy. Gap junctions, also known as electrical synapses, are a type of synapse structure in the broad sense. As previously described, the gap junctions between glioma cells robustly enhance the oncogenic effect of neuronal activity. Hence, drugs targeting gap junctions have huge therapeutic potentials. Current pre-clinical research has demonstrated that the treatment of glioma cell networks with the orally bioavailable gap junction modulator Meclofenamate significantly slows glioma growth and enhances sensitivity to TMZ.21,105,106 Furthermore, the presence of gap junctions between astrocytes and gliomas can promote gliomas migration. Studies have shown that treatment with Meclofenamate and another gap junction inhibitor, tonabersat, results in a significant reduction in glioma migration. 105
There is no doubt that the synapse remains a crucial pathway in CNS-glioma communication. Delving into the molecular mechanisms within this pathway and developing effective targeted drugs against corresponding targets are crucial avenues for future research.
Targeting paracrine factors
Just like synapses, paracrine mechanisms play an indispensable role in CNS-glioma communication. Notable molecular targets include NLGN3, BDNF, IGF-1, as well as various neurotransmitters and their corresponding receptors.
As detailed in Section 2.2, NLGN3 acts as a paracrine molecule modulated by neuronal activity, playing a significant role in promoting glioma progression. The production of NLGN3 is contingent upon the involvement of ADAM10, thus making ADAM10 a primary target for current therapeutic strategies against NLGN3. 43 For instance, GI254023X is recognized as an effective ADAM10 inhibitor, demonstrating notable anti-glioma efficacy and brain penetration in preclinical studies when administered intraperitoneally to mice.43,107 Similarly, INCB3619, another potent ADAM10 inhibitor, has shown promising anti-cancer properties in lung and colon cancer through cellular and mouse model studies. Nevertheless, its efficacy in treating glioma remains to be conclusively determined. Beyond preclinical research, the ADAM10 inhibitor INCB7839 is currently undergoing Phase I clinical trials for children with recurrent or progressive high-grade gliomas, expected to complete by June 30, 2024 (NCT04295759). However, the ability of this drug to cross the BBB is yet to be clarified. Given that NLGN3 triggers a cascade of downstream signaling pathways, including the PI3K/AKT/mTOR pathway, drugs that inhibit this pathway are of considerable clinical interest. The mTOR inhibitor paxalisib was approved by the FDA in 2020 for the treatment of GBM, underscoring its significance. Besides NLGN3, BDNF also plays an essential role through its receptor, TrkB. Larotrectinib and Entrectinib are primary drugs targeting the TrkB receptor. Preclinical studies have shown that Entrectinib effectively blocks TrkB, significantly reducing glioma cell proliferation in mice. 50 Yet, the absence of clinical trials for TrkB-targeting drugs in glioma patients highlights TrkB as an untapped therapeutic avenue.
Targeting other mechanisms
As research on neuro-glioma interactions continues to deepen, some newly emerging targets deserve our attention, including Human semaphorin 4F (SEMA4F), metalloproteinases, and G proteins. SEMA3F belongs to the semaphorin (SEMA) protein family. The SEMA family is a group of secreted or transmembrane proteins that often play a role in axon guidance. 108 Several proteins within the SEMA family have been found to play key roles in TIME regulation. For example, SEMA4D has been shown to be highly expressed in the TIME of glioma and is an important immune regulatory molecule. 109 Targeted drug Pepinemab against SEMA4D has entered a Phase II clinical trial in head and neck cancer patients and has shown promising results. 110 However, overall, the role of the SEMA family in the development of gliomas remains largely unknown, indicating significant therapeutic potential for gliomas. Recent research has discovered that neuronal activity away from the primary glioma site can regulate the infiltration of glioma cells at the primary site, and this process is mediated by SEMA4F. 111 It’s worth noting that there are currently no drugs developed specifically for SEMA4F. Considering the reported roles of the SEMA family in other cancers, the research value of SEMA4F in GBM should not be overlooked.
Meanwhile, the role of metalloproteinases in cancer progression is also worth our attention, and one subtype, MMP, plays a crucial role in epithelial-mesenchymal transition (EMT). 112 EMT is a well-known biological process that plays a pivotal role in cancer invasion. 113 Studies have shown that glioma cells can activate TAA through the Wnt/β-catenin signaling pathway, and in TAA, the expression of MMP is significantly upregulated. Cells use MMP to degrade the basement membrane, change their polarity, rearrange the cell cytoskeleton, and migrate, 112 directly leading to EMT, creating a conducive environment for glioma migration and invasion. 114 Given the critical role of MMP in the glioma invasion process, a novel CAR-T cell therapy, CHM-1101, is currently in Phase I clinical trials (NCT05627323). This therapy can inhibit MMP2, thereby halting the progression of GBM.
Furthermore, we should also pay attention to G proteins, which are widely distributed signal transduction proteins and significantly influence the progression of gliomas. Research indicates that Gαi1 and Gαi3 participate in the transduction of downstream signals activated by NLGN3 or BDNF, thereby affecting patient prognosis.115,116 Given the significant roles of NLGN3 and BDNF in both synaptic and paracrine pathways between CNS and gliomas, G proteins act as an undeniable bridge linking CNS and glioma. Disrupting the functions of these proteins can effectively inhibit the activation of their downstream signaling pathways, thereby achieving the goal of glioma suppression.115,116 Moreover, the Gαi1/3 protein-mediated signaling pathways can also promote cancer angiogenesis. By intervening in these pathways to inhibit neovascularization, we can effectively hinder cancer progression.117,118 This suggests that Gαi1/3 proteins have the potential to become new therapeutic targets.
Representative targets and potential therapeutic strategies for CNS-glioma interaction.

Summary and outlook
The advent of cancer neuroscience has unveiled entirely new avenues for unraveling the intricate mechanisms underlying tumorigenesis. In the case of glioma, its development and progression are profoundly influenced by interactions with normal neurons and neuroglial cells, through established complex communication networks. This review offers a thorough examination of the current understanding of CNS-glioma communication pathways and associated therapeutic approaches. We have detailed the five principal pathways of communication between the CNS and glioma, encompassing synaptic interactions, paracrine signaling, EVs, TNTs, and the CNS- TIME-glioma axis.
Since there have been some breakthroughs in CNS cancer neuroscience in recent years, many research directions are worth noting. For example, EVs, which have gained significant attention in recent years, have been shown to play an important role in CNS-glioma interaction. However, currently published articles have primarily focused on EVs originating from glioma cells and astrocytes, without enough attention to neuron-derived EVs. Actually, neuron-secreted EVs have been demonstrated to have a unique role in the detection and diagnosis of neurological diseases, including Alzheimer’s disease. 122 Although Li et al91,123 have recently confirmed that neuron-derived EVs exert a crucial role in affecting the TIME in glioma and directly influencing glioma cells via miRNAs, current knowledge about neuron-derived EVs is still limited. Therefore, future studies should concentrate on exploring the multiple effects of neuron-derived EVs on glioma. Given the complexity of the sources of EVs within the glioma microenvironment, it remains unclear whether glioma cells can selectively recognize EVs from specific cellular origins. The underlying mechanisms for this potential selectivity warrant further investigation. Additionally, the process of EV uptake involves membrane fusion, and whether this process presents potential targets for pharmacological intervention has yet to be elucidated. 124
In future research, a particularly compelling avenue to explore is the role of neuro-immune interactions in glioma progression. Like the nervous system, the immune system is characterized by its widespread distribution, its sensitivity to changes, and its intricate relationship with cancer. 125 In fact, neurons may regulate the TIME of GBM in multiple ways, with numerous studies providing clues in this regard. For instance, Benjamin Deneen’s research revealed significant changes in the proportion of immune cells around GBM lesions when neurons on the contralateral side were artificially activated in mice models. 111 Additionally, Korvenoja et al 126 observed macrophages and microglial cells ingesting neurons’ secreted EVs in a mouse brain ischemia model. These studies suggest that neurons are highly likely to influence the TIME of GBM directly or indirectly through various pathways. 127 Another noteworthy direction is the study of the human brain connectome, and the first connectome map for the fruit fly brain has already been drawn. 128 Research on the human brain connectome helps us gain a comprehensive understanding of the connections between different types of neurons in the brain, contributing to a holistic understanding of the factors influencing the development of brain tumors. Given the nascent stage of cancer neuroscience, this review provides a summary and synthesis of the major research findings, with the hope of shedding light on new directions for future studies.
Footnotes
Acknowledgments
All figures and graphical abstract are created with Biorender.com.
Author contributions
All authors provided helpful suggestions.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Presidential Foundation of Zhujiang Hospital, Southern Medical University (grant number: yzjj2022ms4).
